Presence of Polyphenols Complex Aromatic “Lignin” in Sargassum spp. from Mexican Caribbean
Abstract
1. Introduction
2. Materials and Methods
2.1. Biomass Collection and Preparation
2.2. Physical-Chemical Characterization
2.2.1. Chemical Composition
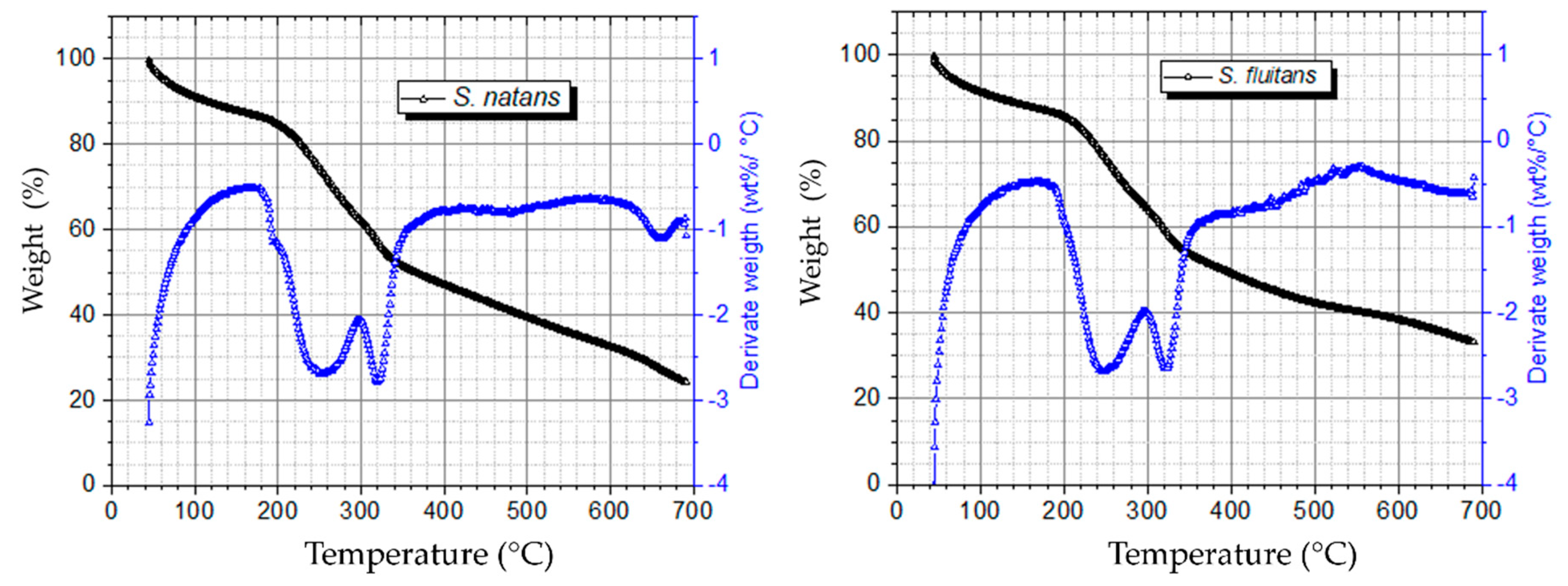
2.2.2. Thermogravimetric Analysis (TGA)
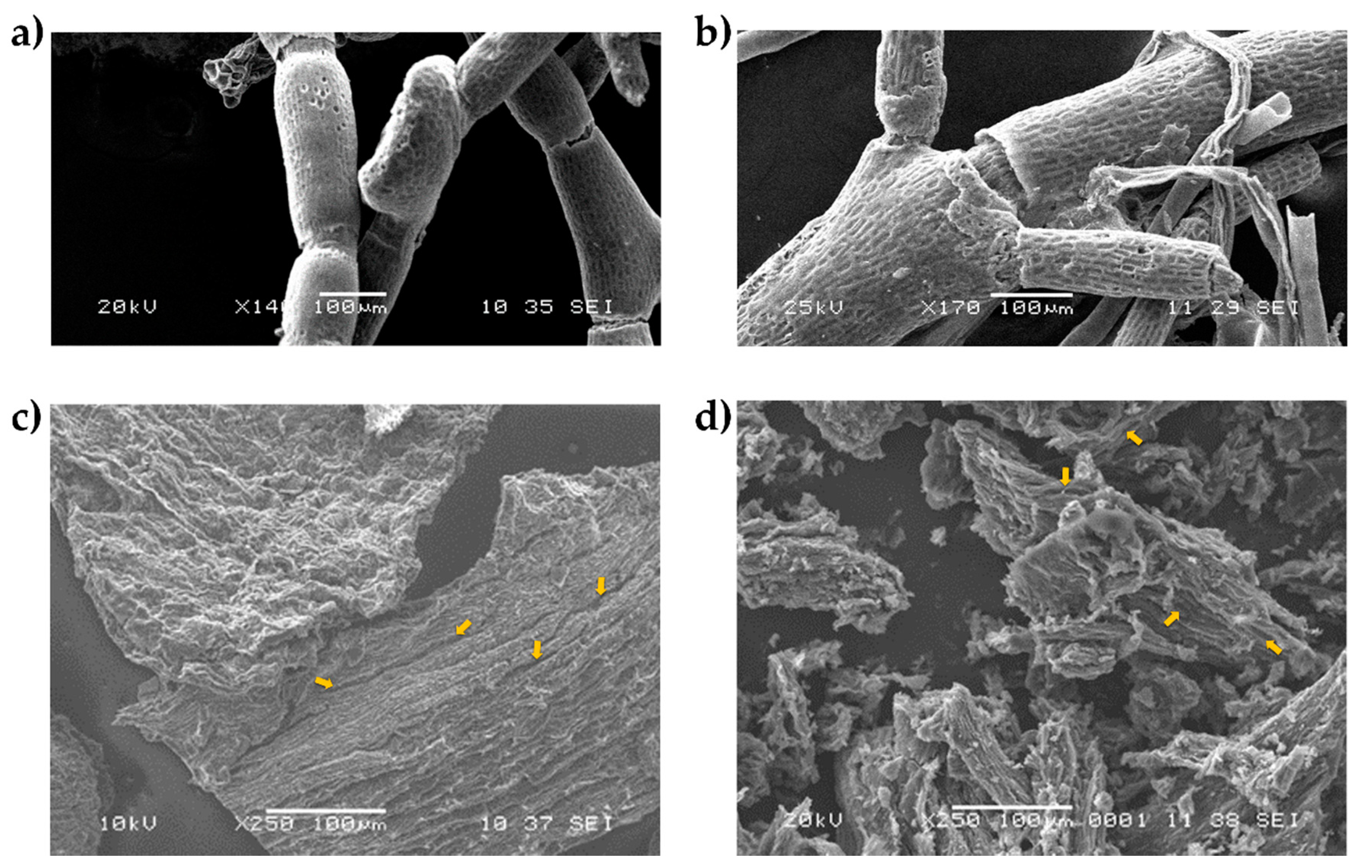
2.2.3. Scanning Electron Microscopy (SEM)
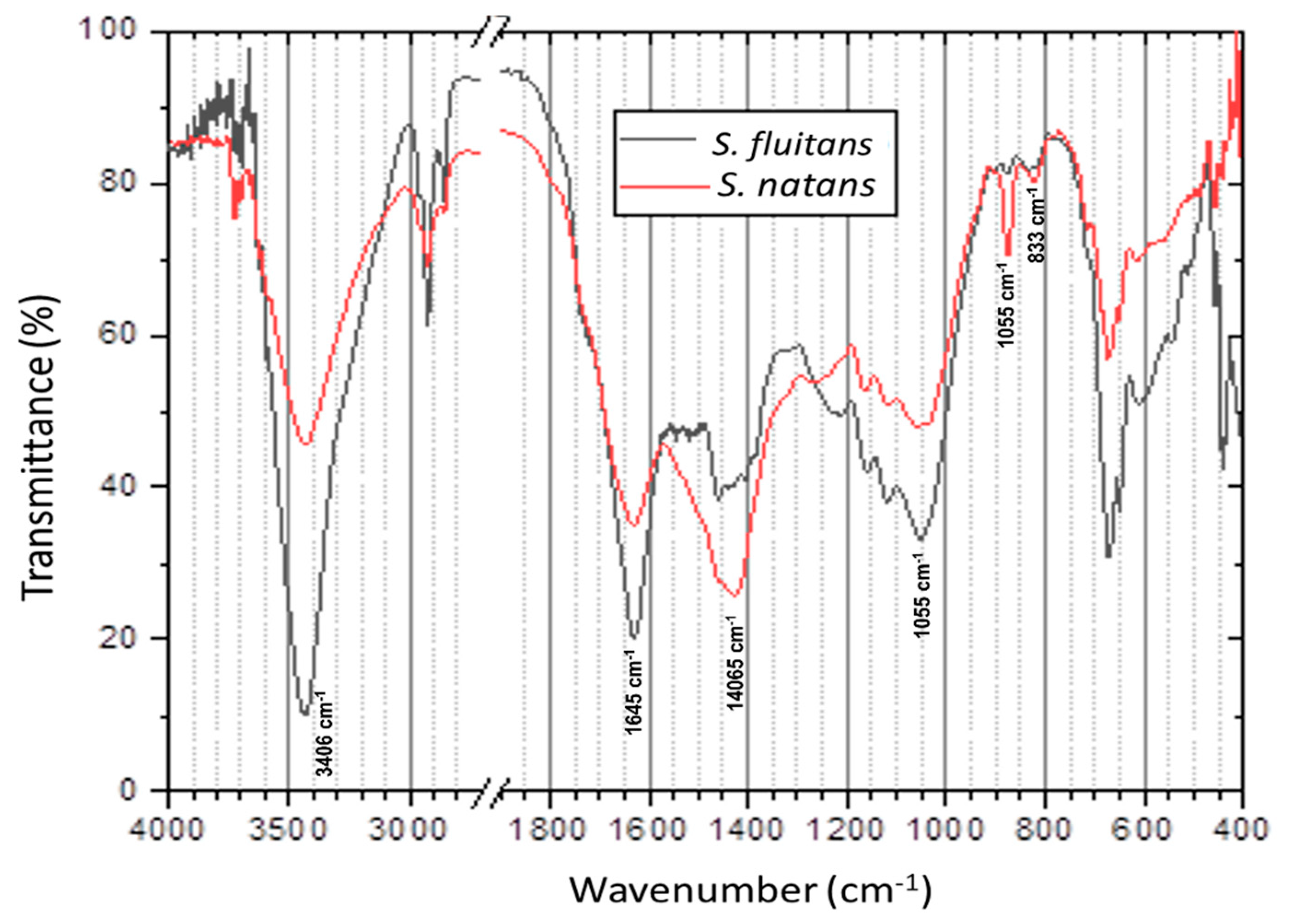
2.2.4. Fourier Transforms Infrared (FT-IR) Analysis
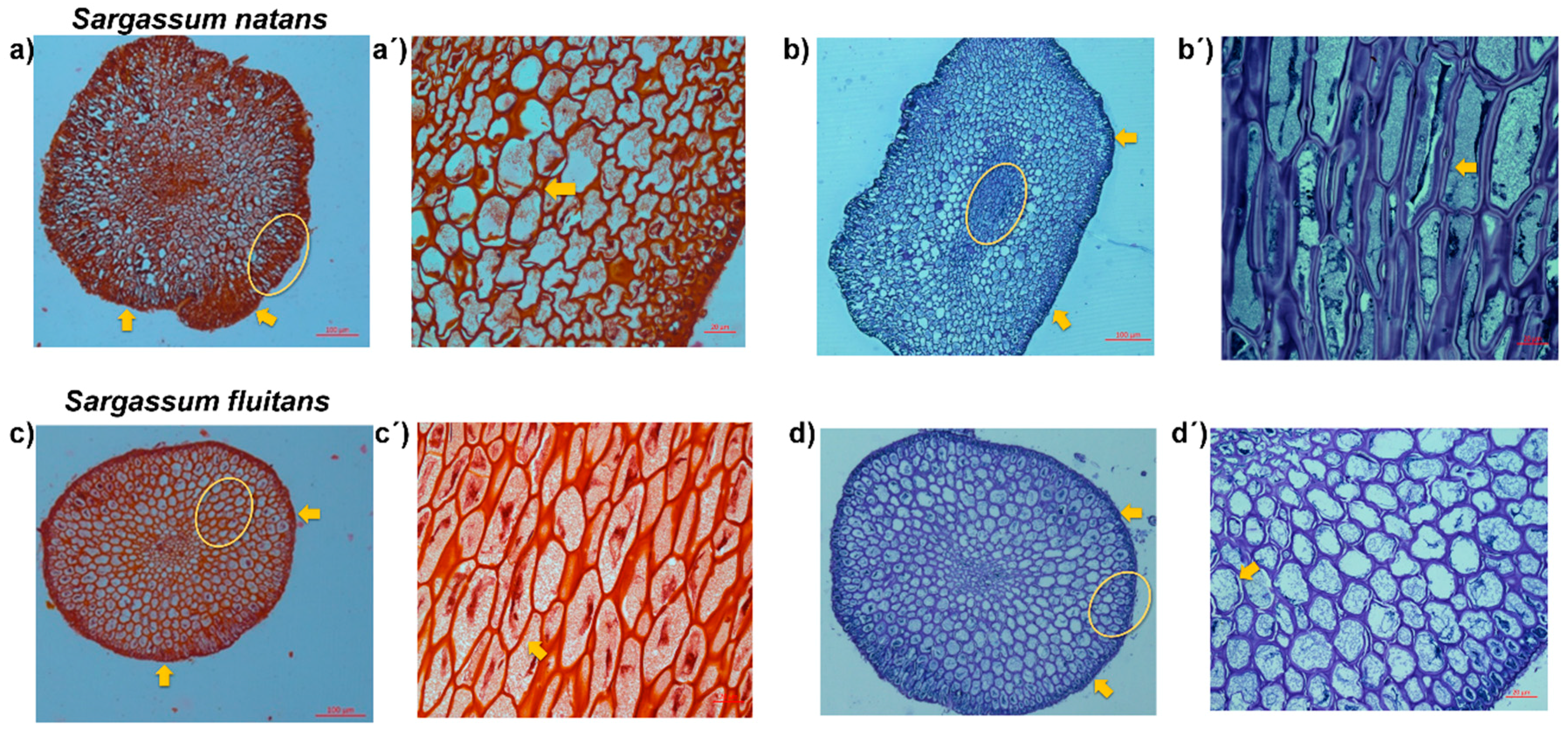
2.2.5. Histological Analysis
2.2.6. Elemental Analysis
3. Results and Discussion
3.1. Biomass Composition and Characterization
3.2. FT-IR Analysis
3.3. Histological Analysis
3.4. Elemental Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Johns, E.M.; Lumpkin, R.; Putman, N.F.; Smith, R.H.; Muller-Karger, F.E.; Rueda, D.; Hu, C.; Wang, M.; Brooks, M.T.; Gramer, L.J. The establishment of a pelagic Sargassum population in the tropical Atlantic: Biological consequences of a basin-scale long distance dispersal event. Prog. Oceanogr. 2020, 182, 102269. [Google Scholar] [CrossRef]
- Johns, E.M.; Muhling, B.A.; Perez, R.C.; Müller-Karger, F.E.; Melo, N.; Smith, R.H.; Lamkin, J.T.; Gerard, T.L.; Malca, E. Amazon River water in the northeastern Caribbean Sea and its effect on larval reef fish assemblages during April 2009. Fish. Oceanogr. 2014, 23, 472–494. [Google Scholar] [CrossRef]
- Oviatt, C.A.; Huizenga, K.; Rogers, C.S.; Miller, W.J. What nutrient sources support anomalous growth and the recent Sargassum mass stranding on Caribbean beaches? A review. Mar. Pollut. Bull. 2019, 145, 517–525. [Google Scholar] [CrossRef]
- Wang, M.; Hu, C.; Barnes, B.B.; Mitchum, G.; Lapointe, B.; Montoya, J.P. The great Atlantic Sargassum belt. Science 2019, 365, 83–87. [Google Scholar] [CrossRef]
- van Tussenbroek, B.I.; Arana, H.A.H.; Rodríguez-Martínez, R.E.; Espinoza-Avalos, J.; Canizales-Flores, H.M.; González-Godoy, C.E.; Barba-Santos, M.G.; Vega-Zepeda, A.; Collado-Vides, L. Severe impacts of brown tides caused by Sargassum spp. on near-shore Caribbean seagrass communities. Mar. Pollut. Bull. 2017, 122, 272–281. [Google Scholar] [CrossRef]
- García-Sánchez, M.; Graham, C.; Vera, E.; Escalante-Mancera, E.; Álvarez-Filip, L.; van Tussenbroek, B.I. Temporal changes in the composition and biomass of beached pelagic Sargassum species in the Mexican Caribbean. Aquat. Bot. 2020, 167, 103275. [Google Scholar] [CrossRef]
- Milledge, J.J.; Harvey, P.J. Golden Tides: Problem or golden opportunity? The valorisation of Sargassum from beach inundations. J. Mar. Sci. Eng. 2016, 4, 60. [Google Scholar] [CrossRef]
- Thompson, T.M.; Young, B.R.; Baroutian, S. Pelagic Sargassum for energy and fertiliser production in the Caribbean: A case study on Barbados. Renew. Sustain. Energy Rev. 2020, 118, 109564. [Google Scholar] [CrossRef]
- Putman, N.F.; Goni, G.J.; Gramer, L.J.; Hu, C.; Johns, E.M.; Trinanes, J.; Wang, M. Simulating transport pathways of pelagic Sargassum from the Equatorial Atlantic into the Caribbean Sea. Prog. Oceanogr. 2018, 165, 205–214. [Google Scholar] [CrossRef]
- Brown, M.T. UV-B radiation and the green tide-forming macroalga Ulva. In Aquatic Ecosystems in a Changing Climate; CRC Press: Boca Raton, FL, USA, 2018; ISBN 0429436130. [Google Scholar]
- Rodríguez-Martínez, R.E.; Roy, P.D.; Torrescano-Valle, N.; Cabanillas-Terán, N.; Carrillo-Domínguez, S.; Collado-Vides, L.; García-Sánchez, M.; van Tussenbroek, B.I. Element concentrations in pelagic Sargassum along the Mexican Caribbean coast in 2018–2019. PeerJ 2020, 8, e8667. [Google Scholar] [CrossRef]
- Holzinger, A.; Pichrtová, M. Abiotic stress tolerance of charophyte green algae: New challenges for omics techniques. Front. Plant Sci. 2016, 7, 678. [Google Scholar] [CrossRef]
- Martone, P.T.; Estevez, J.M.; Lu, F.; Ruel, K.; Denny, M.W.; Somerville, C.; Ralph, J. Discovery of lignin in seaweed reveals convergent evolution of cell-wall architecture. Curr. Biol. 2009, 19, 169–175. [Google Scholar] [CrossRef]
- Moura, J.C.M.S.; Bonine, C.A.V.; de Oliveira Fernandes Viana, J.; Dornelas, M.C.; Mazzafera, P. Abiotic and biotic stresses and changes in the lignin content and composition in plants. J. Integr. Plant Biol. 2010, 52, 360–376. [Google Scholar] [CrossRef]
- Nayaka, S.; Toppo, K.; Verma, S. Adaptation in Algae to Environmental Stress and Ecological Conditions. In Plant Adaptation Strategies in Changing Environment; Springer: Berlin/Heidelberg, Germany, 2017; pp. 103–115. [Google Scholar]
- Sun, D.; Call, D.; Wang, A.; Cheng, S.; Logan, B.E. Geobacter sp. SD-1 with enhanced electrochemical activity in high-salt concentration solutions. Environ. Microbiol. Rep. 2014, 6, 723–729. [Google Scholar] [CrossRef]
- Liu, L.; Lin, L. Effect of Heat Stress on Sargassum fusiforme Leaf Metabolome. J. Plant Biol. 2020, 63, 229–241. [Google Scholar] [CrossRef]
- Rosado-Espinosa, L.A.; Freile-Pelegrín, Y.; Hernández-Nuñez, E.; Robledo, D. A comparative study of Sargassum species from the Yucatan peninsula coast: Morphological and chemical characterisation. Phycologia 2020, 59, 261–271. [Google Scholar] [CrossRef]
- Davis, D.; Simister, R.; Campbell, S.; Marston, M.; Bose, S.; McQueen-Mason, S.J.; Gomez, L.D.; Gallimore, W.A.; Tonon, T. Biomass composition of the golden tide pelagic seaweeds Sargassum fluitans and S. natans (morphotypes I and VIII) to inform valorisation pathways. Sci. Total Environ. 2020, 143134. Available online: https://www.sciencedirect.com/science/article/abs/pii/S004896972036664X (accessed on 9 November 2020).
- Zagoskina, N.V.; Dubravina, G.A.; Alyavina, A.K.; Goncharuk, E.A. Effect of ultraviolet (UV-B) radiation on the formation and localization of phenolic compounds in tea plant callus cultures. Russ. J. Plant Physiol. 2003, 50, 270–275. [Google Scholar] [CrossRef]
- Mannino, A.M.; Micheli, C. Ecological Function of Phenolic Compounds from Mediterranean Fucoid Algae and Seagrasses: An Overview on the Genus Cystoseira sensu lato and Posidonia oceanica (L.) Delile. J. Mar. Sci. Eng. 2020, 8, 19. [Google Scholar] [CrossRef]
- Pliego-Cortés, H.; Bedoux, G.; Boulho, R.; Taupin, L.; Freile-Pelegrín, Y.; Bourgougnon, N.; Robledo, D. Stress tolerance and photoadaptation to solar radiation in Rhodymenia pseudopalmata (Rhodophyta) through mycosporine-like amino acids, phenolic compounds, and pigments in an Integrated Multi-Trophic Aquaculture system. Algal Res. 2019, 41, 101542. [Google Scholar] [CrossRef]
- De Micco, V.; Aronne, G. Combined histochemistry and autofluorescence for identifying lignin distribution in cell walls. Biotech. Histochem. 2007, 82, 209–216. [Google Scholar] [CrossRef]
- Estevez, J.M.; Fernández, P.V.; Kasulin, L.; Dupree, P.; Ciancia, M. Chemical and in situ characterization of macromolecular components of the cell walls from the green seaweed Codium fragile. Glycobiology 2009, 19, 212–228. [Google Scholar] [CrossRef]
- Ferreira, B.G.; Falcioni, R.; Guedes, L.M.; Avritzer, S.C.; Antunes, W.C.; Souza, L.A.; Isaias, R.M.S. Preventing false negatives for histochemical detection of phenolics and lignins in PEG-embedded plant tissues. J. Histochem. Cytochem. 2017, 65, 105–116. [Google Scholar] [CrossRef]
- Camacho, O.; Mattio, L.; Draisma, S.; Fredericq, S.; Diaz-Pulido, G. Morphological and molecular assessment of Sargassum (Fucales, Phaeophyceae). Syst. Biodivers. 2015, 13, 105–130. [Google Scholar] [CrossRef]
- TAPPI. 222 om-02: Acid-insoluble lignin in wood and pulp. In 2002–2003 TAPPI Test Methods; TAPPI: Atlanta, GA, USA, 2002. [Google Scholar]
- TAPPI. Solvent Extractives of Wood and Pulp (Proposed Revision of T 204 cm-97); TAPPI: Atlanta, GA, USA, 2007. [Google Scholar]
- TAPPI. Water Solubility of Wood and Pulp; TAPPI: Atlanta, GA, USA, 1999. [Google Scholar]
- Catzín-Yupit, C.N.; Ramírez-Morillo, I.M.; Pool, F.A.B.; Loyola-Vargas, V.M. Ontogenic development and structure of the embryo, seed, and fruit of Jatropha curcas L.(Euphorbiaceae). South Afr. J. Bot. 2014, 93, 1–8. [Google Scholar] [CrossRef][Green Version]
- Sanchez-Teyer, L.F.; Quiroz-Figueroa, F.; Loyola-Vargas, V.; Infante, D. Culture-induced variation in plants of Coffea arabica cv. Caturra rojo, regenerated by direct and indirect somatic embryogenesis. Mol. Biotechnol. 2003, 23, 107–115. [Google Scholar] [CrossRef]
- Ruzin, S.E. Plant Microtechnique and Microscopy; Oxford University Press: New York, NY, USA, 1999; Volume 198. [Google Scholar]
- Fernández, F.; Boluda, C.J.; Olivera, J.; Gómez, L.A.G.B.; Gómez, E.E.A.M. Prospective elemental analysis of algal biomass accumulated at the Dominican Republic Shores during 2015. Cent. Azucar 2017, 44, 11–22. [Google Scholar]
- Ardalan, Y.; Jazini, M.; Karimi, K. Sargassum angustifolium brown macroalga as a high potential substrate for alginate and ethanol production with minimal nutrient requirement. Algal Res. 2018, 36, 29–36. [Google Scholar] [CrossRef]
- Yazdani, P.; Zamani, A.; Karimi, K.; Taherzadeh, M.J. Characterization of Nizimuddinia zanardini macroalgae biomass composition and its potential for biofuel production. Bioresour. Technol. 2015, 176, 196–202. [Google Scholar] [CrossRef]
- López-Sosa, L.B.; Alvarado-Flores, J.J.; Corral-Huacuz, J.C.; Aguilera-Mandujano, A.; Rodríguez-Martínez, R.E.; Guevara-Martínez, S.J.; Alcaraz-Vera, J.V.; Rutiaga-Quiñones, J.G.; Zárate-Medina, J.; Ávalos-Rodríguez, M.L.; et al. A Prospective Study of the Exploitation of Pelagic Sargassum spp. as a Solid Biofuel Energy Source. Appl. Sci 2020, 10, 8706. [Google Scholar] [CrossRef]
- Ali, I.; Bahadar, A. Red Sea seaweed (Sargassum spp.) pyrolysis and its devolatilization kinetics. Algal Res. 2017, 21, 89–97. [Google Scholar] [CrossRef]
- Burhenne, L.; Messmer, J.; Aicher, T.; Laborie, M.-P. The effect of the biomass components lignin, cellulose and hemicellulose on TGA and fixed bed pyrolysis. J. Anal. Appl. Pyrolysis 2013, 101, 177–184. [Google Scholar] [CrossRef]
- Zhou, H.; Long, Y.; Meng, A.; Li, Q.; Zhang, Y. The pyrolysis simulation of five biomass species by hemi-cellulose, cellulose and lignin based on thermogravimetric curves. Thermochim. Acta 2013, 566, 36–43. [Google Scholar] [CrossRef]
- Abraham, E.; Deepa, B.; Pothan, L.A.; Jacob, M.; Thomas, S.; Cvelbar, U.; Anandjiwala, R. Extraction of nanocellulose fibrils from lignocellulosic fibres: A novel approach. Carbohydr. Polym. 2011, 86, 1468–1475. [Google Scholar] [CrossRef]
- Escobar, B.; Pérez-Salcedo, K.Y.; Alonso-Lemus, I.L.; Pacheco, D.; Barbosa, R. N-doped porous carbon from Sargassum spp. as metal-free electrocatalysts for oxygen reduction reaction in alkaline media. Int. J. Hydrog. Energy 2017, 42, 30274–30283. [Google Scholar] [CrossRef]
- El Moustaqim, M.; El Kaihal, A.; El Marouani, M.; Men-La-Yakhaf, S.; Taibi, M.; Sebbahi, S.; El Hajjaji, S.; Kifani-Sahban, F. Thermal and thermomechanical analyses of lignin. Sustain. Chem. Pharm. 2018, 9, 63–68. [Google Scholar] [CrossRef]
- Silva, T.A.L.; Zamora, H.D.Z.; Varão, L.H.R. Effect of steam explosion pretreatment catalysed by organic acid and alkali on chemical and structural properties and enzymatic hydrolysis of sugarcane bagasse. Waste Biomass Valoriz. 2018, 9, 2191–2201. [Google Scholar] [CrossRef]
- Tapia-Tussell, R.; Avila-Arias, J.; Maldonado, J.D.; Valero, D.; Olguin-Maciel, E.; Pérez-Brito, D.; Alzate-Gaviria, L. Biological pretreatment of mexican caribbean macroalgae consortiums using Bm-2 strain (Trametes hirsuta) and its enzymatic broth to improve biomethane potential. Energies 2018, 11, 494. [Google Scholar] [CrossRef]
- Mitra, P.P.; Loqué, D. Histochemical staining of Arabidopsis thaliana secondary cell wall elements. JoVe (J. Vis. Exp.) 2014, 87, e51381. [Google Scholar]
- Herrera-Ubaldo, H.; de Folter, S. Exploring cell wall composition and modifications during the development of the gynoecium medial domain in Arabidopsis. Front. Plant Sci. 2018, 9, 454. [Google Scholar] [CrossRef] [PubMed]
- Thompson, T.M.; Young, B.R.; Baroutian, S. Efficiency of hydrothermal pretreatment on the anaerobic digestion of pelagic Sargassum for biogas and fertiliser recovery. Fuel 2020, 279, 118527. [Google Scholar] [CrossRef]
| Samples | Humidity | Total Extractives * | Lignin-Like Materials * | Holocellulose * |
|---|---|---|---|---|
| S. natans | 11.10 ± 0.39 | 17.21 ± 0.24 | 29.50 ± 2.35 | 45.39 ± 0.12 |
| S. fluitans | 12.10 ± 0.73 | 18.07 ± 0.16 | 25.40 ± 1.30 | 34.43 ± 0.01 |
| Species | Al | As | Ba | B | Cu | Ni | Zn | Pb | Cd | |
|---|---|---|---|---|---|---|---|---|---|---|
| Cell tissue | S. natans | 61.88 ± 1.70 | 115.66 ± 2.60 | 16.70 ± 0.10 | 228.83 ± 3.97 | 4.51 ± 0.05 | 2.50 ± 0.13 | 80.54 ± 0.53 | ˂LD | 0.47 ± 0.02 |
| S. fluitans | 33.81 ± 1.41 | 76.49 ± 1.25 | 13.73 ± 0.21 | 204.36 ± 2.47 | 3.83 ± 0.09 | ˂LD | 30.80 ± 0.48 | ˂LD | 0.44 ± 0.02 | |
| Leachate | S. natans and S. fluitans | 0.25 ± 0.02 | ˂LD | ˂LD | 3.74 ± 0.17 | ˂LD | ˂LD | ˂LD | ˂LD | ˂LD |
| Limit of Detection(LD) in mg/L | 0.0026 | 0.0070 | 0.0016 | 0.0011 | 0.0065 | 0.0027 | 0.0012 | 0.0058 | 0.0003 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alzate-Gaviria, L.; Domínguez-Maldonado, J.; Chablé-Villacís, R.; Olguin-Maciel, E.; Leal-Bautista, R.M.; Canché-Escamilla, G.; Caballero-Vázquez, A.; Hernández-Zepeda, C.; Barredo-Pool, F.A.; Tapia-Tussell, R. Presence of Polyphenols Complex Aromatic “Lignin” in Sargassum spp. from Mexican Caribbean. J. Mar. Sci. Eng. 2021, 9, 6. https://doi.org/10.3390/jmse9010006
Alzate-Gaviria L, Domínguez-Maldonado J, Chablé-Villacís R, Olguin-Maciel E, Leal-Bautista RM, Canché-Escamilla G, Caballero-Vázquez A, Hernández-Zepeda C, Barredo-Pool FA, Tapia-Tussell R. Presence of Polyphenols Complex Aromatic “Lignin” in Sargassum spp. from Mexican Caribbean. Journal of Marine Science and Engineering. 2021; 9(1):6. https://doi.org/10.3390/jmse9010006
Chicago/Turabian StyleAlzate-Gaviria, Liliana, Jorge Domínguez-Maldonado, Rubí Chablé-Villacís, Edgar Olguin-Maciel, Rosa María Leal-Bautista, Gonzalo Canché-Escamilla, Adán Caballero-Vázquez, Cecilia Hernández-Zepeda, Felipe Alonso Barredo-Pool, and Raúl Tapia-Tussell. 2021. "Presence of Polyphenols Complex Aromatic “Lignin” in Sargassum spp. from Mexican Caribbean" Journal of Marine Science and Engineering 9, no. 1: 6. https://doi.org/10.3390/jmse9010006
APA StyleAlzate-Gaviria, L., Domínguez-Maldonado, J., Chablé-Villacís, R., Olguin-Maciel, E., Leal-Bautista, R. M., Canché-Escamilla, G., Caballero-Vázquez, A., Hernández-Zepeda, C., Barredo-Pool, F. A., & Tapia-Tussell, R. (2021). Presence of Polyphenols Complex Aromatic “Lignin” in Sargassum spp. from Mexican Caribbean. Journal of Marine Science and Engineering, 9(1), 6. https://doi.org/10.3390/jmse9010006






