Soil Organic Carbon Depletion from Forests to Grasslands Conversion in Mexico: A Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Literature Search and Collection of Relevant Information
2.2. Historical Change in Forest and Pastureland Cover in Mexico
2.3. Soil Organic Carbon Pool and Changes
2.4. Data Analysis
3. Results
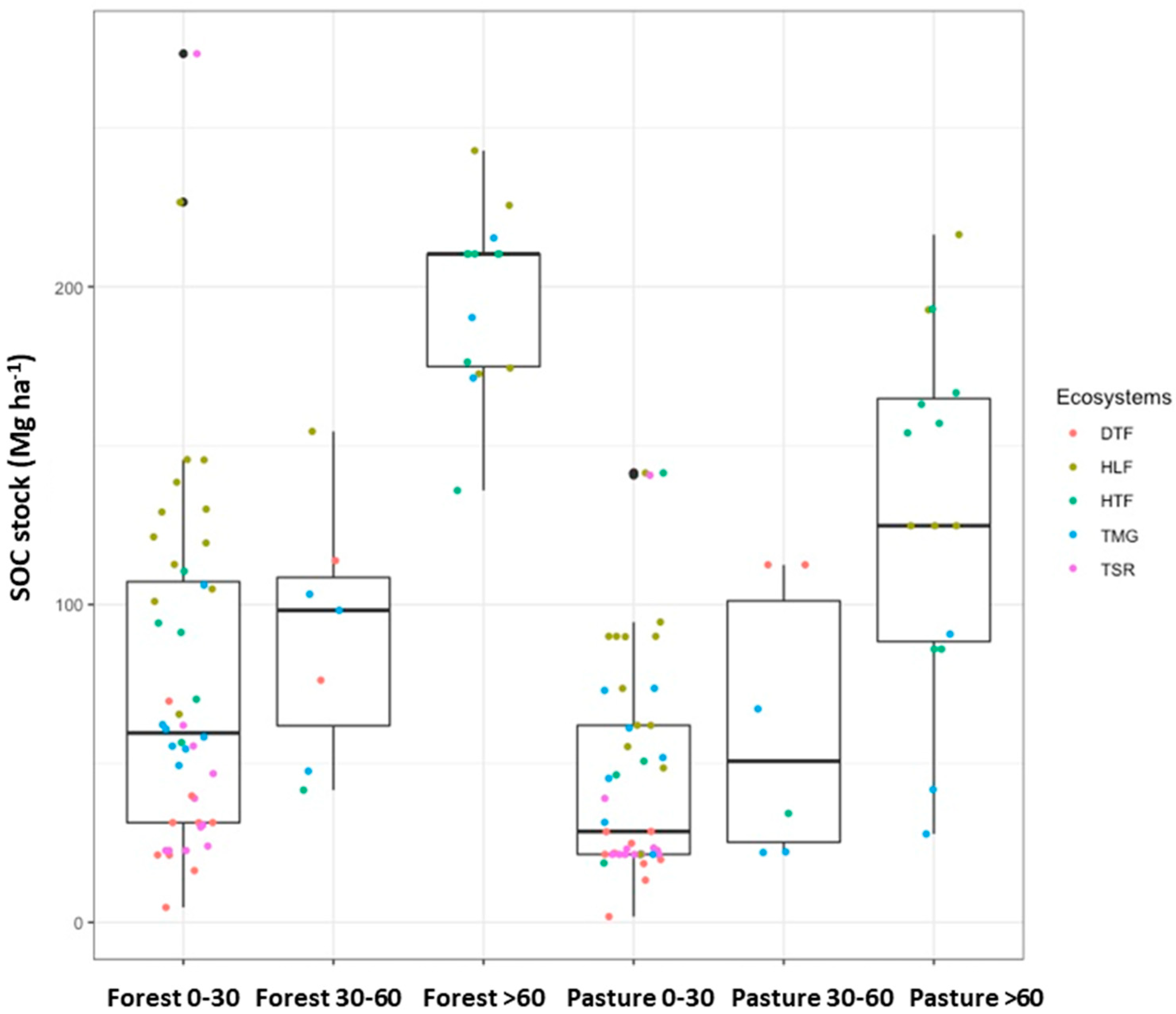
3.1. Soil Organic Carbon Loss from Land Use Change
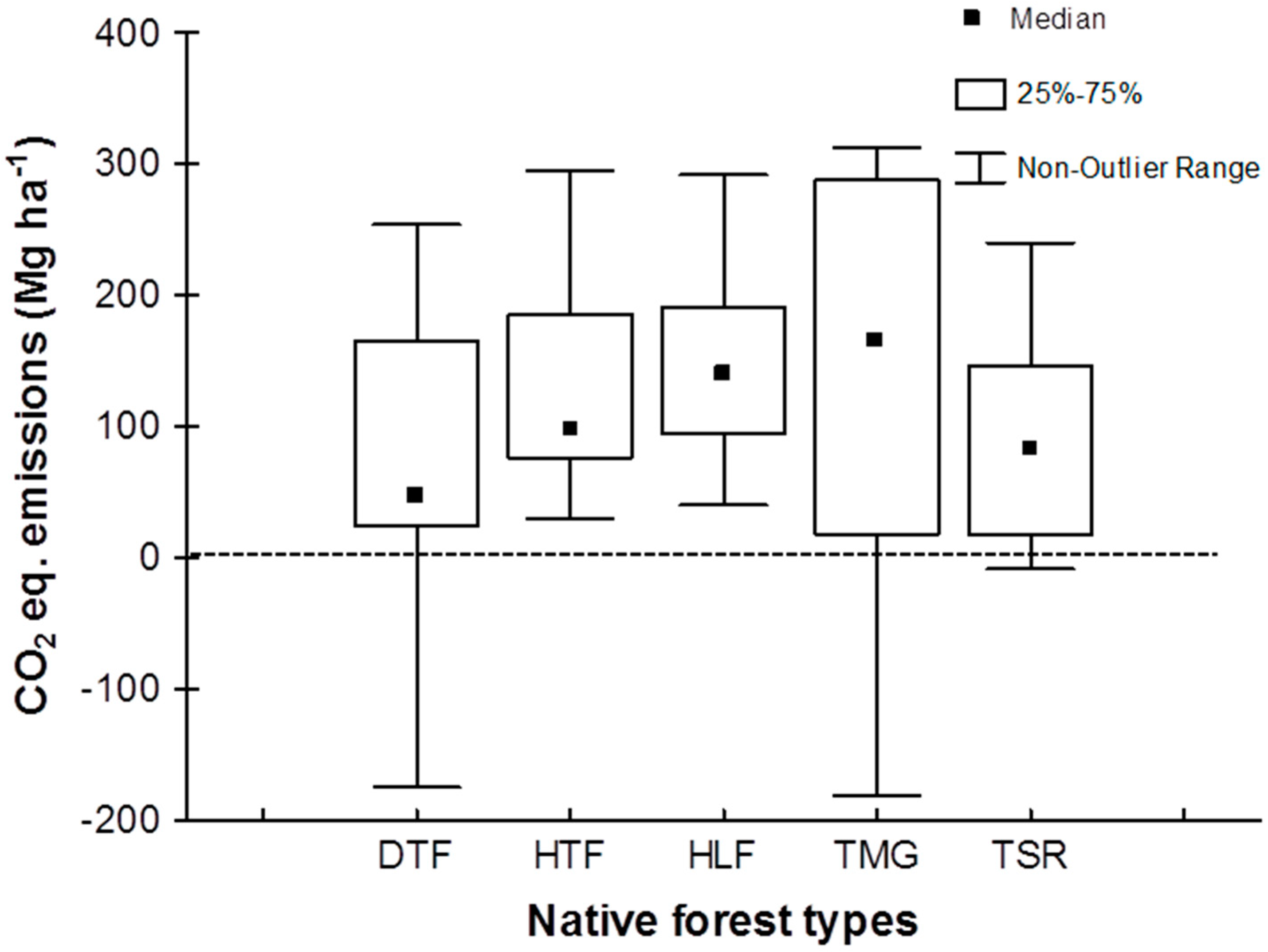
3.2. CO2 Equivalent Emissions Related to Land Conversion
4. Discussion
4.1. Soil Organic Carbon Loss
4.2. Management Recommendations for Carbon Stock Enhancement in Tropical Grasslands
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ciais, P.; Sabine, C.; Bala, G.; Bopp, L.; Brovkin, V.; Canadell, J.; Chhabra, A.; De Fries, R.; Galloway, J.; Heimann, M.; et al. Carbon and Other Biogeochemical Cycles. In Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Stocker, T.F., Qin, D., Plattner, G.-K., Tignor, M., Allen, S.K., Boschung, J., Nauels, A., Xia, Y., Bex, V., Midgley, P.M., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2013. [Google Scholar]
- Elser, J.; Bennett, E. Phosphorus cycle: A broken biogeochemical cycle. Nature 2011, 478, 29–31. [Google Scholar] [CrossRef] [PubMed]
- Foley, J.A.; Ramankutty, N.; Brauman, K.A.; Cassidy, E.S.; Gerber, J.S.; Johnston, M.; Mueller, N.D.; O’Connell, C.; Ray, D.K.; West, P.C. Solutions for a cultivated planet. Nature 2011, 478, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, D.L.; Klein Tank, A.M.G.; Rusticucci, M.; Alexander, L.V.; Brönnimann, S.; Charabi, Y.; Dentener, F.J.; Dlugokencky, E.J.; Easterling, D.R.; Kaplan, A. Observations: Atmosphere and surface. In Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Stocker, T.F., Qin, D., Plattner, G.-K., Tignor, M., Allen, S.K., Boschung, J., Nauels, A., Xia, Y., Bex, V., Midgley, P.M., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2013. [Google Scholar]
- Smith, P.; Bustamante, M.; Anammad, H.; Clark, H.; Dong, H.; Elsidddig, E.A.; Haberl, H.; Harper, R.; House, J.; Jafari, M.; et al. Agriculture, Forestry and Other Land Use (AFOLU). In Climate Change 2014: Mitigation of Climate Change. Contribution of Working Group III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Edenhofer, O., Pichs-Madruga, R., Sokona, Y., Farahani, E., Kadner, S., Seyboth, K., Adler, A., Baum, I., Brunner, S., Eickemeier, P., et al., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2014. [Google Scholar]
- Tubiello, F.N.; Salvatore, M.; Ferrara, A.F.; House, J.; Federici, S.; Rossi, S.; Biancalani, R.; Condor Golec, R.D.; Jacobs, H.; Flammini, A. The contribution of agriculture, forestry and other land use activities to global warming, 1990–2012. Glob. Chang. Biol. 2015, 21, 2655–2660. [Google Scholar] [CrossRef] [PubMed]
- FAO. FAOSTAT: Food and Agriculture Data; Food and Agriculture Organization: Roma, Italy, 2017. [Google Scholar]
- De Jong, B.; Anaya, C.; Masera, O.; Olguín, M.; Paz, F.; Etchevers, J.; Martínez, R.D.; Guerrero, G.; Balbontín, C. Greenhouse gas emissions between 1993 and 2002 from land-use change and forestry in Mexico. For. Ecol. Manag. 2010, 260, 1689–1701. [Google Scholar] [CrossRef]
- Gerber, P.J.; Steinfeld, H.; Henderson, B.; Mottet, A.; Opio, C.; Dijkman, J.; Falcucci, A.; Tempio, G. Tackling Climate Change through Livestock; FAO: Roma, Italy, 2013. [Google Scholar]
- Gómez-Castro, H.; Tewolde, A.M.; Nahed-Toral, J. Análisis de los sistemas ganaderos de doble propósito en el centro de Chiapas, México. Arch. Latinoam. Prod. Anim. 2002, 10, 175–183. [Google Scholar]
- Lal, R. Soil carbon sequestration impacts on global climate change and food security. Science 2004, 304, 1623–1627. [Google Scholar] [CrossRef] [PubMed]
- Navarrete, D.; Sitch, S.; Aragão, L.E.; Pedroni, L. Conversion from forests to pastures in the Colombian Amazon leads to contrasting soil carbon dynamics depending on land management practices. Glob. Chang. Biol. 2016, 22, 3503–3517. [Google Scholar] [CrossRef] [PubMed]
- Velázquez, A.; Mas, J.F.; Gallegos, J.R.D.; Mayorga-Saucedo, R.; Alcántara, P.C.; Castro, R.; Fernández, T.; Bocco, G.; Ezcurra, Y.E.; Palacio, J.L. Patrones y tasas de cambio de uso del suelo en México. Gac. Ecol. 2002, 62, 21–37. [Google Scholar]
- Alkemade, R.; Reid, R.S.; van den Berg, M.; de Leeuw, J.; Jeuken, M. Assessing the impacts of livestock production on biodiversity in rangeland ecosystems. Proc. Natl. Acad. Sci. USA 2013, 110, 20900–20905. [Google Scholar] [CrossRef] [PubMed]
- Aryal, D.R.; Geissen, V.; Ponce-Mendoza, A.; Ramos-Reyes, R.R.; Becker, M. Water quality under intensive banana production and extensive pastureland in tropical Mexico. J. Plant Nutr. Soil Sci. 2012, 175, 553–559. [Google Scholar] [CrossRef]
- Smith, P.; House, J.I.; Bustamante, M.; Sobocká, J.; Harper, R.; Pan, G.; West, P.C.; Clark, J.M.; Adhya, T.; Rumpel, C. Global change pressures on soils from land use and management. Glob. Chang. Biol. 2016, 22, 1008–1028. [Google Scholar] [CrossRef] [PubMed]
- Calle, Z.; Murgueitio, E.; Chará, J.; Molina, C.H.; Zuluaga, A.F.; Calle, A. A strategy for scaling-up Intensive Silvopastoral Systems in Colombia. J. Sustain. For. 2013, 32, 677–693. [Google Scholar] [CrossRef]
- Gaitán, L.; Läderach, P.; Graefe, S.; Rao, I.; van der Hoek, R. Climate-Smart Livestock Systems: An Assessment of Carbon Stocks and GHG Emissions in Nicaragua. PLoS ONE 2016, 11, e0167949. [Google Scholar] [CrossRef] [PubMed]
- Villanueva-López, G.; Martínez-Zurimendi, P.; Ramírez-Avilés, L.; Aryal, D.R.; Casanova-Lugo, F. Live fences reduce the diurnal and seasonal fluctuations of soil CO2 emissions in livestock systems. Agron. Sustain. Dev. 2016, 36, 1–8. [Google Scholar] [CrossRef]
- Cubillos, A.M.; Vallejo, V.E.; Arbeli, Z.; Terán, W.; Dick, R.P.; Molina, C.H.; Molina, E.; Roldan, F. Effect of the conversion of conventional pasture to intensive silvopastoral systems on edaphic bacterial and ammonia oxidizer communities in Colombia. Eur. J. Soil Biol. 2016, 72, 42–50. [Google Scholar] [CrossRef]
- Quintero, M.; Sachet, E.; Wyckhuys, K.A.; Cordingley, J.E.; Kizito, F.; Cruz-García, G.S.; Winowiecki, L.A.; Rajasekharan, M.; Valbuena, D.; Chirinda, N. Ecosystem Action: CIAT’s Ecosystem Services Strategic Initiative; Centro Internacional de Agricultura Tropical (CIAT): Cali, Colombia, 2015. [Google Scholar]
- Aryal, D.R.; De Jong, B.H.; Ochoa-Gaona, S.; Esparza-Olguin, L.; Mendoza-Vega, J. Carbon stocks and changes in tropical secondary forests of southern Mexico. Agric. Ecosyst. Environ. 2014, 195, 220–230. [Google Scholar] [CrossRef]
- Aryal, D.R.; De Jong, B.H.; Ochoa-Gaona, S.; Mendoza-Vega, J.; Esparza-Olguin, L. Successional and seasonal variation in litterfall and associated nutrient transfer in semi-evergreen tropical forests of SE Mexico. Nutr. Cycl. Agroecosyst. 2015, 103, 45–60. [Google Scholar] [CrossRef]
- Aryal, D.R.; De Jong, B.H.J.; Mendoza-Vega, J.; Ochoa-Gaona, S.; Esparza-Olguín, L. Soil Organic Carbon Stocks and Soil Respiration in Tropical Secondary Forests in Southern Mexico. In Global Soil Security; Springer: Cham, Switzerland, 2017; pp. 153–165. [Google Scholar]
- Muñoz-Rojas, M.; Lewandrowski, W.; Erickson, T.E.; Dixon, K.W.; Merritt, D.J. Soil respiration dynamics in fire affected semi-arid ecosystems: Effects of vegetation type and environmental factors. Sci. Total Environ. 2016, 572, 1385–1394. [Google Scholar] [CrossRef] [PubMed]
- Jose, S. Agroforestry for ecosystem services and environmental benefits: An overview. Agrofor. Syst. 2009, 76, 1–10. [Google Scholar] [CrossRef]
- Montagnini, F.; Nair, P.K.R. Carbon sequestration: An underexploited environmental benefit of agroforestry systems. Agrofor. Syst. 2004, 61, 281–295. [Google Scholar] [CrossRef]
- Nair, R.P.K.; Mohan, K.B.; Nair, V.D. Agroforestry as a strategy for carbon sequestration. J. Plant Nutr. Soil Sci. 2009, 172, 10–23. [Google Scholar] [CrossRef]
- Cotler, H.; Ortega-Larrocea, M.P. Effects of land use on soil erosion in a tropical dry forest ecosystem, Chamela watershed, Mexico. Catena 2006, 65, 107–117. [Google Scholar] [CrossRef]
- García-Oliva, F.; Casar, I.; Morales, P.; Maass, J.M. Forest-to-pasture conversion influences on soil organic carbon dynamics in a tropical deciduous forest. Oecologia 1994, 99, 392–396. [Google Scholar] [CrossRef] [PubMed]
- García-Oliva, F.; Lancho, J.F.G.; Montaño, N.M.; Islas, P. Soil carbon and nitrogen dynamics followed by a forest-to-pasture conversion in western Mexico. Agrofor. Syst. 2006, 66, 93–100. [Google Scholar] [CrossRef]
- Jaramillo, V.J.; Kauffman, J.B.; Rentería-Rodríguez, L.; Cummings, D.L.; Ellingson, L.J. Biomass, carbon, and nitrogen pools in Mexican tropical dry forest landscapes. Ecosystems 2003, 6, 609–629. [Google Scholar] [CrossRef]
- Sandoval-Pérez, A.L.; Gavito, M.E.; García-Oliva, F.; Jaramillo, V.J. Carbon, nitrogen, phosphorus and enzymatic activity under different land uses in a tropical, dry ecosystem. Soil Use Manag. 2009, 25, 419–426. [Google Scholar] [CrossRef]
- Segura-Castruita, M.A.; Sánchez-Guzmán, P.; Ortiz-Solorio, C.A.; Gutiérrez-Castorena, M.C. Carbono orgánico de los suelos de México. Terra Latinoam. 2005, 23, 21–28. [Google Scholar]
- Trilleras, J.M.; Jaramillo, V.J.; Vega, E.V.; Balvanera, P. Effects of livestock management on the supply of ecosystem services in pastures in a tropical dry region of western Mexico. Agric. Ecosyst. Environ. 2015, 211, 133–144. [Google Scholar] [CrossRef]
- Campos, A.; Oleschko, K.; Etchevers, J.; Hidalgo, C. Exploring the effect of changes in land use on soil quality on the eastern slope of the Cofre de Perote Volcano (Mexico). For. Ecol. Manag. 2007, 248, 174–182. [Google Scholar] [CrossRef]
- De Jong, B.H.; Ochoa-Gaona, S.; Castillo-Santiago, M.A.; Ramírez-Marcial, N.; Cairns, M.A. Carbon flux and patterns of land-use/land-cover change in the Selva Lacandona, Mexico. AMBIO J. Hum. Environ. 2000, 29, 504–511. [Google Scholar] [CrossRef]
- Geissen, V.; Sánchez-Hernández, R.; Kampichler, C.; Ramos-Reyes, R.; Sepulveda-Lozada, A.; Ochoa-Goana, S.; De Jong, B.H.J.; Huerta-Lwanga, E.; Hernández-Daumas, S. Effects of land-use change on some properties of tropical soils—An example from Southeast Mexico. Geoderma 2009, 151, 87–97. [Google Scholar] [CrossRef]
- Hughes, R.F.; Kauffman, J.B.; Jaramillo, V.J. Ecosystem-scale impacts of deforestation and land use in a humid tropical region of Mexico. Ecol. Appl. 2000, 10, 515–527. [Google Scholar] [CrossRef]
- Jaramillo, V.J.; Ahedo-Hernández, R.; Kauffman, J.B. Root biomass and carbon in a tropical evergreen forest of Mexico: Changes with secondary succession and forest conversion to pasture. J. Trop. Ecol. 2003, 19, 457–464. [Google Scholar] [CrossRef]
- Roa-Fuentes, L.L.; Martínez-Garza, C.; Etchevers, J.; Campo, J. Recovery of soil C and N in a tropical pasture: Passive and active restoration. Land Degrad. Dev. 2015, 26, 201–210. [Google Scholar] [CrossRef]
- Tobón, W.; Martínez-Garza, C.; Campo, J. Soil responses to restoration of a tropical pasture in Veracruz, south-eastern Mexico. J. Trop. For. Sci. 2011, 23, 338–344. [Google Scholar]
- Álvarez-Arteaga, G.; Fajardo, B.G.; Hernández, M.E.O.; Lezama, P.M.; Martínez, J.C. Estimation of carbon stocks under different soil uses in the central highlands of Mexico. Acta Agron. 2017, 66, 21–26. [Google Scholar] [CrossRef]
- Covaleda, S.; Gallardo, J.F.; García-Oliva, F.; Kirchmann, H.; Prat, C.; Bravo, M.; Etchevers, J.D. Land-use effects on the distribution of soil organic carbon within particle-size fractions of volcanic soils in the Transmexican Volcanic Belt (Mexico). Soil Use Manag. 2011, 27, 186–194. [Google Scholar] [CrossRef]
- De Jong, B.H.; Cairns, M.A.; Haggerty, P.K.; Ramirez-Marcial, N.; Ochoa-Gaona, S.; Mendoza-Vega, J.; March-Mifsut, I. Land-use change and carbon flux between 1970s and 1990s in central highlands of Chiapas, Mexico. Environ. Manag. 1999, 23, 373–385. [Google Scholar] [CrossRef]
- Gamboa, A.M.; Galicia, L. Land-use/cover change effects and carbon controls on volcanic soil profiles in highland temperate forests. Geoderma 2012, 170, 390–402. [Google Scholar] [CrossRef]
- González, L.; Etchevers, J.D.; González, J.M.; Paz, F. Soil organic carbon changes at the plot level in hillside systems. Agric. Ecosyst. Environ. 2010, 139, 508–515. [Google Scholar]
- Ordóñez, J.A.B.; de Jong, B.H.; García-Oliva, F.; Aviña, F.L.; Pérez, J.V.; Guerrero, G.; Martínez, R.; Masera, O. Carbon content in vegetation, litter, and soil under 10 different land-use and land-cover classes in the Central Highlands of Michoacan, Mexico. For. Ecol. Manag. 2008, 255, 2074–2084. [Google Scholar] [CrossRef]
- Vela Correa, G.; López Blanco, J.; Rodríguez Gamiño, M.D.L. Niveles de carbono orgánico total en el Suelo de Conservación del Distrito Federal, centro de México. Investig. Geogr. 2012, 77, 18–30. [Google Scholar]
- Ibarra-Flores, F.; Cox, J.R.; Martin-Rivera, M.; Crowl, T.A.; Norton, B.E.; Banner, R.E.; Miller, R.W. Soil physicochemical changes following buffelgrass establishment in Mexico. Arid Soil Res. Rehabil. 1999, 13, 39–52. [Google Scholar] [CrossRef]
- Morales-Romero, D.; Campo, J.; Godinez-Alvarez, H.; Molina-Freaner, F. Soil carbon, nitrogen and phosphorus changes from conversion of thornscrub to buffelgrass pasture in northwestern Mexico. Agric. Ecosyst. Environ. 2015, 199, 231–237. [Google Scholar] [CrossRef]
- Kauffman, J.B.; Trejo, H.H.; Garcia, M.D.C.J.; Heider, C.; Contreras, W.M. Carbon stocks of mangroves and losses arising from their conversion to cattle pastures in the Pantanos de Centla, Mexico. Wetl. Ecol. Manag. 2016, 24, 203–216. [Google Scholar] [CrossRef]
- Adame, M.F.; Kauffman, J.B.; Medina, I.; Gamboa, J.N.; Torres, O.; Caamal, J.P.; Reza, M.; Herrera-Silveira, J.A. Carbon stocks of tropical coastal wetlands within the karstic landscape of the Mexican Caribbean. PLoS ONE 2013, 8, e56569. [Google Scholar] [CrossRef] [PubMed]
- Guerra-Santos, J.J.; Cerón-Bretón, R.M.; Cerón-Bretón, J.G.; Damián-Hernández, D.L.; Sánchez-Junco, R.C.; Carrió, E.D.C.G. Estimation of the carbon pool in soil and above-ground biomass within mangrove forests in Southeast Mexico using allometric equations. J. For. Res. 2014, 25, 129–134. [Google Scholar] [CrossRef]
- Rzedowski, J. Vegetación de México, 1ra. Edición Digital; Comisión Nacional para el Conocimiento y Uso de la Biodiversidad: México, Mexico, 2006. [Google Scholar]
- INEGI. Uso de Suelo y Vegetación: Serie V [WWW Document]. 2013. Available online: http://www.inegi.org.mx/geo/contenidos/recnat/usosuelo/ (accessed on 20 April 2018).
- Fóti, S.; Balogh, J.; Herbst, M.; Papp, M.; Koncz, P.; Bartha, S.; Zimmermann, Z.; Komoly, C.; Szabó, G.; Margóczi, K.; et al. Meta-analysis of field scale spatial variability of grassland soil CO2 efflux: Interaction of biotic and abiotic drivers. Catena 2016, 143, 78–89. [Google Scholar] [CrossRef]
- Zhou, G.; Zhou, X.; He, Y.; Shao, J.; Hu, Z.; Liu, R.; Zhou, H.; Hosseinibai, S. Grazing intensity significantly affects belowground carbon and nitrogen cycling in grassland ecosystems: A meta-analysis. Glob. Chang. Biol. 2016, 23, 1167–1179. [Google Scholar] [CrossRef] [PubMed]
- Batjes, N.H. A world dataset of derived soil properties by FAO-UNESCO soil unit for global modelling. Soil Use Manag. 1997, 13, 9–16. [Google Scholar] [CrossRef]
- Bates, D.; Maechler, M.; Bolker, B.; Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Hothorn, T.; Bretz, F.; Westfall, P. Simultaneous inference in general parametric models. Biom. J. 2008, 50, 346–363. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018; Available online: https://www.R-project.org/ (accessed on 23 October 2018).
- Murty, D.; Kirschbaum, M.U.; Mcmurtrie, R.E.; Mcgilvray, H. Does conversion of forest to agricultural land change soil carbon and nitrogen? A review of the literature. Glob. Chang. Biol. 2002, 8, 105–123. [Google Scholar] [CrossRef]
- Yadav, R.P.; Bisht, J.K.; Bhatt, J.C. Biomass, carbon stock under different production systems in the mid hills of Indian Himalaya. Trop. Ecol. 2017, 58, 15–21. [Google Scholar]
- Shanmugam, S.; Dalal, R.C.; Joosten, H.; Raison, R.J.; Joo, J.K. SOC stock changes and greenhouse gas emissions following tropical land use conversions to plantation crops on mineral soils, with a special focus on oil palm and rubber plantations. Agriculture 2018, 8, 133. [Google Scholar] [CrossRef]
- Aryal, D.R.; Gómez-González, R.R.; Hernández-Nuriasmú, R.; Morales-Ruiz, D.E. Carbon stocks and tree diversity in scattered tree silvopastoral systems in Chiapas, Mexico. Agrofor. Syst. 2018. [Google Scholar] [CrossRef]
- De Jong, B.H. Spatial distribution of biomass and links to reported disturbances in tropical lowland forests of southern Mexico. Carbon Manag. 2013, 4, 601–615. [Google Scholar] [CrossRef]
- Nahed-Toral, J.; Valdivieso-Pérez, A.; Aguilar-Jiménez, R.; Cámara-Cordova, J.; Grande-Cano, D. Silvopastoral systems with traditional management in southeastern Mexico: A prototype of livestock agroforestry for cleaner production. J. Clean. Prod. 2013, 57, 266–279. [Google Scholar] [CrossRef]
- Tonucci, R.G.; Nair, P.K.; Nair, V.D.; Garcia, R.; Bernardino, F.S. Soil carbon storage in silvopasture and related land-use systems in the Brazilian Cerrado. J. Environ. Qual. 2011, 40, 833–841. [Google Scholar] [CrossRef] [PubMed]
- Fearnside, P.M.; Imbrozio Barbosa, R. Soil carbon changes from conversion of forest to pasture in Brazilian Amazonia. For. Ecol. Manag. 1998, 108, 147–166. [Google Scholar] [CrossRef]
- Boval, M.; Angeon, V.; Rudel, T. Tropical grasslands: A pivotal place for a more multi-functional agriculture. Ambio 2017, 46, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Joyce, L.A.; Briske, D.D.; Brown, J.R.; Polley, H.W.; McCarl, B.A.; Bailey, D.W. Climate Change and North American Rangelands: Assessment of Mitigation and Adaptation Strategies. Rangel. Ecol. Manag. 2013, 66, 512–528. [Google Scholar] [CrossRef]
- Villanueva-López, G.; Martínez-Zurimendi, P.; Casanova-Lugo, F.; Ramírez-Avilés, L.; Montañez-Escalante, P.I. Carbon storage in livestock systems with and without live fences of Gliricidia sepium in the humid tropics of Mexico. Agrofor. Syst. 2015, 89, 1083–1096. [Google Scholar] [CrossRef]
- Cárdenas, A.; Moliner, A.; Hontoria, C.; Ibrahim, M. Ecological structure and carbon storage in traditional silvopastoral systems in Nicaragua. Agrofor. Syst. 2018. [Google Scholar] [CrossRef]
- Casanova-Lugo, F.; Petit-Aldana, J.; Solorio-Saánchez, F.; Ramírez-Avilés, L.; Ward, S.E.; Villanueva-López, G.; Aryal, D.R. Carbon stocks in biomass and soils of woody species fodder banks in the dry tropics of Mexico. Soil Use Manag. 2018. [Google Scholar] [CrossRef]
- López-Santiago, J.G.; Casanova-Lugo, F.; Villanueva-López, G.; Díaz-Echeverría, V.F.; Solorio-Sánchez, F.J.; Martínez-Zurimendi, P.; Aryal, D.R.; Chay-Canul, A.J. Carbon storage in a silvopastoral system compared to that in a deciduous dry forest in Michoacán, Mexico. Agrofor. Syst. 2018. [Google Scholar] [CrossRef]
- Aryal, D.R.; Gómez-Castro, H.; Del Carmen-García, N.; José-Ruiz, O.; Molina-Paniagua, L.F.; Jimenez-Trujillo, J.A.; Venegas-Venegas, J.A.; Pinto-Ruiz, R.; Ley de Coss, A.; Guevara-Hernández, F. Carbon storage potential in forest areas within a livestock system of Villaflores, Chiapas, Mexico. Rev. Mex. Cienc. For. 2018, 9, 150–180. [Google Scholar] [CrossRef]
- Dube, F.; Thevathasan, N.V.; Zagal, E.; Gordon, A.M.; Stolpe, N.B.; Espinosa, M. Carbon Sequestration Potential of Silvopastoral and Other Land Use Systems in the Chilean Patagonia. In Carbon Sequestration Potential of Agroforestry Systems: Opportunities and Challenges; Kumar, B.M., Nair, P.K.R., Eds.; Springer: Dordrecht, The Netherlands, 2011; pp. 101–127. [Google Scholar]
- Pandey, C.B.; Singh, G.B.; Singh, S.K.; Singh, R.K. Soil nitrogen and microbial biomass carbon dynamics in native forests and derived agricultural land uses in a humid tropical climate of India. Plant Soil 2010, 333, 453–467. [Google Scholar] [CrossRef]
- Casals, P.; Romero, J.; Rusch, G.M.; Ibrahim, M. Soil organic C and nutrient contents under trees with different functional characteristics in seasonally dry tropical silvopastures. Plant Soil 2014, 374, 643–659. [Google Scholar] [CrossRef]
- Andrade, H.J.; Brook, R.; Ibrahim, M. Growth, production and carbon sequestration of silvopastoral systems with native timber species in the dry lowlands of Costa Rica. Plant Soil 2008, 308, 11–22. [Google Scholar] [CrossRef]
- Ibrahim, M.; Villanueva, C.; Casasola, F.; Rojas, J. Sistemas silvopastoriles como una herramienta para el mejoramiento de la productividad y restauración de la integridad ecológica de paisajes ganaderos. Pastos Forrajes 2006, 29, 383–419. [Google Scholar]
- Adewopo, J.B.; Silveira, M.L.; Xu, S.; Gerber, S.; Sollenberger, L.E.; Martin, T.A. Management intensification impacts on soil and ecosystem carbon stocks in subtropical grasslands. Soil Sci. Soc. Am. J. 2014, 78, 977–986. [Google Scholar] [CrossRef]
- Conant, R.T.; Paustian, K.; Elliott, E.T. Grassland management and conversion into grassland: Effects on soil carbon. Ecol. Appl. 2001, 11, 343–355. [Google Scholar] [CrossRef]
- Medina-Roldán, E.; Arredondo, J.T.; Huber-Sannwald, E.; Chapa-Vargas, L.; Olalde-Portugal, V. Grazing effects on fungal root symbionts and carbon and nitrogen storage in a shortgrass steppe in Central Mexico. J. Arid Environ. 2008, 72, 546–556. [Google Scholar] [CrossRef]
- Lamb, A.; Green, R.; Bateman, I.; Broadmeadow, M.; Bruce, T.; Burney, J. The potential for land sparing to offset greenhouse gas emissions from agriculture. Nat. Clim. Chang. 2016. [Google Scholar] [CrossRef]
- Wollenberg, E.; Richards, M.; Smith, P.; Havlík, P.; Obersteiner, M.; Tubiello, F.N.; Herold, M.; Gerber, P.; Carter, S.; Reisinger, A. Reducing emissions from agriculture to meet the 2 C target. Glob. Chang. Biol. 2016, 22, 3859–3864. [Google Scholar] [CrossRef] [PubMed]
| Principal Land Use/Land Cover | 1976 (km2) a | 1993 (km2) a | 2000 (km2) a | 2013 (km2) b |
|---|---|---|---|---|
| Highland forests | 352,049 | 347,084 | 331,236 | 345,721.49 |
| Humid and dry tropical forests | 377,598 | 352,798 | 314,340 | 357,620.53 |
| Scrublands | 607,472 | 572,118 | 560,791 | 598,883.43 |
| Grasslands | 254,396 | 268,700 | 311,375 | 272,050.63 |
| Natural | 104,779 | 94,947 | 86,240 | 82,477.32 |
| Cultivated and induced | 149,617 | 173,753 | 225,135 | 189,573.31 |
| Annual and perennial cropland | 262,389 | 290,325 | 325,057 | 325,967.30 |
| Wetland | 22,941 | 22,223 | 18,887 | 10,457.18 |
| Estimate | Std. Error | t Value | Pr(>|z|) | Significance 1 | |
|---|---|---|---|---|---|
| (Intercept) | 0.0222 | 0.0040 | 5.5229 | <0.001 | *** |
| LandUses Pasture | 0.0064 | 0.0015 | 4.2810 | <0.001 | *** |
| CategoriesD30–60 | 0.0003 | 0.0032 | 0.1067 | 0.915 | |
| CategoriesD > 60 | −0.0084 | 0.0025 | −3.3496 | <0.001 | *** |
| LandUsesPasture:CategoriesD30–60 | −0.0032 | 0.0037 | −0.8480 | 0.396 | |
| LandUsesPasture:CategoriesD > 60 | −0.0038 | 0.0018 | −2.0919 | 0.036 | * |
| Factors | p Value | Significance 1 | Logical Relation 2 |
|---|---|---|---|
| Pasture 0–30 versus Forest 0–30 | <0.001 | *** | Yes |
| Forest 30–60 versus Forest 0–30 | 0.999 | Yes | |
| Pasture 30–60 versus Forest 0–30 | 0.925 | No | |
| Forest >60 versus Forest 0–30 | 0.008 | *** | Yes |
| Pasture >60 versus Forest 0–30 | 0.181 | No | |
| Forest 30–60 versus Pasture 0–30 | 0.366 | No | |
| Pasture 30–60 versus Pasture 0–30 | 0.972 | Yes | |
| Forest >60 versus Pasture 0–30 | <0.001 | *** | No |
| Pasture >60–Pasture 0–30 | <0.001 | *** | Yes |
| Pasture 30–60 versus Forest 30–60 | 0.920 | No | |
| Forest >60 versus Forest 30–60 | 0.048 | * | Yes |
| Pasture >60 versus Forest 30–60 | 0.341 | No | |
| Forest >60 versus Pasture 30–60 | 0.011 | * | No |
| Pasture >60 versus Pasture 30–60 | 0.100 | Yes | |
| Pasture >60 versus Forest >60 | 0.091 | Yes |
| Estimate | Std. Error | t Value | Pr(>|z|) | Significance 1 | |
|---|---|---|---|---|---|
| (Intercept) | 0.03024 | 0.00533 | 5.67421 | <0.001 | *** |
| LandUses Pasture | 0.00217 | 0.00401 | 0.54281 | 0.587 | |
| Ecosystems HLF | −0.01704 | 0.00568 | −2.99860 | 0.002 | ** |
| Ecosystems HTF | −0.01446 | 0.00586 | −2.46756 | 0.013 | * |
| Ecosystems TMG | −0.01887 | 0.00708 | −2.66579 | 0.007 | ** |
| Ecosystems TSR | −0.00262 | 0.00682 | −0.38423 | 0.700 | |
| LandUses Pasture:Ecosystems HLF | 0.00132 | 0.00418 | 0.31663 | 0.751 | |
| LandUses Pasture:Ecosystems HTF | 0.00002 | 0.00422 | 0.00427 | 0.996 | |
| LandUses Pasture:Ecosystems TMG | 0.00819 | 0.00487 | 1.68164 | 0.092 | |
| LandUses Pasture:Ecosystems TSR | 0.00663 | 0.00566 | 1.17103 | 0.241 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aryal, D.R.; Morales Ruiz, D.E.; Tondopó Marroquín, C.N.; Pinto Ruiz, R.; Guevara Hernández, F.; Venegas Venegas, J.A.; Ponce Mendoza, A.; Villanueva López, G.; Casanova Lugo, F.; Rodríguez Larramendi, L.A.; et al. Soil Organic Carbon Depletion from Forests to Grasslands Conversion in Mexico: A Review. Agriculture 2018, 8, 181. https://doi.org/10.3390/agriculture8110181
Aryal DR, Morales Ruiz DE, Tondopó Marroquín CN, Pinto Ruiz R, Guevara Hernández F, Venegas Venegas JA, Ponce Mendoza A, Villanueva López G, Casanova Lugo F, Rodríguez Larramendi LA, et al. Soil Organic Carbon Depletion from Forests to Grasslands Conversion in Mexico: A Review. Agriculture. 2018; 8(11):181. https://doi.org/10.3390/agriculture8110181
Chicago/Turabian StyleAryal, Deb Raj, Danilo Enrique Morales Ruiz, César Noé Tondopó Marroquín, René Pinto Ruiz, Francisco Guevara Hernández, José Apolonio Venegas Venegas, Alejandro Ponce Mendoza, Gilberto Villanueva López, Fernando Casanova Lugo, Luis Alfredo Rodríguez Larramendi, and et al. 2018. "Soil Organic Carbon Depletion from Forests to Grasslands Conversion in Mexico: A Review" Agriculture 8, no. 11: 181. https://doi.org/10.3390/agriculture8110181
APA StyleAryal, D. R., Morales Ruiz, D. E., Tondopó Marroquín, C. N., Pinto Ruiz, R., Guevara Hernández, F., Venegas Venegas, J. A., Ponce Mendoza, A., Villanueva López, G., Casanova Lugo, F., Rodríguez Larramendi, L. A., Ley de Coss, A., Hernández López, A., Medina Jonapá, F. J., Velázquez Sanabria, C. A., Alcudia Aguilar, A., & Euán Chi, I. (2018). Soil Organic Carbon Depletion from Forests to Grasslands Conversion in Mexico: A Review. Agriculture, 8(11), 181. https://doi.org/10.3390/agriculture8110181






