Selenium Biochemistry and Bioavailability: Implications for Animal Agriculture
Abstract
:1. Introduction
2. Biochemistry of Selenium and Selenoproteins
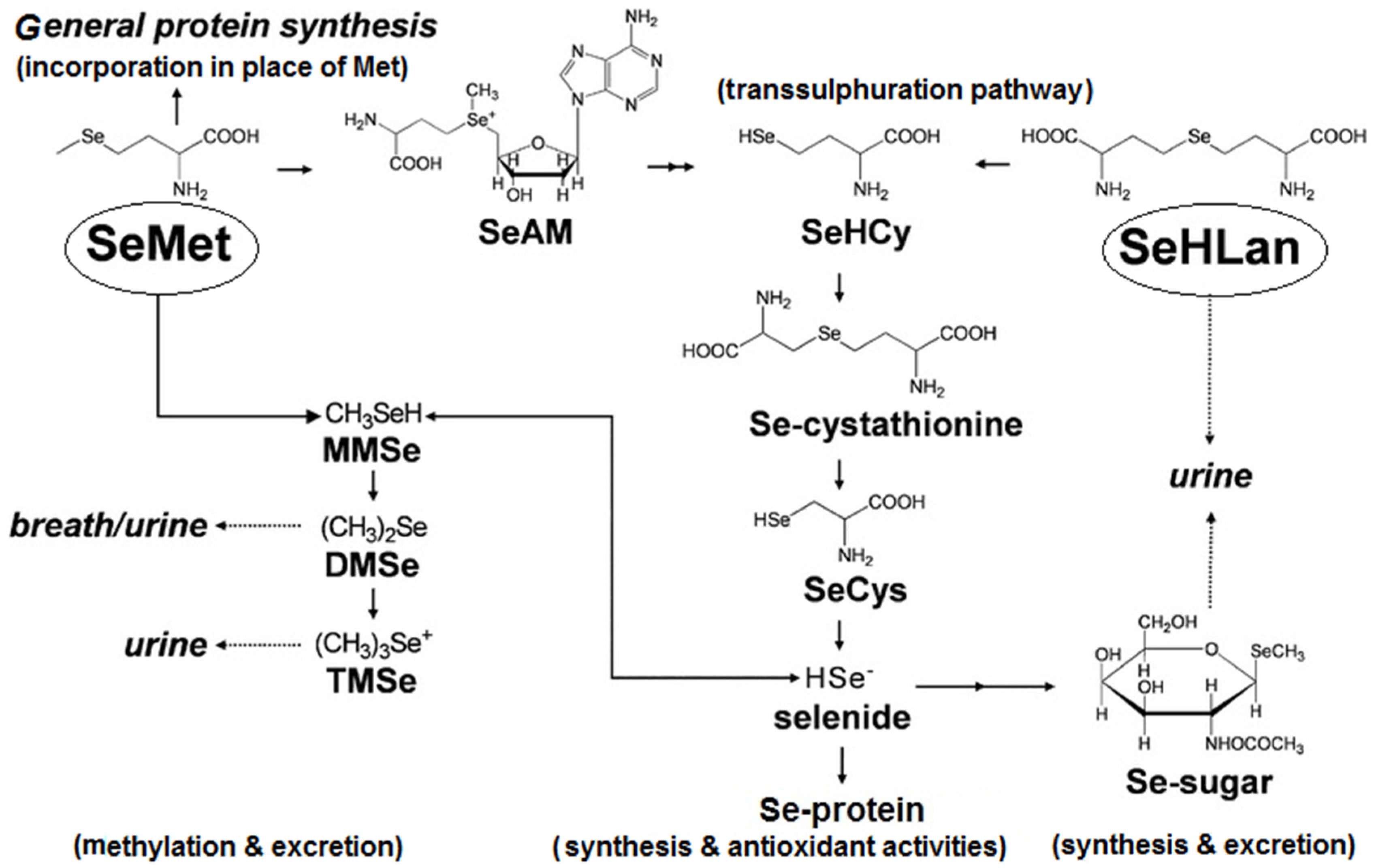
3. Selenium Absorption, Distribution and Metabolic Fate
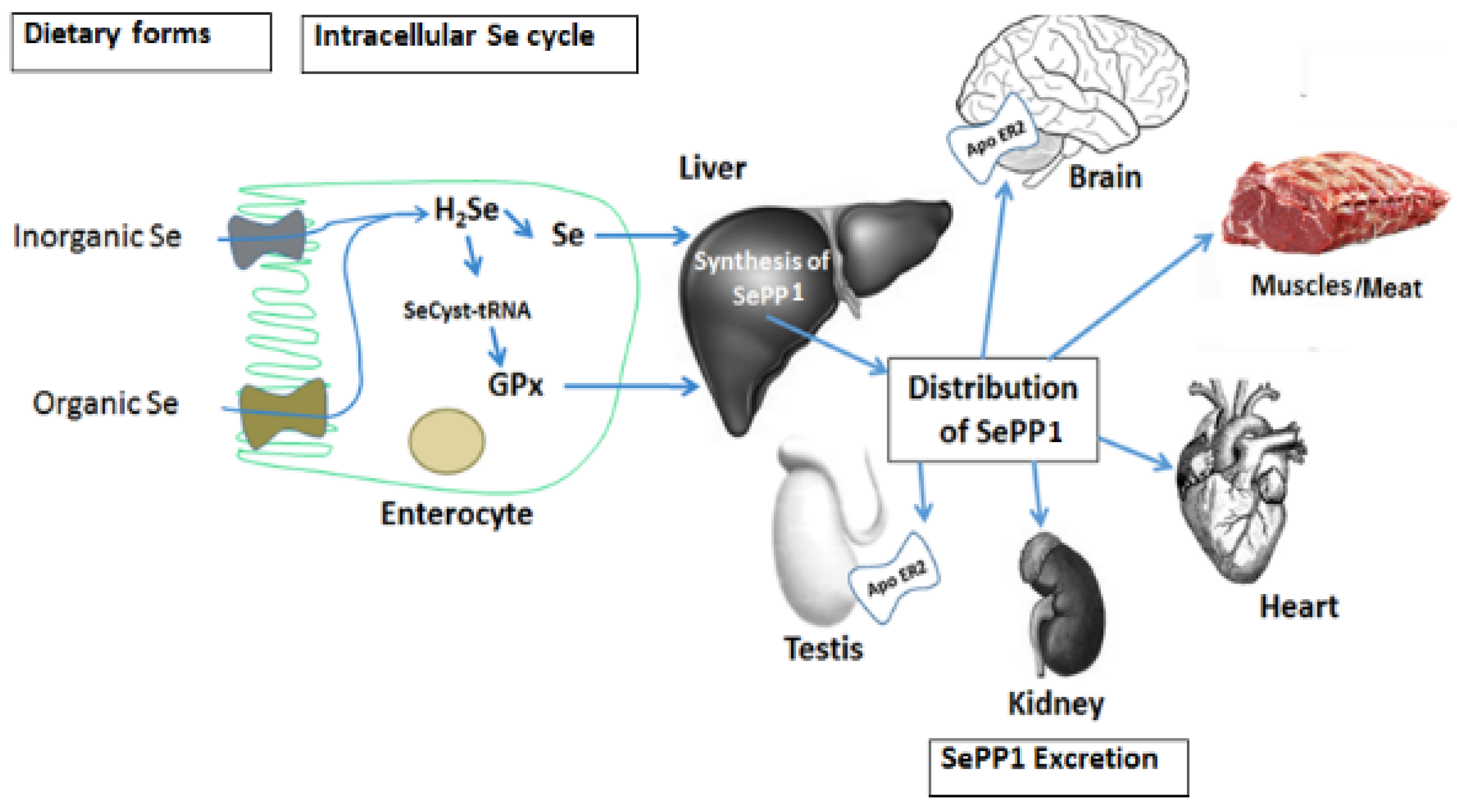
4. Selenium Deficiency and Requirements
5. Dietary Selenium Supplementation
| Name and Content | Nature or Origin | Uses | Reference * |
|---|---|---|---|
| Sodium selenate Sodium selenite Selenase 50 mcg/mL | Synthetic Inorganic | For short-term selenium supplementation; orally in the diet, or by injections for both animals and humans | [12,36,44,45,49,50] |
| Sintomin BIOSEL 2000 | Inactive dry yeast containing high levels of organic selenium | All animals | [51] |
| Selyeast Selenomethionine: 1000, 2000, 3000 | Yeast rich in organic selenium | For use as animal feed. | [22,44,45,47,48] |
| Selemax® (1000, 2000) 70% of total selenium in the form of seleno-methionine | Inactive dry yeast containing organic selenium | All animal species and categories | [22,45,52] |
| SeLECT l(+) Selenomethionine & Vitamin E | Organic, pure selenomethionine | Oral administration humans (capsules), and animals | [22,44,45,49] |
| Sel Plex ™ > 50% of total selenium in the form of seleno-methionine | Yeast rich in organic selenium | All animals | [22,23,24,44,45,50,51] |
| BiOnyc® Tor-Sel Selenohomolanthionine (SeHLan) | Yeast rich in organic selenium | All animals and humans | [53,54] |
| l-Selenomethionine 100% l-selenomethionine | Naturally occurring organoselenium compound made by plants | Predominate form of selenium supplement in food for humans; some use in animals | [22,44,45,53] |
| SeMC ™ Methylselenocysteine 98% | Naturally occurring organoselenium compound made by plants | Humans | [22,53,55] |
6. Conclusions
Conflicts of Interest
References
- Brown, K.M.; Arthur, J.R. Selenium, selenoproteins and human health: A review. Public Health Nutr. 2001, 4, 593–599. [Google Scholar] [CrossRef] [PubMed]
- Bock, A.; Forchhammer, K.; Heider, J.; Leinfelder, W.; Sawers, G.; Veprek, B.; Zinoni, F. Selenocysteine: The 21st amino acid. Mol. Microbiol. 1991, 5, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Hatfield, D.L.; Gladyshev, V.N. How Selenium Has Altered Our Understanding of the Genetic Code. Mol. Cell. Biol. 2002, 22, 3565–3576. [Google Scholar] [CrossRef] [PubMed]
- Reeves, M.A.; Hoffmann, P.R. The human selenoproteome: Recent insights into functions and regulation. Cell. Mol. Life Sci. 2009, 66, 2457–2478. [Google Scholar] [CrossRef] [PubMed]
- Bellinger, F.P.; Raman, A.V.; Reeves, M.A.; Berry, M.J. Regulation and function of selenoproteins in human disease. Biochem. J. 2009, 422, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Burk, R.F.; Hill, K.E.; Motley, A.K. Selenoprotein metabolism and function: Evidence for more than one function for selenoprotein P. J. Nutr. 2003, 133, 1517s–1520s. [Google Scholar] [PubMed]
- Duntas, L.H. The role of selenium in thyroid autoimmunity and cancer. Thyroid 2006, 16, 455–460. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, B.R.; Roberts, B.R.; Bush, A.I.; Hare, D.J. Selenium, selenoproteins and neurodegenerative diseases. Metallomics 2015, 7, 1213–1228. [Google Scholar] [CrossRef] [PubMed]
- Moghadaszadeh, B.; Beggs, A.H. Selenoproteins and their impact on human health through diverse physiological pathways. Physiology 2006, 21, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Köhrle, J.; Jakob, F.; Contempré, B.; Dumont, J.E. Selenium, the thyroid, and the endocrine system. Endocr. Rev. 2005, 26, 944–984. [Google Scholar] [CrossRef] [PubMed]
- Selinus, O.; Fordyce, F. Selenium deficiency and toxicity in the environment. In Essentials of Medical Geology; Springer Netherlands: Heidelberg, Germany, 2013; pp. 375–416. [Google Scholar]
- Oldfield, J.E. Selenium World Atlas: Updated Edition; Selenium-Tellurium Development Association: Grimbergen, Belgium, 2002. [Google Scholar]
- WHO. Guidelines on Food Fortification with Micronutrients; World Health Organization: Geneva, Switzerland, 2006. [Google Scholar]
- Reilly, C. Selenium in Food and Health; Springer Science Media: New York, NY, USA, 2006. [Google Scholar]
- Mangiapane, E.; Pessione, A.; Pessione, E. Selenium and selenoproteins: An overview on different biological systems. Curr. Protein Pept. Sci. 2014, 15, 598–607. [Google Scholar] [CrossRef] [PubMed]
- Rahmanto, A.S.; Davies, M.J. Selenium-containing amino acids as direct and indirect antioxidants. IUBMB Life 2012, 64, 863–871. [Google Scholar] [CrossRef] [PubMed]
- Patching, S.G.; Gardiner, P.H. Recent developments in selenium metabolism and chemical speciation: A review. J. Trace Elem. Med. Biol. 1999, 13, 193–214. [Google Scholar] [CrossRef]
- Steinbrenner, H.; Sies, H. Selenium homeostasis and antioxidant selenoproteins in brain: Implications for disorders in the central nervous system. Arch. Biochem. Biophys. 2013, 536, 152–157. [Google Scholar] [CrossRef] [PubMed]
- Hill, K.E.; Wu, S.; Motley, A.K.; Stevenson, T.D.; Winfrey, V.P.; Capecchi, M.R.; Atkins, J.F.; Burk, R.F. Production of selenoprotein P (Sepp1) by hepatocytes is central to selenium homeostasis. J. Biol. Chem. 2012, 287, 40414–40424. [Google Scholar] [CrossRef] [PubMed]
- Labunskyy, V.M.; Hatfield, D.L.; Gladyshev, V.N. Selenoproteins: Molecular pathways and physiological roles. Physiol. Rev. 2014, 94, 739–777. [Google Scholar] [CrossRef] [PubMed]
- Wastney, M.E.; Combs Jr, G.F.; Canfield, W.K.; Taylor, P.R.; Patterson, K.Y.; Hill, A.D.; Moler, J.E.; Patterson, B.H. A human model of selenium that integrates metabolism from selenite and selenomethionine. J. Nutr. 2011, 141, 708–717. [Google Scholar] [CrossRef] [PubMed]
- Schrauzer, G.N. Nutritional selenium supplements: Product types, quality, and safety. J. Am. Coll. Nutr. 2001, 20, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Bierla, K.; Dernovics, M.; Vacchina, V.; Szpunar, J.; Bertin, G.; Lobinski, R. Determination of selenocysteine and selenomethionine in edible animal tissues by 2D size-exclusion reversed-phase HPLC-ICP MS following carbamidomethylation and proteolytic extraction. Anal. Bioanal. Chem. 2008, 390, 1789–1798. [Google Scholar] [CrossRef] [PubMed]
- Juniper, D.T.; Phipps, R.H.; Bertin, G. Effect of dietary supplementation with selenium-enriched yeast or sodium selenite on selenium tissue distribution and meat quality in commercial-line turkeys. Animal 2011, 11, 1751–1760. [Google Scholar] [CrossRef] [PubMed]
- Santhosh Kumar, B.; Priyadarsini, K.I. Selenium nutrition: How important is it? Biomed. Prev. Nutr. 2014, 4, 333–341. [Google Scholar] [CrossRef]
- Spears, J.W. Trace Mineral Bioavailability in Ruminants. J. Nutr. 2003, 133, 1506S–1509S. [Google Scholar] [PubMed]
- Weiss, W.P. Selenium sources for dairy cattle. In Proceedings of Tri-State Dairy Nutrition Conference, Fort Wayne, IN, USA, May 2005; pp. 61–72.
- Behne, D.; Kyriakopoulos, A. Mammalian selenium-containing proteins. Ann. Rev. Nutr. 2001, 21, 453–473. [Google Scholar] [CrossRef] [PubMed]
- López-Alonso, M.R. Trace Minerals and Livestock: Not Too Much Not Too Little. ISRN Vet. Sci. 2012. [Google Scholar] [CrossRef] [PubMed]
- Combs, G.F. Biomarkers of Selenium Status. Nutrients 2015, 7, 2209–2236. [Google Scholar] [CrossRef] [PubMed]
- Rayman, M.P. The use of high-selenium yeast to raise selenium status: how does it measure up? Br. J. Nutr. 2004, 92, 557–573. [Google Scholar] [CrossRef] [PubMed]
- Turanov, A.A.; Xu, X.-M.; Carlson, B.A.; Yoo, M.-H.; Gladyshev, V.N.; Hatfield, D.L. Biosynthesis of Selenocysteine, the 21st Amino Acid in the Genetic Code, and a Novel Pathway for Cysteine Biosynthesis. Adv. Nutr.: An Int. Rev. J. 2011, 2, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Caprioli, R.M.; Hill, K.E.; Burk, R.F. Loss of selenium from selenoproteins: Conversion of selenocysteine to dehydroalanine in vitro. J. Am. Soc. Mass. Spectrom. 2003, 14, 593–600. [Google Scholar] [CrossRef]
- Combs, G.F.; Scott Jr., M.L. Dietary requirements for vitamin E and selenium measured at the cellular level in the chick. J. Nutr. 1974, 104, 1292–1296. [Google Scholar] [PubMed]
- Scott, M.L.; Noguchi, T.; Combs, G.F., Jr. New evidence concerning mechanisms of action of vitamin E and selenium. Vitam. Horm. 1974, 32, 429–444. [Google Scholar] [PubMed]
- Diplock, A.T. The nutritional and metabollic roles of selenium and vitamin E. Proc. Nutr. Soc. 1974, 33, 315–322. [Google Scholar] [CrossRef] [PubMed]
- National Research Council (US) Subcommittee on Selenium. Selenium in Nutrition: Revised Edition; National Academies Press: Washington, DC, USA, 1983. [Google Scholar]
- Kinoshita, H.; Sugai, K.; Goto, Y.; Nonaka, I. Early onset distal muscular dystrophy. Brain Dev. 1995, 17, 206–209. [Google Scholar] [CrossRef]
- Sharp, B.A.; Van Dreumel, A.A.; Young, L.G. Vitamin E, selenium and methionine supplementation of dystrophogenic diets for pigs. Can. J. Comp. Med. 1972, 36, 398–402. [Google Scholar] [PubMed]
- Sharp, B.A.; Young, L.G.; Van Dreumel, A.A. Dietary induction of mulberry heart disease and hepatosis dietetica in pigs. I. Nutritional aspects. Can. J. Comp. Med. 1972, 36, 371–376. [Google Scholar] [PubMed]
- Jovanović, I.B.; Veličković, M.; Vuković, D.; Milanović, S.; Valčić, O.; Gvozdić, D. Effects of Different Amounts of Supplemental Selenium and Vitamin E on the Incidence of Retained Placenta, Selenium, Malondialdehyde, and Thyronines Status in Cows Treated with Prostaglandin F2α; for the Induction of Parturition. J. Vet. Med. 2013. [Google Scholar] [CrossRef] [PubMed]
- Cheeke, P.R.; Dierenfeld, E.S. Comparative animal nutrition and metabolism; CABI: Wallingford, UK, 2010. [Google Scholar]
- Finley, J.W. Bioavailability of selenium from foods. Nutr. Rev. 2006, 64, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Fisinin, V.I.; Papazyan, T.T.; Surai, P.F. Producing selenium-enriched eggs and meat to improve the selenium status of the general population. Crit. Rev. Biotechnol. 2009, 29, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Schrauzer, G.N.; Surai, P.F. Selenium in human and animal nutrition: resolved and unresolved issues. A partly historical treatise in commemoration of the fiftieth anniversary of the discovery of the biological essentiality of selenium, dedicated to the memory of Klaus Schwarz (1914–1978) on the occasion of the thirtieth anniversary of his death. Crit. Rev. Biotechnol. 2009, 29, 2–9. [Google Scholar] [PubMed]
- Kieliszek, M.; Błażejak, S.; Gientka, I.; Bzducha-Wróbel, A. Accumulation and metabolism of selenium by yeast cells. Appl. Microbiol. Biotechnol. 2015, 99, 5373–5382. [Google Scholar] [CrossRef] [PubMed]
- Suhajda, A.; Hegoczki, J.; Janzso, B.; Pais, I.; Vereczkey, G. Preparation of selenium yeasts I. Preparation of selenium-enriched Saccharomyces cerevisiae. J. Trace Elem. Med. Biol. 2000, 14, 43–47. [Google Scholar] [CrossRef]
- Ouerdane, L.; and Mester, Z. Production and characterization of fully selenomethionine-labeled Saccharomyces cerevisiae. J. Agric. Food Chem. 2008, 56, 11792–11799. [Google Scholar] [CrossRef] [PubMed]
- Cantor, A.H.; Langevin, M.L.; Noguchi, T.; Scott, M.L. Efficacy of selenium in selenium compounds and feedstuffs for prevention of pancreatic fibrosis in chicks. J. Nutr. 1975, 105, 106–111. [Google Scholar]
- Payne, R.L.; Southern, L.L. Comparison of inorganic and organic selenium sources for broilers. Poult. Sci. 2005, 84, 898–902. [Google Scholar] [CrossRef] [PubMed]
- da Silva, ICM; Ribeiro, A.M.L.; Canal, C.W.; Trevizan, L.; Macagnan, M.; Gonçalves, T.A.; Hlavac, N.R.C.; de Almeida, L.L.; Pereira, R.A. The impact of organic and inorganic selenium on the immune system of growing broilers submitted to immune stimulation and heat stress. Rev. Bras. Cienc. Avic. 2010, 12, 247–254. [Google Scholar]
- Oliveira, T.F.B.; Rivera, D.F.R.; Mesquita, F.R.; Braga, H.; Ramos, E.M.; Bertechini, A.G. Effect of different sources and levels of selenium on performance, meat quality, and tissue characteristics of broilers. J. Appl. Poult. Res. 2014, 23, 15–22. [Google Scholar] [CrossRef]
- Tsuji, Y.; Mikami, T.; Anan, Y.; Ogra, Y. Comparison of selenohomolanthionine and selenomethionine in terms of selenium distribution and toxicity in rats by bolus administration. Metallomics 2010, 2, 412–418. [Google Scholar] [CrossRef] [PubMed]
- Celi, P.; Selle, P.H.; Cowieson, A.J. Effects of organic selenium supplementation on growth performance, nutrient utilisation, oxidative stress and selenium tissue concentrations in broiler chickens. Anim. Prod. Sci. 2014, 54, 966–971. [Google Scholar] [CrossRef]
- Suzuki, K.T.; Doi, C.; Suzuki, N. Metabolism of 76Se-methylselenocysteine compared with that of 77Se-selenomethionine and 82Se-selenite. Toxicol. Appl. Pharmacol. 2006, 217, 185–95. [Google Scholar] [CrossRef] [PubMed]
- Anan, Y.; Ogra, Y. Toxicological and pharmacological analysis of selenohomolanthionine in mice. Toxicol. Res. 2013, 2, 115–122. [Google Scholar] [CrossRef]
- Kaufman, N.; Klavins, J.V.; Kinney, T.D. Pancreatic damage induced by excess methionine. Arch. Pathol. 1960, 70, 331–337. [Google Scholar] [PubMed]
- Henman, D.J.; (Riverlea Pty Ltd., Corowa, Australia). Personal communication, 2014.
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shini, S.; Sultan, A.; Bryden, W.L. Selenium Biochemistry and Bioavailability: Implications for Animal Agriculture. Agriculture 2015, 5, 1277-1288. https://doi.org/10.3390/agriculture5041277
Shini S, Sultan A, Bryden WL. Selenium Biochemistry and Bioavailability: Implications for Animal Agriculture. Agriculture. 2015; 5(4):1277-1288. https://doi.org/10.3390/agriculture5041277
Chicago/Turabian StyleShini, Shaniko, Asad Sultan, and Wayne L. Bryden. 2015. "Selenium Biochemistry and Bioavailability: Implications for Animal Agriculture" Agriculture 5, no. 4: 1277-1288. https://doi.org/10.3390/agriculture5041277
APA StyleShini, S., Sultan, A., & Bryden, W. L. (2015). Selenium Biochemistry and Bioavailability: Implications for Animal Agriculture. Agriculture, 5(4), 1277-1288. https://doi.org/10.3390/agriculture5041277





