Architecture of a Hybrid Video/Optical See-through Head-Mounted Display-Based Augmented Reality Surgical Navigation Platform
Abstract
:1. Introduction
2. Materials and Methods
2.1. Theory
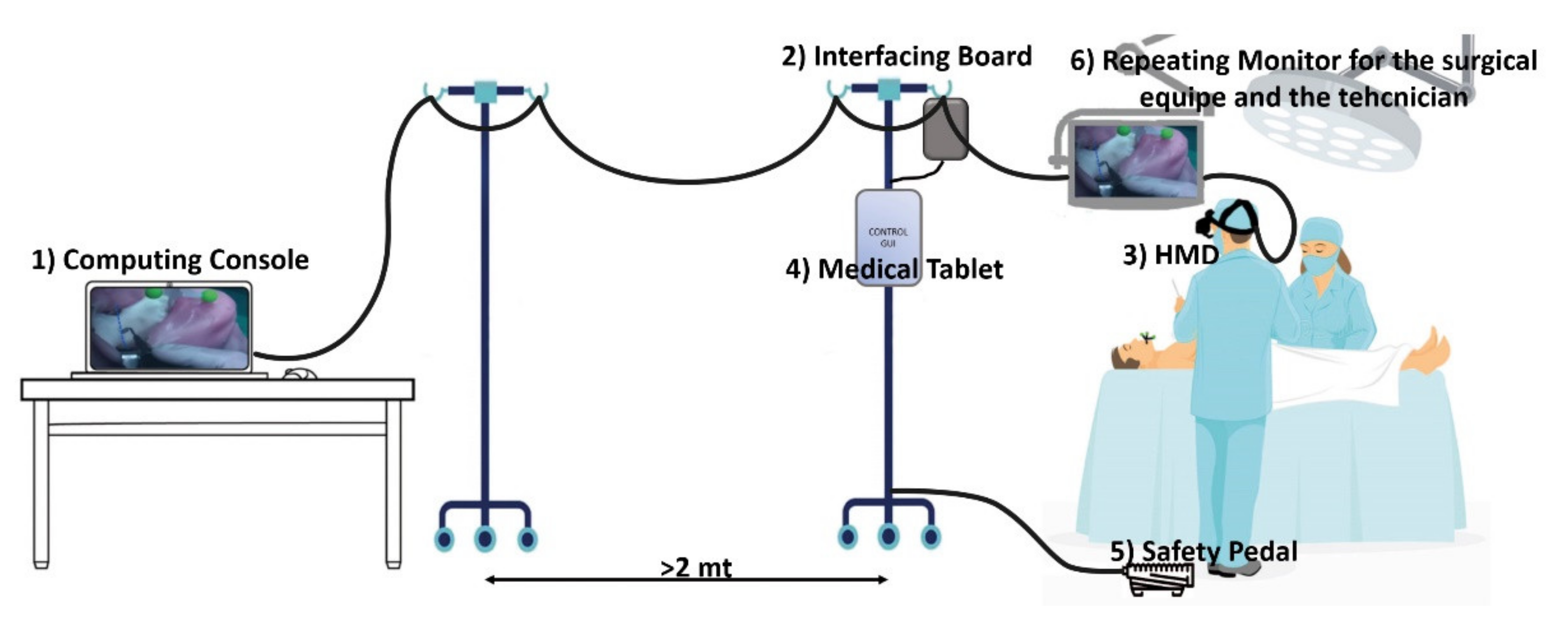
2.2. HMD-Based Surgical Navigation Platform
- -
- Hybrid OST/VST feature
- -
- Displays specifications to provide proper guidance in the peripersonal space
- -
- AR in VST
- -
- Computing Unit
- -
- Cabling and Safety Pedal
- -
- AR Software Framework
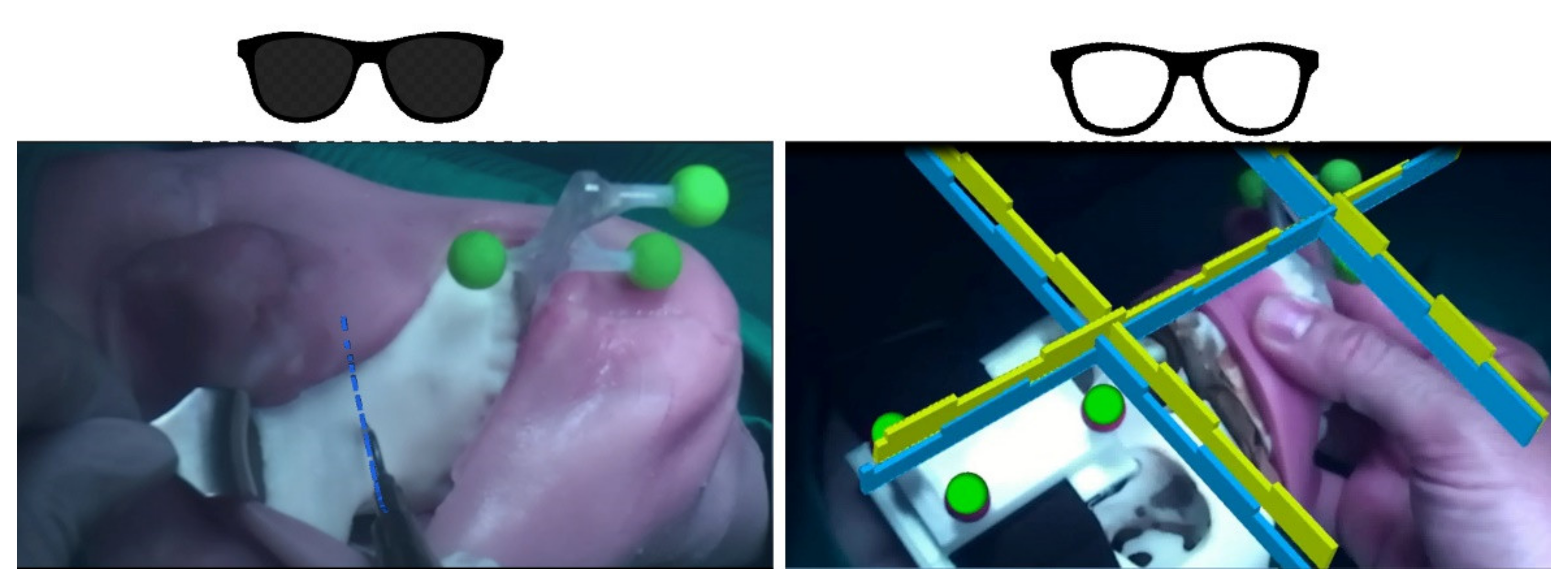
- The AR software framework is conceived for running AR applications for surgical guidance by supporting in situ visualization of medical imaging data, and it is specifically suited for AR stereoscopic headsets, both commercial and custom-made. As comprehensively described in [37], the software can provide both optical and video see-through-based augmentations of the real scene. Under the OST modality, the user sees the real-world scene through the semi-transparent optical combiners without the need for taking the visor off, and with only the computer-generated elements rendered onto the two microdisplays. Under the VST modality, the direct view of the real world is mediated by the external RGB cameras anchored to the headset. The camera frames of the real scene are first digitally blended with the computer-generated content, and the resulting video streams are fed to the microdisplays of the HMD.
- Under VST modality, and for each display side, the rendering pipeline of the virtual camera encapsulates the intrinsic and extrinsic camera parameters of the associated real camera. The intrinsic parameters are determined offline through a standard calibration procedure [39], and they are loaded when the application launches, whereas the extrinsic parameters are obtained through an inside-out marker-based tracking mechanism (as described in point 2.);
- Under OST modality, and for each display side, the intrinsic and extrinsic parameters of the off-axis virtual rendering camera are determined offline for generic user’s eye position through a reliable procedure that relies on a camera used as a replacement of the eye and placed within the eye-box of the OST display [40]. The intrinsic and extrinsic parameters of the two eye/display pinhole camera models obtained from the calibration procedure can be further refined at runtime by the user to account for the actual user’s eye position [38].
- 2.
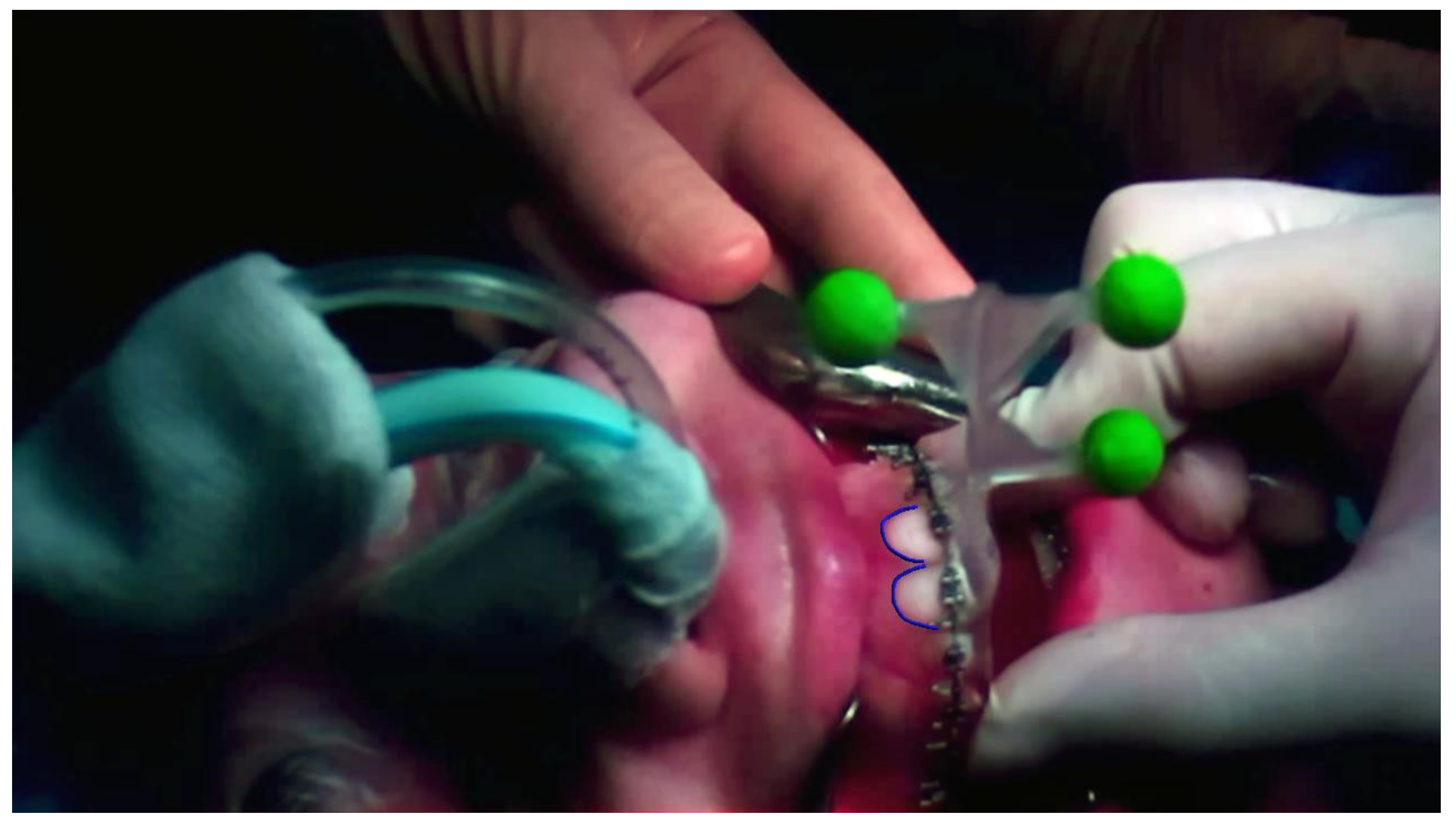
- A dedicated optical self-tracking mechanism: the head-anchored RGB cameras used for implementing the VST augmentation also provide the stereo localization of three colored spherical markers conveniently placed on patient-specific surgical templates or directly on the patient body, thus without requiring obtrusive external trackers or additional tracking cameras [37].
- 3.
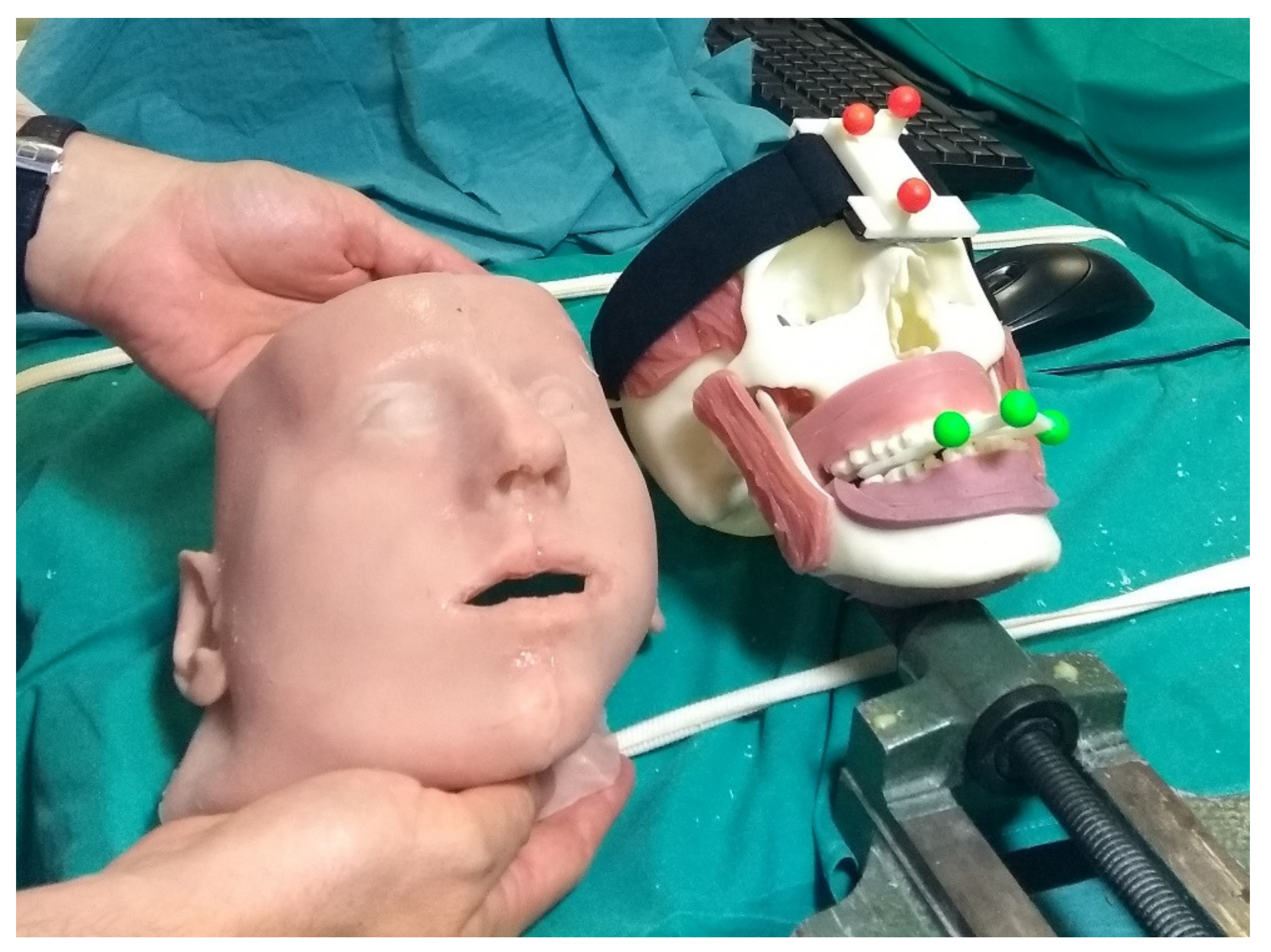
- An automatic image-to-patient registration strategy based on optical markers anchored on patient-specific templates. In particular, in the case of maxillofacial surgery, we designed an occlusal splint that embeds the three optical markers (“maxilla tracker”). The positions of the optical markers are dictated in the reference system of the computed tomography (CT) dataset. By determining their position with respect to the tracking camera, the pose of the 3D virtual planning can be directly computed in a closed-form fashion by solving a standard absolute orientation problem with three points (i.e., estimating the rigid transformation that aligns the two sets of corresponding triplets of 3D points) [37].
- -
- Control Tablet and GUI
- -
- Mechanical Key aspects
- -
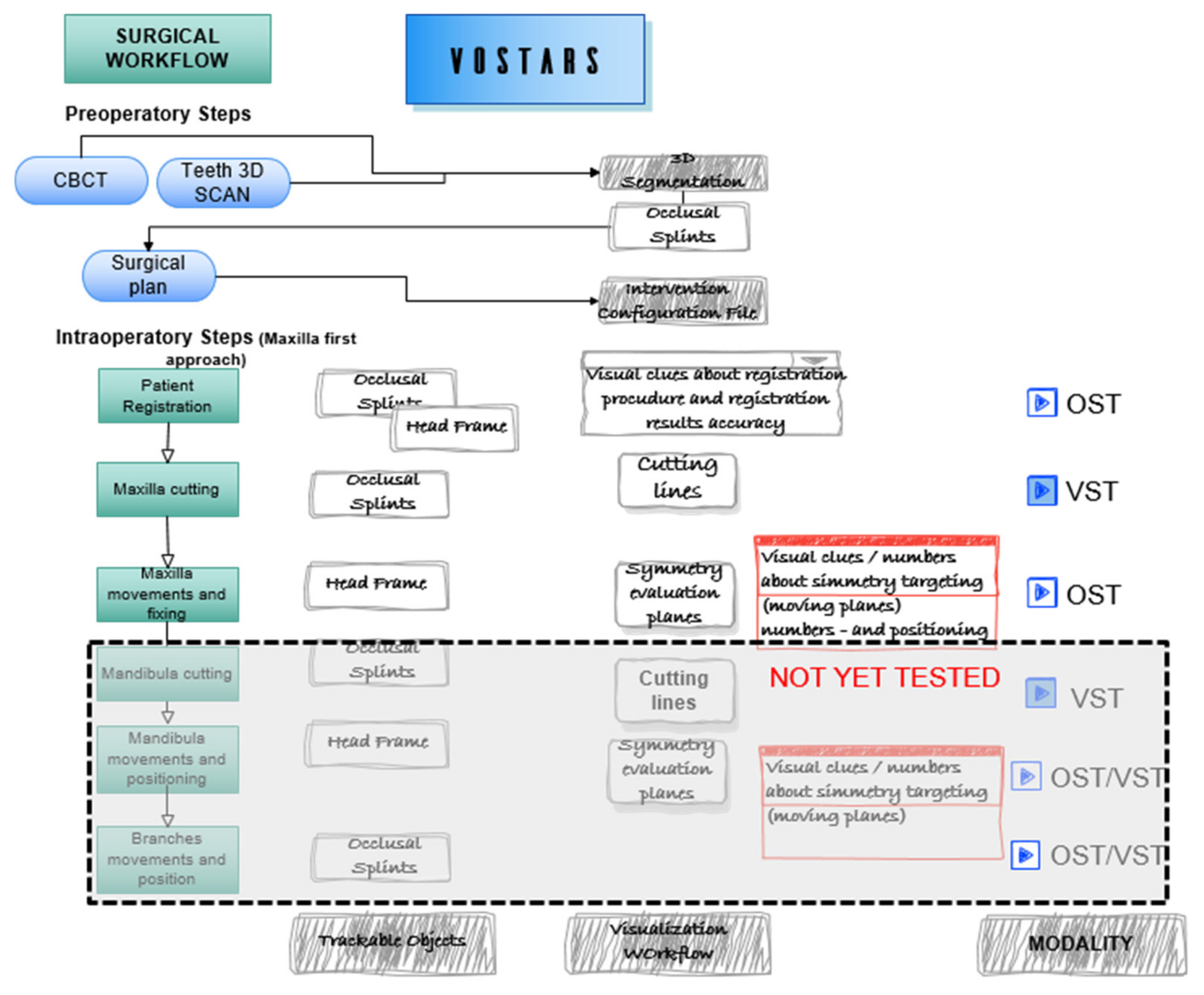
- Visualization Modalities and surgical workflow
- (a)
- VST-oriented tasks
- (b)
- OST-oriented tasks
- -
- Regulatory compliance
- -
- Ethical Committee approval:
- ◦
- “Notification” (statement/documentation on performance and safety of the experimental pre-market medical device) to the government competent authority, i.e., the Italian Ministry of Health.
- ◦
- Fulfillment of a list of conditions for clinical investigations.
- ◦
- Designation of an investigator who exercises a profession that is recognized in the member state and has the necessary scientific knowledge and experience in patient care.
- ◦
- Suitability for clinical investigation of the facilities in which the clinical investigation is to be conducted and their resemblance to the facilities where the device is intended to be used.
2.3. Phantom Trials
2.4. Clinical Trials
2.5. AR Virtual Content
3. Results
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rolland, J.P.; Cakmakci, O. The Past, Present, and Future of Head-Mounted Display Designs; SPIE: Bellingham, WA, USA, 2005; Volume 5638. [Google Scholar]
- Azuma, R.T. A Survey of Augmented Reality. Presence: Teleoperators Virtual Environ. 1997, 6, 355–385. [Google Scholar] [CrossRef]
- Carbone, M.; Condino, S.; Cutolo, F.; Viglialoro, R.M.; Kaschke, O.; Thomale, U.W.; Ferrari, V. Proof of Concept: Wearable Augmented Reality Video See-Through Display for Neuro-Endoscopy. AVR 2018; Lecture Notes in Computer Science; Springer: Cham, Switzerland, 2018; Volume 10851, pp. 95–104. [Google Scholar]
- Vavra, P.; Roman, J.; Zonca, P.; Ihnat, P.; Nemec, M.; Kumar, J.; Habib, N.; El-Gendi, A. Recent Development of Augmented Reality in Surgery: A Review. J. Healthc. Eng. 2017, 2017, 4574172. [Google Scholar] [CrossRef] [PubMed]
- Cutolo, F. Augmented Reality in Image-Guided Surgery. In Encyclopedia of Computer Graphics and Games, Lee, N., Ed.; Springer International Publishing: Cham, Swizterland, 2017; pp. 1–11. [Google Scholar]
- Ferrari, V.; Klinker, G.; Cutolo, F. Augmented Reality in Healthcare. J. Healthc. Eng. 2019, 2019, 9321535. [Google Scholar] [CrossRef] [PubMed]
- Condino, S.; Turini, G.; Viglialoro, R.; Gesi, M.; Ferrari, V. Wearable Augmented Reality Application for Shoulder Rehabilitation. Electronics 2019, 8, 1178. [Google Scholar] [CrossRef] [Green Version]
- Meulstee, J.W.; Nijsink, J.; Schreurs, R.; Verhamme, L.M.; Xi, T.; Delye, H.H.K.; Borstlap, W.A.; Maal, T.J.J. Toward Holographic-Guided Surgery. Surg. Innov. 2019, 26, 86–94. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pratt, P.; Ives, M.; Lawton, G.; Simmons, J.; Radev, N.; Spyropoulou, L.; Amiras, D. Through the HoloLens™ looking glass: Augmented reality for extremity reconstruction surgery using 3D vascular models with perforating vessels. Eur. Radiol. Exp. 2018, 2, 2. [Google Scholar] [CrossRef] [PubMed]
- Tepper, O.M.; Rudy, H.L.; Lefkowitz, A.; Weimer, K.A.; Marks, S.M.; Stern, C.S.; Garfein, E.S. Mixed Reality with HoloLens: Where Virtual Reality Meets Augmented Reality in the Operating Room. Plast. Reconstr. Surg. 2017, 140, 1066–1070. [Google Scholar] [CrossRef] [PubMed]
- Scherl, C.; Stratemeier, J.; Karle, C.; Rotter, N.; Hesser, J.; Huber, L.; Dias, A.; Hoffmann, O.; Riffel, P.; Schoenberg, S.O.; et al. Augmented reality with HoloLens in parotid surgery: How to assess and to improve accuracy. Eur. Arch. Oto-Rhino-Laryngol. 2021, 278, 2473–2483. [Google Scholar] [CrossRef] [PubMed]
- Golse, N.; Petit, A.; Lewin, M.; Vibert, E.; Cotin, S. Augmented Reality during Open Liver Surgery Using a Markerless Non-rigid Registration System. J. Gastrointest. Surg. 2021, 25, 662–671. [Google Scholar] [CrossRef] [PubMed]
- Desselle, M.R.; Brown, R.A.; James, A.R.; Midwinter, M.J.; Powell, S.K.; Woodruff, M.A. Augmented and Virtual Reality in Surgery. Comput. Sci. Eng. 2020, 22, 18–26. [Google Scholar] [CrossRef]
- Neves, C.A.; Vaisbuch, Y.; Leuze, C.; McNab, J.A.; Daniel, B.; Blevins, N.H.; Hwang, P.H. Application of holographic augmented reality for external approaches to the frontal sinus. Int. Forum Allergy Rhinol. 2020, 10, 920–925. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, V.; Carbone, M.; Condino, S.; Cutolo, F. Are augmented reality headsets in surgery a dead end? Expert Rev. Med Devices 2019, 16, 999–1001. [Google Scholar] [CrossRef] [PubMed]
- Condino, S.; Carbone, M.; Piazza, R.; Ferrari, M.; Ferrari, V. Perceptual Limits of Optical See-Through Visors for Augmented Reality Guidance of Manual Tasks. IEEE T Bio-Med. Eng. 2019, 67, 411–419. [Google Scholar] [CrossRef] [PubMed]
- Gabbard, J.L.; Mehra, D.G.; Swan, J.E. Effects of AR Display Context Switching and Focal Distance Switching on Human Performance. IEEE Trans. Vis. Comput. Graph. 2019, 25, 2228–2241. [Google Scholar] [CrossRef] [PubMed]
- Carbone, M.; Piazza, R.; Condino, S. Commercially Available Head-Mounted Displays Are Unsuitable for Augmented Reality Surgical Guidance: A Call for Focused Research for Surgical Applications. Surg. Innov. 2020, 27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Al Janabi, H.F.; Aydin, A.; Palaneer, S.; Macchione, N.; Al-Jabir, A.; Khan, M.S.; Dasgupta, P.; Ahmed, K. Effectiveness of the HoloLens mixed-reality headset in minimally invasive surgery: A simulation-based feasibility study. Surg. Endosc. 2020, 34, 1143–1149. [Google Scholar] [CrossRef] [Green Version]
- Galati, R.; Simone, M.; Barile, G.; De Luca, R.; Cartanese, C.; Grassi, G. Experimental Setup Employed in the Operating Room Based on Virtual and Mixed Reality: Analysis of Pros and Cons in Open Abdomen Surgery. J. Healthc. Eng. 2020, 2020, 8851964. [Google Scholar] [CrossRef] [PubMed]
- Dibble, C.F.; Molina, C.A. Device profile of the XVision-spine (XVS) augmented-reality surgical navigation system: Overview of its safety and efficacy. Expert Rev. Med Devices 2021, 18, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Rolland, J.P.; Fuchs, H. Optical Versus Video See-Through Head-Mounted Displays in Medical Visualization. Presence: Teleoper. Virtual Environ. 2000, 9, 287–309. [Google Scholar] [CrossRef]
- Holliman, N.S.; Dodgson, N.A.; Favalora, G.E.; Pockett, L. Three-Dimensional Displays: A Review and Applications Analysis. IEEE T Broadcast 2011, 57, 362–371. [Google Scholar] [CrossRef]
- Grubert, J.; Itoh, Y.; Moser, K.; Swan, J.E. A Survey of Calibration Methods for Optical See-Through Head-Mounted Displays. IEEE Trans. Vis. Comput. Graph. 2018, 24, 2649–2662. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Itoh, Y.; Klinker, G. Performance and sensitivity analysis of INDICA: INteraction-Free DIsplay CAlibration for Optical See-Through Head-Mounted Displays. In Proceedings of the 2014 IEEE International Symposium on Mixed and Augmented Reality (ISMAR), Munich, Germany, 10–12 September 2014; pp. 171–176. [Google Scholar]
- Cutolo, F.; Cattari, N.; Fontana, U.; Ferrari, V. Optical See-Through Head-Mounted Displays With Short Focal Distance: Conditions for Mitigating Parallax-Related Registration Error. Front. Robot. AI 2020, 7, 572001. [Google Scholar] [CrossRef] [PubMed]
- Raimbaud, P.; Lopez, M.S.A.; Figueroa, P.; Hernandez, J.T. Influence of Depth Cues on Eye Tracking Depth Measurement in Augmented Reality Using the MagicLeap Device. In Proceedings of the 2020 IEEE Conference on Virtual Reality and 3D User Interfaces Abstracts and Workshops (VRW), Atlanta, GA, USA, 22–26 March 2020; pp. 210–214. [Google Scholar]
- Drascic, D.; Milgram, P. Perceptual issues in Augmented Reality. Stereosc. Disp. Virtual Real. Syst. III 1996, 2653, 123–134. [Google Scholar] [CrossRef]
- Condino, S.; Fida, B.; Carbone, M.; Cercenelli, L.; Badiali, G.; Ferrari, V.; Cutolo, A.F. Wearable Augmented Reality Platform for Aiding Complex 3D Trajectory Tracing. Sensors 2020, 20, 1612. [Google Scholar] [CrossRef] [Green Version]
- Cattari, N.; Piazza, R.; D’Amato, R.; Fida, B.; Carbone, M.; Condino, S.; Cutolo, F.; Ferrari, V. Towards a Wearable Augmented Reality Visor for High-Precision Manual Tasks. In Proceedings of the 2020 IEEE International Symposium on Medical Measurements and Applications (MeMeA), Bari, Italy, 1 June–1 July 2020; pp. 1–6. [Google Scholar]
- Cattari, N.; Cutolo, F.; D’amato, R.; Fontana, U.; Ferrari, V. Toed-in vs Parallel Displays in Video See-Through Head-Mounted Displays for Close-Up View. IEEE Access 2019, 7, 159698–159711. [Google Scholar] [CrossRef]
- Cutolo, F.; Fontana, U.; Ferrari, V. Perspective Preserving Solution for Quasi-Orthoscopic Video See-Through HMDs. Technologies 2018, 6, 9. [Google Scholar] [CrossRef] [Green Version]
- State, A.; Ackerman, J.; Hirota, G.; Lee, J.; Fuchs, H. Dynamic virtual convergence for video see-through head-mounted displays: Maintaining maximum stereo overlap throughout a close-range work space. In Proceedings of the Proceedings IEEE and ACM International Symposium on Augmented Reality, New York, NY, USA, 29–30 October 2001; pp. 137–146. [Google Scholar]
- Cutolo, F.; Fontana, U.; Carbone, M.; D’Amato, R.; Ferrari, V. Hybrid Video/Optical See-Through HMD. In Proceedings of the 2017 Ieee International Symposium on Mixed and Augmented Reality (Ismar-Adjunct), Nantes, France, 9–13 October 2017; pp. 52–57. [Google Scholar] [CrossRef] [Green Version]
- OpenCV. Available online: http://opencv.willowgarage.com/documentation/cpp/index.html (accessed on 12 January 2022).
- (VTK), T.V.T. Available online:. Available online: http://www.vtk.org/ (accessed on 12 January 2022).
- Cutolo, F.; Fida, B.; Cattari, N.; Ferrari, V. Software Framework for Customized Augmented Reality Headsets in Medicine. IEEE Access 2020, 8, 706–720. [Google Scholar] [CrossRef]
- Cutolo, F.; Cattari, N.; Carbone, M.; D’Amato, R.; Ferrari, V. Device-Agnostic Augmented Reality Rendering Pipeline for AR in Medicine. In Proceedings of the 2021 IEEE International Symposium on Mixed and Augmented Reality Adjunct (ISMAR-Adjunct), Bari, Italy, 4–8 October 2021; pp. 340–345. [Google Scholar]
- Zhang, Z. A flexible new technique for camera calibration. IEEE T Pattern Anal 2000, 22, 1330–1334. [Google Scholar] [CrossRef] [Green Version]
- Cutolo, F.; Fontana, U.; Cattari, N.; Ferrari, V. Off-Line Camera-Based Calibration for Optical See-Through Head-Mounted Displays. Appl. Sci. 2020, 10, 193. [Google Scholar] [CrossRef] [Green Version]
- VOSTARS. VOSTARS at Work in the Surgical Room. Available online: https://www.youtube.com/watch?v=N8qZUj4amm0 (accessed on 12 January 2022).
- Choi, J.-W.; Lee, J.Y. Update on Orthognathic Surgical Techniques. In The Surgery-First Orthognathic Approach; Springer: Berlin/Heidelberg, Germany, 2021; pp. 149–158. [Google Scholar]
- Naran, S.; Steinbacher, D.M.; Taylor, J.A. Current concepts in orthognathic surgery. Plast. Reconstr. Surg. 2018, 141, 925e–936e. [Google Scholar] [CrossRef] [PubMed]
- Badiali, G.; Ferrari, V.; Cutolo, F.; Freschi, C.; Caramella, D.; Bianchi, A.; Marchetti, C. Augmented reality as an aid in maxillofacial surgery: Validation of a wearable system allowing maxillary repositioning. J. Cranio Maxill. Surg. 2014, 42, 1970–1976. [Google Scholar] [CrossRef] [PubMed]
- Cercenelli, L.; Carbone, M.; Condino, S.; Cutolo, F.; Marcelli, E.; Tarsitano, A.; Marchetti, C.; Ferrari, V.; Badiali, G. The Wearable VOSTARS System for Augmented Reality-Guided Surgery: Preclinical Phantom Evaluation for High-Precision Maxillofacial Tasks. J. Clin. Med. 2020, 9, 3562. [Google Scholar] [CrossRef] [PubMed]
- Hewlett, A.L.; Hohenberger, H.; Murphy, C.N.; Helget, L.; Hausmann, H.; Lyden, E.; Fey, P.D.; Hicks, R. Evaluation of the bacterial burden of gel nails, standard nail polish, and natural nails on the hands of health care workers. Am. J. Infect. Control. 2018, 46, 1356–1359. [Google Scholar] [CrossRef] [PubMed]
- Community BO. Blender—A 3D modelling and rendering package [Internet]. Stichting Blender Foundation, Amsterdam. 2018. Available online: http://www.blender.org (accessed on 12 January 2022).
- Ferrari, V.; Carbone, M.; Cappelli, C.; Boni, L.; Melfi, F.; Ferrari, M.; Mosca, F.; Pietrabissa, A. Value of multidetector computed tomography image segmentation for preoperative planning in general surgery. Surg. Endosc. 2012, 26, 616–626. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jamieson, S. Likert scales: How to (ab)use them. Med. Educ. 2004, 38, 1217–1218. [Google Scholar] [CrossRef] [PubMed]


| Assertion | Surgeon n.1 | Surgeon n.2 | Surgeon n.3 |
|---|---|---|---|
| The weight of the device does affect significantly affect the surgical performance. | 4 | 3 | 3 |
| I can stand with the device for an hour without feeling any discomfort. | 4 | 2 | 3 |
| I am free to move my head without feeling hindered by the device. | 3 | 4 | 3 |
| When the visor is off, the display allows me to see the real-world naturally. | 3 | 4 | 2 |
| When in VST modality, I perceive the real-world naturally. | 3 | 3 | 3 |
| Brightness of the video stream is adequate. | 3 | 4 | 3 |
| I can naturally enjoy an augmented reality experience in terms of brightness. | 4 | 4 | 3 |
| I can naturally enjoy an augmented reality experience in terms of focal distances. | 4 | 4 | 4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carbone, M.; Cutolo, F.; Condino, S.; Cercenelli, L.; D’Amato, R.; Badiali, G.; Ferrari, V. Architecture of a Hybrid Video/Optical See-through Head-Mounted Display-Based Augmented Reality Surgical Navigation Platform. Information 2022, 13, 81. https://doi.org/10.3390/info13020081
Carbone M, Cutolo F, Condino S, Cercenelli L, D’Amato R, Badiali G, Ferrari V. Architecture of a Hybrid Video/Optical See-through Head-Mounted Display-Based Augmented Reality Surgical Navigation Platform. Information. 2022; 13(2):81. https://doi.org/10.3390/info13020081
Chicago/Turabian StyleCarbone, Marina, Fabrizio Cutolo, Sara Condino, Laura Cercenelli, Renzo D’Amato, Giovanni Badiali, and Vincenzo Ferrari. 2022. "Architecture of a Hybrid Video/Optical See-through Head-Mounted Display-Based Augmented Reality Surgical Navigation Platform" Information 13, no. 2: 81. https://doi.org/10.3390/info13020081
APA StyleCarbone, M., Cutolo, F., Condino, S., Cercenelli, L., D’Amato, R., Badiali, G., & Ferrari, V. (2022). Architecture of a Hybrid Video/Optical See-through Head-Mounted Display-Based Augmented Reality Surgical Navigation Platform. Information, 13(2), 81. https://doi.org/10.3390/info13020081











