Predicting Abnormal Respiratory Patterns in Older Adults Using Supervised Machine Learning on Internet of Medical Things Respiratory Frequency Data
Abstract
:1. Introduction
2. Related Works
3. Materials and Methods
3.1. Data Acquisition
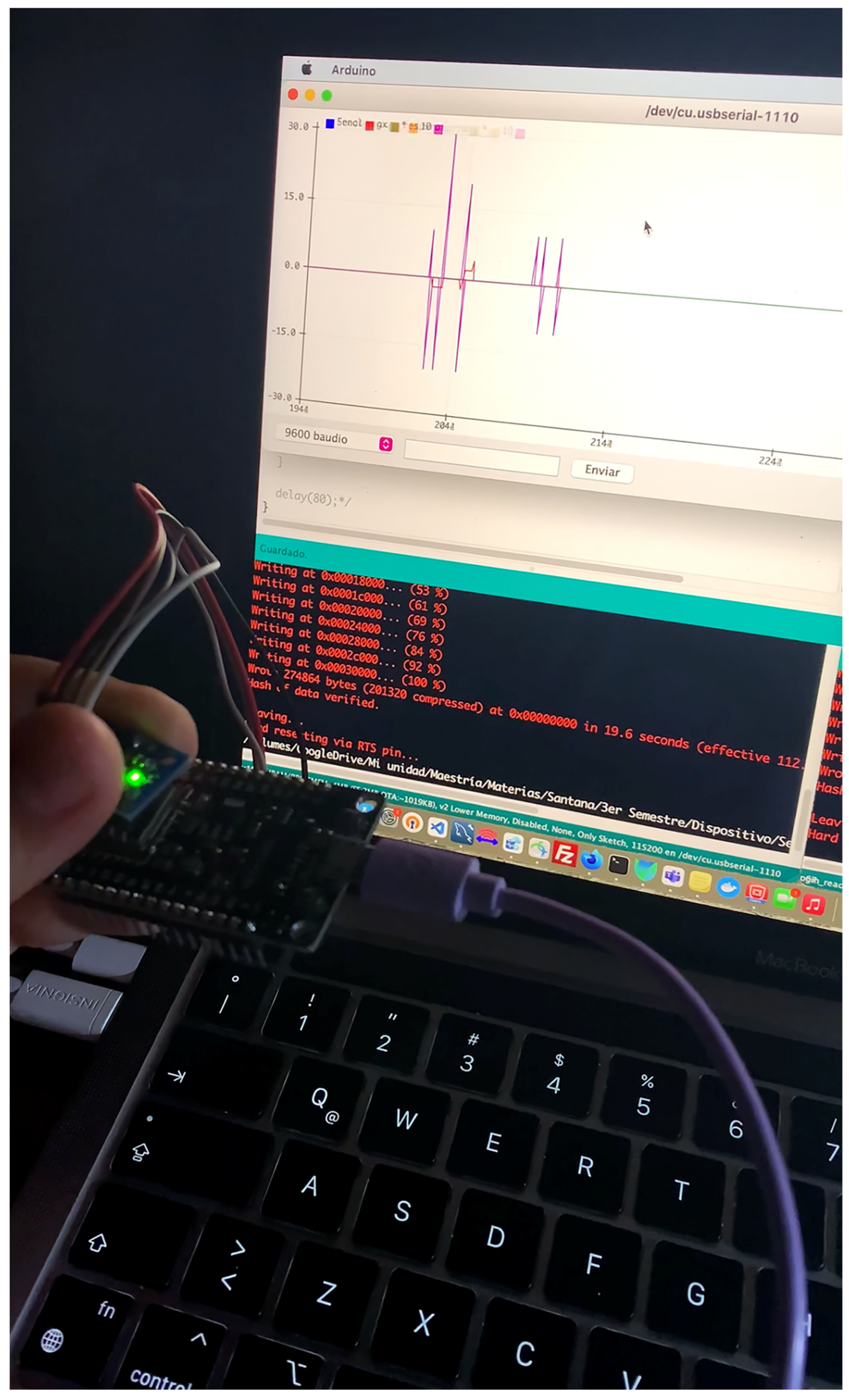
3.1.1. Microphone Sensor Prototype
3.1.2. Gas Sensor Prototype
3.1.3. Movement Sensor Prototype
3.1.4. Radar Sensor Prototype
3.2. Dataset Construction
- Age: The age of the patient (numeric).
- RPM (respirations per minute): Numeric representation of the respiratory rate.
- Sex: The gender of the patient (numeric):
- 0 = female
- 1 = male
- Normal: Indicator of whether the RPM falls within the normal range based on the patient’s age and the ranges specified in the existing literature (numeric):
- 0 = no
- 1 = yes
- Eupnea (normal relaxed breathing): 12–20 RPM
- Normal range > 65 years: 12–25 RPM
- Normal range > 80 years: 10–30 RPM
- Bradypnea (slow respiratory rate): <12 RPM
- Tachypnea (fast respiratory rate): >20 RPM
3.3. Dataset Augmentation
3.4. Machine Learning Models
3.4.1. Data Preprocessing
3.4.2. Model Evaluation
- TP = true positives
- FP = false positives
- FN = false negatives
3.4.3. Hyperparameter Tuning
3.4.4. Comparison
3.4.5. Cross-Validation
4. Results
4.1. Exploratory Data Analysis
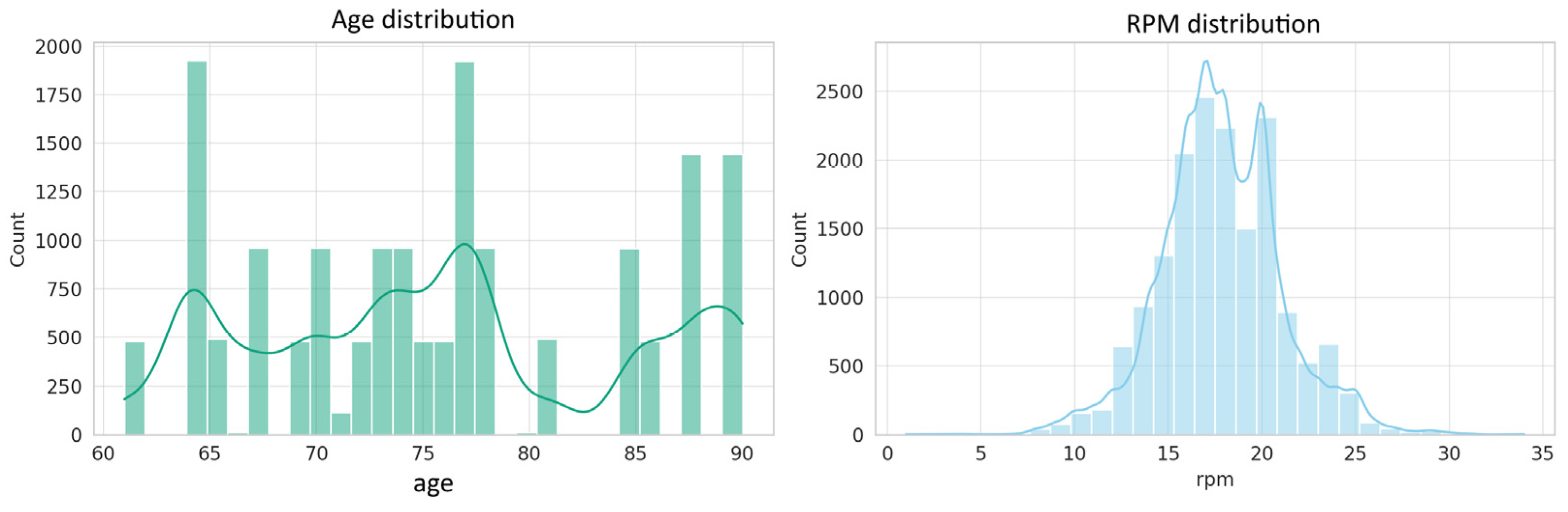
4.1.1. Numeric Variable Distributions
4.1.2. Variable Correlations
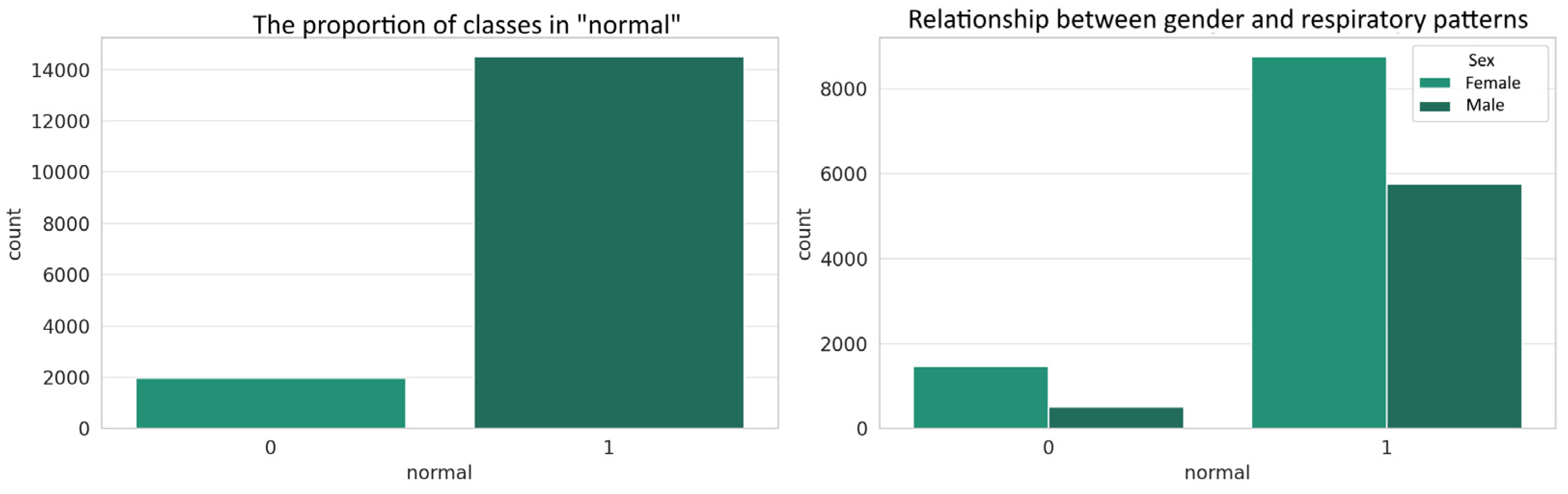
4.2. Class Imbalance
4.3. Feature Scaling
4.4. Data Partitioning
- Training set (features): 13,209 rows by 3 columns.
- Testing set (features): 3303 rows by 3 columns.
- Training set (target): 13,209 rows.
- Testing set (target): 3303 rows.
4.5. Model Validation
- K-nearest neighbors (K-NN)
- ○
- Training time: 0.009 s
- ○
- Prediction time: 0.168 s
- Support vector machine (SVM)
- ○
- Training time: 0.484 s
- ○
- Prediction time: 0.093 s
- Gradient boosting
- ○
- Training time: 0.592 s
- ○
- Prediction time: 0.006 s
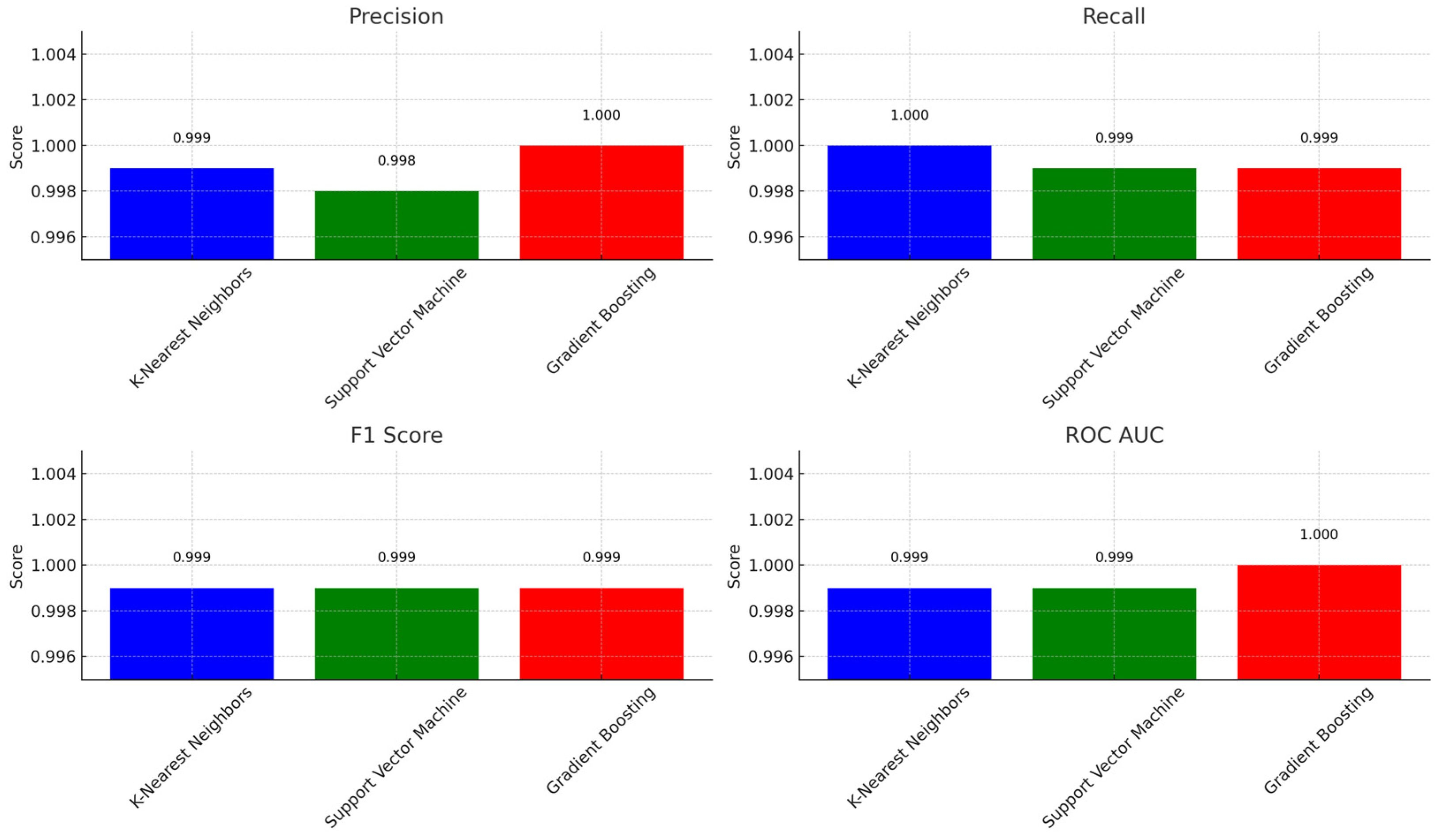
4.5.1. Evaluation Metrics
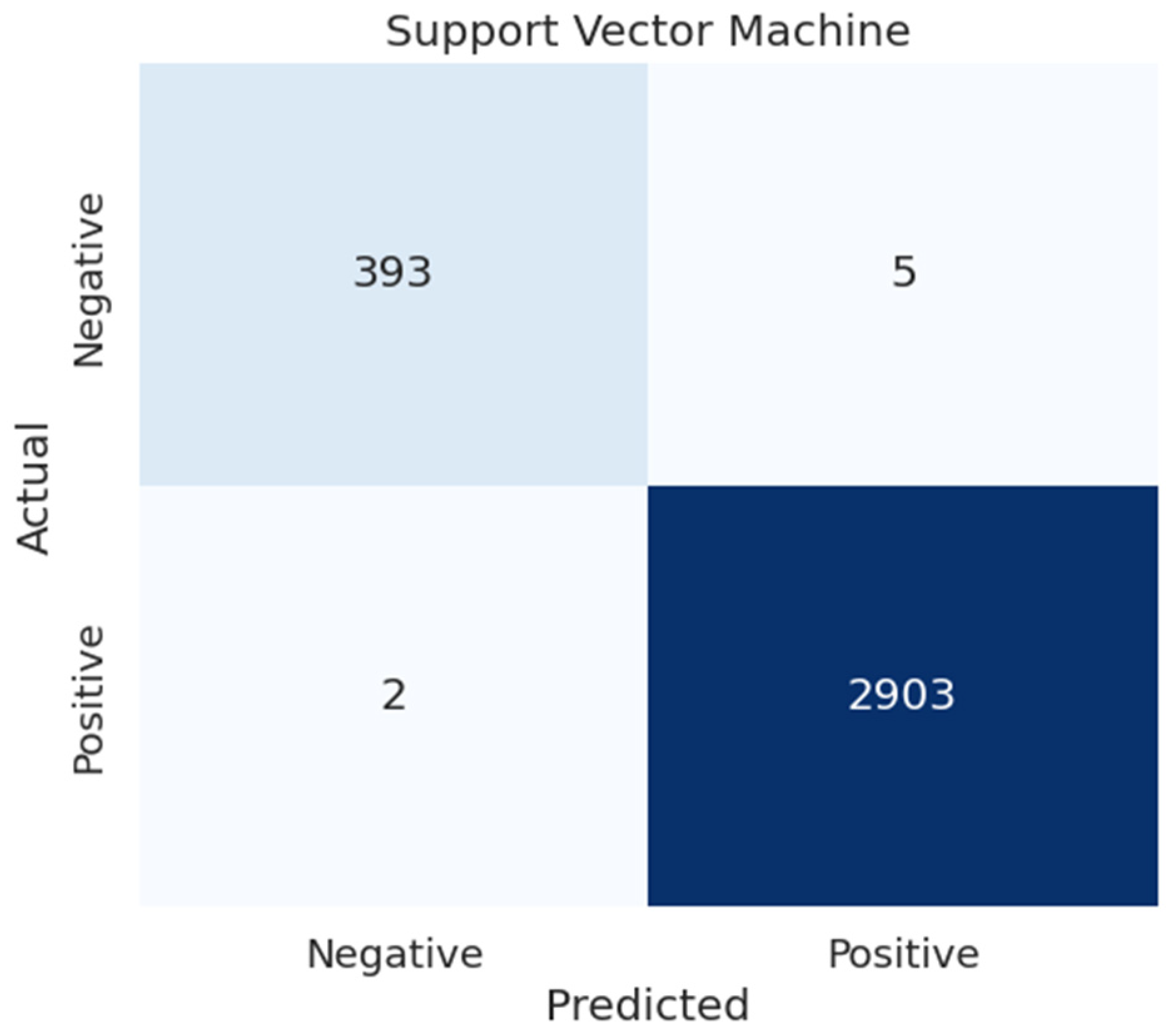
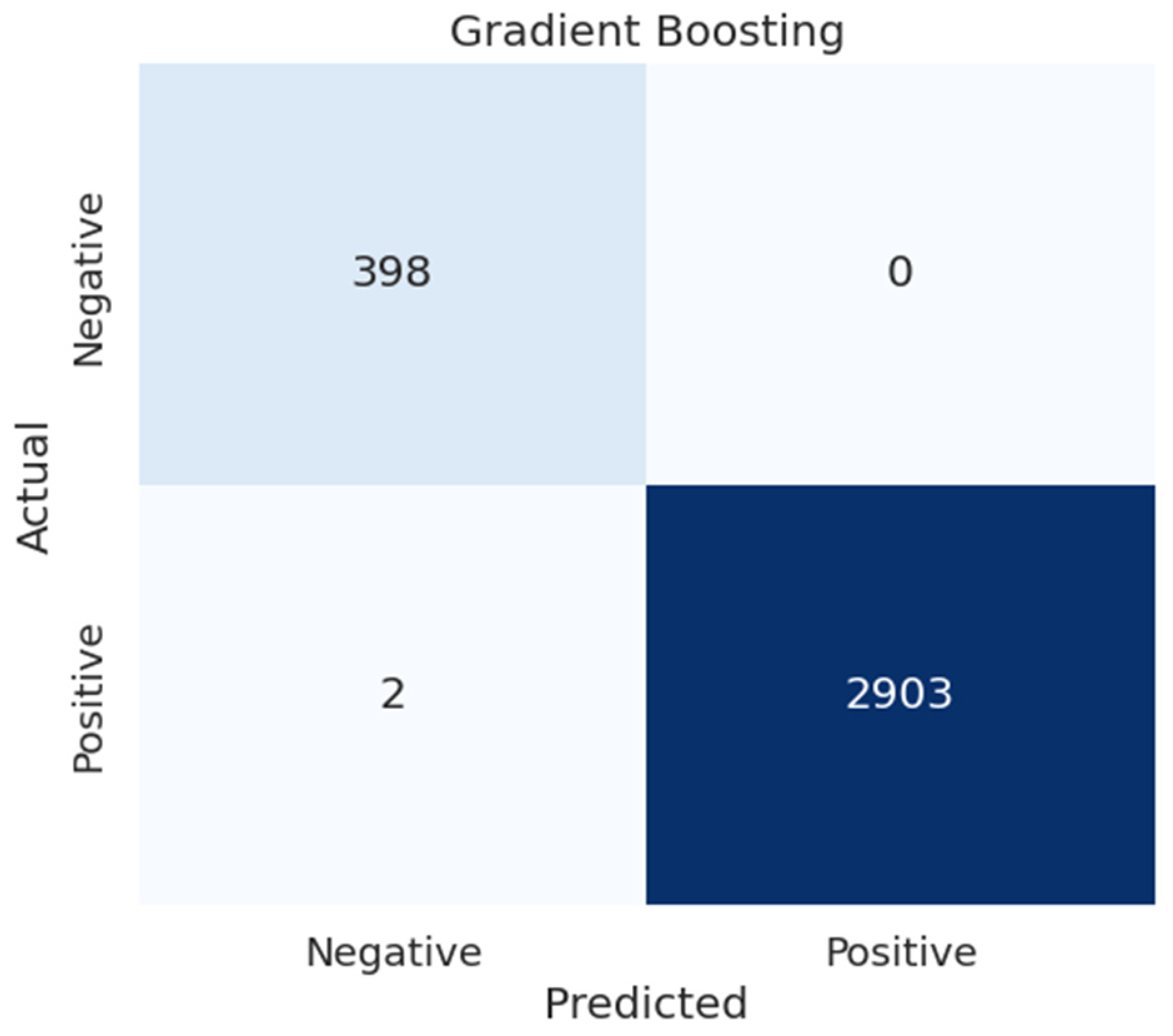
4.5.2. Confusion Matrices
4.5.3. Cross-Validation
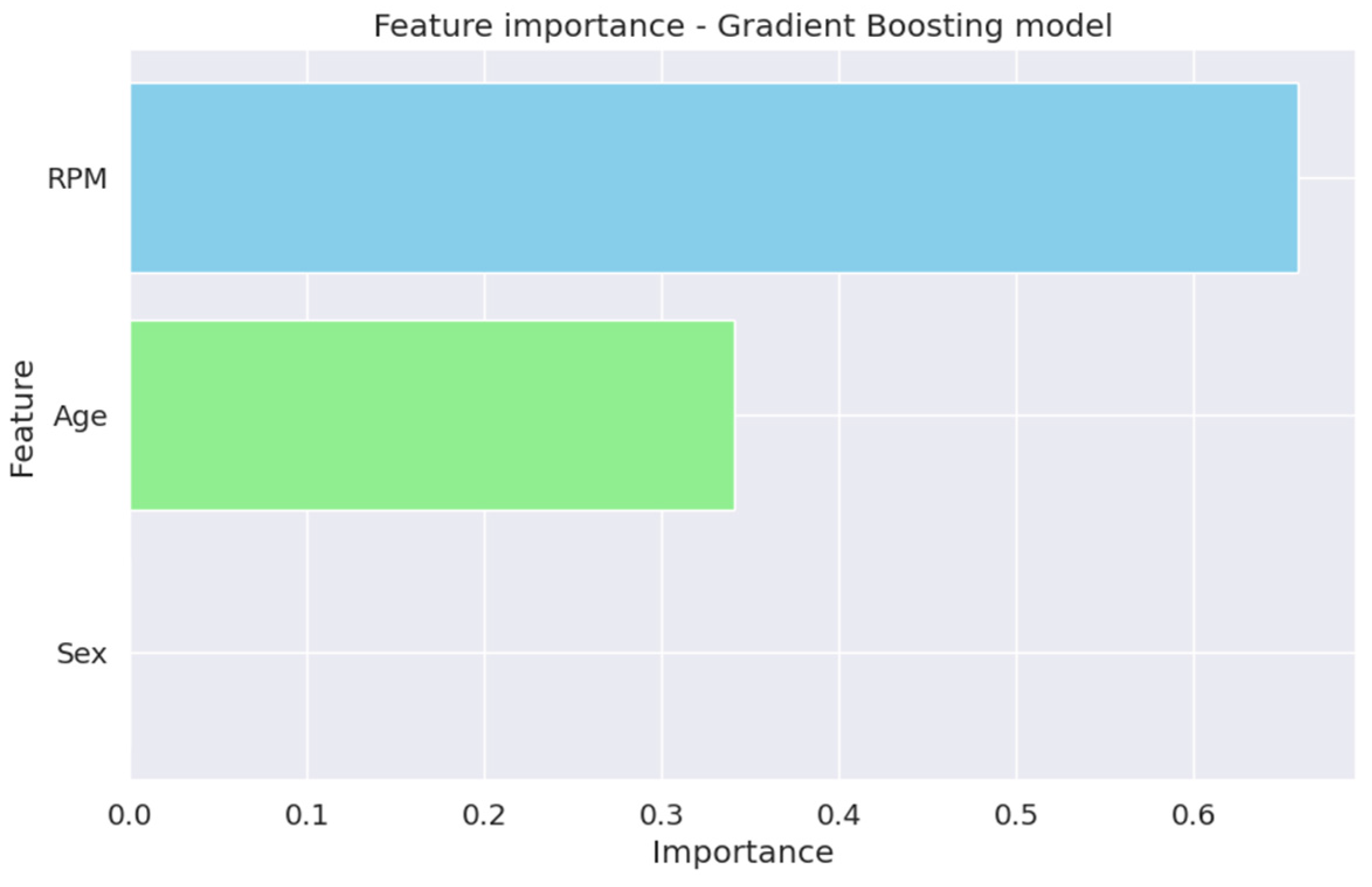
4.5.4. Feature Importance
5. Discussion
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Juárez-Ramírez, C.; Márquez-Serrano, M.; Salgado de Snyder, N.; Pelcastre-Villafuerte, B.E.; Ruelas-González, M.G.; Reyes-Morales, H. Health inequality among vulnerable groups in Mexico: Older adults, indigenous people, and migrants. Rev. Panam. Salud Publica 2014, 35, 284–290. [Google Scholar] [PubMed]
- Rojas, P.M.M.; Díaz, V.R.V.; Sacramento, P.I.; Rodríguez, M.M.; Martínez, R.L.; Delgado, P.G. Mortalidad por enfermedades respiratorias en el adulto mayor. Evolución en un año. Acta Médica Cent. 2016, 10, 33–39. [Google Scholar]
- Ibarra Cornejo, J.L.; Fernández Lara, M.J.; Aguas Alveal, E.V.; Pozo Castro, A.F.; Antillanca Hernández, B.; Quidequeo Reffers, D.G. Efectos Del Reposo Prolongado En Adultos Mayores Hospitalizados. Ann. Fam. Med. 2018, 78, 439. [Google Scholar] [CrossRef]
- Escobar-Rojas, A.; Castillo-Pedroza, J.; Cruz-Hervert, P.; Báez-Saldaña, R. Tendencias de morbilidad y mortalidad por neumonía en adultos mexicanos (1984–2010). Neumol. Y Cirugía Tórax 2015, 74, 4–12. [Google Scholar]
- De Smet, R.; Mellaerts, B.; Vandewinckele, H.; Lybeert, P.; Frans, E.; Ombelet, S.; Lemahieu, W.; Symons, R.; Ho, E.; Frans, J.; et al. Frailty and Mortality in Hospitalized Older Adults with COVID-19: Retrospective Observational Study. J. Am. Med. Dir. Assoc. 2020, 21, 928–932.e1. [Google Scholar] [CrossRef]
- Feehan, J.; Apostolopoulos, V. Is COVID-19 the Worst Pandemic? Maturitas 2021, 149, 56–58. [Google Scholar] [CrossRef]
- Wu, Q.; Li, Q.; Lu, J. A One Health Strategy for Emerging Infectious Diseases Based on the COVID-19 Outbreak. J. Biosaf. Biosecurity 2022, 4, 5–11. [Google Scholar] [CrossRef]
- Arboleda, J.F. Sistema Para El Apoyo a La Atención Domiciliaria Mediante Redes de Sensores Inteligentes. Master’s Thesis, Universidad de Antioquia, Colombia, SC, USA, 2016. [Google Scholar]
- Santana-Mancilla, P.C.; Anido-Rifón, L.E.; Contreras-Castillo, J.; Buenrostro-Mariscal, R. Heuristic Evaluation of an IoMT System for Remote Health Monitoring in Senior Care. Int. J. Environ. Res. Public. Health 2020, 17, 1586. [Google Scholar] [CrossRef]
- Guzman-Sandoval, V.M.; Gaytan-Lugo, L.S.; Santana-Mancilla, P.C. I-Care: An IoMT Remote Monitoring System of Physiological Pain in Pediatric Patients. In Proceedings of the 2021 Mexican International Conference on Computer Science (ENC), IEEE, Morelia, Mexico, 9–11 August 2021; pp. 1–4. [Google Scholar]
- Luque, N.; Ortega, M. Análisis de Sistemas Para Registros Médicos Electrónicos En Clínicas y Su Enfoque al Machine Learning. Bachelor’s Thesis, Universidad Católica San Pablo, Arequipa, Perú, 2020. [Google Scholar]
- Argerich, S.; Herrera, S.; Benito, S.; Giraldo, B.F. Evaluation of Periodic Breathing in Respiratory Flow Signal of Elderly Patients Using SVM and Linear Discriminant Analysis. In Proceedings of the 2016 38th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), IEEE, Orlando, FL, USA, 16–20 August 2016; pp. 4276–4279. [Google Scholar]
- Khan, M.F.; Ghazal, T.M.; Said, R.A.; Fatima, A.; Abbas, S.; Khan, M.A.; Issa, G.F.; Ahmad, M.; Khan, M.A. An IoMT-Enabled Smart Healthcare Model to Monitor Elderly People Using Machine Learning Technique. Comput. Intell. Neurosci. 2021, 2021, 1–10. [Google Scholar] [CrossRef]
- Pham, L.; Ngo, D.; Tran, K.; Hoang, T.; Schindler, A.; McLoughlin, I. An Ensemble of Deep Learning Frameworks for Predicting Respiratory Anomalies. In Proceedings of the 2022 44th Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC), IEEE, Glasgow, Scotland, UK, 11–15 July 2022; pp. 4595–4598. [Google Scholar]
- Wang, Y.; Hu, M.; Zhou, Y.; Li, Q.; Yao, N.; Zhai, G.; Zhang, X.-P.; Yang, X. Unobtrusive and Automatic Classification of Multiple People’s Abnormal Respiratory Patterns in Real Time Using Deep Neural Network and Depth Camera. IEEE Internet Things J. 2020, 7, 8559–8571. [Google Scholar] [CrossRef]
- Jin, Y.; Yu, H.; Zhang, Y.; Pan, N.; Guizani, M. Predictive Analysis in Outpatients Assisted by the Internet of Medical Things. Future Gener. Comput. Syst. 2019, 98, 219–226. [Google Scholar] [CrossRef]
- Bazán, J. BioMakers University. Available online: https://www.biomakers.ai (accessed on 30 September 2023).
- XeThru X4M200 Respiration Sensor. X4M200 Respir. Sens. 2018. Available online: https://www.xethru.com/x4m200-respiration-sensor.html (accessed on 30 September 2023).
- Wheatley, I. Respiratory rate 3: How to take an accurate measurement. Nurs. Times 2018, 114, 21–22. [Google Scholar]
- Fatihah Shamsul Ariffin, F.; Munirah Kamarudin, L.; Ghazali, N.; Nishizaki, H.; Zakaria, A.; Muhammad Mamduh bin Syed Zakaria, S. Inhalation and Exhalation Detection for Sleep and Awake Activities Using Non-Contact Ultra-Wideband (UWB) Radar Signal. J. Phys. Conf. Ser. 2021, 1755, 012038. [Google Scholar] [CrossRef]
- Oscar, E.; Castrejón-Mejía; Pedro, C. Santana-Mancilla Dataset of Respiration Rates in Older Adults. Available online: http://doi.org/10.34740/kaggle/dsv/6652581 (accessed on 20 September 2023).
- Mezquita, Y.; Alonso, R.S.; Casado-Vara, R.; Prieto, J.; Corchado, J.M. A Review of K-NN Algorithm Based on Classical and Quantum Machine Learning. In Distributed Computing and Artificial Intelligence, Special Sessions, 17th International Conference; Rodríguez González, S., González-Briones, A., Gola, A., Katranas, G., Ricca, M., Loukanova, R., Prieto, J., Eds.; Advances in Intelligent Systems and Computing; Springer International Publishing: Cham, Switzerland, 2021; Volume 1242, pp. 189–198. ISBN 978-3-030-53828-6. [Google Scholar]
- Noble, W.S. What Is a Support Vector Machine? Nat. Biotechnol. 2006, 24, 1565–1567. [Google Scholar] [CrossRef]
- Bentéjac, C.; Csörgő, A.; Martínez-Muñoz, G. A Comparative Analysis of Gradient Boosting Algorithms. Artif. Intell. Rev. 2021, 54, 1937–1967. [Google Scholar] [CrossRef]
- Camizuli, E.; Carranza, E.J. Exploratory Data Analysis (EDA). In The Encyclopedia of Archaeological Sciences; López Varela, S.L., Ed.; Wiley: Hoboken, NJ, USA, 2018; pp. 1–7. ISBN 978-0-470-67461-1. [Google Scholar]
- Japkowicz, N.; Stephen, S. The Class Imbalance Problem: A Systematic Study1. Intell. Data Anal. 2002, 6, 429–449. [Google Scholar] [CrossRef]
- Li, D.; Zhang, B.; Li, C. A Feature-Scaling-Based k -Nearest Neighbor Algorithm for Indoor Positioning Systems. IEEE Internet Things J. 2016, 3, 590–597. [Google Scholar] [CrossRef]
- Korjus, K.; Hebart, M.N.; Vicente, R. An Efficient Data Partitioning to Improve Classification Performance While Keeping Parameters Interpretable. PLoS ONE 2016, 11, e0161788. [Google Scholar] [CrossRef]
- Dalianis, H. Evaluation Metrics and Evaluation. In Clinical Text Mining; Springer International Publishing: Cham, Switzerland, 2018; pp. 45–53. ISBN 978-3-319-78502-8. [Google Scholar]
- Joy, T.T.; Rana, S.; Gupta, S.; Venkatesh, S. Hyperparameter Tuning for Big Data Using Bayesian Optimisation. In Proceedings of the 2016 23rd International Conference on Pattern Recognition (ICPR), IEEE, Cancun, Mexico, 4–8 December 2016; pp. 2574–2579. [Google Scholar]
- Montesinos-López, O.A.; Montesinos-López, A.; Cano-Paez, B.; Hernández-Suárez, C.M.; Santana-Mancilla, P.C.; Crossa, J. A Comparison of Three Machine Learning Methods for Multivariate Genomic Prediction Using the Sparse Kernels Method (SKM) Library. Genes 2022, 13, 1494. [Google Scholar] [CrossRef]
- Berrar, D. Cross-Validation. In Encyclopedia of Bioinformatics and Computational Biology; Elsevier: Amsterdam, The Netherlands, 2019; pp. 542–545. ISBN 978-0-12-811432-2. [Google Scholar]
- Bhagat, M.; Bakariya, B. Implementation of Logistic Regression on Diabetic Dataset Using Train-Test-Split, K-Fold and Stratified K-Fold Approach. Natl. Acad. Sci. Lett. 2022, 45, 401–404. [Google Scholar] [CrossRef]
- Saarela, M.; Jauhiainen, S. Comparison of Feature Importance Measures as Explanations for Classification Models. SN Appl. Sci. 2021, 3, 272. [Google Scholar] [CrossRef]
- Siddiqui, H.U.R.; Shahzad, H.F.; Saleem, A.A.; Khan Khakwani, A.B.; Rustam, F.; Lee, E.; Ashraf, I.; Dudley, S. Respiration Based Non-Invasive Approach for Emotion Recognition Using Impulse Radio Ultra Wide Band Radar and Machine Learning. Sensors 2021, 21, 8336. [Google Scholar] [CrossRef] [PubMed]
- Ying, X. An Overview of Overfitting and Its Solutions. J. Phys. Conf. Ser. 2019, 1168, 022022. [Google Scholar] [CrossRef]




| Age | RPM | Sex | Normal |
|---|---|---|---|
| −0.776 | −0.256 | −0.778 | 1 |
| −0.083 | −0.256 | 1.286 | 1 |
| −1.007 | −0.256 | −0.778 | 1 |
| 0.264 | 0.364 | −0.778 | 1 |
| 1.650 | −0.566 | −0.778 | 1 |
| Model | Precision | Recall | F1 Score | ROC-AUC |
|---|---|---|---|---|
| K-nearest neighbors | 0.999 | 1.000 | 0.999 | 0.999 |
| Support vector machine | 0.998 | 0.999 | 0.999 | 0.999 |
| Gradient boosting | 1.000 | 0.999 | 0.999 | 1.000 |
| Model | Precision Mean | Precision Std | Recall Mean | Recall Std |
|---|---|---|---|---|
| K-nearest neighbors | 0.999 | 0.001 | 1.000 | 0.000 |
| Support vector machine | 0.998 | 0.001 | 0.999 | 0.001 |
| Gradient boosting | 1.000 | 0.000 | 1.000 | 0.000 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santana-Mancilla, P.C.; Castrejón-Mejía, O.E.; Fajardo-Flores, S.B.; Anido-Rifón, L.E. Predicting Abnormal Respiratory Patterns in Older Adults Using Supervised Machine Learning on Internet of Medical Things Respiratory Frequency Data. Information 2023, 14, 625. https://doi.org/10.3390/info14120625
Santana-Mancilla PC, Castrejón-Mejía OE, Fajardo-Flores SB, Anido-Rifón LE. Predicting Abnormal Respiratory Patterns in Older Adults Using Supervised Machine Learning on Internet of Medical Things Respiratory Frequency Data. Information. 2023; 14(12):625. https://doi.org/10.3390/info14120625
Chicago/Turabian StyleSantana-Mancilla, Pedro C., Oscar E. Castrejón-Mejía, Silvia B. Fajardo-Flores, and Luis E. Anido-Rifón. 2023. "Predicting Abnormal Respiratory Patterns in Older Adults Using Supervised Machine Learning on Internet of Medical Things Respiratory Frequency Data" Information 14, no. 12: 625. https://doi.org/10.3390/info14120625
APA StyleSantana-Mancilla, P. C., Castrejón-Mejía, O. E., Fajardo-Flores, S. B., & Anido-Rifón, L. E. (2023). Predicting Abnormal Respiratory Patterns in Older Adults Using Supervised Machine Learning on Internet of Medical Things Respiratory Frequency Data. Information, 14(12), 625. https://doi.org/10.3390/info14120625










