Effect of Magnesium on Gas Exchange and Photosynthetic Efficiency of Coffee Plants Grown under Different Light Levels
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
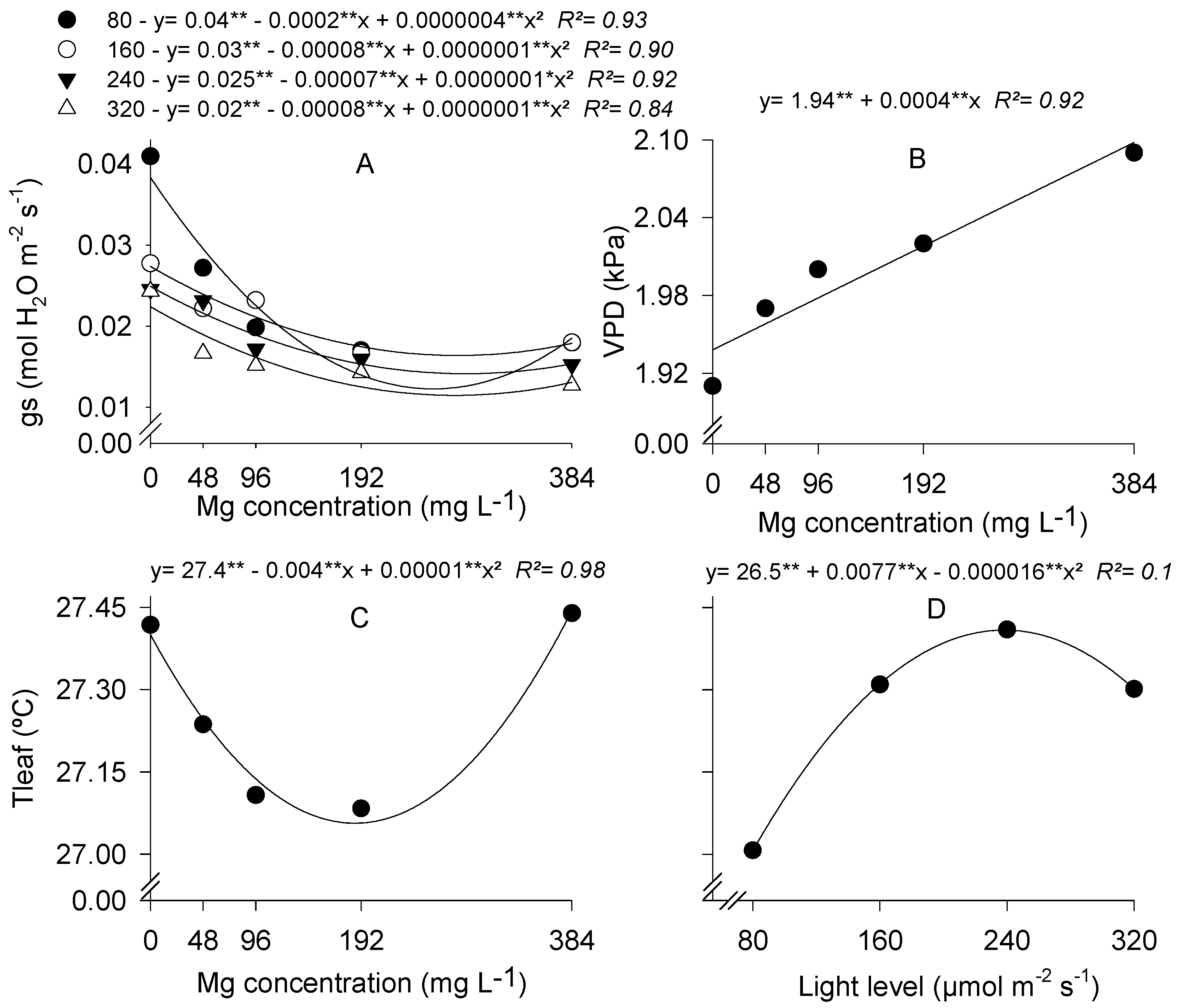
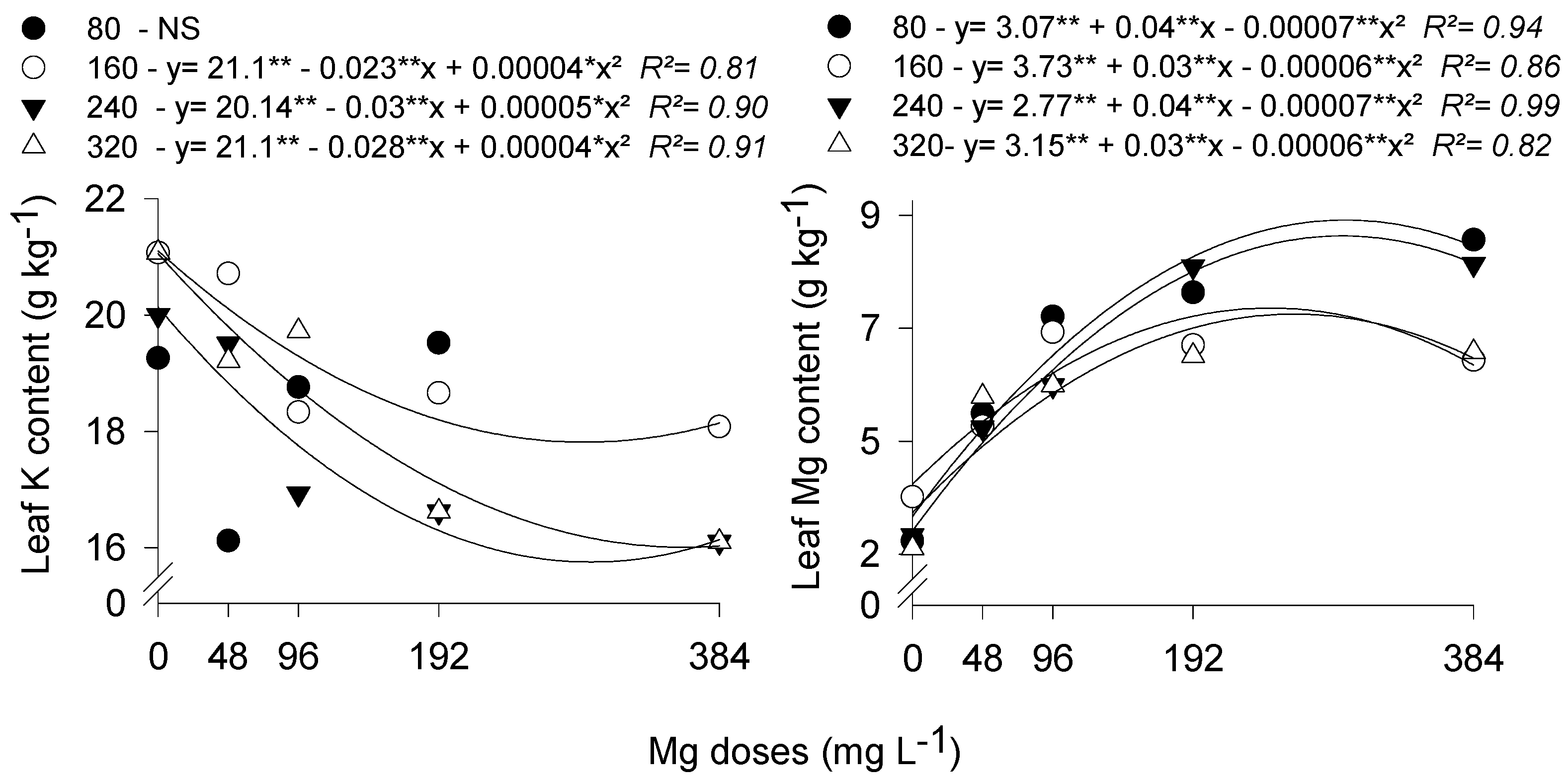
3.1. Stomatal Conductance and Leaf Temperature
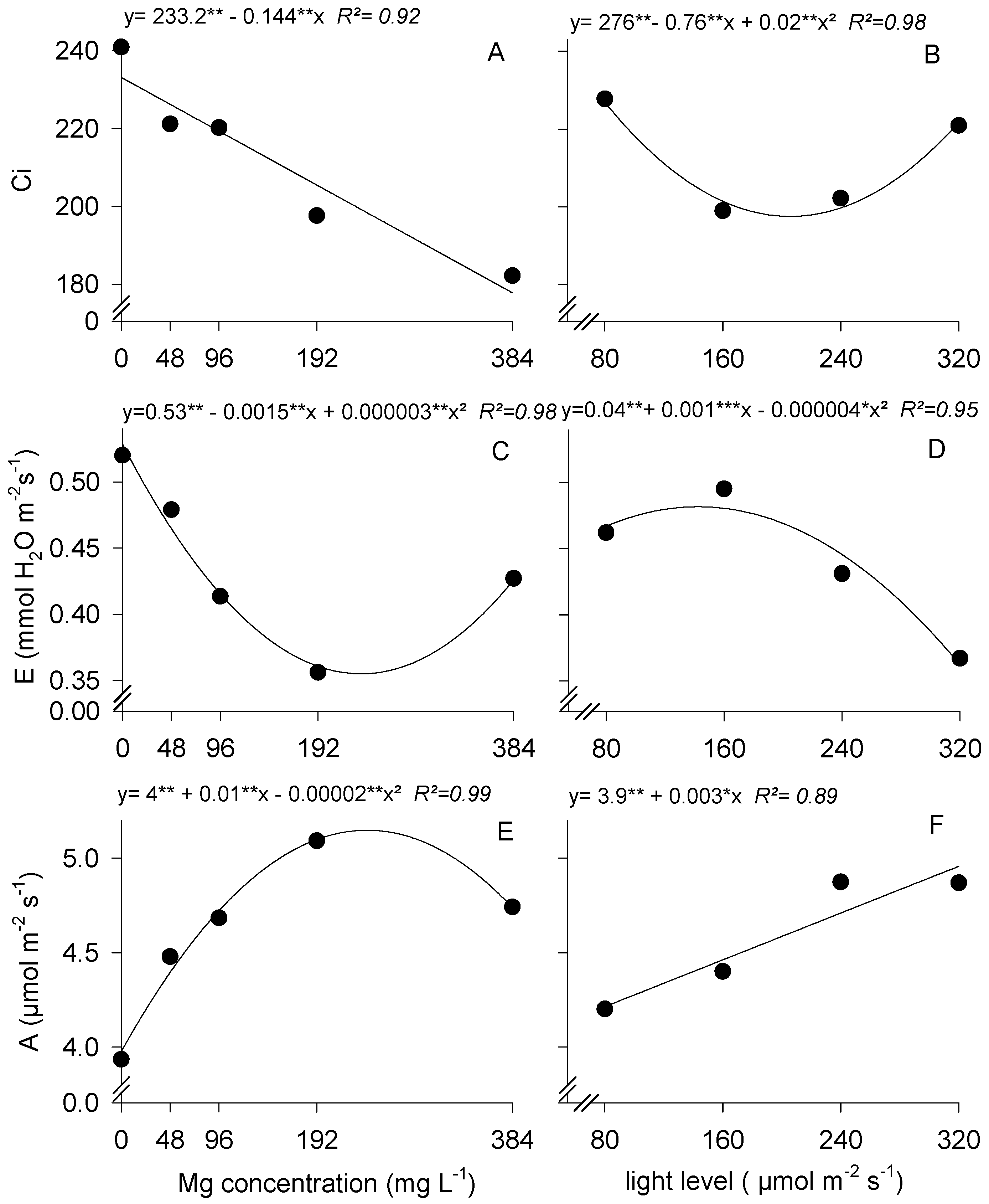
3.2. Gas Exchange
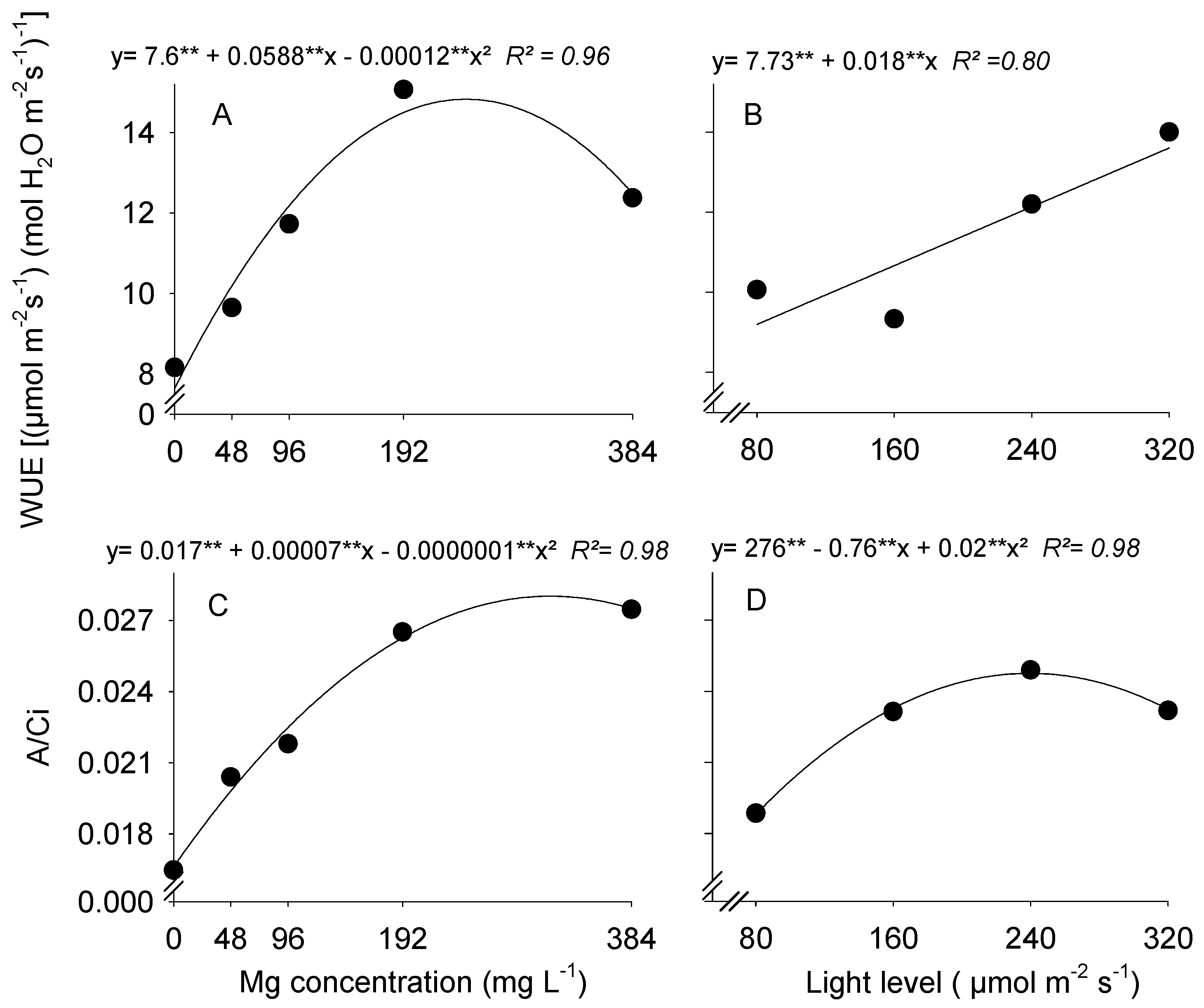
3.3. Water Use Efficiency and Instantaneous Carboxylation Efficiency
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Damatta, F.M.; Ronchi, C.P.; Maestri, M.; Barros, R.S. Ecophysiology of coffee growth and production. Braz. J. Plant Physiol. 2007, 19, 485–510. [Google Scholar] [CrossRef]
- Larcher, W. Ecofisiologia Vegetal [Plant Ecophysiology]; Rima: São Carlos, Brazil, 2000. [Google Scholar]
- Ronquim, J.C. Assimilação de Carbono e Fluorescência da Clorofila do Cafeeiro (Coffea arábica L.) sob Condições Contrastantes de Irradiância, Temperatura e Disponibilidade de CO2 [Carbon Assimilation and Chlorophyll Fluorescence of Coffee Trees (Coffea arábica L.) under Contrasting Conditions of Irradiance, Temperature and CO2 Availability]. Ph.D. Thesis, Federal University of São Carlos, São Carlos, Brazil, 2007. [Google Scholar]
- Damatta, F.M. Ecophysiological constraints on the production of shaded and unshaded coffee: A review. Field Crops Res. 2004, 86, 99–114. [Google Scholar] [CrossRef]
- Silveira, J.A.; Silva, S.L.F.; Silva, E.N.; Viégas, R.A. Mecanismos biomoleculares envolvidos com a resistência ao estresse salino em plantas [Biomolecular mechanisms involved in plant saline stress resistance]. In Manejo da Salinidade na Agricultura: Estudos Básicos e Aplicados [Salinity Management in Agriculture: Basic and Applied Studies]; Gheyi, H., Dias, N.S., Lacerda, C.F., Eds.; INCTSal: Fortaleza, Brazil, 2010; pp. 161–180. [Google Scholar]
- Taiz, L.; Zeiger, E. Fisiologia Vegetal, 5th ed.; Artmed: Porto Alegre, Brazil, 2013. [Google Scholar]
- Araújo, S.A.C.; Deminicis, B.B. Fotoinibição da fotossíntese [Photosynthesis Inhibition]. Rev. Bras. Biociênc. 2009, 7, 463–472. [Google Scholar]
- Oliveira, K.M.G.; Carvalho, L.G.; Lima, L.A.; Gomes, R.C.C. Modelagem para estimativa da orientação de linhas de plantio de cafeeiros [Modeling for estimating the orientation of planting rows of coffee trees]. Eng. Agrícola 2012, 32, 293–305. [Google Scholar] [CrossRef]
- Salati, E.; Santos, A.A.; Nobre, C. As Mudanças Climáticas Globais e seus Efeitos nos Ecossistemas Brasileiros [Global Climate Change and Its Effects on Brazilian Ecosystems]. 2012. Available online: www.comciencia.br/reportagens/clima/clima14.htm (accessed on 13 January 2015).
- Morais, H.; Caramori, P.H.; Ribeiro, A.M.A.; Gomes, J.C.; Koguishi, M.S. Microclimatic characterization and productivity of coffee plants grown under shade of pigeon pea in Southern Brazil. Pesqui. Agropecu. Bras. 2006, 41, 763–770. [Google Scholar] [CrossRef]
- Cakmak, I.; Yazici, A.M. Magnesium: A forgotten element in crop production. Better Crops Plant Food 2010, 94, 23–25. [Google Scholar]
- Cakmak, I.; Kirkby, E.A. Role of magnesium in carbon partitioning and alleviating photooxidative damage. Physiol. Plant. 2008, 133, 692–704. [Google Scholar] [CrossRef] [PubMed]
- Hoagland, D.R.; Arnon, D.I. The Water-Culture Method for Growing Plants without Soil; Agricultural Experiment Station: Berkley, CA, USA, 1950. [Google Scholar]
- Jaimez, R.E.; Rada, F.; Garcia-Núñez, C.; Azócar, A. Seasonal variations in leaf gas exchange of platain cv. Hartón (Musa AAB) under different soil water conditions in a humid tropical region. Sci. Hortic. 2005, 104, 79–89. [Google Scholar] [CrossRef]
- Melo, A.S.; Silva Júnior, C.D.; Fernandes, P.D.; Sobral, L.; Brito, M.E.B.; Dantas, J.D.M. Alterações das características fisiológicas da bananeira sob condições de fertirrigação [Alteration of the physiologic characteristics in banana under fertigation conditions]. Ciênc. Rural 2009, 39, 733–741. [Google Scholar] [CrossRef]
- Ferreira, D.F. SISVAR: A computer statistical analysis system. Ciênc. Agrotecnol. 2011, 35, 1039–1042. [Google Scholar] [CrossRef]
- Dias, K.G.L. Nutrição, Bioquímica e Fisiologia de Cafeeiros Supridos com Magnésio [Nutrition, Biochemistry and Physiology in Coffee Plants Supplied with Mg]. Ph.D. Thesis, Federal University of Lavras, Lavras-MG, Brazil, 2015. [Google Scholar]
- Marschner, H. Mineral Nutrition of Higher Plants, 3rd ed.; Academic Press: London, UK, 2012. [Google Scholar]
- Ding, Y.; Xu, G. Low magnesium with high potassium supply changes sugar partitioning and root growth pattern prior to visible magnesium deficiency in leaves of Rice (Oryza sativa L.). Am. J. Plant Sci. 2011, 2, 601–608. [Google Scholar] [CrossRef]
- Wallau, R.L.R.; Soares, A.P.; Camargos, S.L. Concentração e acúmulo de macronutrientes em mudas de mogno cultivadas em solução nutritiva. Rev. Ciênc. Agroambient. 2008, 6, 1–12. [Google Scholar]
- Marques, D.J.; Broetto, F.; Silva, E.C.; Carvalho, J.G. Dinâmica de cátions na raiz e folhas de berinjela cultivada sobre doses crescentes de potássio oriundas de duas fontes. Idesia (Arica) 2011, 29, 69–77. [Google Scholar] [CrossRef]
- Veigas, I.J.M.; Sousa, G.O.; Silva, A.F.; Carvalho, J.G.; Lima, M. M. Composição mineral e sintomas visuais de deficiências de nutrientes em plantas de pimenta-longa (Piper hispidinervum C. DC.). Acta Amaônica 2013, 43, 43–50. [Google Scholar] [CrossRef]
- Yong, J.W.H.; Wong, S.C.; Farquhar, G.D. Stomatal responses to changes in vapour pressure difference between leaf and air. Plant Cell Environ. 1997, 20, 1213–1216. [Google Scholar] [CrossRef]
- Marenco, R.A.; Siebke, K.; Farquhar, G.D.; Ball, M.C. Hydraulically based stomatal oscillations and stomatal patchiness in Gossypium hirsutum. Funct. Plant Biol. 2006, 33, 1103–1113. [Google Scholar] [CrossRef]
- Chaves, A.R.M.; Martins, S.C.V.; Batista, K.D.; Celin, E.F.; DaMatta, F.M. Varying leaf-to-fruit ratios affect branch growth and dieback, with little to no effect on photosynthesis, carbohydrate or mineral pools, in different canopy positions of field-grown coffee trees. Environ. Exp. Bot. 2012, 77, 207–218. [Google Scholar] [CrossRef]
- Cavatte, P.C.; Mrtins, S.V.C.; Wolfgramm, R.; DaMatta, F.M. Physiological responses of two coffee (Coffea canephora) genotypes to soil water deficit. In Droughts: Causes, Effects and Predictions; Sanchez, J.M., Ed.; Nova Science Publishers: New York, NY, USA, 2008; pp. 306–330. [Google Scholar]
- Dias, D.P.; Marenco, R.A. Fotossíntese e fotoinibição em mogno e acariquara em função da luminosidade e temperatura foliar [Photosynthesis and photoinhibition in mahogany and acariquara as a function of irradiance and leaf temperature]. Pesqui. Agropecu. Bras. 2007, 42, 305–311. [Google Scholar] [CrossRef]
- Concenço, G.; Ferreira, E.A.; Silva, A.A.; Ferreira, F.A.; Galon, L.; Reis, M.R.; D’Antonino, L.; Vargas, L.; Silva, L.V.B.D. Fotossíntese de biótipos de azevém sob condição de competição [Photosynthesis of ryegrass biotypes under different competition levels]. Planta Daninha 2008, 26, 595–600. [Google Scholar]
- Mariano, K.R.; Barreto, L.S.; Silva, A.H.B.; Neiva, G.K.P.; Ribeiro, A.J.; Amorim, S.M.C. Fotossíntese e tolerância protoplasmática foliar em Myracrodruonurundeuva fr. All. submetida ao déficit hídrico [Photosynthesis and leaf protoplasmatic tolerance in Myracrodruonurundeuva fr. All. under water deficit]. Caatinga 2009, 22, 72–77. [Google Scholar]
- Gutierrez, M.V.; Meinzer, F.C. Carbon isotope discrimination and photosynthetic gas exchange in coffee hedgerows during canopy development. Aust. J. Plant Physiol. 1994, 21, 207–219. [Google Scholar] [CrossRef]
- Lee, D.W.; Baskaran, K.; Mansor, M.; Mohamad, H.; Yap, S.K. Irradiance and spectral quality affect Asian tropical rain forest tree seedling development. Ecology 1996, 77, 568–580. [Google Scholar] [CrossRef]
- Baliza, D.P.; Cunha, R.L.; Castro, A.M.; Barbosa, J.P.R.A.D.; Pires, M.F.; Gomes, R.A. Trocas gasosas e características estruturais adaptativas de cafeeiros cultivados em diferentes níveis de radiação [Gas exchange and adaptative structural characteristics of coffee plants grown in different levels of radiation]. Coffee Sci. 2012, 7, 250–258. [Google Scholar]
- Assad, E.D.; Pinto, H.S.; Zullo, J.R.J.; Ávila, A.M.H. Impacto das mudanças climáticas no zoneamento agroclimático do café no Brasil [Climate changes impact the agroclimatic zonning of coffee in Brazil]. Pesqui. Agropec. Bras. 2004, 39, 1057–1064. [Google Scholar] [CrossRef]
- Silva, D.M.; Brandão, I.R.; Alves, J.D.; Santos, M.O.; Souza, K.R.D.; Silveira, H.R.O. Physiological and biochemical impacts of magnesium-deficiency in two cultivars of coffee. Plant Soil 2014, 128, 133–150. [Google Scholar] [CrossRef]
- Prado, R.M. Nutrição de Plantas [Plant Nutrition]; Editora da Unesp: São Paulo, Brazil, 2008. [Google Scholar]
- Jia, Y.; Yang, X.; Islam, E.; Feng, Y. Effects of potassium deficiency on chloroplast ultrastructure and chlorophyll fluorescence in inefficient and efficient genotypes of rice. J. Plant Nutr. 2008, 31, 2105–2118. [Google Scholar] [CrossRef]
- Catuchi, T.A.; Vitolo, H.F.; Bertolli, S.C.; Souza, G.M. Tolerance to water deficiency between two soybean cultivars: Transgenic versus conventional. Ciênc. Rural 2011, 31, 373–378. [Google Scholar] [CrossRef]
- Logan, B.A.; Kornyeyev, D.; Hardison, J.; Holaday, A.S. The role of antioxidant enzymes in photoprotection. Photosynth. Res. 2006, 88, 119–132. [Google Scholar] [CrossRef] [PubMed]
- Pandey, V.; Dixit, V.; Shyam, R. Chromium effect on ROS generation and detoxification in pea (Pisun sativum) leaf chloroplasts. Protoplasma 2010, 236, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, M.W. The effect of hydrogen peroxide on CO2 fixation of isolated chloroplast. Biochim. Biophys. Acta 1976, 440, 476–482. [Google Scholar] [CrossRef]
- Machado, E.C.; Schmidt, P.T.; Medina, C.L.; Ribeiro, R.V. Respostas da fotossíntese de três espécies de citros a fatores ambientais [Photosynthetic responses of three citrus species to environmental factors]. Pesqui. Agropec. Bras. 2005, 40, 1161–1170. [Google Scholar] [CrossRef]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dias, K.G.d.L.; Guimarães, P.T.G.; Neto, A.E.F.; Silveira, H.R.O.d.; Lacerda, J.J.d.J. Effect of Magnesium on Gas Exchange and Photosynthetic Efficiency of Coffee Plants Grown under Different Light Levels. Agriculture 2017, 7, 85. https://doi.org/10.3390/agriculture7100085
Dias KGdL, Guimarães PTG, Neto AEF, Silveira HROd, Lacerda JJdJ. Effect of Magnesium on Gas Exchange and Photosynthetic Efficiency of Coffee Plants Grown under Different Light Levels. Agriculture. 2017; 7(10):85. https://doi.org/10.3390/agriculture7100085
Chicago/Turabian StyleDias, Kaio Gonçalves de Lima, Paulo Tácito Gontijo Guimarães, Antônio Eduardo Furtini Neto, Helbert Rezende Oliveira de Silveira, and Julian Junio de Jesus Lacerda. 2017. "Effect of Magnesium on Gas Exchange and Photosynthetic Efficiency of Coffee Plants Grown under Different Light Levels" Agriculture 7, no. 10: 85. https://doi.org/10.3390/agriculture7100085
APA StyleDias, K. G. d. L., Guimarães, P. T. G., Neto, A. E. F., Silveira, H. R. O. d., & Lacerda, J. J. d. J. (2017). Effect of Magnesium on Gas Exchange and Photosynthetic Efficiency of Coffee Plants Grown under Different Light Levels. Agriculture, 7(10), 85. https://doi.org/10.3390/agriculture7100085




