Multimarker Approach to Evaluate the Exposure to Electromagnetic Fields at 27 GHz on Danio rerio Larvae
Abstract
1. Introduction
2. Materials and Methods
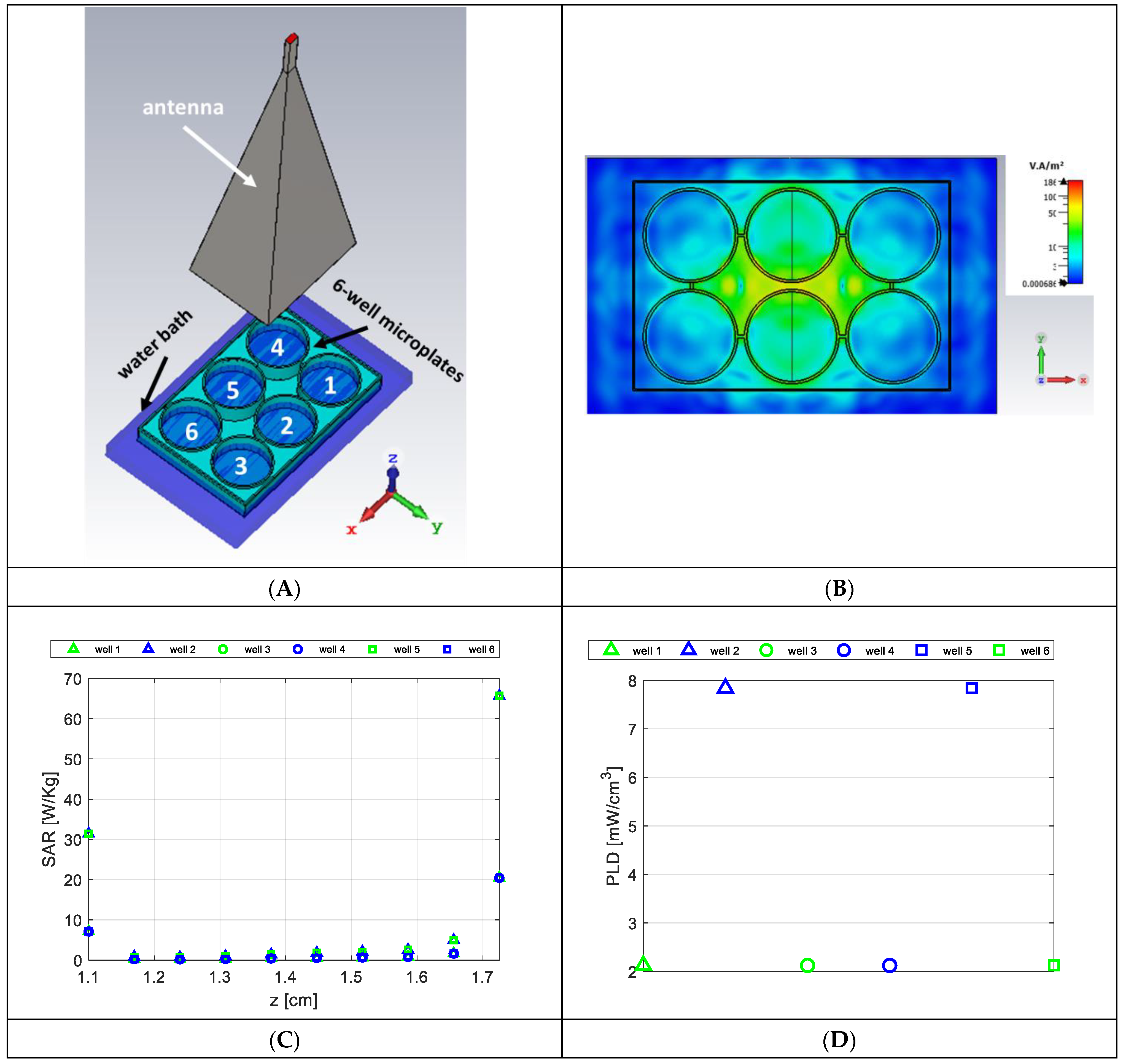
2.1. Exposure Setup Description for Numerical Dosimetry
- -
- electric field density power [W/m2];
- -
- local specific absorption rate, defined as [W/Kg]; and
- -
- power loss density (PLD) into exposed aqueous samples [W/m3].
2.2. Experimental Procedure
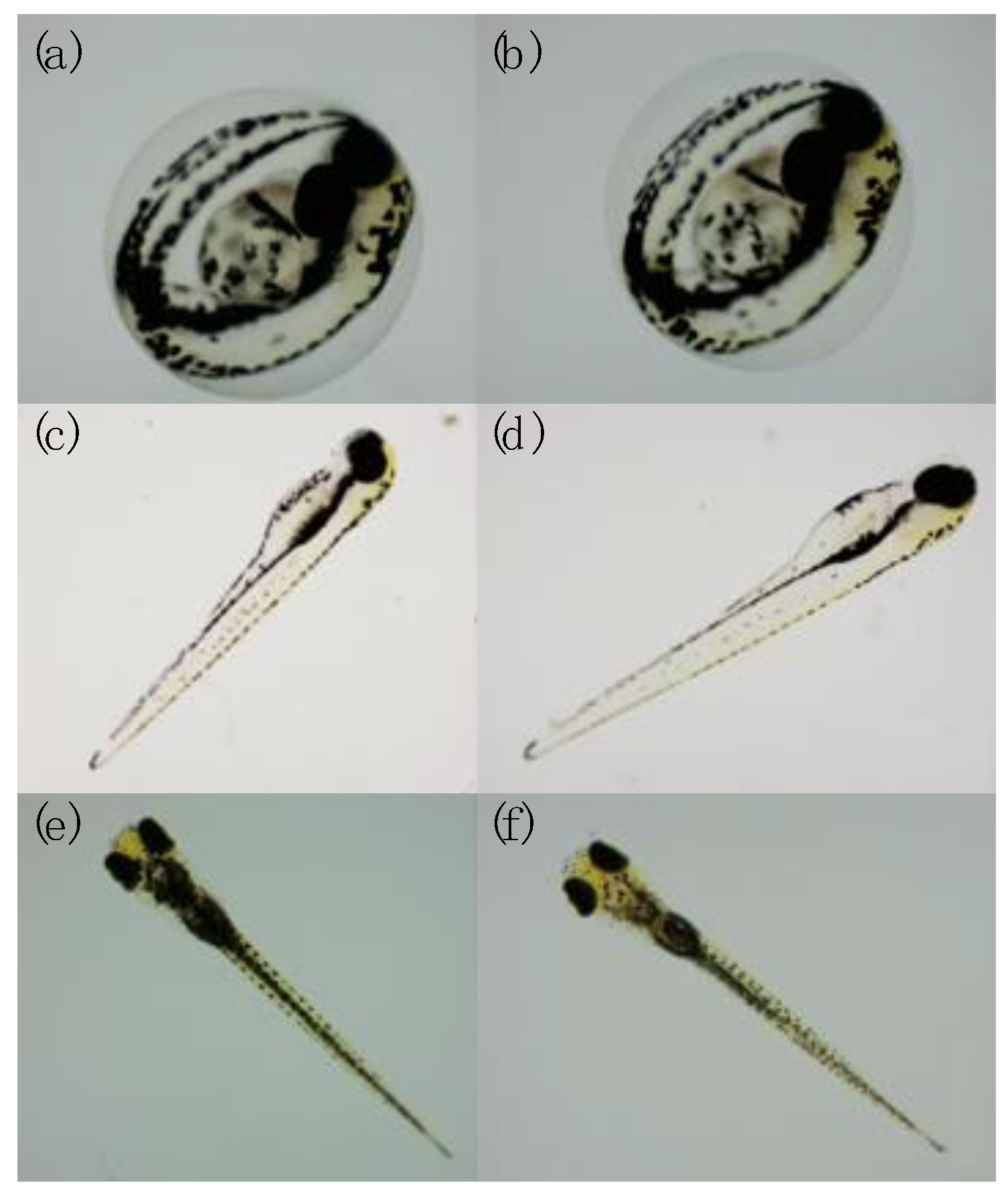
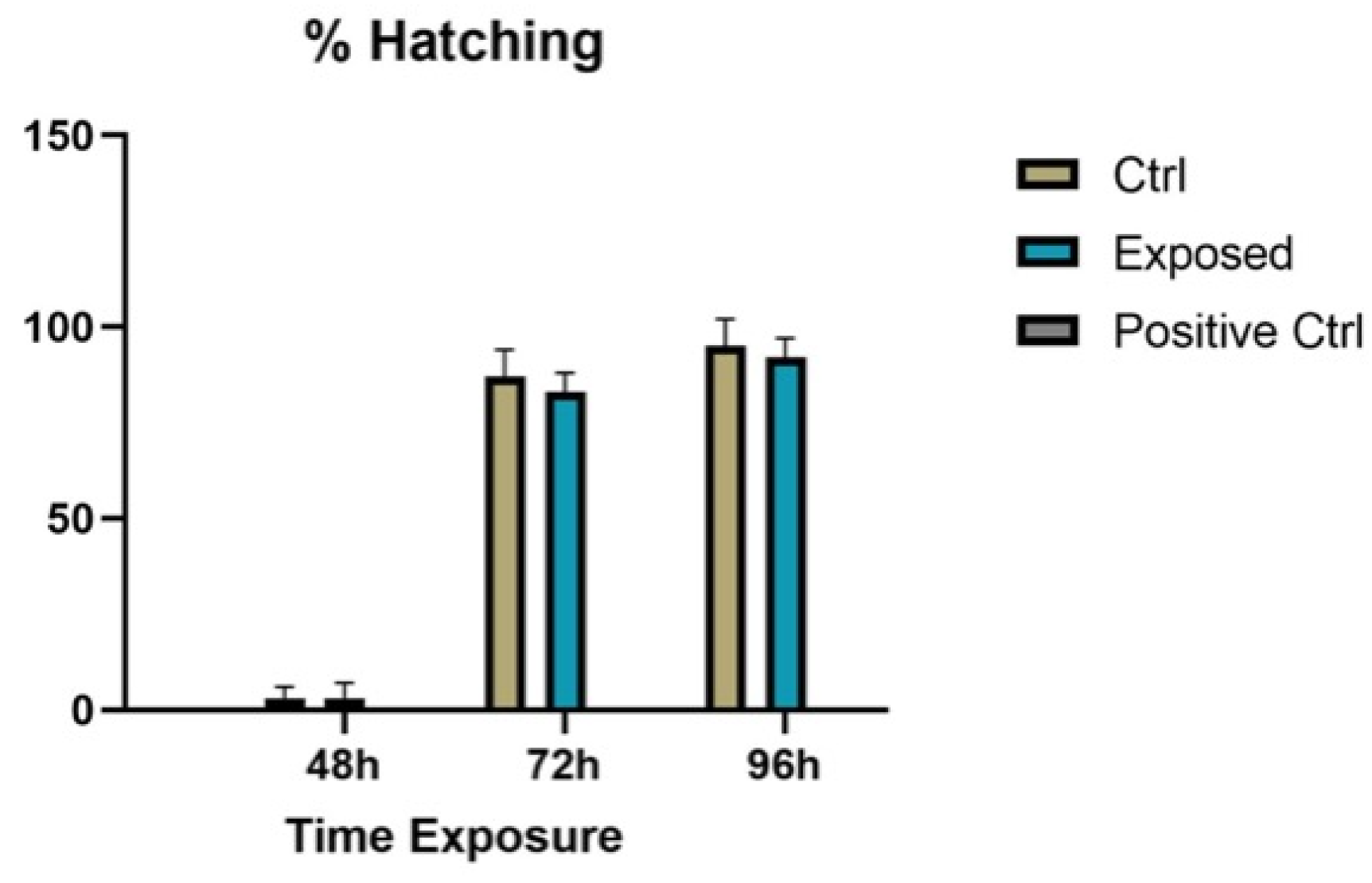
2.3. Observation of Endpoints
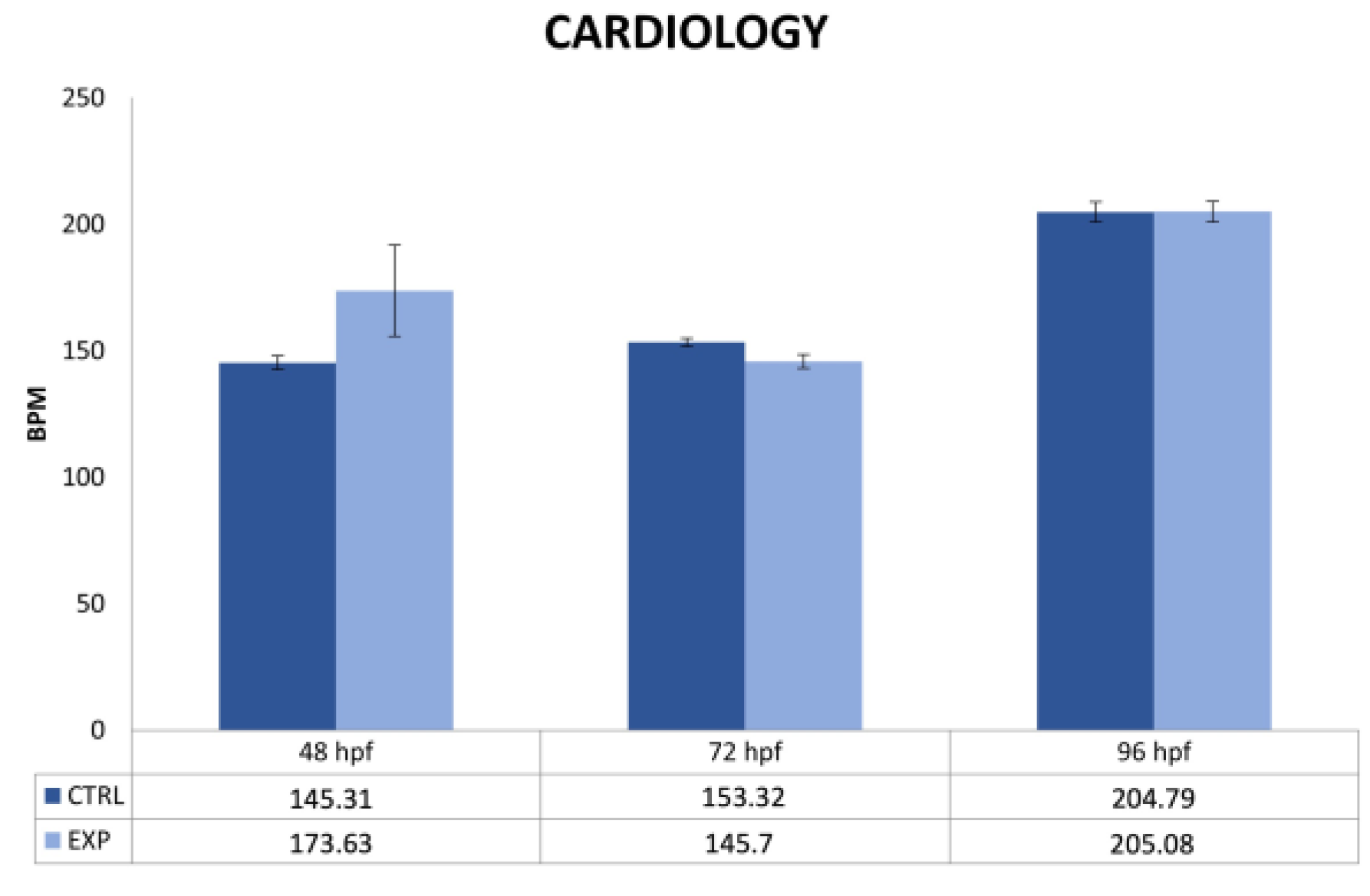
2.4. Cardiology Measurements
2.5. Immunohistochemical Analysis and Evaluation of Intracellular Reactive Oxygen Species (ROS)
2.6. Statistical Analyses
3. Results
3.1. Numerical Dosimetry Analyses
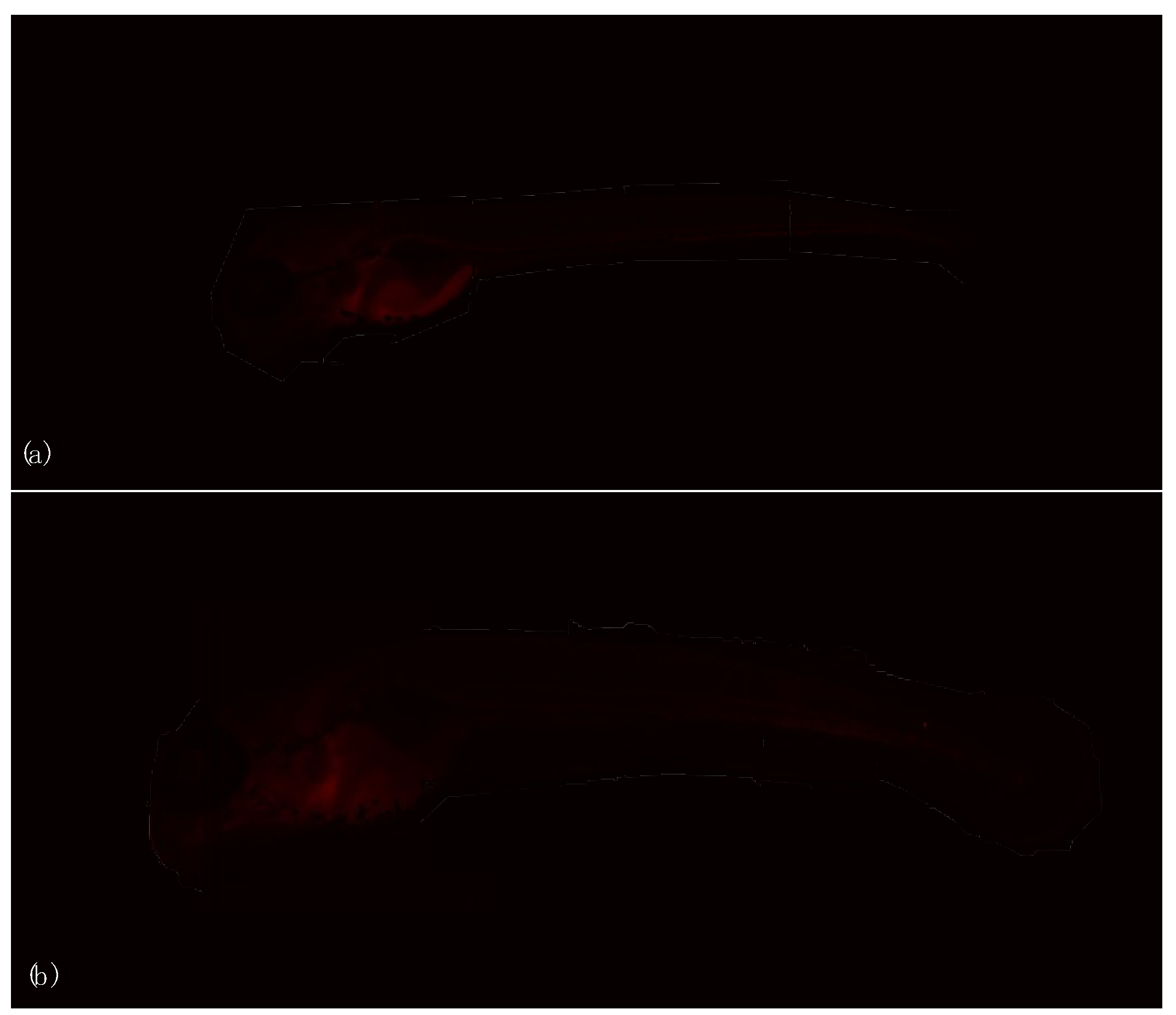
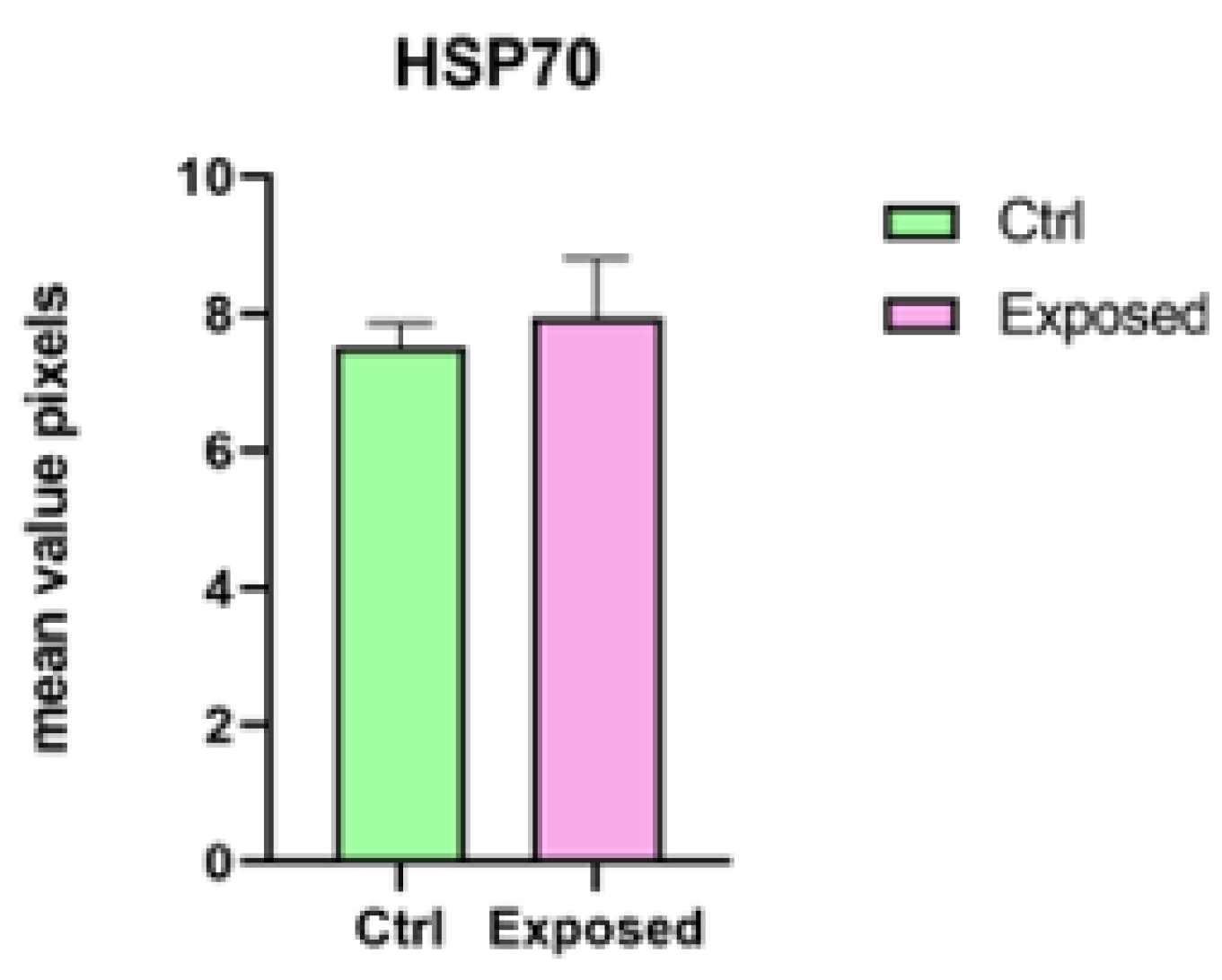
3.2. Endpoints and Biomarkers Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mattsson, M.O.; Simkó, M.; Foster, K.R. 5G new radio requires the best possible risk assessment studies: Perspective and recommended guidelines. Front. Comms. Net. 2021, 49, 724772. [Google Scholar] [CrossRef]
- Attaran, M. The impact of 5G on the evolution of intelligent automation and industry digitization. J. Ambient Intell. Human. Comput. 2021, 1–17. [Google Scholar] [CrossRef]
- Miller, A.B.; Sears, M.E.; Morgan, L.L.; Davis, D.L.; Hardell, L.; Oremus, M.; Soskolne, C.L. Risks to Health and Well-Being from Radio-Frequency Radiation Emitted by Cell Phones and Other Wireless Devices. Front. Public Health. 2019, 7, 223. [Google Scholar] [CrossRef]
- Sienkiewicz, Z.; Calderón, C.; Broom, K.A.; Addison, D.; Gavard, A.; Lundberg, L.; Maslanyj, M. Are Exposures to Multiple Frequencies the Key to Future Radiofrequency Research? Front. Public Health 2017, 5, 328. [Google Scholar] [CrossRef]
- Foster, K.R.; Vijayalaxmi. Needed: More Reliable Bioeffects Studies at “High Band” 5G Frequencies. Front. Comms. Net. 2021, 2, 721925. [Google Scholar] [CrossRef]
- Larsen, A.I.; Skotte, J. Hazardous health effects of microwaves and radio waves. Ugeskr Laeger. 1994, 156, 1618–1623. [Google Scholar] [PubMed]
- Simkó, M.; Remondini, D.; Zeni, O.; Scarfi, R.M. Quality Matters: Systematic Analysis of Endpoints Related to ‘Cellular Life’ In Vitro Data of Radiofrequency Electromagnetic Field Exposure. Int. J. Environ. Res. Public Health. 2016, 13, 701. [Google Scholar] [CrossRef] [PubMed]
- Leszczynski, D. Physiological Effects of Millimeter-Waves on Skin and Skin Cells: An Overview of the To-Date Published Studies. Rev. Environ. Health 2020, 35, 493–515. [Google Scholar] [CrossRef]
- Wood, A.; Mate, R.; Karipidis, K. Meta-Analysis of In Vitro and In Vivo Studies of the Biological Effects of Low-Level Millimetre Waves. J. Expo. Sci. Environ. Epidemiol. 2021, 31, 606–613. [Google Scholar] [CrossRef]
- Brundo, M.V.; Pecoraro, R.; Marino, F.; Salvaggio, A.; Tibullo, D.; Saccone, S.; Bramanti, V.; Buccheri, M.A.; Impellizzeri, G.; Scuderi, V.; et al. Toxicity evaluation of new engineered nanomaterials in zebrafish. Front. Physiol. 2016, 7, 130. [Google Scholar] [CrossRef]
- Pecoraro, R.; Salvaggio, A.; Marino, F.; Caro, G.D.; Capparucci, F.; Lombardo, B.M.; Messina, G.; Scalisi, E.M.; Tummino, M.; Loreto, F.; et al. Metallic nano-composite toxicity evaluation by zebrafish embryo toxicity test with identification of specific exposure biomarkers. Curr. Protoc. Toxicol. 2017, 74, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Hulot, G.; Finlay, C.C.; Constable, C.G.; Olsen, N.; Mandea, M. The magnetic field of planet Earth. Space Sci. Rev. 2010, 152, 159–222. [Google Scholar] [CrossRef]
- Krylov, V.V.; Izyumov, Y.G.; Izvekov, E.I.; Nepomnyashchikh, V.A. Magnetic fields and fish behavior. Biol. Bull. Rev. 2014, 4, 222–231. [Google Scholar] [CrossRef]
- Putman, N.F.; Lohmann, K.J.; Putman, E.M.; Quinn, T.P.; Klimley, A.P.; Noakes, D.L. Evidence for geomagnetic imprinting as a homing mechanism in Pacific salmon. Curr. Biol. 2013, 23, 312–316. [Google Scholar] [CrossRef]
- Brothers, J.R.; Lohmann, K.J. Evidence that magnetic navigation and geomagnetic imprinting shape spatial genetic variation in sea turtles. Curr. Biol. 2018, 28, 1325–1329. [Google Scholar] [CrossRef]
- Andrulewicz, E.; Napierska, D.; Otremba, Z. The environmental effects of the installation and functioning of the submarine SwePol Link HVDC transmission line: A case study of the Polish Marine Area of the Baltic Sea. J. Sea Res. 2003, 49, 337–345. [Google Scholar] [CrossRef]
- Lin, L.; Yu, H. Offshore wave energy generation devices: Impacts on ocean bio-environment. Acta Ecol. Sin. 2012, 32, 117–122. [Google Scholar] [CrossRef]
- Veith, A.; Moorthy, B. Role of cytochrome P450s in the generation and metabolism of reactive oxygen species. Curr. Opin. Toxicol. 2018, 7, 44–51. [Google Scholar] [CrossRef]
- Yamashita, M.; Yabu, T.; Ojima, N. Stress protein HSP70 in fish. Aqua-BioScience Monogr. 2010, 3, 111–141. [Google Scholar] [CrossRef]
- Morimoto, R.I.; Tissieres, A.; Georgopoulos, C.P. The stress response, function of the proteins, and perspectives. In Stress Proteins in Biology and Medicine; Cold Spring Harbor Laboratory Press: New York, NY, USA, 1990; pp. 1–36. [Google Scholar]
- Yamashita, M.; Hojo, M. Generation of a transgenic zebrafish model overexpressing heat shock protein HSP70. Mar. Biotechnol. 2004, 6, S1–S7. [Google Scholar]
- Ojima, N.; Yamashita, M.; Watabe, S. Comparative expression analysis of two paralogous Hsp70s in rainbow trout cells exposed to heat stress. Biochim. Biophys. Acta Gene Struct. Expr. 2005, 1681, 99–106. [Google Scholar] [CrossRef]
- International Commission on Non-Ionizing Radiation Protection (ICNIRP). Guidelines for limiting exposure to electromagnetic fields (100 kHz to 300 GHz). Health Phys. 2020, 118, 483–524. [Google Scholar] [CrossRef]
- OECD. Guideline for the Testing of Chemicals. Fish Embryo Toxicity (FET); N.236; OECD: Paris, France, 2013. [Google Scholar]
- Chen, X.; Zhong, Z.; Xu, Z.; Chen, L.; Wang, Y. 2′,7′-Dichlorodihydrofluorescein as a fluorescent probe for reactive oxygen species measurement: Forty years of application and controversy. Free Radic. Res. 2010, 44, 587–604. [Google Scholar] [CrossRef]
- Mugoni, V.; Camporeale, A.; Santoro, M.M. Analysis of Oxidative Stress in Zebrafish Embryos. J. Vis. Exp. 2014, 89, 51328. [Google Scholar]
- Bankhead, P. Analyzing fluorescence microscopy images with ImageJ. ImageJ 2014, 1, 10–1109. [Google Scholar]
- Kimmel, C.B.; Ballard, W.W.; Kimmel, S.R.; Ullmann, B.; Schilling, T.F. Stages of embryonic development of the zebrafish. Dev. Dynam. 1995, 203, 253–310. [Google Scholar] [CrossRef] [PubMed]
- Asharani, P.V.; Wu, Y.L.; Gong, Z.; Valiyaveettil, S. Toxicity of silver nanoparticles in zebrafish models. Nanotechnology 2008, 19, 255102. [Google Scholar] [CrossRef] [PubMed]
- Wei., Y.; Meng, Y.; Huang, Y.; Liu, Z.; Zhong, K.; Ma, J.; Zhang, W.; Li, Y.; Lu, H. Development toxicity and cardiotoxicity in zebrafish from exposure to iprodione. Chemosphere 2021, 263, 127860. [Google Scholar] [CrossRef]
- Georgiou, C.D.; Margaritis, L.H. Oxidative Stress and NADPH Oxidase: Connecting Electromagnetic Fields, Cation Channels and Biological Effects. Int. J. Mol. Sci. 2021, 22, 10041. [Google Scholar] [CrossRef]
- Gu, J.; Wang, H.; Zhou, L.; Fan, D.; Shi, L.; Ji, G.; Gu, A. Oxidative stress in bisphenol AF-induced cardiotoxicity in zebrafish and the protective role of N-acetyl N-cysteine. Sci. Total Environ. 2020, 731, 139190. [Google Scholar] [CrossRef]
- Guerriero, G.; D’Errico, G.; Di Giaimo, R.; Rabbito, D.; Olanrewaju, O.S.; Ciarcia, G. Reactive oxygen species and glutathione antioxidants in the testis of the soil biosentinel Podarcis sicula (Rafinesque 1810). Environ. Sci. Pollut. Res. Int. 2018, 25, 18286–18296. [Google Scholar] [CrossRef] [PubMed]
- Alkis, M.E.; Bilgin, H.M.; Akpolat, V.; Dasdag, S.; Yegin, K.; Yavas, M.C.; Akdag, M.Z. Effect of 900-, 1800, and 2100-MHz radiofrequency radiation on DNA and oxidative stress in brain. Electromagn. Biol. Med. 2019, 38, 32–47. [Google Scholar] [CrossRef] [PubMed]
- Üstündağ, Ü.V.; Özen, M.S.; Ünal, İ.; Ateş, P.S.; Alturfan, A.A.; Akalın, M.; Sancak, E.; Emekli-Alturfan, E. Oxidative stress and apoptosis in electromagnetic waves exposed Zebrafish embryos and protective effects of conductive nonwoven fabric. Cell. Mol. Biol. 2020, 66, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Zhao, Y.; Nel, A.E.; Lin, S. Zebrafish: An in vivo model for nano EHS studies. Small 2013, 9, 1608–1618. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.M.; Lim, H.M.; Ro, H.S.; Ki, G.E.; Seo, Y.K. Pulsed electromagnetic fields increase pigmentation through the p-ERK/p-p38 pathway in zebrafish (Danio rerio). Int. J. Mol. Sci. 2018, 19, 3211. [Google Scholar] [CrossRef]
- Li, Y.; Liu, X.; Liu, K.; Miao, W.; Zhou, C.; Li, Y.; Wu, H. Extremely low-frequency magnetic fields induce developmental toxicity and apoptosis in zebrafish (Danio rerio) embryos. Biol. Trace Elem. Res. 2014, 162, 324–332. [Google Scholar] [CrossRef] [PubMed]
- Piccinetti, C.C.; De Leo, A.; Cosoli, G.; Scalise, L.; Randazzo, B.; Cerri, G.; Olivotto, I. Measurement of the 100 MHz EMF radiation in vivo effects on zebrafish D. rerio embryonic development: A multidisciplinary study. Ecotoxicol. Environ. Saf. 2018, 154, 268–279. [Google Scholar] [CrossRef]
- Dasgupta, S.; Wang, G.; Simonich, M.T.; Zhang, T.; Truong, L.; Liu, H.; Tanguay, R.L. Impacts of high dose 3.5 GHz cellphone radiofrequency on zebrafish embryonic development. PLoS ONE 2020, 15, e0235869. [Google Scholar] [CrossRef]
- Fey, D.P.; Jakubowska, M.; Greszkiewicz, M.; Andrulewicz, E.; Otremba, Z.; Urban-Malinga, B. Are magnetic and electromagnetic fields of anthropogenic origin potential threats to early life stages of fish? Aquat. Toxicol. 2019, 209, 150–158. [Google Scholar] [CrossRef]
- Di Carlo, A.; White, N.; Guo, F.; Garrett, P.; Litovitz, T. Chronic electromagnetic field exposure decreases HSP70 levels and lowers cytoprotection. J. Cell. Biochem. 2022, 84, 447–454. [Google Scholar] [CrossRef]
- Patruno, A.; Tabrez, S.; Pesce, M.; Shakil, S.; Kamal, M.A.; Reale, M. Effects of extremely low frequency electromagnetic field (ELF-EMF) on catalase, cytochrome P450 and nitric oxide synthase in erythro-leukemic cells. Life Sci. 2015, 121, 117–123. [Google Scholar] [CrossRef] [PubMed]


| Component | Material | Dielectric Constant | Loss Tangent | Mass Density |
|---|---|---|---|---|
| Horn antenna | Perfect electric conductor (good metal) | - | ∞ | - |
| Aqueous sample and water bath | Distilled water | 28.47 | 1.25 | 1000 [Kg/m3] |
| Six-well microplates | Polystyrene | 2.5 | 0 | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pecoraro, R.; Pavone, S.C.; Scalisi, E.M.; Ignoto, S.; Sica, C.; Indelicato, S.; Capparucci, F.; Iaria, C.; Salvaggio, A.; Sorbello, G.; et al. Multimarker Approach to Evaluate the Exposure to Electromagnetic Fields at 27 GHz on Danio rerio Larvae. J. Mar. Sci. Eng. 2023, 11, 693. https://doi.org/10.3390/jmse11040693
Pecoraro R, Pavone SC, Scalisi EM, Ignoto S, Sica C, Indelicato S, Capparucci F, Iaria C, Salvaggio A, Sorbello G, et al. Multimarker Approach to Evaluate the Exposure to Electromagnetic Fields at 27 GHz on Danio rerio Larvae. Journal of Marine Science and Engineering. 2023; 11(4):693. https://doi.org/10.3390/jmse11040693
Chicago/Turabian StylePecoraro, Roberta, Santi Concetto Pavone, Elena Maria Scalisi, Sara Ignoto, Carmen Sica, Stefania Indelicato, Fabiano Capparucci, Carmelo Iaria, Antonio Salvaggio, Gino Sorbello, and et al. 2023. "Multimarker Approach to Evaluate the Exposure to Electromagnetic Fields at 27 GHz on Danio rerio Larvae" Journal of Marine Science and Engineering 11, no. 4: 693. https://doi.org/10.3390/jmse11040693
APA StylePecoraro, R., Pavone, S. C., Scalisi, E. M., Ignoto, S., Sica, C., Indelicato, S., Capparucci, F., Iaria, C., Salvaggio, A., Sorbello, G., Di Donato, L., & Brundo, M. V. (2023). Multimarker Approach to Evaluate the Exposure to Electromagnetic Fields at 27 GHz on Danio rerio Larvae. Journal of Marine Science and Engineering, 11(4), 693. https://doi.org/10.3390/jmse11040693













