Long-Term Variations in Species Composition of Bloom-Forming Toxic Pseudo-nitzschia Diatoms in the North-Western Sea of Japan during 1992–2015
Abstract
1. Introduction
2. Study Area and Sampling
3. Statistical Analysis
4. Results
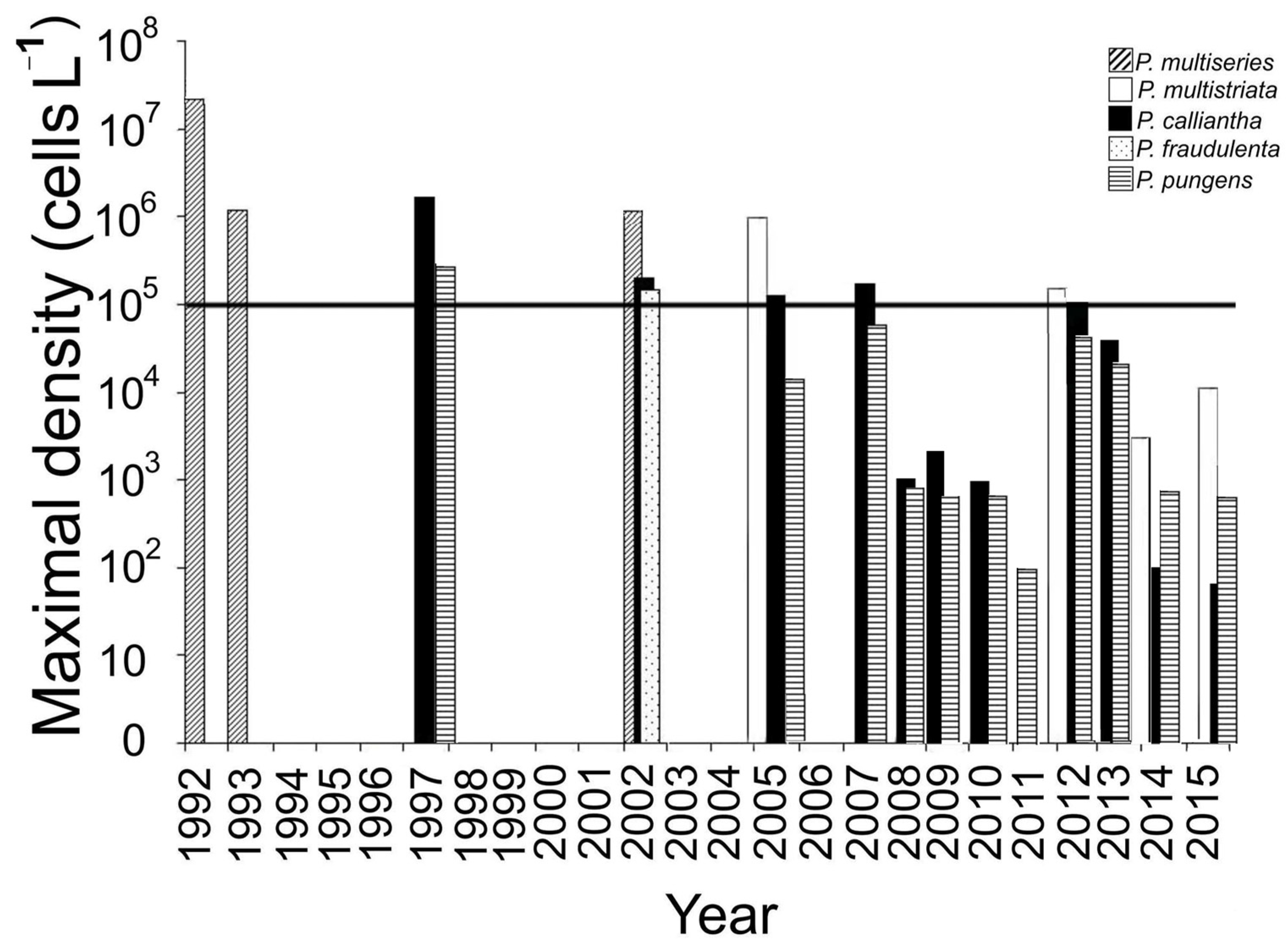
4.1. The Bloom Events
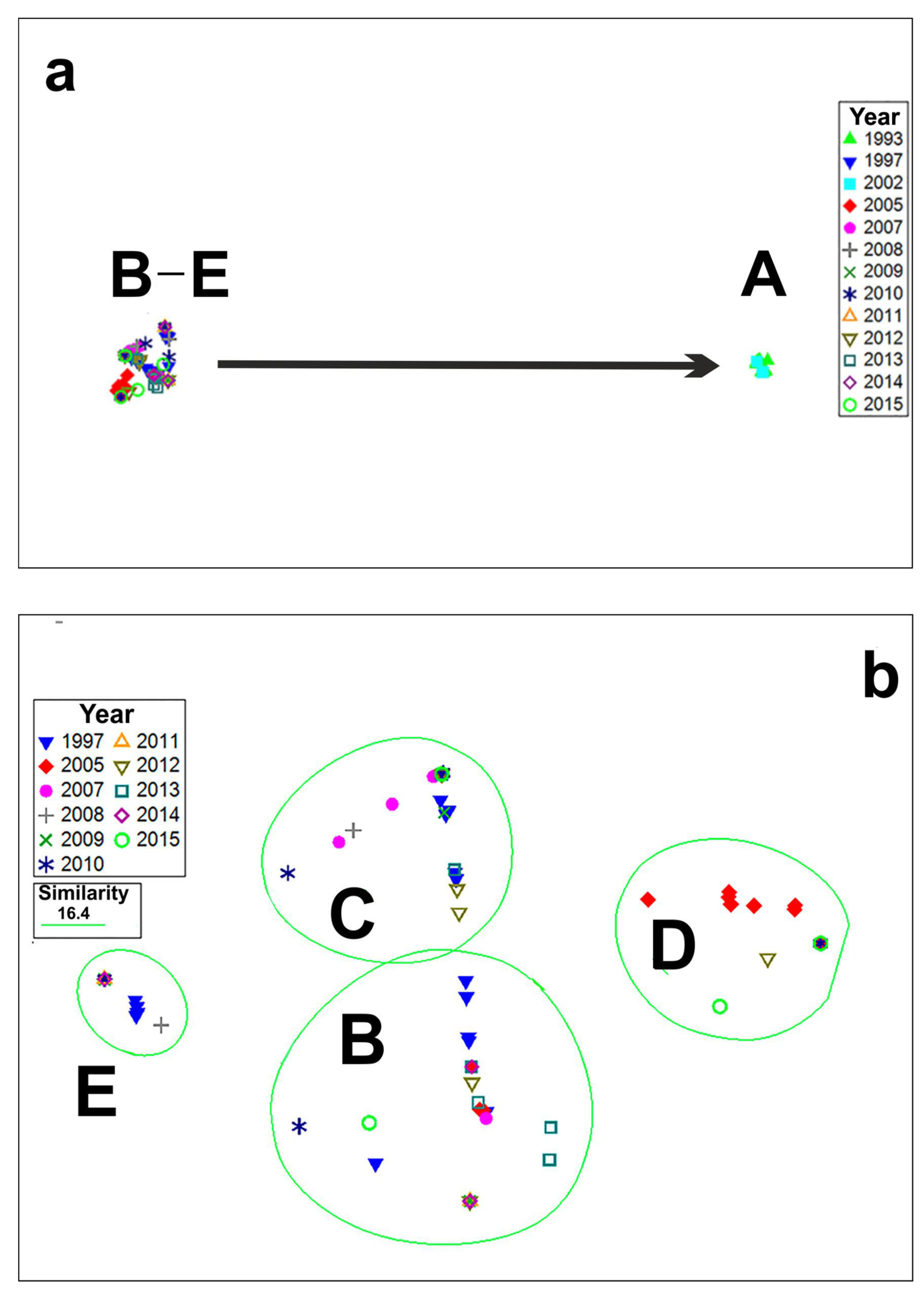
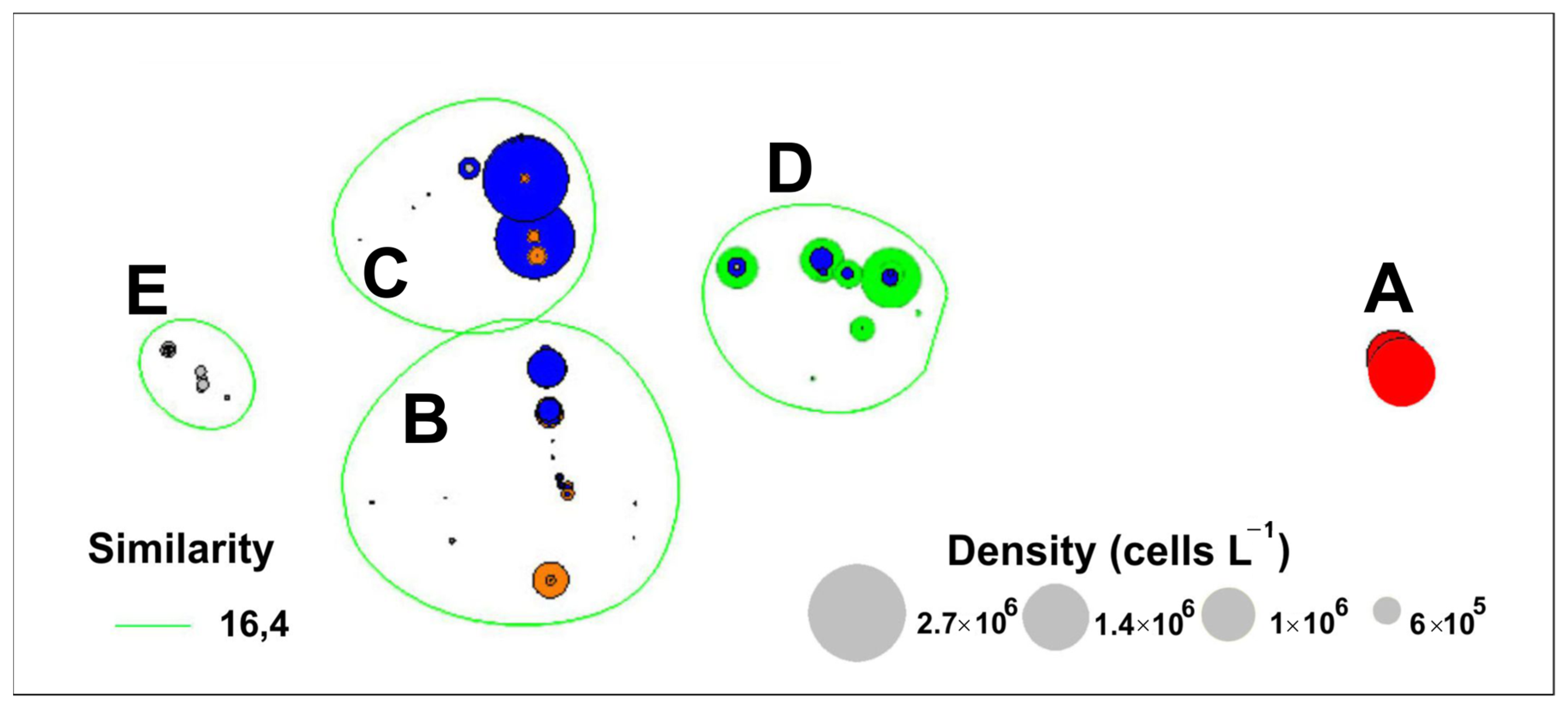
4.2. Long-Term Variations in Species Composition of Bloom-Forming Pseudo-nitzschia
4.3. A Shift towards the Dominance of Pseudo-nitzschia multistriata and Pseudo-nitzchia calliantha and Long-Term Variations in Water Temperature
5. DA Content of Pseudo-nitzschia Cultures
6. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bates, S.S.; Hubbard, K.A.; Lundholm, N.; Montresor, M.; Leaw, C.P. Pseudo-nitzschia, Nitzschia, and domoic acid: New research since 2011. Harmful Algae 2018, 79, 3–43. [Google Scholar] [CrossRef] [PubMed]
- Lundholm, N.; Clarke, A.; Ellegaard, M. A 100-year record of changing Pseudo-nitzschia species in a sill-fjord in Denmark related to nitrogen loading and temperature. Harmful Algae 2010, 9, 449–457. [Google Scholar] [CrossRef]
- Cabrini, M.; Fornasaro, D.; Cossarini, G.; Lipizer, M.; Virgilio, D. Phytoplankton temporal changes in a coastal northern Adriatic site during the last 25 years. Estuar. Coast. Shelf Sci. 2012, 115, 113–124. [Google Scholar] [CrossRef]
- Totti, C.; Romagnoli, T.; Accoroni, S.; Coluccelli, A.; Pellegrini, M.; Campanelli, A.; Grilli, F.; Marini, M. Phytoplankton communities in the northwestern Adriatic Sea: Interdecadal variability over a 30-years period (1988–2016) and relationships with meteoclimatic drivers. J. Mar. Syst. 2019, 193, 137–153. [Google Scholar] [CrossRef]
- Derolez, V.; Soudant, D.; Malet, N.; Chiantella, C.; Richard, M.; Abadie, E.; Aliaume, C.; Bec, B. Two decades of oligotrophication: Evidence for a phytoplankton community shift in the coastal lagoon of Thau (Mediterranean Sea, France). Estuar. Coast. Shelf Sci. 2020, 241, 106810. [Google Scholar] [CrossRef]
- Lefebvre, A.; Dezécache, C. Trajectories of changes in phytoplankton biomass, Phaeocystis globosa and diatom (incl. Pseudo-nitzschia sp.) abundances re-lated to nutrient pressures in the Eastern English Channel, Southern North Sea. J. Mar. Sci. Eng. 2020, 8, 401. [Google Scholar] [CrossRef]
- Huang, C.; Lin, X.; Lin, J.; Du, H.; Dong, Q. Population dynamics of Pseudo-nitzschia pungens in Zhelin Bay, China. J. Mar. Biol. Assoc. U. K. 2008, 89, 663–668. [Google Scholar] [CrossRef]
- Adrianov, A.V.; Kussakin, O.G. A Check-List of biota of the Peter the Great Bay, the Sea of Japan; Dalnauka: Vladivostok, Russia, 1998; p. 349. (In Russian) [Google Scholar]
- Semina, G.I. Phytoplankton of the Pacific Ocean; Publ. House Nauka: Moscow, Russia, 1974; p. 239. (In Russian) [Google Scholar]
- Orlova, T.; Morozova, T.; Kameneva, P.; Shevchenko, O. Harmful algal blooms on the Russian east coast and their possible economic impacts. In Proc. Workshop on Economic Impacts of Harmful Algal Blooms on Fisheries and Aquaculture; PICES Scientific Report, no. 47; Trainer, V.L., Yoshida, T., Eds.; PICES: Sidney, BC, Canada, 2014; pp. 41–58. [Google Scholar]
- Orlova, T.Y.; Stonik, I.V.; Aizdaicher, N.A.; Bates, S.S.; Léger, C.; Fehling, J. Toxicity, morphology and distribution of Pseudo-nitzschia calliantha, P. multistriata and P. multiseries (Bacillariophyta) from the northwestern Sea of Japan. Bot. Mar. 2008, 51, 297–306. [Google Scholar] [CrossRef]
- Stonik, I.V.; Orlova, T.Y.; Lundholm, N. Diversity of Pseudo-nitzschia H. Peragallo from the western North Pacific. Diatom Res. 2011, 26, 121–134. [Google Scholar] [CrossRef]
- Orlova, T.Y.; Zhukova, N.V.; Stonik, I.V. Bloom-forming Diatom Pseudo-nitzschia pungens in Amurskii Bay (the Sea of Japan): Morphology, ecology and biochemistry. In Harmful and Toxic Algal Blooms; Yasumoto, T., Oshima, Y., Fukuyo, Y., Eds.; Intergovernmental Oceanographic Commission of UNESCO: Paris, France, 1996; pp. 147–150. [Google Scholar]
- Stonik, I.V.; Isaeva, M.P.; Aizdaicher, N.A.; Balakirev, E.S.; Ayala, F.J. Morphological and genetic identification of Pseudo-nitzschia H. Peragallo, 1900 (Bacillariophyta) from the Sea of Japan. Russ. J. Mar. Biol. 2018, 44, 192–201. [Google Scholar] [CrossRef]
- Stonik, I.V.; Orlova, T.Y.; Chikalovets, I.V.; Aizdaicher, N.A.; Aleksanin, A.I.; Kachur, V.A.; Morozova, T.V. Pseudo-nitzschia species (Bacillariophyceae) and the domoic acid concentration in Pseudo-nitzschia cultures and bivalves from the northwestern Sea of Japan, Russia. Nova Hedwig. 2019, 108, 73–93. [Google Scholar] [CrossRef]
- Stonik, I.V.; Orlova, T.Y.; Shevchenko, O.G. Morphology and ecology of the species of the genus Pseudo-nitzschia (Bacillariophyta) from Peter the Great Bay, Sea of Japan. Russ. J. Mar. Biol. 2001, 27, 362–366. [Google Scholar] [CrossRef]
- Grigoriev, R.V.; Zuenko, Y.I. Climatic distribution of water temperature and salinity in the Amur Bay (Japan Sea). Izv. TINRO 2005, 143, 179–188, (In Russian with English Abstract). [Google Scholar]
- Zuenko, Y.I.; Rachkov, V.I. Climatic changes of temperature, salinity and nutrients in the Amur Bay of the Japan Sea. Izv. TINRO 2015, 183, 186–199, (In Russian with English Abstract). [Google Scholar]
- Stonik, I.V.; Orlova, T.Y.; Propp, L.N.; Demchenko, N.L.; Skriptsova, A.V. An autumn bloom of diatoms of the genus Pseudo-nitzschia H. Peragallo, 1900 in Amursky Bay, the Sea of Japan. Russ. J. Mar. Biol. 2012, 38, 211–217. [Google Scholar] [CrossRef]
- Clarke, K.; Green, R. Statistical design and analysis for a ‘biological effects’ study. Mar. Ecol. Prog. Ser. 1988, 46, 213–226. [Google Scholar] [CrossRef]
- Clarke, K.R.; Warwick, R.M. Software PRIMER; Primer-E Ltd.: Plymouth, UK, 2001. [Google Scholar]
- Stonik, I.V.; Orlova, T.Y.; Begun, A.A. Potentially toxic diatoms Pseudo-nitzschia fraudulenta and P. calliantha from Russian waters of East/Japan Sea and Sea of Okhotsk. Ocean Sci. J. 2008, 43, 25–30. [Google Scholar] [CrossRef]
- Bates, S.S.; Garrison, D.L.; Horner, R.A. Bloom dynamics and physiology of domoic-acid-producing Pseudo-nitzschia species. In Physiological Ecology of Harmful Algal Blooms; Proc. of the NATO Advanced Study Institute on “The Physiological Ecology of Harmful Algae Blooms”, Held at the Bermuda Biological Station for Research, Bermuda, 27 May–6 June 1996; Anderson, D.M., Cembella, A.D., Hallegraeff, G.M., Eds.; Springer: Berlin, Germany, 1966; Series G: Ecolological Sciences, 1996; Volume 41, pp. 267–292. [Google Scholar]
- Hasle, G.R. Pseudo-nitzschia pungens and P. multiseries (Bacillariophyceae): Nomenclatural history, morphology, and distribution. J. Phycol. 1995, 31, 428–435. [Google Scholar] [CrossRef]
- Hasle, G.R.; Lange, C.B.; Syvertsen, E.E. A review of Pseudo-nitzschia, with special reference to the Skagerrak, North Atlantic, and adjacent waters. Helgol. Mar. Res. 1996, 50, 131–175. [Google Scholar] [CrossRef]
- Jester, R.; Lefebvre, K.; Langlois, G.; Vigilant, V.; Baugh, K.; Silver, M.W. A shift in the dominant toxin-producing algal species in central California alters phycotoxins in food webs. Harmful Algae 2009, 8, 291–298. [Google Scholar] [CrossRef]
- Lee, J.H.; Baik, J.H. Neurotoxin-producing Pseudo-nitzschia multiseries (Hasle) Hasle, in the coastal waters of Southern Korea II. Production of domoic acid. Algae 1997, 12, 31–38. [Google Scholar]
- Fehling, J.; Davidson, K.; Bolch, C.; Tett, P. Seasonality of Pseudo-nitzschia spp. (Bacillariophyceae) in western Scottish waters. Mar. Ecol. Prog. Ser. 2006, 323, 91–105. [Google Scholar] [CrossRef]
- Lewis, N.I.; Bates, S.S.; McLachlan, J.L.; Smith, J.C. Temperature effects on growth, domoic acid production, and morphology of the diatom Nitzschia pungens f. multiseries. In Toxic Phytoplankton Blooms in the Sea; Smayda, T.J., Shimizu, Y., Eds.; Elsevier: Amsterdam, The Netherlands, 1993; pp. 601–606. [Google Scholar]
- Smith, J.C.; McLachlan, J.L.; Cormier, P.G.; Pauley, K.E.; Bouchard, N. Growth and domoic acid production and retention by Nitzschia pungens forma multiseries at low temperatures. In Toxic Phytoplankton Blooms in the Sea; Smayda, T.J., Shimizu, Y., Eds.; Elsevier: Amsterdam, The Netherlands, 1993; pp. 631–636. [Google Scholar]
- Cho, E.S.; Kotaki, Y.; Park, J.G. The comparison between toxic Pseudo-nitzschia multiseries (Hasle) Hasle and non-toxic P. pungens (Grunow) Hasle isolated from Jinhae Bay, Korea. Algae 2001, 16, 275–285. [Google Scholar]
- Smayda, T.J. Novel and nuisance phytoplankton blooms in the sea: Evidence for a global epidemic. In Toxic Marine Phytoplankton; Grane’li, E., Sundstrøm, B., Edler, L., Anderson, D.M., Eds.; Elsevier: New York, NY, USA, 1990; pp. 29–40. [Google Scholar]
- Hallegraeff, G.M. A review of harmful algal blooms and their apparent global increase. Phycologia 1993, 32, 79–99. [Google Scholar] [CrossRef]
- Parsons, M.L.; Dortch, Q. Sedimentological evidence of an increase in Pseudo-nitzschia (Bacillariophyceae) abundance in response to coastal eutrophication. Limnol. Oceanogr. 2002, 47, 551–558. [Google Scholar] [CrossRef]
- Shatilina, T.A.; Anzhina, G.I. Variability of the Far-Eastern monsoon intensity in 1948−2010. Izv. TINRO 2011, 167, 146–159, (In Russian with English Abstract). [Google Scholar]
- Lundholm, N.; Hansen, P.J.; Kotaki, Y. Effect of pH on growth and domoic acid production by potentially toxic diatoms of the genera Pseudo-nitzschia and Nitzschia. Mar. Ecol. Prog. Ser. 2004, 273, 1–15. [Google Scholar] [CrossRef]
- Kotaki, Y.; Koike, K.; Sato, S.; Ogata, T.; Fukuyo, Y.; Kodama, M. Confirmation of domoic acid production of Pseudo-nitzschia multiseries isolated from Ofunato Bay, Japan. Toxicon 1999, 37, 677–682. [Google Scholar] [CrossRef]


| Area | Station | Dates | No. of Samples | Latitude N | Longitude E |
|---|---|---|---|---|---|
| Amursky Bay | 1 | 20 April 1993–17 December 1993 | 14 | 43°12′ | 131°54′ |
| 1 | 4 June 1997–5 December 1997 | 21 | 43°12′ | 131°54′ | |
| 1 | 6 June 2002–11 September 2002 | 4 | 43°12′ | 131°54′ | |
| 1 | 14 June 2005–30 December 2005 | 31 | 43°12′ | 131°54′ | |
| 1 | 26 January 2007–29 December 2015 | 205 | 43°12′ | 131°54′ | |
| 2 | 25 June 1992–25 October 1992 | 9 | 43°10′ | 131°52′ | |
| 3 | 31 August 2002–20 November 2005 | 39 | 43°11′ | 131°54′ | |
| 4 | 10 May 2001–20 October 2002 | 11 | 43°01′ | 131°47′ | |
| Ussuriisky Bay | 5 | 13 November 2012–19 December 2012 | 2 | 43°04′ | 131°57′ |
| 5 | 11 November 2013–23 December 2013 | 2 | 43°04′ | 131°57′ | |
| 5 | 15 October 2015–5 November 2015 | 2 | 43°04′ | 131°57′ | |
| 6 | 11 November 2013–23 December 2013 | 2 | 43°05′ | 131°58′ | |
| 6 | 15 October 2015–5 November 2015 | 2 | 43°05′ | 131°58′ | |
| 7 | 13 November 2012–19 December 2012 | 2 | 43°11′ | 132°06′ | |
| 7 | 11 November 2013–23 December 2013 | 2 | 43°11′ | 132°06′ | |
| 7 | 15 October 2015–5 November 2015 | 2 | 43°11′ | 132°06′ |
| Species | Station | Dates | Maximum Concentration (Cells L−1) | Conditions | Data from |
|---|---|---|---|---|---|
| Amursky Bay | |||||
| Pseudo-nitzschia multiseries | 2 | June 1992 | 1.1–3.5 × 107 | After heavy rains at SST of 15.3–16.1 °C | [13] |
| Pseudo-nitzschia multiseries | 1 | June 1993 | 1–1.2 × 106 | SST of 14.2–15.8 °C | [16] |
| Pseudo-nitzschia multiseries | 1 | September 2002 | 1.4 × 106 | SST of 12.5–14 °C | [12] |
| Pseudo-nitzschia calliantha Pseudo-nitzschia pungens | 1 | November 1997 | 2.7 × 1065.9 × 105 | SST of 5–6 °C and SSS of 34.5 | [16] |
| Pseudo-nitzschia fraudulenta Pseudo-nitzschia calliantha | 3 | October– November 2002 | 2.1 × 1051.5 × 105 | Water temperature of 6–16 °C and SSS of 28.8–33.5 | [22] |
| Pseudo-nitzschia multistriata Pseudo-nitzschia calliantha | 1 | October 2005 | 0.9 × 1061.2 × 105 | SST of 6–12 °C and SSS of 31–33.5. | [19] |
| Pseudo-nitzschia calliantha | 1 | September 2007 | 1.7 × 105 | SST of 20 °C | [15] |
| Pseudo-nitzschia multistriata | 1 | September 2012 | 2.5 × 105 | SST of 17.8 °C and SSS of 28. | [15] |
| Pseudo-nitzschia calliantha | 1 | November 2012 | 1.1 × 105 | SST of 6.8 °C | [15] |
| Ussuriisky Bay | |||||
| Pseudo-nitzschia calliantha | 5 | November 2012 | 1.4 × 106 | SST of 6 °C | Unpublished data |
| Pseudo-nitzschia calliantha | 6 | November 2012 | 1.1 × 106 | SST of 6.5 °C | Unpublished data |
| Pseudo-nitzschia calliantha | 7 | November 2012 | 1.2 × 106 | SST of 6 °C | Unpublished data |
| Pseudo-nitzschia calliantha | 7 | November 2013 | 1.9 × 105 | SST of 5 °C | Unpublished data |
| Pseudo-nitzschia calliantha | 6 | October 2015 | 3.5 × 105 | SST of 10.4 °C | Unpublished data |
| Pseudo-nitzschia multistriata | 7 | October 2015 | 1.7 × 105 | SST of 14.3 °C | Unpublished data |
| Cluster | R-Statistics | Significance Level |
|---|---|---|
| A, B | 1 | 0.1% |
| A, C | 1 | 0.1% |
| A, D | 1 | 0.1% |
| A, E | 1 | 0.1% |
| B, E | 0.999 | 0.1% |
| B, C | 0.976 | 0.1% |
| B, D | 1 | 0.1% |
| E, C | 0.997 | 0.1% |
| E, D | 1 | 0.1% |
| C, D | 1 | 0.1% |
| Factor | Pseudo-nitzschia spp. | Pseudo-nitzchia multistriata | Pseudo-nitzchia calliantha |
|---|---|---|---|
| NH4 | −0.521 | −0.487 | −0.691 |
| Salinity | −0.445 | −0.441 | −0.232 |
| Water temperature | 0.428 | 0.432 | 0.204 |
| Species | r |
|---|---|
| Pseudo-nitzchia calliantha | 0.278 |
| Pseudo-nitzchia pungens | 0.188 |
| Pseudo-nitzchia multistriata | 0.154 |
| Pseudo-nitzschia spp. | 0.464 |
| Species and Temperature Differences | Months and Years of Bloom Events | |
|---|---|---|
| September | October | |
| Pseudo-nitzchia multistriata | nd | 2005 |
| Pseudo-nitzchia calliantha | 2007 | nd |
| Difference of mean monthly SST | 0.9 °C | 1.4 °C |
| Species | Maximum DA Concentration (Picogram DA Cell−1) |
|---|---|
| Pseudo-nitzchia multiseries | 20.8 (Peter the Great Bay, Russia a) |
| 1.03–2.4 (Jinhal and Chinhae bays, South Korea b) | |
| 1.15–140 (Okkiray Bay, Japan c) | |
| 5.7 (Ofunato Bay, Japan d) | |
| Pseudo-nitzchia pungens var. pungens | 0.0004 e |
| Pseudo-nitzchia calliantha | 0.44 e |
| Pseudo-nitzchia multistriata | 0.57 e |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stonik, I.V. Long-Term Variations in Species Composition of Bloom-Forming Toxic Pseudo-nitzschia Diatoms in the North-Western Sea of Japan during 1992–2015. J. Mar. Sci. Eng. 2021, 9, 568. https://doi.org/10.3390/jmse9060568
Stonik IV. Long-Term Variations in Species Composition of Bloom-Forming Toxic Pseudo-nitzschia Diatoms in the North-Western Sea of Japan during 1992–2015. Journal of Marine Science and Engineering. 2021; 9(6):568. https://doi.org/10.3390/jmse9060568
Chicago/Turabian StyleStonik, Inna V. 2021. "Long-Term Variations in Species Composition of Bloom-Forming Toxic Pseudo-nitzschia Diatoms in the North-Western Sea of Japan during 1992–2015" Journal of Marine Science and Engineering 9, no. 6: 568. https://doi.org/10.3390/jmse9060568
APA StyleStonik, I. V. (2021). Long-Term Variations in Species Composition of Bloom-Forming Toxic Pseudo-nitzschia Diatoms in the North-Western Sea of Japan during 1992–2015. Journal of Marine Science and Engineering, 9(6), 568. https://doi.org/10.3390/jmse9060568






