Exfoliating Whales–Sandy Bottom Contact Behaviour of Humpback Whales
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Clapham, P.J. The humpback whale. In Cetacean Societies, Field Studies of Dolphins and Whales; The University of Chicago: Chicago, IL, USA, 2000; pp. 173–196. [Google Scholar]
- Eisenmann, P.; Fry, B.; Holyoake, C.; Coughran, D.; Nicol, S.; Bengtson Nash, S. Isotopic Evidence of a Wide Spectrum of Feeding Strategies in Southern Hemisphere Humpback Whale Baleen Records. PLoS ONE 2016, 11, e0156698. [Google Scholar] [CrossRef] [PubMed]
- Wedekin, L.L.; Engel, M.H.; Andriolo, A.; Prado, P.I.; Zerbini, A.N.; Marcondes, M.M.C.; Kinas, P.G.; Simões-Lopes, P.C. Running fast in the slow lane: Rapid population growth of humpback whales after exploitation. Mar. Ecol. Prog. Ser. 2017, 575, 195–206. [Google Scholar] [CrossRef]
- Pallin, L.J.; Baker, C.S.; Steel, D.; Kellar, N.M.; Robbins, J.; Johnston, D.W.; Nowacek, D.P.; Read, A.J.; Friedlaender, A.S. High pregnancy rates in humpback whales (Megaptera novaeangliae) around the Western Antarctic Peninsula, evidence of a rapidly growing population. R. Soc. Open Sci. 2018, 5, 180017. [Google Scholar] [CrossRef]
- Fleming, A.; Jackson, J. Global Review of Humpback Whales (Megaptera novaeangliae). 2011. Available online: https://repository.library.noaa.gov/view/noaa/4489 (accessed on 10 January 2023).
- Duarte, C.M.; Agusti, S.; Barbier, E.; Britten, G.L.; Castilla, J.C.; Gattuso, J.-P.; Fulweiler, R.W.; Hughes, T.P.; Knowlton, N.; Lovelock, C.E.; et al. Rebuilding marine life. Nature 2020, 580, 39–51. [Google Scholar] [CrossRef]
- Calambokidis, J.; Fahlbusch, J.A.; Szesciorka, A.R.; Southall, B.L.; Cade, D.E.; Friedlaender, A.S.; Goldbogen, J.A. Differential Vulnerability to Ship Strikes Between Day and Night for Blue, Fin, and Humpback Whales Based on Dive and Movement Data From Medium Duration Archival Tags. Front. Mar. Sci. 2019, 6, 543. [Google Scholar] [CrossRef]
- Meynecke, J.-O.; Seyboth, E.; De Bie, J.; Menzel Barraqueta, J.-L.; Chama, A.; Prakash Dey, S.; Lee, S.B.; Tulloch, V.; Vichi, M.; Findlay, K.; et al. Responses of humpback whales to a changing climate in the Southern Hemisphere: Priorities for research efforts. Mar. Ecol. 2020, 41, e12616. [Google Scholar] [CrossRef]
- Reinke, J.; Lemckert, C.; Meynecke, J.-O. Coastal fronts utilized by migrating humpback whales, Megaptera novaeangliae, on the Gold Coast, Australia. J. Coast. Res. 2016, 75, 552–557. [Google Scholar] [CrossRef]
- Hazen, E.L.; Friedlaender, A.S.; Thompson, M.A.; Ware, C.R.; Weinrich, M.T.; Halpin, P.N.; Wiley, D.N. Fine-scale prey aggregations and foraging ecology of humpback whales Megaptera novaeangliae. Mar. Ecol. Prog. Ser. 2009, 395, 75–89. [Google Scholar] [CrossRef]
- Parra, G.J. Observations of an Indo-Pacific humpback dolphin carrying a sponge: Object play or tool use? Mammalia 2007, 71, 147–149. [Google Scholar] [CrossRef]
- Woodward, B.L.; Winn, J.P. Apparent lateralized behaviour in gray whales feeding off the central British Columbia coast. Mar. Mammal Sci. 2006, 22, 64–73. [Google Scholar] [CrossRef]
- Wiley, D.; Ware, C.; Bocconcelli, A.; Cholewiak, D.; Friedlaender, A.; Thompson, M.; Weinrich, M. Underwater components of humpback whale bubble-net feeding. Behaviour 2011, 148, 575–602. [Google Scholar]
- McMillan, C.J.; Towers, J.R.; Hildering, J. The innovation and diffusion of “trap-feeding,” a novel humpback whale foraging strategy. Mar. Mammal Sci. 2019, 35, 779–796. [Google Scholar] [CrossRef]
- Owen, K.; Dunlop, R.; Donnelly, D. Seaweed interactions by humpback whales (Megaptera novaeangliae): A form of object play? Aquat. Mamm. 2012, 38, 418. [Google Scholar] [CrossRef]
- Shea, B.D.; Gallagher, A.J. Humpback Whale Instigates Object Play with a Lion’s Mane Jellyfish. Oceans 2021, 2, 386–392. [Google Scholar] [CrossRef]
- Pitman, R.L.; Deecke, V.B.; Gabriele, C.M.; Srinivasan, M.; Black, N.; Denkinger, J.; Durban, J.W.; Mathews, E.A.; Matkin, D.R.; Neilson, J.L.; et al. Humpback whales interfering when mammal-eating killer whales attack other species: Mobbing behavior and interspecific altruism? Mar. Mammal Sci. 2017, 33, 7–58. [Google Scholar] [CrossRef]
- Fortune, S.M.E.; Koski, W.R.; Higdon, J.W.; Trites, A.W.; Baumgartner, M.F.; Ferguson, S.H. Evidence of molting and the function of “rock-nosing” behavior in bowhead whales in the eastern Canadian Arctic. PLoS ONE 2017, 12, e0186156. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.; Aubin, D.; Hammill, M. Rubbing behaviour of belugas, Delphinapterus leucas, in a high Arctic estuary. Can. J. Zool. 2011, 70, 2405–2409. [Google Scholar] [CrossRef]
- Pinheiro, F.C.F.; Pinheiro, H.T.; Siciliano, S.; Santos, R.G. Bottom contact behaviour by humpback whales in Brazilian waters: First underwater observations at Trindade Island. Mar. Biodivers. Rec. 2016, 9, 64. [Google Scholar] [CrossRef]
- Ware, C.; Friedlaender, A.S.; Nowacek, D.P. Shallow and deep lunge feeding of humpback whales in fjords of the West Antarctic Peninsula. Mar. Mammal Sci. 2011, 27, 587–605. [Google Scholar] [CrossRef]
- Canning, C.; Crain, D.; Eaton, T.S.; Nuessly, K.; Friedlaender, A.; Hurst, T.; Parks, S.; Ware, C.; Wiley, D.; Weinrich, M. Population-level lateralized feeding behaviour in North Atlantic humpback whales, Megaptera novaeangliae. Anim. Behav. 2011, 82, 901–909. [Google Scholar] [CrossRef]
- Friedlaender, A.S.; Hazen, E.L.; Nowacek, D.P.; Halpin, P.N.; Ware, C.; Weinrich, M.; Hurst, T.; Wiley, D. Diel changes in humpback whale Megaptera novaeangliae feeding behavior in response to sand lance Ammodytes spp. behavior and distribution. Mar. Ecol. Prog. Ser. 2009, 395, 91–100. [Google Scholar] [CrossRef]
- Hain, J.H.W.; Ellis, S.L.; Kenney, R.D.; Clapham, P.J.; Gray, B.K.; Weinrich, M.T.; Babb, I.G. Apparent bottom feeding by humpback whales on Stellwagen Bank. Mar. Mammal Sci. 1995, 11, 464–479. [Google Scholar] [CrossRef]
- Ware, C.; Arsenault, R.; Plumlee, M.; Wiley, D. Visualizing the Underwater Behavior of Humpback Whales. IEEE Comput. Graph. Appl. 2006, 26, 14–18. [Google Scholar] [CrossRef] [PubMed]
- Andrews, R.D.; Baird, R.W.; Calambokidis, J.; Goertz, C.E.C.; Gulland, F.M.D.; Heide-Jorgensen, M.P.; Hooker, S.K.; Johnson, M.; Mate, B.; Mitani, Y.; et al. Best practice guidelines for cetacean tagging. J. Cetacean Res. Manag. 2019, 20, 27–66. [Google Scholar] [CrossRef]
- Johnson, M.; Tyack, P.; Nowacek, D.; Shorter, A. A digital acoustic recording tag for measuring the response of marine mammals to sound. J. Acoust. Soc. Am. 2000, 108, 2582–2583. [Google Scholar] [CrossRef]
- Friedlaender, A.S.; Goldbogen, J.A.; Nowacek, D.P.; Read, A.J.; Johnston, D.; Gales, N. Feeding rates and under-ice foraging strategies of the smallest lunge filter feeder, the Antarctic minke whale (Balaenoptera bonaerensis). J. Exp. Biol. 2014, 217, 2851. [Google Scholar] [CrossRef] [PubMed]
- Baird, R. Studying diving behavior of whales and dolphins using suction-cup attached tags. Whalewatcher 1998, 31, 3–7. [Google Scholar]
- Ratsimbazafindranahaka, M.N.; Huetz, C.; Andrianarimisa, A.; Reidenberg, J.S.; Saloma, A.; Adam, O.; Charrier, I. Characterizing the suckling behavior by video and 3D-accelerometry in humpback whale calves on a breeding ground. PeerJ 2022, 10, e12945. [Google Scholar] [CrossRef]
- Tackaberry, J.E.; Cade, D.E.; Goldbogen, J.A.; Wiley, D.N.; Friedlaender, A.S.; Stimpert, A.K. From a calf’s perspective: Humpback whale nursing behavior on two US feeding grounds. PeerJ 2020, 8, e8538. [Google Scholar] [CrossRef]
- Linsky, J.M.J.; Wilson, N.; Cade, D.E.; Goldbogen, J.A.; Johnston, D.W.; Friedlaender, A.S. The scale of the whale: Using video-tag data to evaluate sea-surface ice concentration from the perspective of individual Antarctic minke whales. Anim. Biotele. 2020, 8, 31. [Google Scholar] [CrossRef]
- Garrigue, C.; Clapham, P.J.; Geyer, Y.; Kennedy, A.S.; Zerbini, A.N. Satellite tracking reveals novel migratory patterns and the importance of seamounts for endangered South Pacific humpback whales. R. Soc. Open Sci. 2015, 2, 150489. [Google Scholar] [CrossRef]
- Weinstein, B.G.; Double, M.; Gales, N.; Johnston, D.W.; Friedlaender, A.S. Identifying overlap between humpback whale foraging grounds and the Antarctic krill fishery. Biol. Conserv. 2017, 210, 184–191. [Google Scholar] [CrossRef]
- Cerchio, S.; Trudelle, L.; Zerbini, A.N.; Charrassin, J.B.; Geyer, Y.; Mayer, F.X.; Andrianarivelo, N.; Jung, J.L.; Adam, O.; Rosenbaum, H.C. Satellite telemetry of humpback whales off Madagascar reveals insights on breeding behavior and long-range movements within the southwest Indian Ocean. Mar. Ecol. Prog. Ser. 2016, 562, 193–209. [Google Scholar] [CrossRef]
- Santora, J.A.; Mantua, N.J.; Schroeder, I.D.; Field, J.C.; Hazen, E.L.; Bograd, S.J.; Sydeman, W.J.; Wells, B.K.; Calambokidis, J.; Saez, L.; et al. Habitat compression and ecosystem shifts as potential links between marine heatwave and record whale entanglements. Nat. Commun. 2020, 11, 536. [Google Scholar] [CrossRef] [PubMed]
- Chapman, D.M. Coastal erosion and the sediment budget, with special reference to the gold coast, Australia. Coast. Eng. 1980, 4, 207–227. [Google Scholar] [CrossRef]
- Meynecke, J.O.; Vindenes, S.; Teixeira, D. Monitoring humpback whale (Megaptera novaeangliae) behaviour in a highly urbanised coastline: Gold Coast, Australia. In Global Challenges in Integrated Coastal Zone Management; Moksness, E., Dahl, E., Støttrup, J., Eds.; Wiley: Chichester, UK, 2013; pp. 101–113. [Google Scholar]
- McCulloch, S.; Meynecke, J.O.; Franklin, T.; Franklin, W.; Chauvenet, A.L.M. Humpback whale (Megaptera novaeangliae) behaviour determines habitat use in two Australian bays. Mar. Freshw. Res. 2021, 72, 1251–1267. [Google Scholar] [CrossRef]
- Torre-Williams, L.; Martinez, E.; Meynecke, J.O.; Reinke, J.; Stockin, K.A. Presence of newborn humpback whale (Megaptera novaeangliae) calves in Gold Coast Bay, Australia. Mar. Freshw. Behav. Physiol. 2019, 52, 199–216. [Google Scholar] [CrossRef]
- Goldbogen, J.A.; Cade, D.E.; Boersma, A.T.; Calambokidis, J.; Kahane-Rapport, S.R.; Segre, P.S.; Stimpert, A.K.; Friedlaender, A.S. Using Digital Tags with Integrated Video and Inertial Sensors to Study Moving Morphology and Associated Function in Large Aquatic Vertebrates. Anat. Rec. 2017, 300, 1935–1941. [Google Scholar] [CrossRef] [PubMed]
- Goldbogen, J.A.; Friedlaender, A.S.; Calambokidis, J.; McKenna, M.F.; Simon, M.; Nowacek, D.P. Integrative Approaches to the Study of Baleen Whale Diving Behavior, Feeding Performance, and Foraging Ecology. BioScience 2013, 63, 90–100. [Google Scholar] [CrossRef]
- Savoca, M.; Czapanskiy, M.; Kahane-Rapport, S.; Gough, W.; Fahlbusch, J.; Bierlich, K.; Segre, P.; Di Clemente, J.; Penry, G.; Wiley, D.; et al. Baleen whale prey consumption based on high-resolution foraging measurements. Nature 2021, 599, 85–90. [Google Scholar] [CrossRef]
- Cade, D.E.; Gough, W.T.; Czapanskiy, M.F.; Fahlbusch, J.A.; Kahane-Rapport, S.R.; Linsky, J.M.J.; Nichols, R.C.; Oestreich, W.K.; Wisniewska, D.M.; Friedlaender, A.S.; et al. Tools for integrating inertial sensor data with video bio-loggers, including estimation of animal orientation, motion, and position. Anim. Biotele. 2021, 9, 34. [Google Scholar] [CrossRef]
- Wilson, R.P.; Liebsch, N.; Davies, I.M.; Quintana, F.; Weimerskirch, H.; Storch, S.; Lucke, K.; Siebert, U.; Zankl, S.; Müller, G.; et al. All at sea with animal tracks; methodological and analytical solutions for the resolution of movement. Deep Sea Res. Part II Top. Stud. Oceanogr. 2007, 54, 193–210. [Google Scholar] [CrossRef]
- Valani, R.; Meynecke, J.-O.; Olsen, M.T. Presence and movement of humpback whale (Megaptera novaeangliae) mother-calf pairs in the Gold Coast, Australia. Mar. Freshw. Behav. Physiol. 2020, 53, 251–263. [Google Scholar] [CrossRef]
- Félix, F.; Botero-Acosta, N. Distribution and behaviour of humpback whale mother–calf pairs during the breeding season off Ecuador. Mar. Ecol. Prog. Ser. 2011, 426, 277–287. [Google Scholar] [CrossRef]
- Condy, P.R.; Aarde, R.J.V.; Bester, M.N. The seasonal occurrence and behaviour of Killer whales Orcinus orca, at Marion Island. J. Zool. 1978, 184, 449–464. [Google Scholar] [CrossRef]
- Cartwright, R.; Sullivan, M. Behavioral ontogeny in humpback whale (Megaptera novaeangliae) calves during their residence in Hawaiian waters. Mar. Mammal Sci. 2009, 25, 659–680. [Google Scholar] [CrossRef]
- Zoidis, A.M.; Lomac-MacNair, K.S.; Chomos-Betz, A.E.; Day, A.J.; Sasha McFarland, A. Effects of sex, seasonal period, and sea state on calf behavior in Hawaiian humpback whales (Megaptera novaeangliae). Aquat. Mamm. 2014, 40, 44–58. [Google Scholar] [CrossRef]
- Deakos, M.H.; Branstetter, B.K.; Mazzuca, L.; Fertl, D.; Mobley, J.R., Jr. Two Unusual Interactions Between a Bottlenose Dolphin (Tursiops truncatus) and a Humpback Whale (Megaptera novaeangliae) in Hawaiian Waters. Aquat. Mamm. 2010, 36. [Google Scholar] [CrossRef]
- Fertl, D.; Fulling, G. Interactions between marine mammals and turtles. Mar. Turt. Newsl. 2007, 115, 4–8. [Google Scholar]
- Matsui, K.; Khalil, A.M.; Takeda, K.-i. Do horses prefer certain substrates for rolling in grazing pasture? J. Equine Vet. Sci. 2009, 29, 590–594. [Google Scholar] [CrossRef]
- Zoe, R.B.; John, K.E.M.; Wendy, C.T. Dust-bathing behaviours of African herbivores and the potential risk of inhalation of Anthrax. J. Wildl. Dis. 2018, 54, 34–44. [Google Scholar] [CrossRef]
- Pitman, R.L.; Durban, J.W.; Joyce, T.; Fearnbach, H.; Panigada, S.; Lauriano, G. Skin in the game: Epidermal molt as a driver of long-distance migration in whales. Mar. Mammal Sci. 2020, 36, 565–594. [Google Scholar] [CrossRef]
- Félix, F.; Haase, B.; Aguirre, W.E. Spondylitis in a humpback whale (Megaptera novaeangliae) from the southeast Pacific. Dis. Aquat. Org. 2007, 75, 259–264. [Google Scholar] [CrossRef]
- Rowntree, V.J. Feeding, distribution, and reproductive behavior of cyamids (Crustacea: Amphipoda) living on humpback and right whales. Can. J. Zool. 1996, 74, 103–109. [Google Scholar] [CrossRef]
- Apprill, A.; Mooney, T.A.; Lyman, E.; Stimpert, A.K.; Rappé, M.S. Humpback whales harbour a combination of specific and variable skin bacteria. Environ. Microbiol. Rep. 2011, 3, 223–232. [Google Scholar] [CrossRef]
- Félix, F.; Bearson, B.; Falconí, J. Epizoic barnacles removed from the skin of a humpback whale after a period of intense surface activity. Mar. Mammal Sci. 2006, 22, 979–984. [Google Scholar] [CrossRef]
- Gibson, R.; Barnes, M. Evolution and ecology of cleaning symbioses in the sea. Oceanography and Marine Biology: An Annual Review; CRC Press: Boca Raton, FL, USA, 2000; Volume 38, p. 311. [Google Scholar]
- Morlock, G.E.; Ziltener, A.; Geyer, S.; Tersteegen, J.; Mehl, A.; Schreiner, T.; Kamel, T.; Brümmer, F. Evidence that Indo-Pacific bottlenose dolphins self-medicate with invertebrates in coral reefs. iScience 2022, 25, 104271. [Google Scholar] [CrossRef] [PubMed]
- Pack, A.A.; Herman, L.M.; Spitz, S.S.; Hakala, S.; Deakos, M.H.; Herman, E.Y. Male humpback whales in the Hawaiian breeding grounds preferentially associate with larger females. Anim. Behav. 2009, 77, 653–662. [Google Scholar] [CrossRef]
- Herman, E.Y.; Herman, L.M.; Pack, A.A.; Marshall, G.; Shepard, C.M. When whales collide: Crittercam offers insight into the competitive behavior of humpback whales on their Hawaiian wintering grounds. Mar. Technol. Soc. J. 2007, 41, 35–43. [Google Scholar] [CrossRef]
- Iwata, T.; Biuw, M.; Aoki, K.; Miller, P.J.O.M.; Sato, K. Using an omnidirectional video logger to observe the underwater life of marine animals: Humpback whale resting behaviour. Behav. Process. 2021, 186, 104369. [Google Scholar] [CrossRef]
- Cartwright, R.; Gillespie, B.; LaBonte, K.; Mangold, T.; Venema, A.; Eden, K.; Sullivan, M. Between a rock and a hard place: Habitat selection in female-calf humpback whale (Megaptera novaeangliae) pairs on the Hawaiian breeding grounds. PLoS ONE 2012, 7, e38004. [Google Scholar] [CrossRef] [PubMed]
- Ona, J.; Duque, E.; Garland, E.C.; Seger, K.; Narvaez, M.; Maldonado, J.; Denkinger, J. A Giant’s Dance: Underwater Social and Vocal Behavior of Humpback Whales (Megaptera novaeangliae) Recorded on the Northern Coast of Ecuador. Aquat. Mamm. 2019, 45, 456–464. [Google Scholar] [CrossRef]
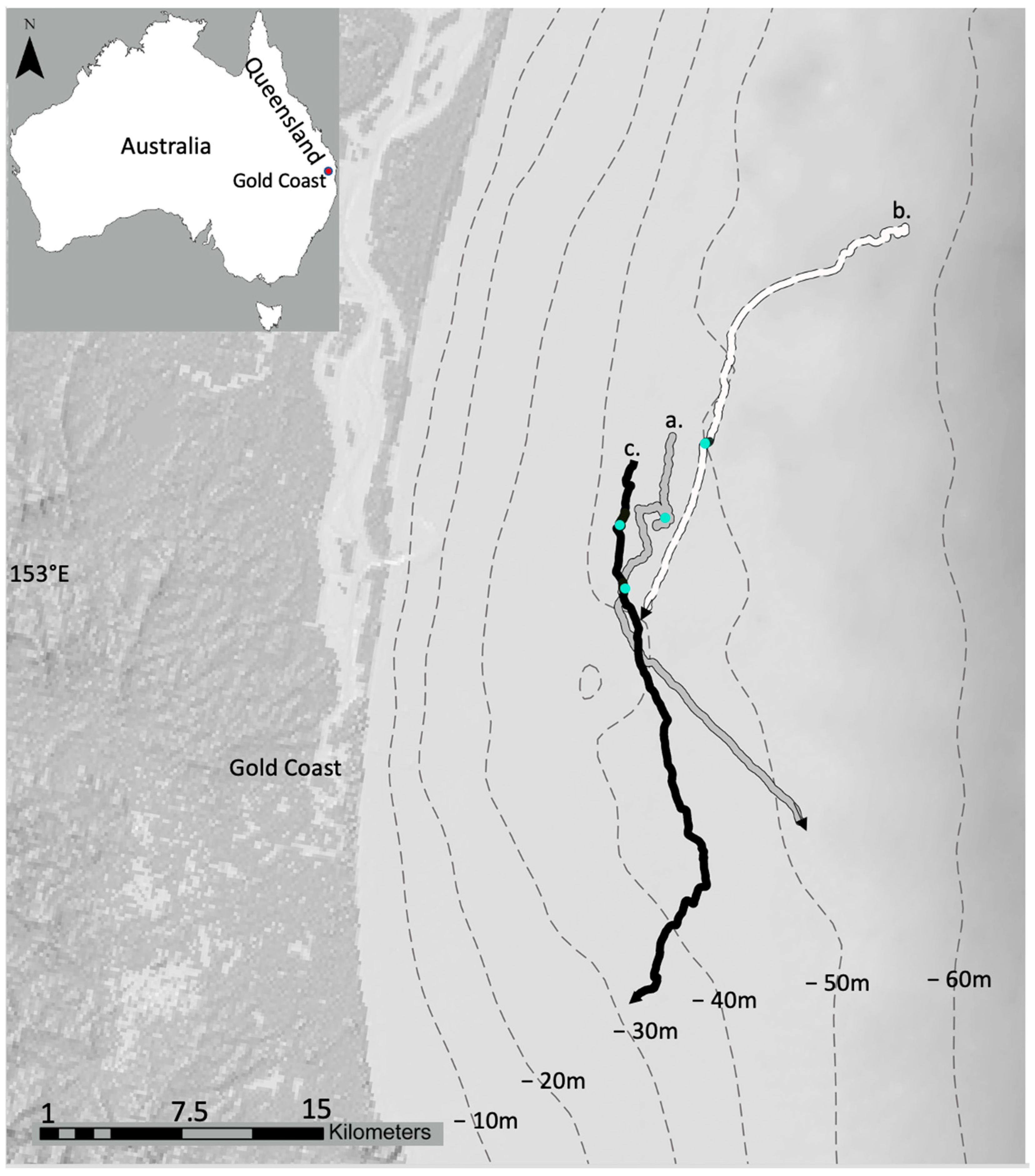


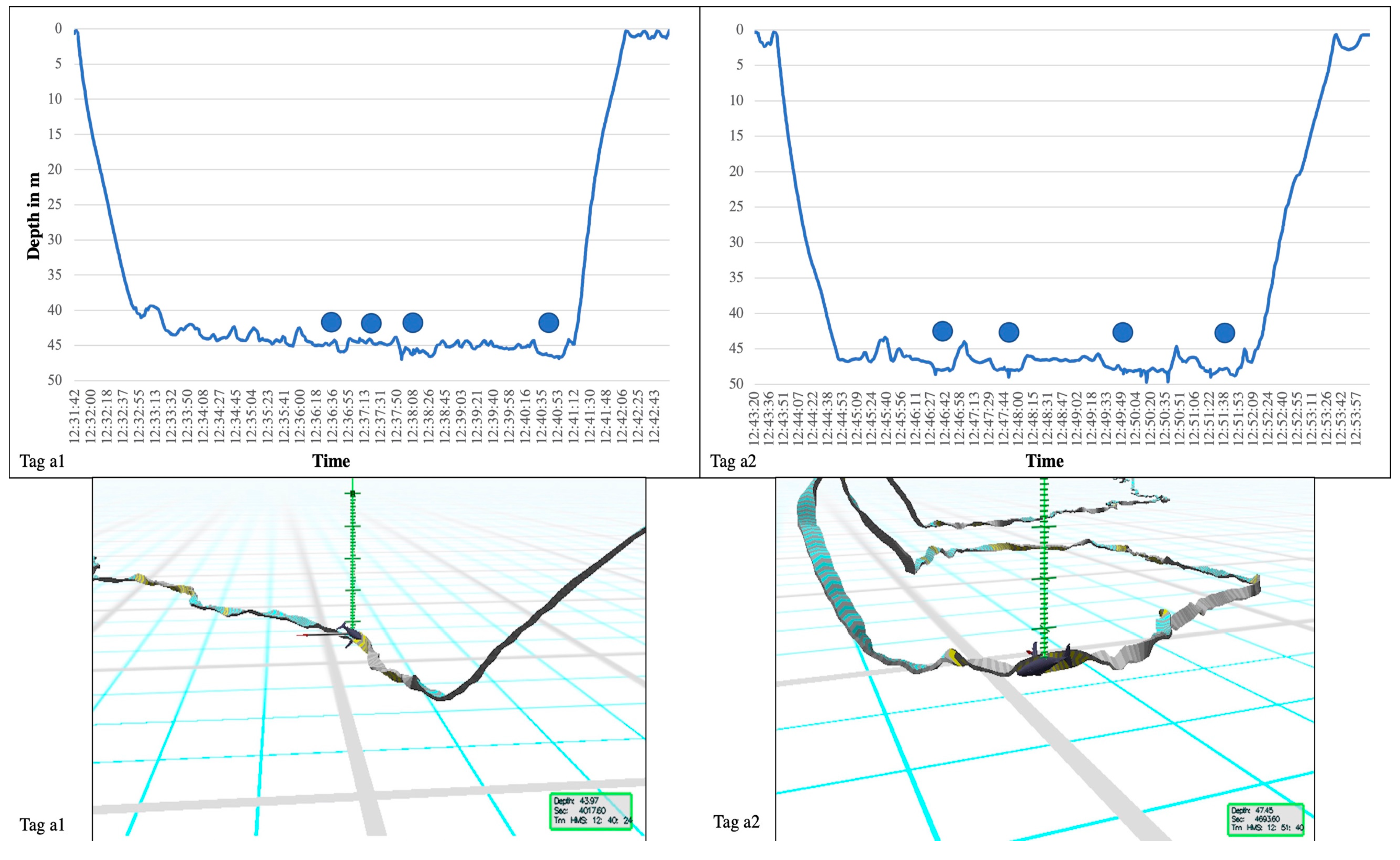
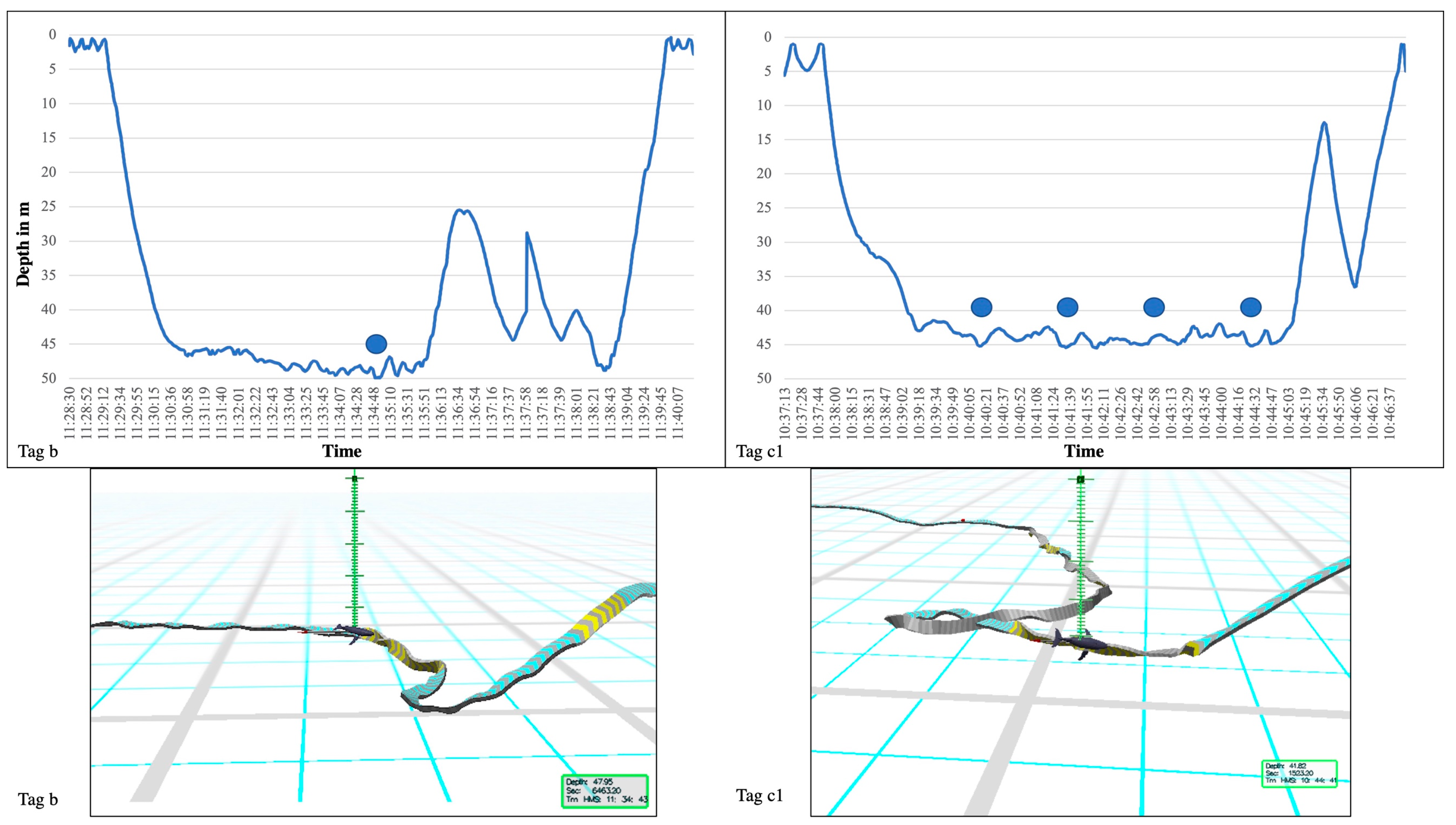
| Tag ID | Date Tagged | Tag On | TAG OFF | Tag on in Hrs | Aver. Speed km/h | Distance in km | Number of Sand Rolls |
|---|---|---|---|---|---|---|---|
| a | 31 August 21 | 11:33 | 18:01 | 6:28 | 2.9 (1.8) | 18.8 (11.8) | 8 |
| b | 6 October 21 | 9:47 | 13:00 | 3:13 | 5.4 (4.6) | 16.9 (14.7) | 1 |
| c | 26 September 22 | 10:19 | 13:59 | 4:17 | 4.7 (3.8) | 19.6 (16.1) | 5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meynecke, J.-O.; Gustafon, J.; Cade, D.E. Exfoliating Whales–Sandy Bottom Contact Behaviour of Humpback Whales. J. Mar. Sci. Eng. 2023, 11, 600. https://doi.org/10.3390/jmse11030600
Meynecke J-O, Gustafon J, Cade DE. Exfoliating Whales–Sandy Bottom Contact Behaviour of Humpback Whales. Journal of Marine Science and Engineering. 2023; 11(3):600. https://doi.org/10.3390/jmse11030600
Chicago/Turabian StyleMeynecke, Jan-Olaf, Johan Gustafon, and David E. Cade. 2023. "Exfoliating Whales–Sandy Bottom Contact Behaviour of Humpback Whales" Journal of Marine Science and Engineering 11, no. 3: 600. https://doi.org/10.3390/jmse11030600
APA StyleMeynecke, J.-O., Gustafon, J., & Cade, D. E. (2023). Exfoliating Whales–Sandy Bottom Contact Behaviour of Humpback Whales. Journal of Marine Science and Engineering, 11(3), 600. https://doi.org/10.3390/jmse11030600








