Abstract
The aim of this paper is to present information gathered from studies regarding the current status and challenges of microplastics (MPs) in Mediterranean coastal counties. MPs are considered emerging pollutants, and their effect on fish and on the final consumer through the trophic food chain are of great concern. Studies from almost all of the Mediterranean coastal countries are gathered and discussed. The source of MPs, as well as their transfer and accumulation are reviewed. In addition, the laboratory techniques for sampling, analysis and characterization of MPs are presented. Moreover, the current regulations for MPs restrictions in Europe will be discussed. Finally, the authors present the current challenges on the topic and provide recommendations for future work.
1. Introduction
Plastics have an essential role in everyday life, but despite that, they pose a potential health risk for both humans and the environment. Plastics in dimensions of less than 5 mm are named microplastics (MPs), which can be deliberately added to products (e.g., cosmetics) or can be formed when plastics of a larger size break down (e.g., from plastic litter in the seas). Microplastics originate from various polymer-based materials. Currently, the Mediterranean Sea is affected by significant plastic pollution [1]. Although the Mediterranean Sea is one of the most heavily investigated maritime regions worldwide, information on marine litter is relatively limited and poorly documented [2]. MPs were found to be more than 80% of the total plastic pollution in the Mediterranean [3]. Concentrations of plastic debris on the surface waters of the Mediterranean Sea, specifically at basin scale, are presented in Figure 1. Awareness of the environmental, economic, social, public safety and individual health risks posed by microplastic pollution is increasing and this leads to the development of various and diverse sets of regulatory tools at local, national, regional, and international levels [4].
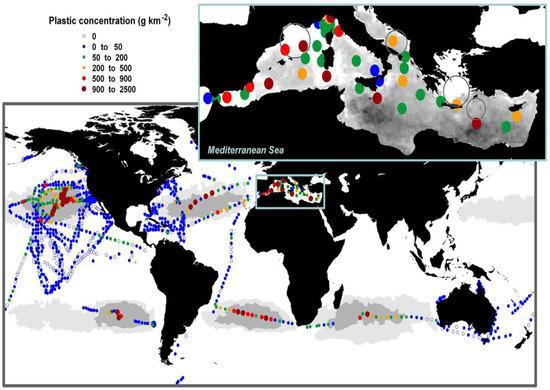
Figure 1.
Concentrations of plastic debris in surface waters of the Mediterranean Sea at basin scale (zoomed in the top right corner inset), and compared to the plastic concentrations reported for oceans globally. Reprinted with permission taken by Public Library of Science (PLoS) [3].
MPs enter the aquatic environment either as a readily manufactured ingredient through the treated wastewater effluent, or as product of plastic photodegradation or wear breakdowns. Examples of already manufactured MPs are some raw materials in cosmetic products, or fibers from synthetic clothes. A questionnaire-based survey of wastewater treatment plants (WWTPs) in Greece [5] showed that WWTPs are a potential way for plastic to escape in different aquatic environments, and that WWTPs cannot remove plastics around the size of a cotton swab stick or below. In addition, generation and accumulation of MP pollution tends to occur in areas such as closed bays, gulfs and seas that are surrounded by densely populated coastlines [6,7].
Once released in the marine environment, high-density plastics (e.g., polyester) tend to settle and accumulate in the sediment, while low-density microplastics (e.g., polyethylene) float on the sea surface [8]. Since MPs are not naturally removed from the marine environment, they can be ingested by aquatic biota. MPs can aid the delivery of persistent organic pollutants (POPs), which are added during manufacturing or adsorbed and concentrated from the surrounding seawater [9]. As a result, MPs laden with high levels of POPs can be transferred via the food chain. Studies proposed that vertical settling from surface accumulations causes MPs to move to the seafloor; however, a recent publication demonstrated that the spatial distribution and final destination of MPs is strongly controlled by the bottom current (i.e., near-bed thermohaline currents) [10]. Although surface currents distribute the plastics on the sea surface, most of the plastic litter ends up in the deep sea and not on the ocean surface [10,11].
In recent years, the number of analyses proposed to properly characterize the contamination of the natural environment by microplastics has increased [12] and the use of statistical approaches for reducing the number of microplastics required for analysis can be a robust and efficient tool [13].
In this review, information gathered from studies in 18 Mediterranean coastal countries will be discussed. The source of MPs, their transfer and accumulation are reviewed. In addition, the laboratory techniques for sampling, analysis and characterization of MPs are presented. Moreover, the current regulations for MPs restrictions in Europe will be discussed. Finally, the authors present the current challenges on the topic, and provide recommendations for future work.
2. Sampling and Analysis
2.1. Sampling Methods
Recent years have shown a steady increase in literature on the topic, showing a diverse spatial and temporal distribution of MP concentrations in the Mediterranean’s surface waters. The sampling of microplastics takes place in maritime waters (sea surface, water column, sediments and living aquatic organisms) [14]. There are three ways of collecting microplastic samples from the aquatic ecosystem: Bulk sampling, selective sampling and volume-reduced sampling. The primary disadvantage of selective sampling method is that the size of discoverable MPs is highly restricted and microplastics are mixed with other debris, so there is a significant risk οf overlooking them. Βulk samples refer to samples where the entire volume of the sample is taken without reducing it during the sampling procedure. Bulk sampling is suitable when microplastics cannot be easily identified visually because they are covered by sediment particles, so it is necessary to filter large volumes of sediment/water [14,15]. However, bulk sampling only allows for small sample sizes, which might render the sample less representative. The volume of the bulk sample is mostly reduced during sampling, preserving only that part of the sample that is of interest for extra treatment. Therefore, volume-reduced sampling has the advantage of being able to cover great quantities of samples during the sampling process [15]. The disadvantage, however, is that the majority of the sample is rejected, resulting in significant loss in microplastics. Among these three methods, volume-reduced sampling is the most popular for water samples, selective method is mostly used in beach sampling, while the bulk method is mainly used to collect sediment sample and occasionally water samples. Further processing under laboratory conditions requires the use of the bulk and volume-reduced samples [16,17,18,19]. From specific depths οf water column or water surface samples can be collected. There are various approximations, which are used for the sampling of microplastics, the majority of which are based on the volume-reduced method [19,20].
Samples of water can be obtained at specific depths from the surface or from the water column [14]. There are various methods that are used for water microplastics sampling. Neuston networks and manta trawls are the most common devices for surface water sampling, while the other techniques are mainly used for water column sampling, bongo networks, plankton networks, continuous plankton net recorders (CPR), and multiple opening networks [15,21]. Alternative devices for microplastics, such as water retention tubes or water intake pumps, are often used in surface water or water column sampling. It was found that most of the sampling techniques commonly used are only applicable on a selection of specific microplastics size ranges [15]. Using varying mesh sizes for sampling makes the analysis of measurement data challenging, as the amount of particles per km2 or m3 of water may be considered units of measurement for abundances of microplastics in water. Since sampling microplastics, which are in the samples for further detection and quantification, should be collected. Techniques such as separation of mass, sieving, digestion and filtration are critical for the separation of microplastics from other materials [19,22].
2.2. Analysis
Particle analysis, or characterization, can be split into two different sub-types. Morphological or physical categorization take sizes, colors and shape into account, while chemical classification concentrates on polymer types and additives. Various techniques are viable for microplastic identification, from an uncomplicated visual identification to analytical techniques based on the chemical composition of the polymer [23]. This is usually conducted on specific instruments such as scanning electron microscopy (SEM), Raman spectroscopy and pyrolysis-gas chromatography–mass spectrometry (Pyr–GC–MS) and Fourier transform infrared spectroscopy (FTIR) [24].
2.2.1. Scanning Electron Microscopy (SEM)
Using a scanning electron microscope (SEM) creates an image of a small surface of a sample, by firing a high intensity of electrons at the sample surface and scanning it in a raster scan pattern [24]. The sample is imaged with lots of surface details (0.5 nm revolution) by the electrons at very high magnifications. The surface morphology of high resolution microplastics images is examined, so as to differentiate them from other organic or inorganic impurities [24]. The combined used of SEM and energy dispersive X-ray spectroscopy can provide plenty of information about the elemental composition of microplastics and the additives they contain [24,25].
2.2.2. Raman Spectroscopy/FTIR and Fluorescence Spectroscopy
Another widely employed and extremely reliable method for polymer detection of microplastics from many environmental matrices is Raman spectroscopy. Identification of microplastics by an irradiating monochromatic laser in a suspicious sample results in a variable wavelength of a backscattered light due to the absorption, reflection or scattering of the specific molecular structure and atomic composition of the material. This famous shift from Raman will generate an individual spectrum for any polymer [21,23,26]. FTIR has a high degree of efficacy and is one of the most widely used methods in the chemical characterization of microplastics. FTIR is primarily used in screening systems for microplastics in two ways: Evaluating a collection of subsamples to validate the effects of visual detection, or scanning all the detected particles. FTIR can not only reliably classify the polymer forms of microplastics, but also provide details on microplastics’ physiochemical weathering by evaluating their oxidation strength [27]. FTIR remains a promising tool to classify toxic microplastics in a chemical way. Raman spectroscopy shares some of the advantages of FTIR, such as high reliability, good screening performance, low demand for samples, and being environmentally friendly [28]. Raman spectroscopy is useful in higher spatial analysis, broader spectral spectrum, and thinner spectral bonds as opposed to FTIR techniques. The key downside of Raman strategies is that they are easily disturbed by the existence of microplastics-related pigments, contaminants or attached chemicals, which may have a detrimental effect on the precision of the detection. Both Raman and FTIR are ideal non-destructive methods for detecting particles of MPs in environmental samples and determining the observed particles’ chemical and morphological characteristics [26,28,29,30]. Both methods have similar drawbacks, such as complicated sample preparation and the occurrence of interferences induced by particle surface alterations [28]. A combination of Raman spectroscopy with microscopy makes it feasible to recognize microplastics down to 1 mm in size, a degree of accuracy which is extremely difficult for other spectroscopic techniques to attain [24].
It is theoretically possible to detect microplastics tens of microns in size using micro-FTIR and Raman spectroscopes [31]), but repeated trials are often required to obtain reliable spectra of very small and weathered plastic particles. Both the FTIR and Raman spectroscopy methods require expensive instruments and are time-consuming when a large number of microplastic particles are present on the filter paper. This is not only the case for environmental samples with complex matrices but also for the quantification of controlled laboratory experimental samples. Therefore, it is becoming increasingly important to develop alternative methods to facilitate identification of microplastics for field monitoring and laboratory studies on the toxicity, accumulation, weathering, etc. of microplastics. Plastics are comprised of hydrocarbons derived from petroleum, natural gas or biomass, and are hydrophobic. Lipophilic dyes can be used to visualize microplastics under a fluorescence microscope [9]. Nile Red (9-diethylamino-5H-benzo[α]phenoxazine-5-one; hereafter, NR), a fluorescent dye, has been used to stain neutral lipids in biological samples [32] and can be used to stain synthetic polymers in polymer chemistry [33]. NR is a hydrophobic fluorophore that specifically binds to neutral lipids and is strongly fluorescent only in the presence of a hydrophobic environment [34]. There are many published studies in which the aforementioned analysis was carried out for MPs detection [23,35,36]. Fluorescence is a critical limitation of Raman. In MP analysis, surface alterations ranging from sorption of humic substances to surface oxidation (aging) or biofouling are a major source of fluorescence. During the dwell time of MP particles in (aqueous) environments, numerous alterations of the particle surface can likely occur. Also, artificial dyes in colored plastic particles can impede detection and may result in the generation of spectra of dyes instead of the polymers. To circumvent problems with fluorescence, more sample preparation steps are required. Additionally, humic substances can be removed by treatment with bases, like potassium hydroxide and sodium hydroxide solutions.
2.2.3. Thermo-Analytical Methods
In synthetic polymer analytics, even thermo-analytical approaches are widely used. Methodologies for detecting MPs were developed on the basis of thermo-analytical methods such as thermogravimetry, differential scanning, calorimetry etc. [23,30]. Promising findings obtained by integrating thermogravimetry with differential calorimetry scanning (TGA-DSC), pyrolysis gas chromatography-mass spectrometry (Py-GC-MS), and thermal extraction desorption gas chromatography-mass spectrometry (TED-GC-MS), which is a modification of this. Py-GC-MS and TED-GC-MS use pyrolysis for polymer detection and quantification, while TGA-DSC is based on heat changes during the solid-liquid transformation of polymer. The same rationale exists for DSC and TGA-DSC: Elevated temperatures induce the transformation of a polymer sample in phase. However, there are inconveniences with respect to TGA-DSC [25,37]. Owing to overlapping transition temperatures, not each polymer type could be clearly separated. Production parameters (additives, impurities, and branching of polymer chains which could impact the recognition of complex polymers) also affect transition temperatures [23,37]. High sample volumes for TED-GC-MS compared with Py-GC-MS can be added from 0.5 to 100 mg, resulting in improved sensitivities. Measurement times are about 2.5 h. per study [38]. In comparison to spectroscopic approaches, thermo-analytical methods are destructive, capable only of chemical characterization, and cannot describe morphological properties as distributions of size and particle size [38].
3. Discussion
Following a clockwise order, the MP-related literature from the coastal Mediterranean countries is discussed. Literature from the following countries is presented; Spain, France, Italy, Slovenia, Croatia, Bosnia and Herzegovina, Montenegro, Albania, Greece, Turkey, Lebanon, Israel, Egypt, Tunisia, Algeria, Morocco, Malta and Cyprus.
Spain. A recent study has confirmed the widespread distribution of MPs in surface sediments from the Spanish Mediterranean continental shelf [39]. MPs were extracted from bulk sediments by density separation. The number of MPs/kg of d.w. (dry weight) were from 45.9 ± 23.9 MPs/kg d.w. observed at Palma de Mallorca to 280.3 ± 164.9 MPs/kg d.w. noted at Málaga, with an average value of 113.2 ± 88.9 MPs/kg d.w. Moreover, MP size distribution for fragments showed that 85% was lower than 0.5 mm, and in the case of fibers, three intervals (0.5–1, 1–2, 2–5 mm) had a similar distribution (35, 34 and 31%, respectively) [39]. Sediment samples were obtained during the autumn of 2013, in the coastal shallow waters of Mallorca Island and Cabrera Island [40]. Sediments from marine protected areas contained up to 0.90 ± 0.10 MPs/g, indicating that MPs move from source areas to endpoint areas. Beach sediment from Denia and Barcelona [41] showed average abundances of 156 and 148 MPs/kg d.w.
France. The French Mediterranean coastline is subject to permanent waste inputs, such as those occurring on the continental shelf of the Golf of Lion [6]. Schmidt et al. [42] reported that the overall average MP abundance of samples collected at the Golf of Lion was 112·103 items km−2, which is in the same range as other areas in the northwestern basin [6]. For the Bay of Marseille, it is estimated that the total MP load consists of 519·103–101·106 items. Authors reported significant temporal and spatial heterogeneity for MP abundances, and stated that anthropogenic pressures are significant in the area [42]. Beach sediment from Cassis showed average values of 124 MP/kg d.w.
Italy. From a study on MPs along the Tuscany coast, the results showed an average concentration of 0.26 items/m3 in the water column samples, and 41.1 g/km2 and 69,161.3 items/km2 of floating microplastics, with an increase along with the distance to the coast [43]. In the coastal waters of South Tuscany [44], MPs are the majority of analyzed sediments, with the maximum MP items/kg being 1069 in Albania’s sandy shore, while the minimum was detected in a surficial sample from Talamone harbor (42 items/kg). Results from a WWTP in Northern Italy highlighted that, despite the high MP removal efficiency (i.e., 84%), MPs cannot be controlled completely at the final effluent [45]. The results of a large-scale survey [46,47] of neustonic micro- and mesoplastics floating in Mediterranean waters (close to Italy but not limited to it), provided detailed information on their abundance and geographical distribution (Figure 2).
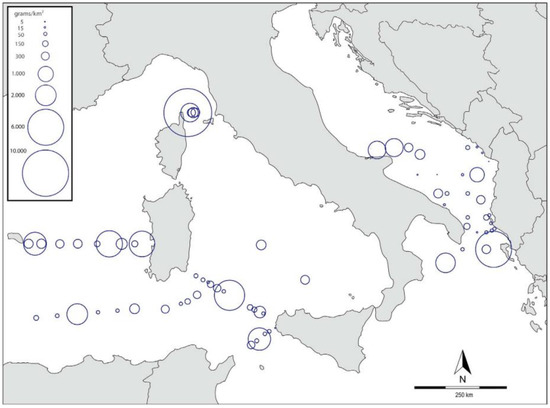
Figure 2.
Map of the central-western Mediterranean Sea showing the location of all sampling stations and the distribution of un-corrected plastic densities expressed as grams of plastic per km2. Redrawn from [46].
Slovenia. Slovenia’s coastline, located on the Gulf of Trieste, by the Adriatic Sea, is less than 50 km long. Sediment samples were collected at the strandlines of nine locations along the coast of Slovenia, and analyzed using infrared spectroscopy [48]. MP abundances at the strandlines of Slovenian beaches were unexpectedly low, with tourism and aquaculture assumed to be the major microplastic contributors.
Croatia. Sediments from the natural park of Telaščica bay showed that MPs were the principal litter group in sediments. Compared to Venice, Italy [49], low to medium range of contamination by MPs was evidenced for the sediments from Croatia [50]. Moreover, a study on trace metal concentrations in plastic pellets in the island of Vis at Eastern Adriatic Sea, Croatia, showed that the trace metal concentrations in plastic pellets are greater than the concentrations reported for seawater in the investigated area, which indicates that plastic pellets sorb metals from the marine environment [51].
Bosnia and Herzegovina. Even though the Bosnia and Herzegovina coastline is very short (24.5 km), due to the karstic nature of its inland pertaining to the Adriatic region and rapid rainfall infiltration of pollutants, as well as the poor waste water management, the pollution impact to marine environment is expected to be high. The first assessment and application of common methodology for marine microplastics pollution in Bosnia and Herzegovina was performed in 2014 [52]. Monitoring of sea surface microplastics has been performed by using a manta net with 308 µm pore size on 2 sampling sites. The identification and separation of microplastics particles was performed using a stereomicroscope. MPs were characterized using a FTIR microscope.
Montenegro. Urbanization and tourism, industry and maritime activities are the main sources of marine and coastal pollution in Montenegro. A study in Velikaplaža near Ulcinj revealed that artificial polymer items are by far the most common litter material found on the beach [53].
Albania. Recent studies [54,55] showed that plastic materials accounted for the majority of marine litter items found, with a percentage of 65% of traceable marine litter items originating from land-based sources. To the knowledge of the authors, no research on microplastics has been performed yet, however due to the large quantity of plastics on the Albanian coastline, the presence of MPs is inevitable.
Greece. At the northern beaches of the Kea island [56], MPs exceeded the amount of 300 items per m2, showing a direct relation to beach orientation and wind regime rather than to proximity to land based sources. The authors emphasize the importance of water circulation as the major mechanism of microplastic dispersion for the presence of MPs worldwide [56]. From a later study close to Pilion [41], the concentration range of MPs varied from 150 to 300 MP/kg d.w.
A study on MPs from Lesvos island [57] examined the long-term phenanthrene distribution properties of new plastic pellets obtained from local plastic producers compared to eroded plastic pellets found in Lesvos island beaches, in order to determine differences in the sorptive behavior of the various materials. Results showed that the adsorption of phenanthrene on propylene MPs increased with salinity.
Plastic pellets were used to determine the diffuse pollution of some Greek beaches, and the observed differences among pellets from various sampling sites were related to the pollution occurring at each site [58]. Plastic pellets collected in Saronikos Gulf beaches demonstrated much higher pollutant loading than the ones collected in a remote island or close to an agricultural area [58].
The occurrence and the spatial distribution of MPs in the shallow marine sediments of Samos island were investigated [59]. The results show higher concentrations of MPs at the Beach compared to the Intertidal zone, indicating that transport by hydrodynamics and in situ deposition by humans may be important processes explaining the lateral distribution of MPs. In addition, the distribution of MPs could indicate that spatial distribution is not only governed by size, but perhaps a morphology and chemical composition as well.
Turkey. An average of 0.9 MP were found per liter of effluent from three WWTPs amounting to around 180 × 106 particles per day to Mersin Bay [60]. This shows that despite their ability to remove 55–97% of microplastics, WWTPs are one of the main sources of MPs to the northeast Mediterranean Sea [60]. MP distribution at surface waters in the Sea of Marmara showed that the average level of MPs on the surface was 1.263 item/m2 [61]. MP contamination in the Datça Peninsula was eminent, with the highest contamination found in Aktur Beach (2073.3 ± 648.6 particles/kgd.w.) which could be considered as a hotspot for the whole region in terms of MP pollution [62]. From a study in Mersin Bay [63], 94% of all collected MPs from the sea were in the size range between 0.1 and 2.5 mm. The quantity of MPs in surface water samples ranged from 16,339 to 520,213 per km2.
Lebanon. The first study on plastic pellets along the cost of Lebanon is reported to have taken place in the late 1970s [64]. A MP evaluation of the Lebanese coast was conducted in three cities (Tripoli, Beirut, Sidon) and showed that Lebanese sediments, waters and biota are exceedingly contaminated in MPs [65]. Specifically, the water sample from Sidon contained MPs (6.7 MPs/m3) while Tripoli showed the highest MPs in sediments (4.68 MPs/g). When it comes to biota, the number of MPs found in the digestive track of European anchovies (Engraulis encrasicolus) was high (83.4% and 86.3% in anchovies and spiny oysters, respectively). Both anchovies and oysters from the Beirut region had the highest ingested MPs/individual (2.9 ± 1.9 and 8.3 ± 4.4 MPs/individual, respectively) [65]. Authors compared MPs pollution found in the Levantine Basin with other Western Mediterranean regions, and concluded that MPs contamination in Lebanon is higher. Finally, the study highlighted the role of landfills in the entry of MPs into the coastal marine environment.
Israel. An early 1990s study showed that more than 70% of the coastal litter was plastic [66]. A thorough study across the Israeli Mediterranean coastal water showed very high abundances of MPs [67]. MPs (0.3–5.0 mm) were found in all samples, with a mean abundance of 7.68 ± 2.38 particles/m3 or 1,518,340 particles/km2.
Egypt. MP contamination in fish from the Nile River was examined [68]. Over 75% of the fish sampled contained MPs in their digestive tract, with the most abundant MP type being fibers, followed by films. The chemical composition of MPs was non-destructively identified using attenuated total reflectance Fourier transform infrared (ATR-FTIR) spectroscopy. Authors mentioned that such a high level of MP ingestion is rarely found, and that fish sampled from the Nile River in Cairo are potentially among the highest-risk groups of consuming MPs worldwide [68]. Shabaka et al. [69] mentioned that the lack of proper solid waste management in Egypt has resulted in the accumulation of marine plastic litter, and that MP pollution has a large geographical extent due to dispersal by surface water currents. Authors used differential scanning calorimetry (DSC) as a cost-effective method to identify MPs in Eastern Harbor, Alexandria, and they reported ten different polymers in seawater and shoreline sediments.
Tunisia. MPs in sediments in the northern coast of complex lagoon-channel of Bizerte were investigated [70]. Authors showed that the number of MPs was at the range of 3000–18,000 items/kg dry sediment. The distribution of MPs was investigated in the sediments of five sampling sites from the northern Tunisian coast during June 2017, and representative MPs from five sites were isolated for polymer identification using FTIR-ATR [71]. The results showed that MPs that were recovered, from all sediment samples, showed extensive distribution along the Tunisian coast. Concentrations varied from 141.20 ± 25.98 to 461.25 ± 29.74 items/kg d.w. Authors mentioned that one main source of MPs entering the marine environment is the wastewater infrastructure which, in Tunisia, is poorly managed and unable to keep up with the rapidly growing population [71].
Algeria. In Algeria, the coastal population increased by 112% in the past 30 years, and currently represents a densely populated coastline [47]. Although there is no available data on microplastics, anthropogenic marine debris in Algerian basin had mean values of 52.9 ± 11.4 items/km2, respectively.
Morocco. The ingestion of microscopic debris (<5 mm) by marine organisms has been studied. The evaluation of the ingestion phenomenon of MPs in the fish demonstrated that 10% of fish samples had ingested MPs [72].
Malta. Plastic pellets are abundant on the beaches of Malta, especially on the backshores of beaches with a westerly aspect. After qualitative examination of plastic pellets, authors reported that MPs are similar to those reported for other regions of the Mediterranean in surveys spanning three decades, suggesting that they are a general and persistent characteristic of the region [73]. The first detailed investigation of MPs in Malta was conducted [74] and identification of polymer type was carried out by means of qualitative density tests. Higher MP concentrations were recorded at 10 m up-shore as opposed to the strandline. The local level of occurrence of MP seems to be lower when compared to other European locations studied so far, but that can be explained by the fact that in their study, MPs below 1 mm were not included in the data.
Cyprus. A recent study showed that the beaches of Cyprus are exposed to very high MPs abundances, with the majority of them coming from industrial spills, followed by fragments from the breakdown of larger plastics [75]. MPs were found in all sampling points, and authors mentioned that a high abundance of MPs could alter life history parameters of marine turtles. Microplastics (<5 mm) were found at all locations and depths, with particularly high abundance in superficial sand. The top 2 cm of sand presented grand mean ± SD particle counts of 45,497 ± 11,456 particles m−3 (range 637–131,939 particles m−3) [75].
4. Regulations
Many consumers are unaware that MPs are in their products, and of their possible route to the wastewater treatment plant effluent and from there to the seas. Various local, regional, and transnational initiatives have been developed toward the mitigation of plastic pollution [4]. Microplastic hazards are numerous and still undetermined. In some cases replacement of MPs can be easily applied, but in other cases substitutions may come with more uncertainty, performance questions and costs [76]. According to Mitrano and Wohlleben [76], an unbiased assessment of the hazard, fate, and societal benefits of primary microplastics throughout the regulation process is needed and regulation should be enforceable and focused, and linked to hazards before alternative and more environmentally conscious materials replace microplastics.
The EU has highlighted the necessity to adopt measures to preserve the Mediterranean Sea, since the contamination in the Mediterranean Sea is critical, mainly due to overexploitation of the resources and global warming [77]. The first Mediterranean Action Plan was implemented by the European Community in 1975 [78], followed by the Protocol on Integrated Coastal Zone Management in the Mediterranean [79] adopted during the Barcelona Convention in 1995. The European Union and 21 countries are involved and one of the main goals is working with contracting parties and partners to fulfill the vision of a healthy Mediterranean Sea and Coast that underpin sustainable development in the region [78]. The EU’s Marine Strategy Framework Directive (MSFD) aim was to more effectively protect the marine environment across Europe. The MSFD was adopted on 2008 [80]. The knowledge gained from implementing this directive was a driving force leading to the adoption of the Single Use Plastics Directive.
The first-ever European Strategy for Plastics in a Circular Economy [81] adopted in 2018 will transform the way plastic products are designed, used, produced and recycled in the EU. This new strategy will lead to better design of plastic products, higher plastic waste recycling rates, delivering greater added value for a more competitive, resilient plastics industry.
Starting in 2018, the European Chemical Agency (ECHA) examined the need for an EU-wide restriction on the placement and marketing or use of intentionally added MPs, and in 2019, ECHA proposed a wide-ranging restriction on intentional uses of microplastics in products placed on the EU market, to avoid or reduce their release to the environment. The ECHA’s proposal is not limited to MPs in cosmetics, but also applies to any other consumer product placed on the EU market. This proposal is estimated to cut down emissions of MPs by at least 85% and prevent the release of 400,000 tons of MPs for the upcoming 20 years after the restriction. Discussions with Member States and voting should begin in 2021, and if an agreement is reached, a restriction will begin from 2022 [82].
5. Techno-Economical Aspects
Nowadays the use of plastics is ever-present. Globally, the overall annual plastics demand has grown tremendously (~230 times). This rise in plastics production and use represents a greater simple understanding and awareness of their characteristics, which has allowed better control of their properties, and has lead to the growth of a significant number of new polymers [83]. The world population grew from about 2.5 billion people in 1950 to 7.7 billion people today. Compared to the conventional production of plastics, the amount produced per capita each year on average across the global population has shown that the quantity of plastics each capita produced over this timeframe has risen by around 50 times. While this calculation is not the same for people with high incomes relative to countries with low incomes, where the consumption of plastics is drastically different, what it showcases is the dependence we have on plastics [84]. Development in the use of microplastics has detrimental effects, the most obvious of which is the illegal leakage of plastics to the atmosphere due to massive quantities of plastics that ultimately end up in the seas. Currently, the lifecycle for most plastics is changed to a linear economy approach whereby plastics are made, used once and discarded, which have become known as single use plastics [84,85]. This consumption model is at the root of the latest problems with plastics contamination. Such techniques include maximizing their reuse and perchance potentially by extracting the materials found in plastics and microplastics, contributing to the reversal of the linearity of usage to which so many of the new plastics are subject. This circularity of the distribution of goods is fundamental to the idea of a circular economy [85]. The abovementioned European Strategy for Plastics in a Circular Economy [81] is part of Europe’s transition towards a circular economy, and will also contribute to reaching the sustainable development goals, the global climate commitments and the EU’s industrial policy objectives. This strategy will reduce marine litter by supporting more sustainable and safer consumption and production patterns for plastics.
The use of economic instruments to prioritize waste prevention and recycling at the national level for every Mediterranean country is essential. Internalizing the environmental costs of landfilling and incineration through high or gradually rising fees or taxes could improve the economics of plastic recycling [86].
6. Conclusions
In the Mediterranean basin, MPs were found in all coastal countries. From the literature, MP levels are diverse, and further studies are needed to comprehend the distribution dynamics of MPs in coastal areas. Strict regulations are needed to avoid plastics getting into the environment, by cutting the use of MPs from the source. Even if the use of MPs were prohibited from now on, the plastics are already present and their degradation will generate MPs. Any restriction that will be proposed will influence the industry, because it will necessitate the use of alternative materials such as biodegradable materials.
Author Contributions
Methodology, G.F., E.I., A.T., A.C.M. and G.Z.K.; writing-original draft preparation, G.F., E.I., A.T., A.C.M. and G.Z.K.; writing—review and editing, G.F., E.I., A.T., A.C.M. and G.Z.K.; supervision, G.F. and G.Z.K.; All authors have read and agreed to the published version of the manuscript.
Funding
This work was financed by the project of Kavala Port S.A. “Assessment and measures of microplastics pollution in the marine environment of Kavala region” (acronym: MICROPLAKA; project code: 30344).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available upon request from the corresponding author.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Martellini, T.; Guerranti, C.; Scopetani, C.; Ugolini, A.; Chelazzi, D.; Cincinelli, A. A snapshot of microplastics in the coastal areas of the Mediterranean Sea. TrAC Trends Anal. Chem. 2018, 109, 173–179. [Google Scholar] [CrossRef]
- Cincinelli, A.; Martellini, T.; Guerranti, C.; Scopetani, C.; Chelazzi, D.; Giarrizzo, T. A potpourri of microplastics in the sea surface and water column of the Mediterranean Sea. Trac Trends Anal. Chem. 2019, 110, 321–326. [Google Scholar] [CrossRef]
- Cózar, A.; Sanz-Martín, M.; Martí, E.; González-Gordillo, J.I.; Ubeda, B.; Gálvez, J.Á.; Irigoien, X.; Duarte, C.M. Plastic Accumulation in the Mediterranean Sea. PLoS ONE 2015, 10, e0121762. [Google Scholar] [CrossRef] [PubMed]
- da Costa, J.P.; Mouneyrac, C.; Costa, M.; Duarte, A.C.; Rocha-Santos, T. The Role of Legislation, Regulatory Initiatives and Guidelines on the Control of Plastic Pollution. Front. Environ. Sci. 2020, 8. [Google Scholar] [CrossRef]
- Mourgkogiannis, N.; Kalavrouziotis, I.K.; Karapanagioti, H.K. Questionnaire-based survey to managers of 101 wastewater treatment plants in Greece confirms their potential as plastic marine litter sources. Mar. Pollut. Bull. 2018, 133, 822–827. [Google Scholar] [CrossRef]
- Collignon, A.; Hecq, J.-H.; Glagani, F.; Voisin, P.; Collard, F.; Goffart, A. Neustonic microplastic and zooplankton in the North Western Mediterranean Sea. Mar. Pollut. Bull. 2012, 64, 861–864. [Google Scholar] [CrossRef] [PubMed]
- Eriksen, M.; Lebreton, L.C.M.; Carson, H.S.; Thiel, M.; Moore, C.J.; Borerro, J.C.; Galgani, F.; Ryan, P.G.; Reisser, J. Plastic Pollution in the World’s Oceans: More than 5 Trillion Plastic Pieces Weighing over 250,000 Tons Afloat at Sea. PLoS ONE 2014, 9, e111913. [Google Scholar] [CrossRef] [PubMed]
- Van Cauwenberghe, L.; Devriese, L.; Galgani, F.; Robbens, J.; Janssen, C.R. Microplastics in sediments: A review of techniques, occurrence and effects. Mar. Environ. Res. 2015, 111, 5–17. [Google Scholar] [CrossRef]
- Andrady, A.L. Microplastics in the marine environment. Mar. Pollut. Bull. 2011, 62, 1596–1605. [Google Scholar] [CrossRef]
- Kane, I.A.; Clare, M.A.; Miramontes, E.; Wogelius, R.; Rothwell, J.J.; Garreau, P.; Pohl, F. Seafloor microplastic hotspots controlled by deep-sea circulation. Science 2020, 368, 1140–1145. [Google Scholar] [CrossRef]
- Choy, C.A.; Robison, B.H.; Gagne, T.O.; Erwin, B.; Firl, E.; Halden, R.U.; Hamilton, J.A.; Katija, K.; Lisin, S.E.; Rolsky, C.; et al. The vertical distribution and biological transport of marine microplastics across the epipelagic and mesopelagic water column. Sci. Rep. 2019, 9, 7843. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Q.; Tan, Z.; Wang, J.; Peng, J.; Li, M.; Zhan, Z. Extraction, enumeration and identification methods for monitoring microplastics in the environment. Estuar. Coast. Shelf Sci. 2016, 176, 102–109. [Google Scholar] [CrossRef]
- Kedzierski, M.; Villain, J.; Falcou-Préfol, M.; Kerros, M.E.; Henry, M.; Pedrotti, M.L.; Bruzaud, S. Microplastics in Mediterranean Sea: A protocol to robustly assess contamination characteristics. PLoS ONE 2019, 14, e0212088. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Wang, J. Investigation of microplastics in aquatic environments: An overview of the methods used, from field sampling to laboratory analysis. TrAC Trends Anal. Chem. 2018, 108, 195–202. [Google Scholar] [CrossRef]
- Crawford, C.B.; Quinn, B. 8-Microplastic collection techniques. In Microplastic Pollutants; Crawford, C.B., Quinn, B., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 179–202. [Google Scholar]
- Zhang, K.; Su, J.; Xiong, X.; Wu, X.; Wu, C.; Jiantong, L. Microplastic pollution of lakeshore sediments from remote lakes in Tibet plateau, China. Environ. Pollut. 2016, 219, 450–455. [Google Scholar] [CrossRef]
- Tsang, Y.Y.; Mak, C.W.; Liebich, C.; Lam, S.W.; Sze, E.T.P.; Chan, K.M. Microplastic pollution in the marine waters and sediments of Hong Kong. Mar. Pollut. Bull. 2017, 115, 20–28. [Google Scholar] [CrossRef]
- Su, L.; Xue, Y.; Li, L.; Yang, D.; Kolandhasamy, P.; Li, D.; Shi, H. Microplastics in Taihu Lake, China. Environ. Pollut. 2016, 216, 711–719. [Google Scholar] [CrossRef]
- Hidalgo-Ruz, V.; Gutow, L.; Thompson, R.C.; Thiel, M. Microplastics in the Marine Environment: A Review of the Methods Used for Identification and Quantification. Environ. Sci. Technol. 2012, 46, 3060–3075. [Google Scholar] [CrossRef]
- Wang, W.; Ndungu, A.W.; Li, Z.; Wang, J. Microplastics pollution in inland freshwaters of China: A case study in urban surface waters of Wuhan, China. Sci. Total Environ. 2017, 575, 1369–1374. [Google Scholar] [CrossRef]
- Silva, A.B.; Bastos, A.S.; Justino, C.I.L.; da Costa, J.P.; Duarte, A.C.; Rocha-Santos, T.A.P. Microplastics in the environment: Challenges in analytical chemistry-A review. Anal. Chim. Acta 2018, 1017, 1–19. [Google Scholar] [CrossRef]
- Zhang, K.; Xiong, X.; Hu, H.; Wu, C.; Bi, Y.; Wu, Y.; Zhou, B.; Lam, P.K.S.; Liu, J. Occurrence and Characteristics of Microplastic Pollution in Xiangxi Bay of Three Gorges Reservoir, China. Environ. Sci. Technol. 2017, 51, 3794–3801. [Google Scholar] [CrossRef] [PubMed]
- Huppertsberg, S.; Knepper, T.P. Instrumental analysis of microplastics—benefits and challenges. Anal. Bioanal. Chem. 2018, 410, 6343–6352. [Google Scholar] [CrossRef] [PubMed]
- Crawford, C.B.; Quinn, B. 10-Microplastic identification techniques. In Microplastic Pollutants; Crawford, C.B., Quinn, B., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 219–267. [Google Scholar]
- Fries, E.; Dekiff, J.H.; Willmeyer, J.; Nuelle, M.-T.; Ebert, M.; Remy, D. Identification of polymer types and additives in marine microplastic particles using pyrolysis-GC/MS and scanning electron microscopy. Environ. Sci. Process. Impacts 2013, 15, 1949–1956. [Google Scholar] [CrossRef] [PubMed]
- Shim, W.J.; Hong, S.H.; Eo, S.E. Identification methods in microplastic analysis: A review. Anal. Methods 2017, 9, 1384–1391. [Google Scholar] [CrossRef]
- Corcoran, P.L.; Biesinger, M.C.; Grifi, M. Plastics and beaches: A degrading relationship. Mar. Pollut. Bull. 2009, 58, 80–84. [Google Scholar] [CrossRef]
- Araujo, C.F.; Nolasco, M.M.; Ribeiro, A.M.P.; Ribeiro-Claro, P.J.A. Identification of microplastics using Raman spectroscopy: Latest developments and future prospects. Water Res. 2018, 142, 426–440. [Google Scholar] [CrossRef]
- Käppler, A.; Fischer, D.; Oberbeckmann, S.; Schernewski, G.; Labrenz, M.; Eichhorn, K.-J.; Voit, B. Analysis of environmental microplastics by vibrational microspectroscopy: FTIR, Raman or both? Anal. Bioanal. Chem. 2016, 408, 8377–8391. [Google Scholar] [CrossRef]
- Majewsky, M.; Bitter, H.; Eiche, E.; Horn, H. Determination of microplastic polyethylene (PE) and polypropylene (PP) in environmental samples using thermal analysis (TGA-DSC). Sci. Total Environ. 2016, 568, 507–511. [Google Scholar] [CrossRef]
- Lenz, R.; Enders, K.; Stedmon, C.A.; MacKenzie, D.M.A.; Nielsen, T.G. A critical assessment of visual identification of marine microplastic using Raman spectroscopy for analysis improvement. Mar. Pollut. Bull. 2015, 100, 82–91. [Google Scholar] [CrossRef]
- Greenspan, P.; Fowler, S.D. Spectrofluorometric studies of the lipid probe, nile red. J. Lipid Res. 1985, 26, 781–789. [Google Scholar] [CrossRef]
- Jee, A.-Y.; Park, S.; Kwon, H.; Lee, M. Excited state dynamics of Nile Red in polymers. Chem. Phys. Lett. 2009, 477, 112–115. [Google Scholar] [CrossRef]
- Greenspan, P.; Mayer, E.P.; Fowler, S.D. Nile red: A selective fluorescent stain for intracellular lipid droplets. J. Cell Biol. 1985, 100, 965–973. [Google Scholar] [CrossRef] [PubMed]
- Shim, W.J.; Song, Y.K.; Hong, S.H.; Jang, M. Identification and quantification of microplastics using Nile Red staining. Mar. Pollut. Bull. 2016, 113, 469–476. [Google Scholar] [CrossRef] [PubMed]
- Scircle, A.; Cizdziel, J.V. Detecting and Quantifying Microplastics in Bottled Water using Fluorescence Microscopy: A New Experiment for Instrumental Analysis and Environmental Chemistry Courses. J. Chem. Educ. 2020, 97, 234–238. [Google Scholar] [CrossRef]
- Dümichen, E.; Eisentraut, P.; Bannick, C.G.; Barthel, A.-K.; Senz, R.; Braun, U. Fast identification of microplastics in complex environmental samples by a thermal degradation method. Chemosphere 2017, 174, 572–584. [Google Scholar] [CrossRef]
- Duemichen, E.; Braun, U.; Senz, R.; Fabian, G.; Sturm, H. Assessment of a new method for the analysis of decomposition gases of polymers by a combining thermogravimetric solid-phase extraction and thermal desorption gas chromatography mass spectrometry. J. Chromatogr. A 2014, 1354, 117–128. [Google Scholar] [CrossRef]
- Filgueiras, A.V.; Gago, J.; Campillo, J.A.; León, V.M. Microplastic distribution in surface sediments along the Spanish Mediterranean continental shelf. Environ. Sci. Pollut. Res. 2019, 26, 21264–21273. [Google Scholar] [CrossRef]
- Alomar, C.; Estarellas, F.; Deudero, S. Microplastics in the Mediterranean Sea: Deposition in coastal shallow sediments, spatial variation and preferential grain size. Mar. Environ. Res. 2016, 115, 1–10. [Google Scholar] [CrossRef]
- Lots, F.A.E.; Behrens, P.; Vijver, M.G.; Horton, A.A.; Bosker, T. A large-scale investigation of microplastic contamination: Abundance and characteristics of microplastics in European beach sediment. Mar. Pollut. Bull. 2017, 123, 219–226. [Google Scholar] [CrossRef]
- Schmidt, N.; Thibault, D.; Galgani, F.; Paluselli, A.; Sempéré, R. Occurrence of microplastics in surface waters of the Gulf of Lion (NW Mediterranean Sea). Prog. Oceanogr. 2018, 163, 214–220. [Google Scholar] [CrossRef]
- Baini, M.; Fossi, M.C.; Galli, M.; Caliani, I.; Campani, T.; Finoia, M.G.; Panti, C. Abundance and characterization of microplastics in the coastal waters of Tuscany (Italy): The application of the MSFD monitoring protocol in the Mediterranean Sea. Mar. Pollut. Bull. 2018, 133, 543–552. [Google Scholar] [CrossRef] [PubMed]
- Cannas, S.; Fastelli, P.; Guerranti, C.; Renzi, M. Plastic litter in sediments from the coasts of south Tuscany (Tyrrhenian Sea). Mar. Pollut. Bull. 2017, 119, 372–375. [Google Scholar] [CrossRef] [PubMed]
- Magni, S.; Binelli, A.; Pittura, L.; Avio, C.G.; Della Torre, C.; Parenti, C.C.; Gorbi, S.; Regoli, F. The fate of microplastics in an Italian Wastewater Treatment Plant. Sci. Total Environ. 2019, 652, 602–610. [Google Scholar] [CrossRef] [PubMed]
- Suaria, G.; Avio, C.G.; Mineo, A.; Lattin, G.L.; Magaldi, M.G.; Belmonte, G.; Moore, C.J.; Regoli, F.; Aliani, S. The Mediterranean Plastic Soup: Synthetic polymers in Mediterranean surface waters. Sci. Rep. 2016, 6, 37551. [Google Scholar] [CrossRef]
- Suaria, G.; Aliani, S. Floating debris in the Mediterranean Sea. Mar. Pollut. Bull. 2014, 86, 494–504. [Google Scholar] [CrossRef]
- Korez, Š.; Gutow, L.; Saborowski, R. Microplastics at the strandlines of Slovenian beaches. Mar. Pollut. Bull. 2019, 145, 334–342. [Google Scholar] [CrossRef]
- Vianello, A.; Boldrin, A.; Guerriero, P.; Moschino, V.; Rella, R.; Sturaro, A.; Da Ros, L. Microplastic particles in sediments of Lagoon of Venice, Italy: First observations on occurrence, spatial patterns and identification. Estuar. Coast. Shelf Sci. 2013, 130, 54–61. [Google Scholar] [CrossRef]
- Blašković, A.; Fastelli, P.; Čižmek, H.; Guerranti, C.; Renzi, M. Plastic litter in sediments from the Croatian marine protected area of the natural park of Telaščica bay (Adriatic Sea). Mar. Pollut. Bull. 2017, 114, 583–586. [Google Scholar] [CrossRef]
- Maršić-Lučić, J.; Lušić, J.; Tutman, P.; Bojanić Varezić, D.; Šiljić, J.; Pribudić, J. Levels of trace metals on microplastic particles in beach sediments of the island of Vis, Adriatic Sea, Croatia. Mar. Pollut. Bull. 2018, 137, 231–236. [Google Scholar] [CrossRef]
- Fusco, M.; Dzajic-Valjevac, M.; Dzonlic, M.; Horvat, P.; Krzan, A.; Gajić, A. First Assessment of Microplastics Pollution in the Aquatorium of Bosnia and Herzegovina. In Proceedings of the International Workshop: Science Meets Policy for the Cleaner Adriatic, Sarajevo, Bosnia and Herzegovina, 16 April 2016; p. 17. [Google Scholar]
- Šilc, U.; Küzmič, F.; Caković, D.; Stešević, D. Beach litter along various sand dune habitats in the southern Adriatic (E Mediterranean). Mar. Pollut. Bull. 2018, 128, 353–360. [Google Scholar] [CrossRef]
- Kolitari, J.; Gjyli, L.; Carbonara, P. Preliminary results and impact of marine litter in Albanian adriatic area. J. Environ. Prot. Ecol. 2016, 17, 922–931. [Google Scholar]
- Gjyli, L.; Vlachogianni, T.; Kolitari, J.; Matta, G.; Metalla, O.; Gjyli, S. Marine litter on the Albanian coastline: Baseline information for improved management. Ocean Coast. Manag. 2020, 187, 105108. [Google Scholar] [CrossRef]
- Kaberi, H.; Zeri, C.; Mousdis, G.; Papadopoulos, A.; Streftaris, N. Microplastics along the shoreline of a Greek island (Kea isl., Aegean Sea): Types and densities in relation to beach orientation, characteristics and proximity to sources. In Proceedings of the 4th International Conference on Environmental Management, Engineering, Planning and Economics (CEMEPE) and SECOTOXAt, Mykonos Island, Greece, 24–28 June 2013; pp. 197–202. [Google Scholar]
- Karapanagioti, H.K.; Klontza, I. Testing phenanthrene distribution properties of virgin plastic pellets and plastic eroded pellets found on Lesvos island beaches (Greece). Mar. Environ. Res. 2008, 65, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Karapanagioti, H.K.; Endo, S.; Ogata, Y.; Takada, H. Diffuse pollution by persistent organic pollutants as measured in plastic pellets sampled from various beaches in Greece. Mar. Pollut. Bull. 2011, 62, 312–317. [Google Scholar] [CrossRef] [PubMed]
- De Ruijter, V.N.; Milou, A.; Costa, V. Assessment of microplastics distribution and stratification in the shallow marine sediments of Samos island, Eastern Mediterranean sea, Greece. Mediterr. Mar. Sci. 2019, 20, 736–744. [Google Scholar] [CrossRef]
- Akarsu, C.; Kumbur, H.; Gökdağ, K.; Kıdeyş, A.E.; Sanchez-Vidal, A. Microplastics composition and load from three wastewater treatment plants discharging into Mersin Bay, north eastern Mediterranean Sea. Mar. Pollut. Bull. 2020, 150, 110776. [Google Scholar] [CrossRef]
- Tunçer, S.; Artüz, O.B.; Demirkol, M.; Artüz, M.L. First report of occurrence, distribution, and composition of microplastics in surface waters of the Sea of Marmara, Turkey. Mar. Pollut. Bull. 2018, 135, 283–289. [Google Scholar] [CrossRef]
- Yabanlı, M.; Yozukmaz, A.; Şener, İ.; Ölmez, Ö.T. Microplastic pollution at the intersection of the Aegean and Mediterranean Seas: A study of the Datça Peninsula (Turkey). Mar. Pollut. Bull. 2019, 145, 47–55. [Google Scholar] [CrossRef]
- Güven, O.; Gökdağ, K.; Jovanović, B.; Kıdeyş, A.E. Microplastic litter composition of the Turkish territorial waters of the Mediterranean Sea, and its occurrence in the gastrointestinal tract of fish. Environ. Pollut. 2017, 223, 286–294. [Google Scholar] [CrossRef]
- Shiber, J.G. Plastic pellets on the coast of Lebanon. Mar. Pollut. Bull. 1979, 10, 28–30. [Google Scholar] [CrossRef]
- Kazour, M.; Jemaa, S.; Issa, C.; Khalaf, G.; Amara, R. Microplastics pollution along the Lebanese coast (Eastern Mediterranean Basin): Occurrence in surface water, sediments and biota samples. Sci. Total Environ. 2019, 696, 133933. [Google Scholar] [CrossRef] [PubMed]
- Golik, A.; Gertner, Y. Litter on the israeli coastline. Mar. Environ. Res. 1992, 33, 1–15. [Google Scholar] [CrossRef]
- van der Hal, N.; Ariel, A.; Angel, D.L. Exceptionally high abundances of microplastics in the oligotrophic Israeli Mediterranean coastal waters. Mar. Pollut. Bull. 2017, 116, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Khan, F.R.; Shashoua, Y.; Crawford, A.; Drury, A.; Sheppard, K.; Stewart, K.; Sculthorp, T. The Plastic Nile’: First Evidence of Microplastic Contamination in Fish from the Nile River (Cairo, Egypt). Toxics 2020, 8, 22. [Google Scholar] [CrossRef] [PubMed]
- Shabaka, S.H.; Ghobashy, M.; Marey, R.S. Identification of marine microplastics in Eastern Harbor, Mediterranean Coast of Egypt, using differential scanning calorimetry. Mar. Pollut. Bull. 2019, 142, 494–503. [Google Scholar] [CrossRef]
- Abidli, S.; Toumi, H.; Lahbib, Y.; Trigui El Menif, N. The First Evaluation of Microplastics in Sediments from the Complex Lagoon-Channel of Bizerte (Northern Tunisia). Water Air Soil Pollut. 2017, 228, 262. [Google Scholar] [CrossRef]
- Abidli, S.; Antunes, J.C.; Ferreira, J.L.; Lahbib, Y.; Sobral, P.; Trigui El Menif, N. Microplastics in sediments from the littoral zone of the north Tunisian coast (Mediterranean Sea). Estuar. Coast. Shelf Sci. 2018, 205, 1–9. [Google Scholar] [CrossRef]
- Alshawafi, A.; Analla, M.; Alwashali, E.; Ahechti, M.; Aksissou, M. Impacts of Marine Waste, Ingestion of Microplastic in the Fish, Impact on Fishing Yield, M’diq, Morocco. Int. J. Mar. Biol. Res. 2018, 3, 1–14. [Google Scholar] [CrossRef]
- Turner, A.; Holmes, L. Occurrence, distribution and characteristics of beached plastic production pellets on the island of Malta (central Mediterranean). Mar. Pollut. Bull. 2011, 62, 377–381. [Google Scholar] [CrossRef]
- Deidun, A.; Axiak, V.; Bezzina, M.; Lomax, C.; Edwards, K. First Full Investigation of Microplastics on Maltese Sandy Beaches. In Proceedings of the 13th International MEDCOAST Congress on Coastal and Marine Sciences, Engineering, Management and Conservation, MEDCOAST 2017, Mellieha, Malta, 31 October–1 November 2017; pp. 664–674. [Google Scholar]
- Duncan, E.M.; Arrowsmith, J.; Bain, C.; Broderick, A.C.; Lee, J.; Metcalfe, K.; Pikesley, S.K.; Snape, R.T.E.; van Sebille, E.; Godley, B.J. The true depth of the Mediterranean plastic problem: Extreme microplastic pollution on marine turtle nesting beaches in Cyprus. Mar. Pollut. Bull. 2018, 136, 334–340. [Google Scholar] [CrossRef]
- Mitrano, D.M.; Wohlleben, W. Microplastic regulation should be more precise to incentivize both innovation and environmental safety. Nat. Commun. 2020, 11, 5324. [Google Scholar] [CrossRef] [PubMed]
- Llorca, M.; Álvarez-Muñoz, D.; Ábalos, M.; Rodríguez-Mozaz, S.; Santos, L.H.M.L.M.; León, V.M.; Campillo, J.A.; Martínez-Gómez, C.; Abad, E.; Farré, M. Microplastics in Mediterranean coastal area: Toxicity and impact for the environment and human health. Trends Environ. Anal. Chem. 2020, 27, e00090. [Google Scholar] [CrossRef]
- UN Environment. Available online: https://www.unenvironment.org/unepmap (accessed on 11 January 2021).
- Protocol on Integrated Coastal Zone Management in the Mediterranean. Available online: https://eur-lex.europa.eu/eli/prot/2009/89/oj (accessed on 11 January 2021).
- Our Oceans, Seas and Coasts: The Marine Strategy Framework Directive. Available online: https://ec.europa.eu/environment/marine/eu-coast-and-marine-policy/marine-strategy-framework-directive/index_en.htm (accessed on 11 January 2021).
- European Strategy for Plastics. Available online: https://ec.europa.eu/environment/waste/plastic_waste.htm (accessed on 11 January 2021).
- ECHA. Available online: https://echa.europa.eu/hot-topics/microplastics (accessed on 10 January 2021).
- Briassoulis, D.; Pikasi, A.; Hiskakis, M. Recirculation potential of post-consumer /industrial bio-based plastics through mechanical recycling-Techno-economic sustainability criteria and indicators. Polym. Degrad. Stab. 2020, 109217. [Google Scholar] [CrossRef]
- Helmer Pedersen, T.; Conti, F. Improving the circular economy via hydrothermal processing of high-density waste plastics. Waste Manag. 2017, 68, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Millette, S.; Williams, E.; Hull, C.E. Materials flow analysis in support of circular economy development: Plastics in Trinidad and Tobago. Resour. Conserv. Recycl. 2019, 150, 104436. [Google Scholar] [CrossRef]
- Communication from the Commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions: A European Strategy for Plastics in a Circular Economy. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A52018DC0028 (accessed on 11 January 2021).
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).