Aerobic and Anaerobic Transformations in Estrogens and Nutrients in Swine Manure: Environmental Consequences
Abstract
:1. Introduction
2. Materials and Methods

3. Results
3.1. Fresh Manure Characteristics
3.2. Effects of Aerobic Digestion
| Estradiol (E2) | Estrone (E1) | Estriol (E3) | Total | ||||
|---|---|---|---|---|---|---|---|
| Conjugated μg/L | Free μg/L | Conjugated μg/L | Free μg/L | Conjugated μg/L | Free μg/L | μg/L | |
| Manure Liquid | |||||||
| n = 23 | 43 ± 17 G 78% | 13 ± 11 | 165 ± 4 * G 20% | 3 ± 7 | 15 ± 13 # G 90% | 17 ± 12 | 310 ± 117 |
| E2Eq Ph E2Eq Ah | 13 ± 12 71 ± 22 † | ||||||
| Manure Solid | |||||||
| n = 23 | 143 ± 63 G 80% | 210 ± 111 | 229 ± 138 G 30% | 413 ± 211 | 45 ± 8 # G 90% | 18 ± 13 * | 973 ± 471 |
| E2Eq Ph E2Eq Ah | 53 ± 11 97 ± 32 † | ||||||
| KD | 3.3 ± 1.7 | 16 ± 5 * | 1.4 ± 0.9 | 105 ± 21 * | 3 ± 2 | 1 ± 0.8 | |
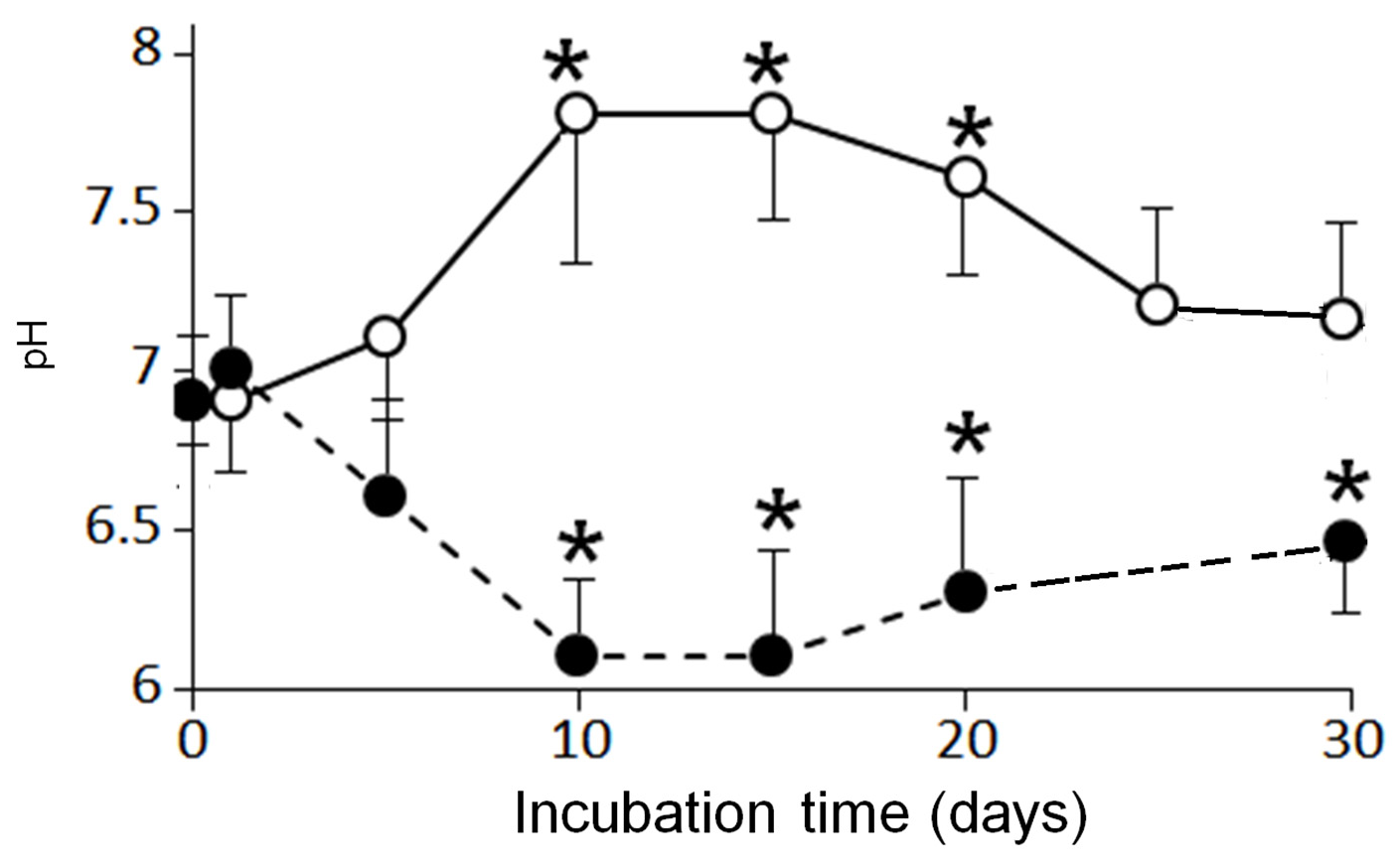
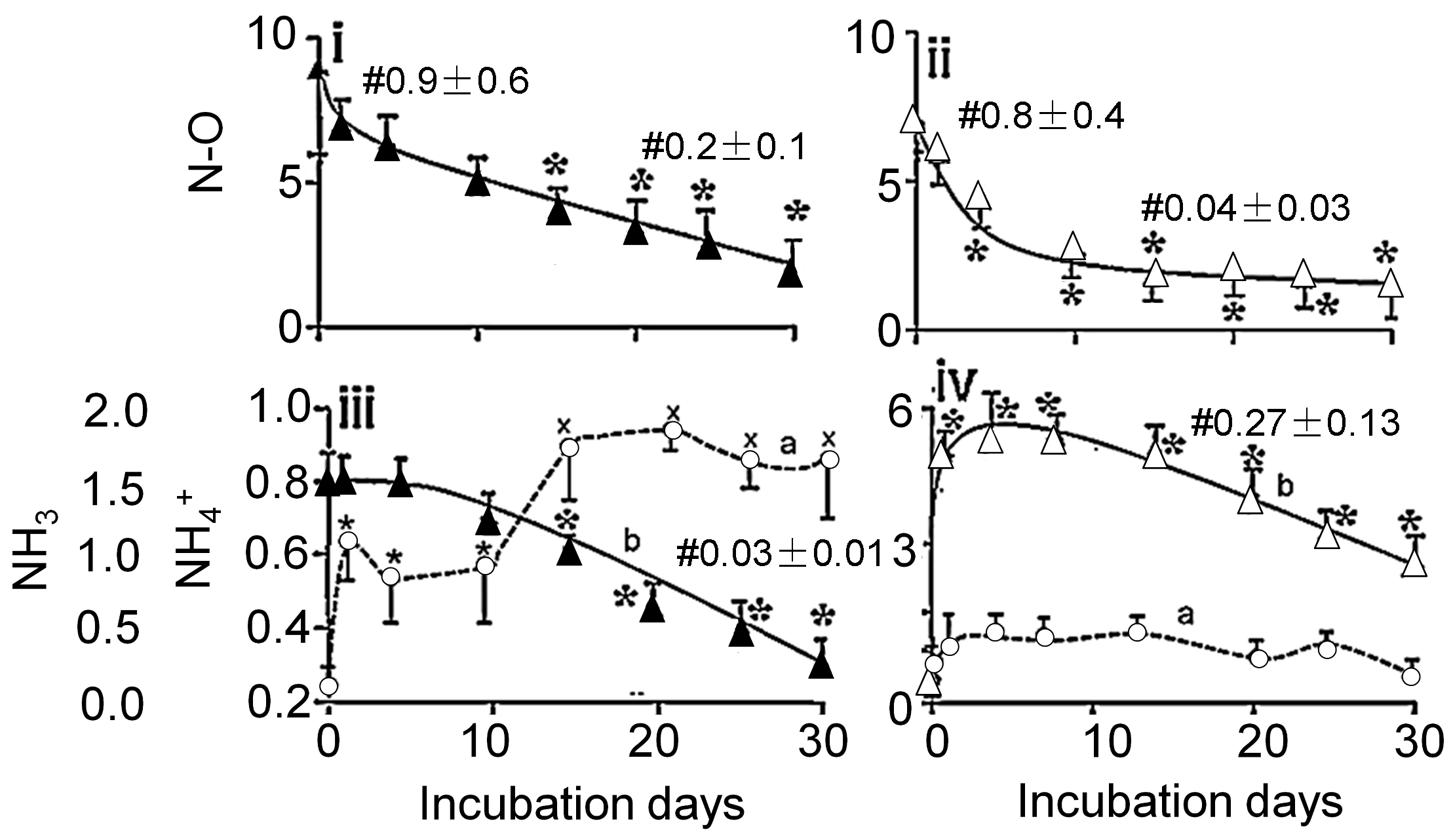
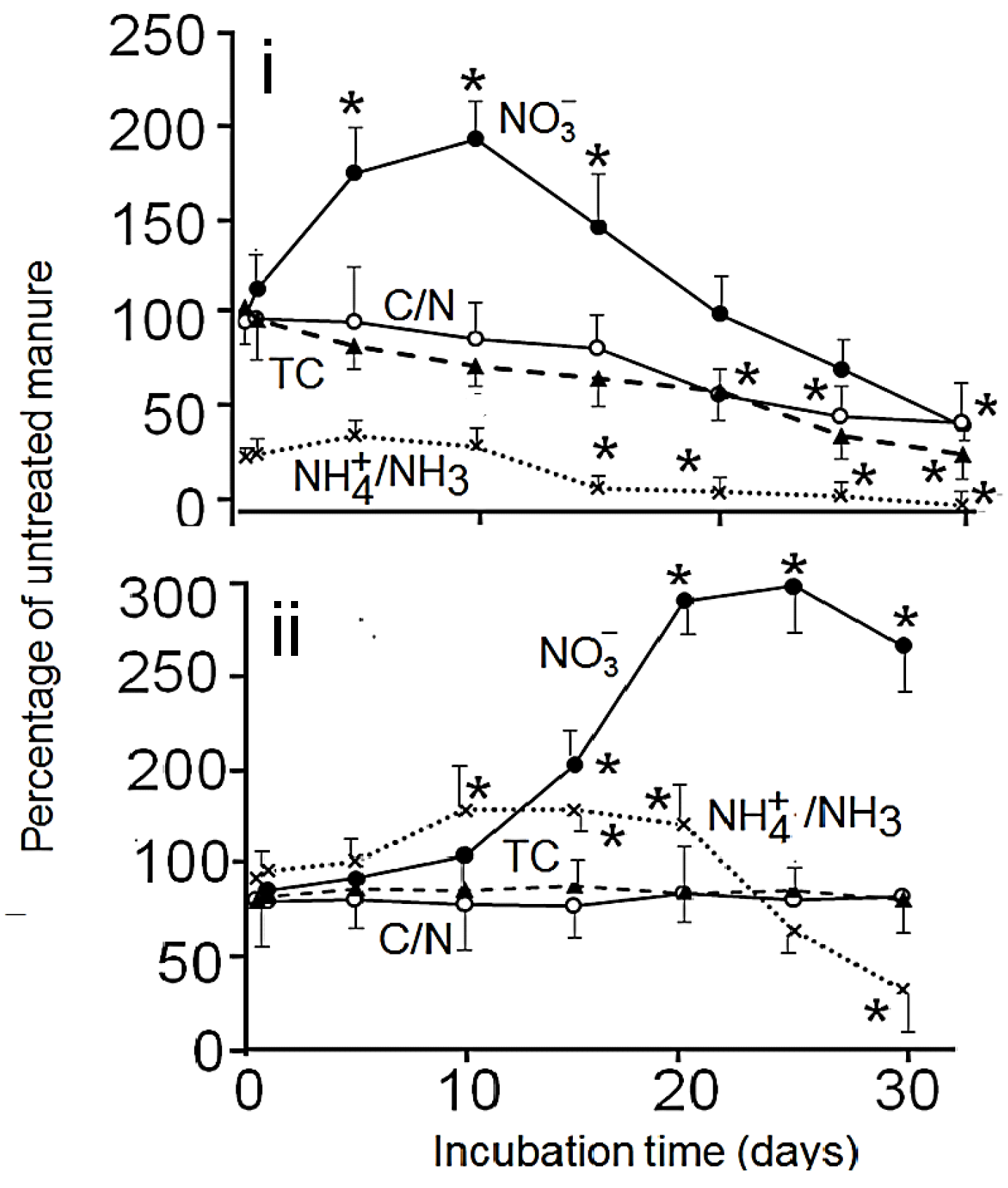
3.3. Effects of Anaerobic Digestion
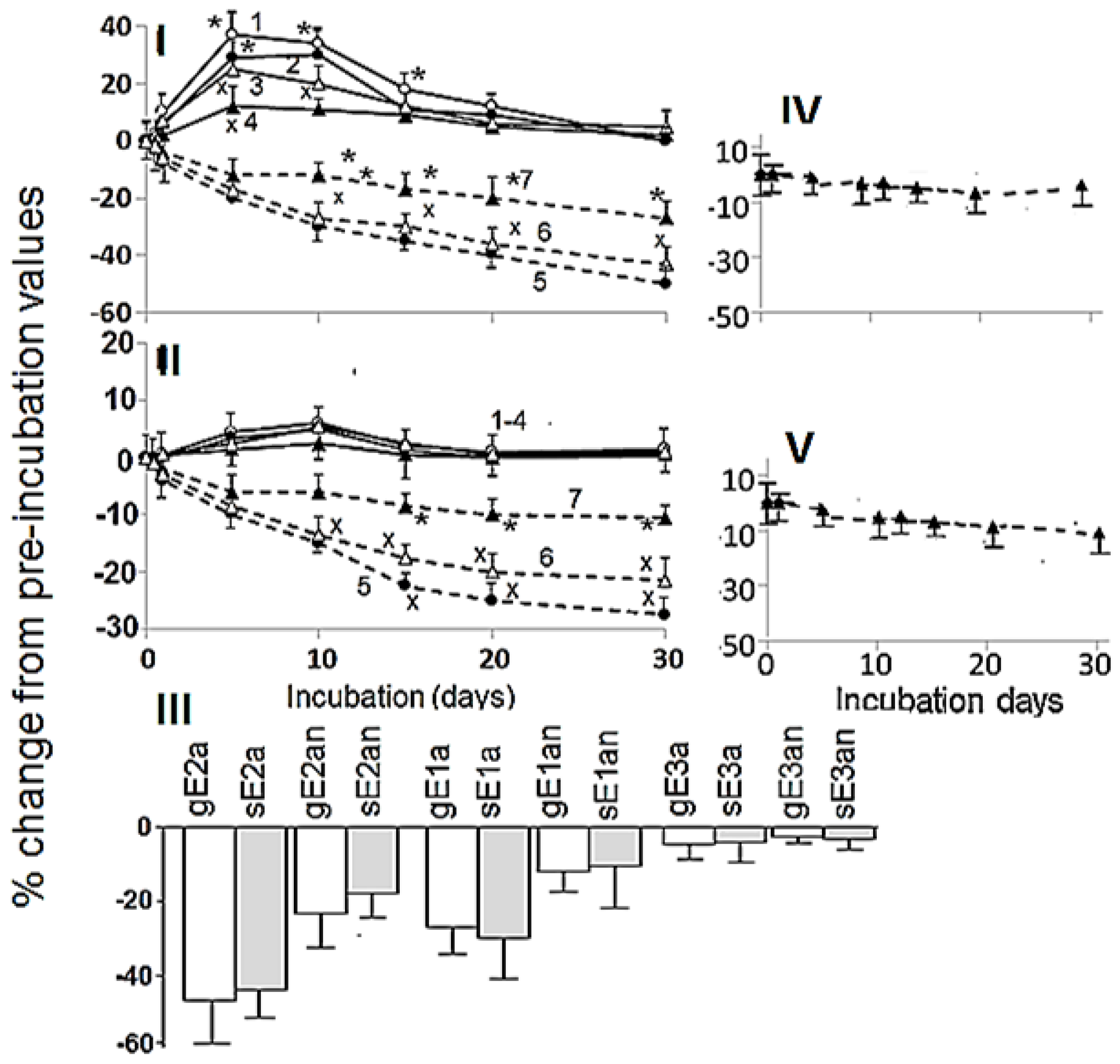
4. Discussion
4.1. Manure Characteristics
- Manure liquid: cE1G > cE2G > cE1S > cE2S, cE3G, fE2, fE3 > fE1 > cE3S
- Manure Solid: fE1 > fE2 > cE1S > cE1G > cE2G > fE3, cE3G > cE2S, cE3S
4.2. Plant Nutrients in Aerobic and Anaerobic Environment
- Heterotrophic bacteria mediated ammonification and/or denitrification of amino acids convert O-N into NH4+. Bacteria deamination of amino acids occur via oxidative deamination or anoxic deamination via sehydratase or sulfhydrase enzymes [39].
- Oxidative nitrification converts NH3 or NH4+ to nitrites (NO2−) according to the following equation: NH3 + O2 → NO2− + 3H+ + 2e−. Nitrosomonas, Nitrosococcus, and Nitrosospira are most frequently associated with this reaction, however, Nitrosolobus and Nitrosovibrio can also autotrophically oxidize ammonia [40]. This is followed by the oxidation of nitrites to nitrates (NO3−) by Nitrobacter, Nitrospira, Nitrococcus [41] according to the equation: NO2− + H2O → NO3− + 2H+ + 2e−.
- Anaerobic oxidation (Anammox) of NH4+ to N2 with NO2− as the electron acceptor [42].NH4+ + NO2− → N2 + 2H2O.
- Denitrification in the presence of easily available carbon as an energy and carbon source and carried out by numerous facultative bacteria such as Pseudomonas [43]. In this process, nitrates and nitrites are reduced to nitrogen gas, such as N2O and N2, when a proton donor (energy source such as carbon) is available. The process involve the following pathway:NO3− → NO2− → NO → N2O → N2.In this process microorganisms first reduce nitrates to nitrites and then produce nitric oxide, nitrous oxide and nitrogen gas. A source of organic carbon is an important component of the denitrification process.
4.3. Effects of Aerobic and Anaerobic Digestions on Estrogen Concentrations and Estrogenic Activity
4.4. Environmental Consequences
5. Conclusions
Supplementary Files
Supplementary File 1Acknowledgements
Author Contributions
Conflicts of Interest
References
- Hribar, C. Understanding Concentrated Environmental Health Animal Feeding Operations and Their Impact on Communities. Available online: http://www.cdc.gov/nceh/ehs/Docs/Understanding_CAFOs_NALBOH.pdf (accessed on 29 July 2015).
- Hamilton, D.W.; Luce, W.G.; Heald, A.D. Production and Characteristics of Swine Manure. Available online: http://agrienvarchive.ca/bioenergy/download/F-1735_swine_man_char_OK.pdf (accessed on 28 July 2015).
- Khanal, S.K.; Xie, B.; Thompson, M.L.; Sung, S.; Ong, S-K.; van Leeuwen, J. Fate, transport, and biodegradation of natural estrogens in the environment and engineered systems. Environ. Sci. Technol. 2006, 40, 6537–6546. [Google Scholar] [CrossRef] [PubMed]
- Weida, W.J. Economic Implications of Confined Animal Feeding Operations. Available online: http://www.sraproject.org/wp-content/uploads/2007/12/economicimplicationsofcafos.pdf (accessed on 28 July 2015).
- Liney, K.E.; Jobling, S.; Shears, J.A.; Simpson, P.; Tyler, C.R. Assessing the sensitivity of different life stages for sexual disruption in roach (Rutilus rutilus) exposed to effluents from wastewater treatment works. Environ. Health Perspect. 2005, 113, 1299–1307. [Google Scholar] [CrossRef] [PubMed]
- Hutchinson, T.H.; Ankley, G.T.; Segner, H.; Tyler, C.R. Screening and testing for endocrine disruption in fish—Biomarkers as “signposts” not “traffic lights” in risk assessment. Environ. Health Perspect. 2006, 114, 106–114. [Google Scholar] [CrossRef] [PubMed]
- Combalbert, S.; Hernandez-Raquet, G. Occurrence, fate, and biodegradation of estrogens in sewage and manure. Appl. Microbiol. Biotechnol. 2010, 86, 1671–1692. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.K.; Gupta, S.; Kumar, K.; Gupta, S.K.; Chander, Y.; Gupta, A.; Saxena, R. Quantitative analysis of conjugated and free estrogens in swine manure: Solutions to overcome analytical problems due to matrix effects. J. Chromatogr. A 2013, 1305, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Panter, G.H.; Thompson, R.S.; Beresford, N.; Sumpter, J.P. Transformation of a non-oestrogenic steroid metabolite to an oestrogenically active substance by minimal bacterial activity. Chemosphere 1999, 38, 3579–3596. [Google Scholar] [CrossRef]
- Ritter, J.K. Roles of glucuronidation and UDP-glucuronosyltransferases in xenobiotic bioactivation reactions. Chem. Biol. Interact. 2000, 129, 201–208. [Google Scholar] [CrossRef]
- Masse, D.I.; Croteaua, F.; Masses, L. The fate of crop nutrients during digestion of swine manure in psychrophilic anaerobic sequencing batch reactor. Bioresource Technol. 2006, 98, 2819–2823. [Google Scholar] [CrossRef] [PubMed]
- Krom, M.D. Spectrophotometric determination of ammonia: A study of a modified Berthelot reaction using salicylate and dichloroisocyanurate. Analyst 1980, 105, 305–316. [Google Scholar] [CrossRef]
- Zeng, Y.; de Guardia, A.; Daumoin, M.; Benoist, J.-C. Characterizing the transformation and transfer of nitrogen during the aerobic treatment of organic wastes and digestates. Waste Manag. 2012, 32, 2239–2247. [Google Scholar] [CrossRef] [PubMed]
- Kirchmann, H.; Witter, E. Ammonia volatilization during aerobic and anaerobic manure decomposition. Plant Soil 1989, 115, 35–41. [Google Scholar] [CrossRef]
- Amberger, A.; Huber, J.; Rank, M. Gulleausbringung: Vorsicht, Ammoniakerverluste. DLG Miteilungen 1987, 20, 1084–1086. [Google Scholar]
- Hobbs, P.J.; Misselbrook, T.H.; Cumby, T.R. Production and emission of odours and gases from ageing pig waste. J. Agric. Engng. Res. 1999, 72, 291–298. [Google Scholar] [CrossRef]
- Zahn, J.A.; Hatfield, J.L.; Do, Y.S.; Pfeiffer, R.L. Characterization of volatile organic emissions and wastes from a swine production facility. J. Environ. Qual. 1997, 26, 1687–1696. [Google Scholar] [CrossRef]
- Gale, E.S.; Sullivan, D.M.; Cogger, C.G.; Bary, A.I.; Hemphill, D.D.; Myhre, E.A. Estimating plant-available nitrogen release from manures, composts, and specialty products. J. Environ. Qual. 2006, 35, 2321–2332. [Google Scholar] [CrossRef] [PubMed]
- Van Kessel, J.S.; Reeves, J.B.; Meisinger, J.J. Nitrogen and carbon mineralization of potential manure components. J. Environ. Qual. 2000, 29, 1669–1677. [Google Scholar] [CrossRef]
- Westerman, P.W.; Huffman, R.L.; Feng, J.S. Swine-lagoon seepage in sandy soil. Trans. ASAE 1995, 38, 1749–1760. [Google Scholar] [CrossRef]
- Buijsman, E.; Mass, H.J.; Asman, W.A. Anthropogenic NH3 emissions in Europe. Atmos. Epyiron. 1987, 11, 1009–1022. [Google Scholar] [CrossRef]
- Raman, D.R.; Williams, E.L.; Layton, A.C.; Burns, R.T.; Easter, J.P.; Daugherty, A.S.; Mullen, D.M.; Sayler, G.S. Estrogen content of dairy and swine wastes. Environ. Sci. Technol. 2004, 38, 3567–3573. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Shi, J.; Liu, X.; Xinmin, Z.; Qingcai, C. Occurrence of free estrogens, conjugated estrogens, and bisphenol A in fresh livestock excreta and their removal by composting in North China. Environ. Sci. Pol. Res. 2014, 21, 9939–9947. [Google Scholar] [CrossRef] [PubMed]
- Furuichi, T.; Kannan, K.; Suzuki, K.; Tanaka, S.; Giesy, J.P.; Masunaga, S. Occurrence of estrogenic compounds in and removal by a swine farm waste treatment plant. Environ. Sci. Technol. 2006, 40, 7896–7902. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.C.; Williams, R.J.; Matthiessen, P. The potential steroid hormone contribution of farm animals to freshwaters, the United Kingdom as a case study. Sci. Total Environ. 2006, 362, 166–178. [Google Scholar] [CrossRef] [PubMed]
- Lee, L.S.; Strock, T.J.; Sarmah, A.K.; Rao, P.S. Sorption and dissipation of testosterone, estrogens, and their primary transformation products in soils and sediment. Environ. Sci. Technol. 2003, 37, 4098–4105. [Google Scholar] [CrossRef] [PubMed]
- Hakk, H.; Sikora, L. Dissipation of 17β-estradiol in composted poultry litter. J. Environ. Qual. 2011, 40, 1560–1566. [Google Scholar] [CrossRef] [PubMed]
- Stumpe, B.; Marschner, B. Long-term sewage sludge application and wastewater irrigation on the mineralization and sorption of 17β-estradiol and testosterone in soils. Sci. Total Environ. 2007, 374, 282–291. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, S.L.; Francis, X.M.; Casey, F.X.M.; Hakk, H.; Smith, D.J.; Padmanabhan, G. Fate and transformation of an estrogen conjugate and its metabolites in agricultural soils. Environ. Sci. Technol. 2012, 46, 11047–11053. [Google Scholar] [CrossRef] [PubMed]
- Shore, L.S.; Reichmann, O.; Shemesh, M.; Wenzel, A.; Litaor, M.I. Washout of accumulated testosterone in a watershed. Sci. Total Environ. 2004, 332, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Sarmah, A.K.; Scherr, F. Mediator saolution influence on the sorption potential of sulfo-conjugated esterogenic steroid hormone and its metabolite in New Zealand dairy farm soil. In Proceedings of the 19th World Congress of Soil Science, Soil Solutions for a Changing World, Brisbane, Australia, 1–6 August 2010; International Union of Soil Sciences: Rome, Italy, 2010; pp. 102–105. [Google Scholar]
- Zitnick, K.K.; Shappel, N.W.; Hakk, H.; DeSutter, T.M.; Khan, E.E.; Casey, FXM. Effects of liquid swine manure on dissipation of 17β-estradiol in soil. J. Hazard. Mat. 2011, 186, 1111–1117. [Google Scholar] [CrossRef] [PubMed]
- Dinova, M.V. Transformation of steroids by actinobacteria: A review. Appl. Biochem. Microbiol. 2001, 43, 1–14. [Google Scholar] [CrossRef]
- Nikaeen, M.; Nafez, A.H.; Bina, B.; Nabavi, B.F.; Hassanzadeh, A. Respiration and enzymatic activities as indicators of stabilization of sewage sludge composting. Waste Manag. 2015, 39, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Liu, X.; Huang, Y.; Huang, H. Inoculation with nitrogen turnover bacterial agent appropriately increasing nitrogen and promoting maturity in pig manure composting. Waste Manag. 2015, 39, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Kulikowska, D.; Gusiatin, Z.M. Sewage sludge composting in a two-stage system: Carbon and nitrogen transformations and potential ecological risk assessment. Waste Manag. 2015, 38, 312–320. [Google Scholar] [CrossRef] [PubMed]
- Kuypers, M.M.; Sliekers, A.O.; Lavik, G.; Schmid, M.; Jørgensen, B.B.; Kuenen, J.G.; Sinninghe, D.J.S.; Strous, M.; Jetten, M.S. Anaerobic ammonium oxidation by anammox bacteria in the Black Sea. Nature 2003, 422, 608–611. [Google Scholar] [CrossRef] [PubMed]
- Oleszkiewicz, J.A. A comparison of anaerobic treatment of low concentration piggery wastewaters. Agric. Wastes 1983, 8, 215–231. [Google Scholar] [CrossRef]
- Orlygsson, J.; Houwen, F.P.; Svensson, B.H. Thermophilic anaerobic amino acid degradation: deamination rates and end-product formation. [CrossRef]
- Watson, S.W.; Valos, F.W.; Waterbury, J.B. The family nitrobacteraceae. In The Prokaryotes; Starr, M.P., Ed.; Springer-Verlag: Berlin, Germany, 1981. [Google Scholar]
- Anthonisen, A.C.; Loehr, R.C.; Prakasam, T.B.S. Inhibition of nitrification by ammonia and nitrous-acid. J. Water Pol. Control Fed. 1976, 48, 835–852. [Google Scholar]
- Yamamoto, T.; Wakamatsu, S.; Qiao, S.; Hira, D.; Fujii, T.; Furukawa, K. Partial nitritation and anammox of a livestock manure digester liquor and analysis of its microbial community. Bioresour. Technol. 2011, 102, 2348–2351. [Google Scholar] [CrossRef] [PubMed]
- Naik, S.; Setty, Y.P. Effect of Carbon Sources on Biological Denitrification of Wastewater by Immobilized Pseudomonas Stutzeri Bacteria in a Fluidized Bed Bio Reactor (FBBR). Available online: http://ipcbee.com/vol23/23-CCEA2011-BEM30014.pdf (accessed on 24 August 2015).
- Lqynachatn, E.; Bartholomeww, V.; Wolluma, G. Nitrogen transformations in aerated swine manure slurries. J. Environ. Qual. 1976, 5, 293–297. [Google Scholar]
- Evans, M.R.; Hissetert, R.; Smith, M.P.W.; Ellamd, D.F.; Baines, S. Effect of microorganism residence time on aerobic treatment of piggery waste. Agric. Wastes 1979, 1, 67–85. [Google Scholar] [CrossRef]
- Evans, M.R.; Smith, M.P.W.; Deans, E.A.; Svoboda, I.F.; Thacker, F.E. Nitrogen and aerobic treatment of slurry. Agric. Wastes 1986, 15, 205–213. [Google Scholar] [CrossRef]
- Garrawaj, Y.L. Investigations on the aerobic treatment of pig slurry. Agric. Wastes 1982, 4, 131–142. [Google Scholar] [CrossRef]
- Blouin, M.; Bisailloj, N.-G.; Beaudet, R.; Ishaque, M. Nitrification of swine waste. Can. J. Microbiol. 1990, 36, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Hino, T.; M atsumoto, Y.; Nagano, S.; Sugimoto, H.; Fukumori, Y.; Murata, T.; Iwata, S.; Shiro, Y. Structural basis of biological N2O generation by bacterial nitric oxide reductase. Science 2010, 330, 1666–1670. [Google Scholar] [CrossRef] [PubMed]
- Moenne-Loccoz, P.; Fee, J.A. Catalyzing NO to N2O in the nitrogen cycle. Science 2010, 330, 1632–1633. [Google Scholar] [CrossRef] [PubMed]
- Canfield, D.E.; Glazer, A.N.; Falkowski, P.G. The evolution and future of Earth’s nitrogen cycle. Science 2010, 330, 192–196. [Google Scholar] [CrossRef] [PubMed]
- Sommer, S.G. Effect of composting on nutrient loss and nitrogen availability of cattle deep litter Eur. J. Agron. 2001, 14, 123–133. [Google Scholar]
- Fukumoto, Y.; Osada, T.; Hanajima, D.; Haga, K. Patterns and quantities of NH3, N2O and CH4 emissions during swine manure composting without forced aeration—Effect of compost pile scale. Bioresour. Technol. 2003, 89, 109–114. [Google Scholar] [CrossRef]
- Szanto, G.L.; Hamelers, H.M.; Rulkens, W.H.; Veeken, A.H.M. NH3, N2O and CH4 emissions during passively aerated composting of straw-rich pig manure. Bioresour. Technol. 2007, 98, 2659–2670. [Google Scholar] [CrossRef] [PubMed]
- Ravishankara, A.R.; Daniel, J.S.; Portmann, R.W. Nitrous oxide (N2O): The dominant ozone-depleting substance emitted in the 21st century. Science 2009, 326, 123–125. [Google Scholar] [CrossRef] [PubMed]
- Kelleher, B.P.; Leahy, J.J.; Henihan, A.M.; O’dwyer, T.F.; Sutton, D.; Leahy, M.J. Advances in poultry litter disposal technology—A review. Bioresource Technol. 2002, 83, 27–36. [Google Scholar] [CrossRef]
- Moller, K. Effects of anaerobic digestion on soil carbon and nitrogen turnover, N emissions, and soil biological activity. A review. Agron. Sustain. Dev. 2014, 35, 1021–1041. [Google Scholar] [CrossRef]
- Seto, B. The stickland reaction. In Diversity of Bacterial Respiratory Systems; Knowles, C.J., Ed.; CRC Press: Boca Raton, FL, USA, 1980; Volume 2, pp. 49–64. [Google Scholar]
- Pain, B.F.; Misselbrook, T.H.; Clarkson, C.R.; Rees, Y.J. Odor and ammonia emissions following the spreading of anaerobically digested pig slurry on grassland. Agric. Wastes 1990, 34, 259–267. [Google Scholar]
- Chantigny, M.H.; Rochette, P.; Angers, D.A.; Masse, D.; Cote, D. Ammonia volatilization and selected soil characteristics following application of anaerobically digested pig slurry. Soil Sci. Soc. Am. J. 2004, 68, 306–312. [Google Scholar] [CrossRef]
- Chantigny, M.H.; Angers, D.A.; Rochette, P.; Belanger, G.; Masse, D.I.; Cote, D. Gaseous nitrogen emissions and forage nitrogen uptake on soils fertilized with raw and treated swine manure. J. Environ. Qual. 2007, 36, 1864–1872. [Google Scholar] [CrossRef] [PubMed]
- Chantigny, M.H.; Angers, D.A.; Belanger, G.; Rochette, P.; Eriksen-Hamel, N.; Bittman, S.; Buckley, K.; Masse, D.; Gasser, M.O. Yield and nutrient export of grain corn fertilized with raw and treated liquid swine manure. Agron. J. 2008, 100, 1303–1309. [Google Scholar] [CrossRef]
- Chantigny, M.H.; MacDonald, J.D.; Beaupre, C.; Rochette, P.; Angers, D.A.; Masse, D.; Parent, L.E. Ammonia volatilization following surface application of raw and treated liquid swine manure. Nutr. Cycl. Agroecosyst. 2009, 85, 275–286. [Google Scholar] [CrossRef]
- Möller, K.; Stinner, W. Effects of different manuring systems with and without biogas digestion on soil mineral nitrogen content and on gaseous nitrogen losses (ammonia, nitrous oxides). Eur. J. Agron. 2009, 30, 1–16. [Google Scholar] [CrossRef]
- Ni, K.; Pacholski, A.; Kage, H. Analysis of ammonia losses after field application of biogas slurries by an empirical model. J. Plant Nutr. Soil Sci. 2012, 175, 253–264. [Google Scholar] [CrossRef]
- Yoshimoto, T.; Nagi, F.; Fujimoto, J.; Watanabe, K.; Mizukoshi, H.; Makino, T.; Kimora, K.; Saino, H.; Sawada, H.; Omura, H. Degradation of estrogens by Rhodococcus zopfi and Rhodococcus equi isolates from activated sludge in wastewater treatment plan. Appl. Environ. Microbiol. 2004, 70, 5283–5289. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.-P.; Roh, H.; Chu, K.-H. 17β-Estradiol-Degrading bacterial isolated from activated sludge. Environ. Sci. Technol. 2007, 41, 486–492. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, V.; Lim, J.-W.; Stabnikova, O.; Gin, K.Y.-H. Biodegradation of estrogens by facultative anaerobic iron-reducing bacteria. Process. Biochem. 2010, 45, 284–287. [Google Scholar] [CrossRef]
- Lee, H.B.; Liu, D. Degradation of 17β-Estradiol and its metabolites by sewage bacteria. Water Air Soil Pollut. 2002, 134, 351–366. [Google Scholar] [CrossRef]
- Isabelle, M.; Villemur, R.; Juteau, P.; Lepine, F. Isolation of estrogen-degrading bacteria from an activated sludge bioreactor treating swine waste, including a strain that converts estrone to β-estradiol. Can. J. Microbiol. 2011, 57, 550–568. [Google Scholar] [CrossRef] [PubMed]
- Barbier, O.; Albert, C.; Martineau, I.; Vallee, M.; High, K.; Labrie, F.; Hum, D.W.; Labrie, C.; Belanger, A. Glucuronidation of the nonsteroidal antiestrogen EM-652 (SCH 57068), by human and monkey steroid conjugating UDP-glucuronosyltransferase enzymes. Mol. Pharmacol. 2001, 59, 636–645. [Google Scholar] [PubMed]
- D’Ascenzo, G.; di Corcia, A.; Gentili, A.; Mancin, R.; Mastropasqua, R.; Nazzari, M.; Samperi, R. Fate of natural estrogen conjugates in municipal sewage transport and treatment facilities. Sci. Total Environ. 2003, 302, 199–209. [Google Scholar] [CrossRef]
- Hutchins, S.R.; White, M.V.; Hudson, F.M.; Fine, D.D. Analysis of lagoon samples from different concentrated animal feeding operations for estrogens and estrogen conjugates. Environ. Sci. Technol. 2007, 41, 738–744. [Google Scholar] [CrossRef] [PubMed]
- Furimsky, A.M.; Green, G.A.; Sharp, L.E.H.; Catz, P.; Adjei, A.A.; Parman, T.; Kapetanovic, I.M.; Weinshilboum, R.M.; Iyer, L.V. Effect of resveratrol on 17β-estradiol sulfation by human hepatic and jejunal S9 and recombinant sulfotransferase 1E1. Drug Metab. Disp. 2008, 36, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Czajka, C.P.; Londry, K.L. Anaerobic biotransformation of estrogens. Sci. Total Environ. 2006, 367, 932–941. [Google Scholar] [CrossRef] [PubMed]
- Yost, E.E.; Meyer, M.T.; Dietze, J.E.; Meissner, B.M.; Davis, L.W.; Williams, C.M.; Lee, B.; Kullman, S.W. Comprehensive assessment of hormones, phytoestrogens, and estrogenic activity in an anaerobic swine waste lagoon. Environ. Sci. Technol. 2013, 47, 13781–13790. [Google Scholar] [CrossRef] [PubMed]
- De Mes, T.; Kujawa-Roeleveld, K.; Zeeman, G.; Lettinga, G. Anaerobic biodegradation of estrogens—Hard to digest. Water Sci. Technol. 2008, 57, 1177–1182. [Google Scholar] [CrossRef] [PubMed]
- Andersen, H.; Siegrist, H.; Halling-Sorensen, B.; Ternes, T.A. Fate of estrogens in a municipal sewage treatment plant. Environ. Sci. Technol. 2003, 37, 4021–4026. [Google Scholar] [CrossRef] [PubMed]
- Ternes, T.A.; Kreckel, P.; Mueller, J. Behaviour and occurrence of estrogens in municipal sewage treatment plants—II. Aerobic batch experiments with activated sludge. Sci. Total Environ. 1999, 225, 91–99. [Google Scholar] [CrossRef]
- Shappell, N.W.; Billey, L.; Forbes, D.; Matheny, T.A.; Poach, M.E.; Reddy, G.B.; Hunt, P.G. Estrogenic activity and steroid hormones in swine wastewater through a lagoon constructed-wetland system. Environ. Sci. Technol. 2007, 41, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Pholchan, P.; Mnes, J.; Donnelly, T.; Sallis, P.J. Fate of estrogens during the biological treatment of synthetic wastewater in a nitrite-accumulating sequencing batch reactor. Environ. Sci. Technol. 2008, 42, 6141–6147. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Singh, A.K. Aerobic and Anaerobic Transformations in Estrogens and Nutrients in Swine Manure: Environmental Consequences. Agriculture 2015, 5, 697-712. https://doi.org/10.3390/agriculture5030697
Singh AK. Aerobic and Anaerobic Transformations in Estrogens and Nutrients in Swine Manure: Environmental Consequences. Agriculture. 2015; 5(3):697-712. https://doi.org/10.3390/agriculture5030697
Chicago/Turabian StyleSingh, Ashok K. 2015. "Aerobic and Anaerobic Transformations in Estrogens and Nutrients in Swine Manure: Environmental Consequences" Agriculture 5, no. 3: 697-712. https://doi.org/10.3390/agriculture5030697
APA StyleSingh, A. K. (2015). Aerobic and Anaerobic Transformations in Estrogens and Nutrients in Swine Manure: Environmental Consequences. Agriculture, 5(3), 697-712. https://doi.org/10.3390/agriculture5030697





