Combining Visual Contrast Information with Sound Can Produce Faster Decisions
Abstract
1. Introduction
2. Materials and Methods
2.1. Research Ethics and Participants
2.2. Image and Sound Conditions
2.3. Experimental Design
2.4. Procedure and Task Instructions
3. Results
3.1. Perceptual Decisions Relative to Expected Depth Effects (“Nearer”)
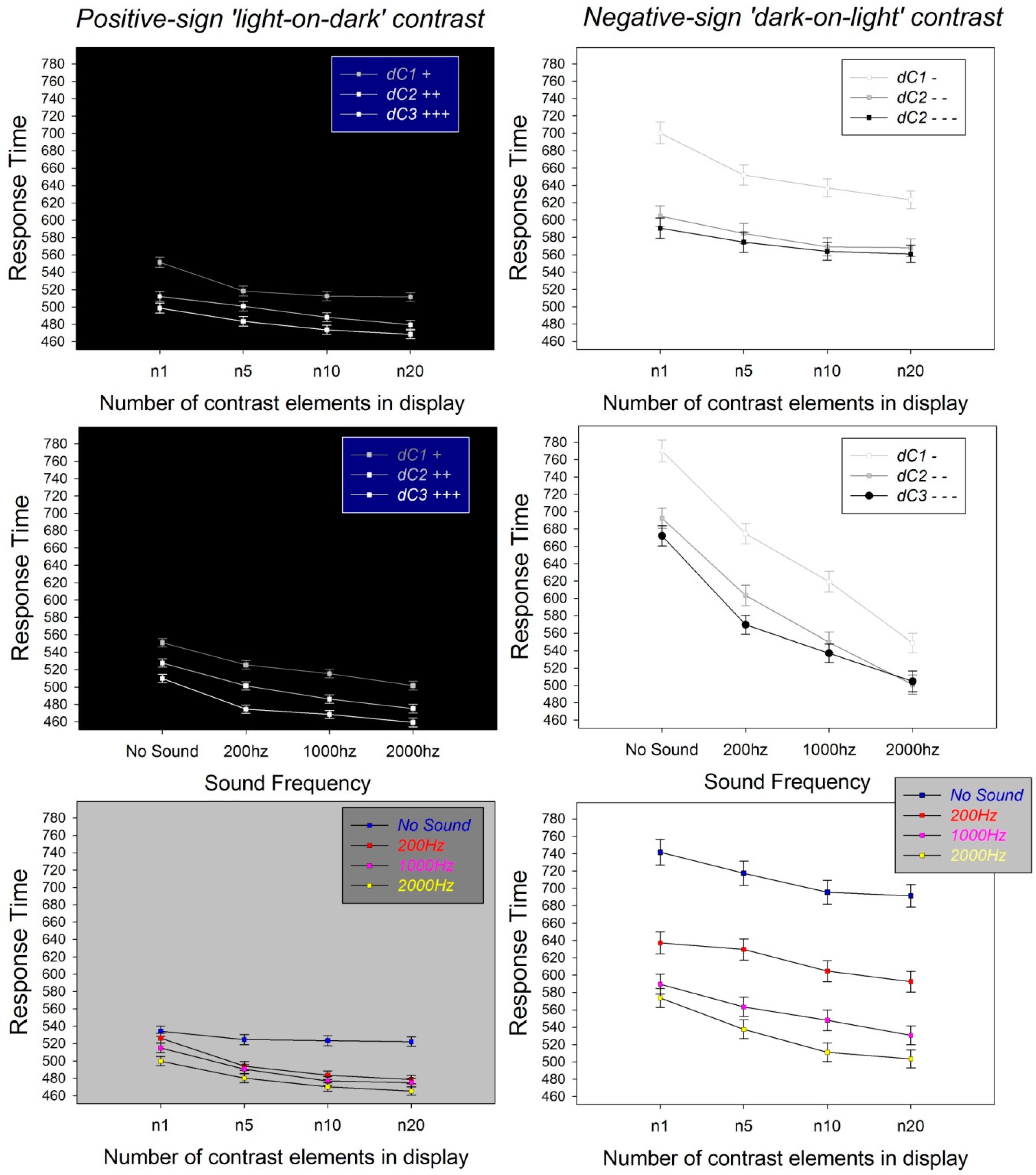
3.2. Effects of Experimental Factors on Response Times
3.3. Contrast Polarity
3.4. Contrast Difference (dC) in a Pattern Pair
3.5. Number of Contrast Elements in a Pattern Pair
3.6. Sound Frequency
3.7. Interactions
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Piéron, H. The Sensations; Yale University Press: New Haven, CT, USA, 1952. [Google Scholar]
- Pins, D.; Bonnet, C. On the relation between stimulus intensity and processing time: Pieron’s law and choice reaction time. Percept. Psychophys. 1996, 58, 390–400. [Google Scholar] [CrossRef] [PubMed]
- Bonnet, C.; Gurlekian, J.; Harris, P. Reaction time and visual area: Searching for the determinants. Bull. Psychon. Soc. 1992, 30, 396–398. [Google Scholar] [CrossRef][Green Version]
- Cattell, J.M. The influence of the intensity of the stimulus on the length of the reaction time. Brain 1886, 8, 512–515. [Google Scholar] [CrossRef]
- Exner, S. Ueber die zu einer Gesichtswahrnehmung noetige Zeit. Sitz. Der Kais. Akad. Der Wiss. 1868, 57, 601–632. [Google Scholar]
- Wundt, W. Grundzüge der Physiologischen Psychologie; Engelmann: Leipzig, Germany, 1874. [Google Scholar]
- Chocholle, R. Variation des temps de réaction auditifs en fonction de l’intensité a diverses fréquences. L’année Psychol. 1940, 41, 65–124. [Google Scholar] [CrossRef]
- Stevens, S.S. Psychophysics: Introduction to Its Perceptual Neural and Social Prospects; Wiley: New York, NY, USA, 1975. [Google Scholar]
- Altmann, J. Acoustic weapons: A prospective assessment. Sci. Glob. Secur. 2001, 9, 165–234. [Google Scholar] [CrossRef]
- Beckerman, J. The Sonic Boom. How Sound Transforms the Way We Think, Feel, and Buy; Houghton Mifflin Harcourt: Boston, MA, USA; New York, NY, USA, 2014. [Google Scholar]
- Hanes, D.; Schall, J. Neural control of voluntary movement initiation. Science 1996, 274, 427–430. [Google Scholar] [CrossRef]
- Batmaz, A.U.; de Mathelin, M.; Dresp-Langley, B. Getting nowhere fast: Trade-off between speed and precision in training to execute image-guided hand-tool movements. BMC Psychol. 2016, 4, 55. [Google Scholar] [CrossRef]
- Batmaz, A.U.; de Mathelin, M.; Dresp-Langley, B. Seeing virtual while acting real: Visual display and strategy effects on the time and precision of eye-hand coordination. PLoS ONE 2017, 12, e0183789. [Google Scholar] [CrossRef]
- Dresp-Langley, B. Principles of perceptual grouping: Implications for image-guided surgery. Front. Psychol. 2015, 6, 1565. [Google Scholar] [CrossRef]
- Farnè, M. Brightness as an indicator to distance: Relative brightness per se or contrast with the background? Perception 1977, 6, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Egusa, H. Effects of brightness, hue, and saturation on the perceived depth between adjacent regions in the visual field. Perception 1983, 12, 167–175. [Google Scholar] [CrossRef] [PubMed]
- Dresp, B.; Durand, S.; Grossberg, S. Depth perception from pairs of overlapping cues in pictorial displays. Spat. Vis. 2002, 15, 255–276. [Google Scholar] [PubMed]
- Guibal, C.; Dresp, B. Interaction of color and geometric cues in depth perception: When does “red” mean “near”? Psychol. Res. 2004, 10, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Qiu, F.T.; Sugihara, T.; von der Heydt, R. Figure-ground mechanisms provide structure for selective attention. Nat. Neurosci. 2007, 11, 1492–1499. [Google Scholar] [CrossRef] [PubMed]
- Dresp, B.; Fischer, S. Asymmetrical contrast effects induced by luminance and colour configurations. Percept. Psychophys. 2001, 63, 1262–1270. [Google Scholar] [CrossRef]
- Dresp-Langley, B.; Reeves, A. Simultaneous brightness and apparent depth from true colors on grey: Chevreul revisited. See. Perceiving 2012, 25, 597–618. [Google Scholar] [CrossRef]
- Dresp-Langley, B.; Reeves, A. Color and figure-ground: From signals to qualia. In Perception Beyond Gestalt: Progress in Vision Research; Magnussen, S., Greenlee, M., Werner, J., Geremek, A., Eds.; Psychology Press: Abingdon, UK, 2014; pp. 159–171. [Google Scholar]
- Dresp-Langley, B.; Reeves, A. Effects of saturation and contrast polarity on the figure-ground organization of color on gray. Front. Psychol. 2004, 5, 1136. [Google Scholar] [CrossRef]
- Dresp-Langley, B.; Grossberg, S. Neural Computation of Surface Border Ownership and Relative Surface Depth from Ambiguous Contrast Inputs. Front. Psychol. 2016, 7, 1102. [Google Scholar] [CrossRef]
- Von der Heydt, R. Figure–ground and the emergence of proto-objects in the visual cortex. Front. Psychol. 2015, 6, 1695. [Google Scholar] [CrossRef]
- Dresp-Langley, B.; Reeves, A. Colour for behavioural success. i-Perception 2018, 9, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Spillmann, L.; Dresp-Langley, B.; Tseng, C.H. Beyond the classic receptive field: The effect of contextual stimuli. J. Vis. 2015, 15, 7. [Google Scholar] [CrossRef] [PubMed]
- Pápai, M.S.; Soto-Faraco, S. Sounds can boost the awareness of visual events through attention without cross-modal integration. Sci. Rep. 2017, 7, 41684. [Google Scholar] [CrossRef] [PubMed]
- Oxenham, A.J. Pitch perception. J. Neurosci. 2012, 32, 13335–13338. [Google Scholar] [CrossRef] [PubMed]
- Green, D.M.; Swets, J.A. Signal Detection Theory and Psychophysics; Krieger Publishing: Huntington, NY, USA, 1973. [Google Scholar]
- Wandeto, J.M.; Dresp-Langley, B. The quantization error in a Self-Organizing Map as a contrast and color specific indicator of single-pixel change in large random patterns. Neural Netw. 2019, 119, 273–285. [Google Scholar] [CrossRef] [PubMed]
- Schiller, P.H.; Sandell, J.H.; Maunsell, J.H. Functions of the ON and OFF channels of the visual system. Nature 1986, 322, 824–825. [Google Scholar] [CrossRef]

| R | G | B | Lum | LumC | dC | ||
|---|---|---|---|---|---|---|---|
| Dark image background | 5 | 5 | 5 | 2.5 (cd/m2) | |||
| Light image background | 250 | 250 | 250 | 95 (cd/m2) | |||
| Positive-sign light-on-dark pairs | |||||||
| ‘dC +’ | lighter patterns | 250 | 250 | 250 | 95 (cd/m2) | 37 | |
| darker patterns | 150 | 150 | 150 | 52 (cd/m2) | 20 | 17 | |
| ‘dC ++’ | lighter patterns | 250 | 250 | 250 | 95 (cd/m2) | 37 | |
| darker patterns | 100 | 100 | 100 | 30 (cd/m2) | 11 | 26 | |
| ‘dC +++’ | lighter patterns | 250 | 250 | 250 | 95 (cd/m2) | 37 | |
| darker patterns | 50 | 50 | 50 | 10 (cd/m2) | 3 | 34 | |
| Negative-sign dark-on-light pairs | |||||||
| ‘dC –’ | darker patterns | 5 | 5 | 5 | 2.5 (cd/m2) | 37 | |
| lighter patterns | 50 | 50 | 50 | 10 (cd/m2) | 8.5 | 28.5 | |
| ‘dC– –’ | darker patterns | 5 | 5 | 5 | 2.5 (cd/m2) | 37 | |
| lighter patterns | 100 | 100 | 100 | 30 (cd/m2) | 2.2 | 34.8 | |
| ‘dC– – –’ | darker patterns | 5 | 5 | 5 | 2.5 (cd/m2) | 37 | |
| lighter patterns | 150 | 150 | 150 | 52 (cd/m2) | 0.8 | 36.2 | |
| Factor | df | F | p |
|---|---|---|---|
| Polarity | 1 | 231.926 | <0.001 |
| Nelements | 3 | 3.397 | <0.017 |
| dC | 2 | 24.990 | <0.001 |
| Sound Frequency | 3 | 49.835 | <0.001 |
| Location | 1 | 2.404 | 0.121 NS |
| Interactions | |||
| Nelements × dC | 6 | 0.872 | 0.515 NS |
| Nelements × Sound Frequency | 9 | 0.307 | 0.973 NS |
| dC × Sound Frequency | 6 | 0.727 | 0.628 NS |
| Nelements × Polarity | 3 | 0.845 | 0.535 NS |
| dC × Polarity | 2 | 3.891 | <0.021 |
| Sound Frequency × Polarity | 3 | 20.880 | <0.001 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dresp-Langley, B.; Monfouga, M. Combining Visual Contrast Information with Sound Can Produce Faster Decisions. Information 2019, 10, 346. https://doi.org/10.3390/info10110346
Dresp-Langley B, Monfouga M. Combining Visual Contrast Information with Sound Can Produce Faster Decisions. Information. 2019; 10(11):346. https://doi.org/10.3390/info10110346
Chicago/Turabian StyleDresp-Langley, Birgitta, and Marie Monfouga. 2019. "Combining Visual Contrast Information with Sound Can Produce Faster Decisions" Information 10, no. 11: 346. https://doi.org/10.3390/info10110346
APA StyleDresp-Langley, B., & Monfouga, M. (2019). Combining Visual Contrast Information with Sound Can Produce Faster Decisions. Information, 10(11), 346. https://doi.org/10.3390/info10110346





