Development of Bioelectrochemical Systems to Promote Sustainable Agriculture
Abstract
:1. Introduction
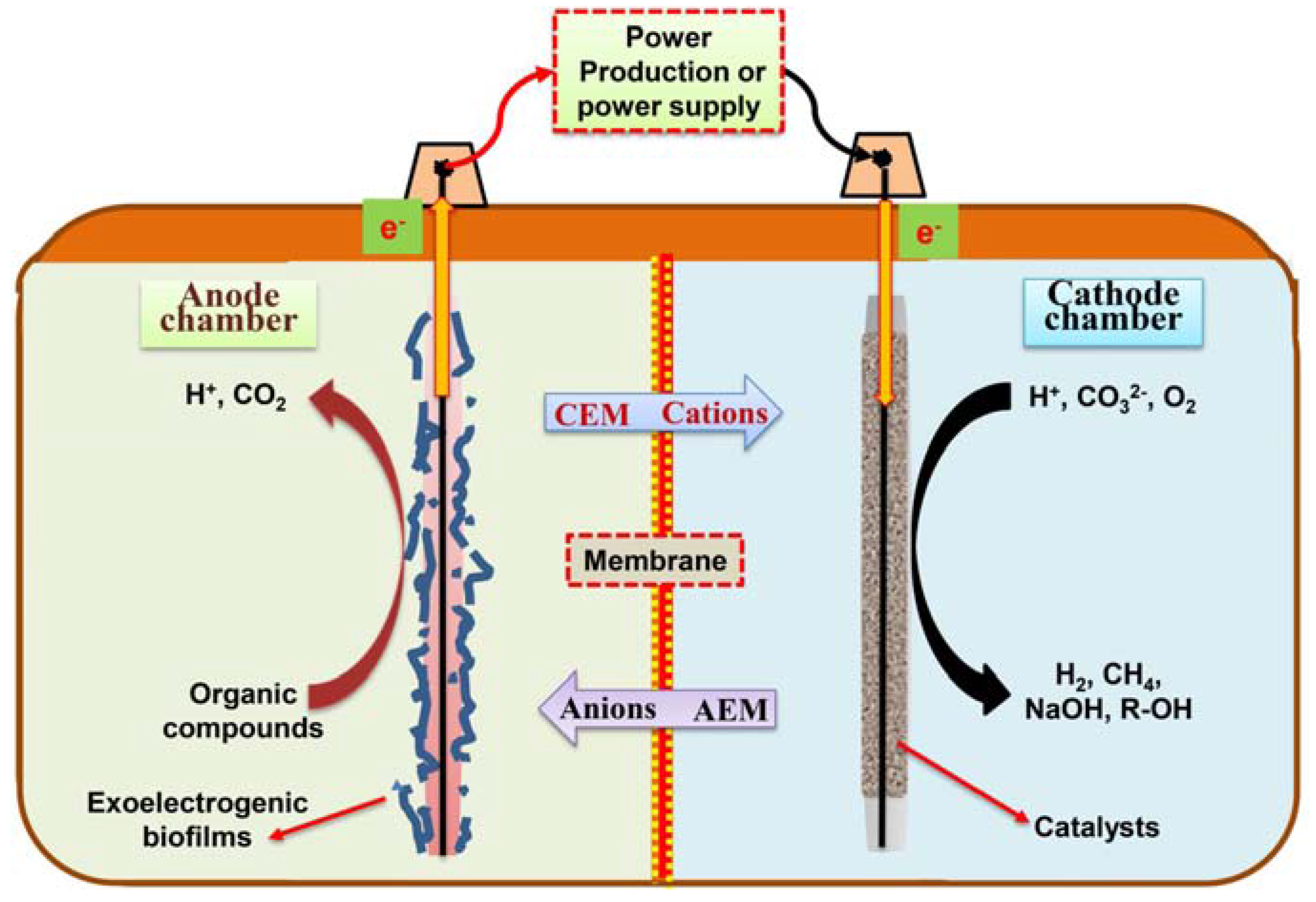
2. BES for Agricultural Waste Management
2.1. BES as a Standalone Technology
2.1.1. Animal Waste
| MFC Type | Feedstock | External Resistance | Max Area Power Density | Max Volume Power Density | Original COD | COD Removal | Ref. |
|---|---|---|---|---|---|---|---|
| Ω | mW·m−2 | W·m−3 | mg·L−1 | % | |||
| S-MFC | cattle manure | 470 | 36.6 | 0.2 | 1000 | - | [24] |
| T-MFC | cattle manure | 470 | 67 | 0.3 | 1000 | - | [24] |
| T-MFC | manure sludge | - | 5 | - | - | - | [27] |
| S-MFC | Cow manure | 1000 | 349 | - | - | - | [25] |
| S-MFC | dairy manure | 1000 | 189 | 4.7 | - | - | [28] |
| T-MFC | cow waste slurry | 460 | 0.34 | - | 1010 | 84 | [26] |
| S-MFC | swine wastewater | 200 | 261 | 1.2 | 8320 | 90 | [15] |
| T-MFC | swine wastewater | 1000 | 45 | - | 8320 | - | [15] |
| S-MFC | swine wastewater | 1000 | 228 | - | 8270 | 84 | [29] |
| S-MFC | swine wastewater | 10 | - | 1.08 | 12980 | 0.523 kg COD m−3·day−1 | [30] |
2.1.2. Plant Waste
| MFC Type | Substrate | Strains or Culture | Anode Material | Max. Power Density mW·m−2 | COD Removal % | Ref. |
|---|---|---|---|---|---|---|
| MFC | 3 g·L−1 D-0 | Clostridium cellulolyticum & Clostridium thermocellum | Pt-PTFA | 130 A·m−3 * | - | [41] |
| T-MFC | 1 g·L−1 CMC | Clostridium cellulolyticum & Geobacter sulfurreducens | graphite plates | 143 | 38 | [42] |
| T-MFC | 1 g·L−1 MN301 | 59.2 | 27 | |||
| T-MFC | 7.5 g·L−1 Sigmacell 20 | ruman | graphite plates | 55 | [45] | |
| T-MFC | 1.5 g·L−1 Sigmacell 20 | cellulolytic & exoelectrogenic bacteria | carbon paper | 880 | 50–70 | [36] |
| S-MFC | carbon paper | 1070 | 50–70 | |||
| 3-T-MFC | 1 g·L−1 rice straw powder | Nocardiopsis sp. KNU & Streptomyces enissocaesilis KNU | carbon paper | 490 | - | [46] |
| U-tube MFC | cellulose | Enterobacter cloacae | carbon cloth | 4.9 | - | [39] |
2.2. BES as a Supplementary Technology
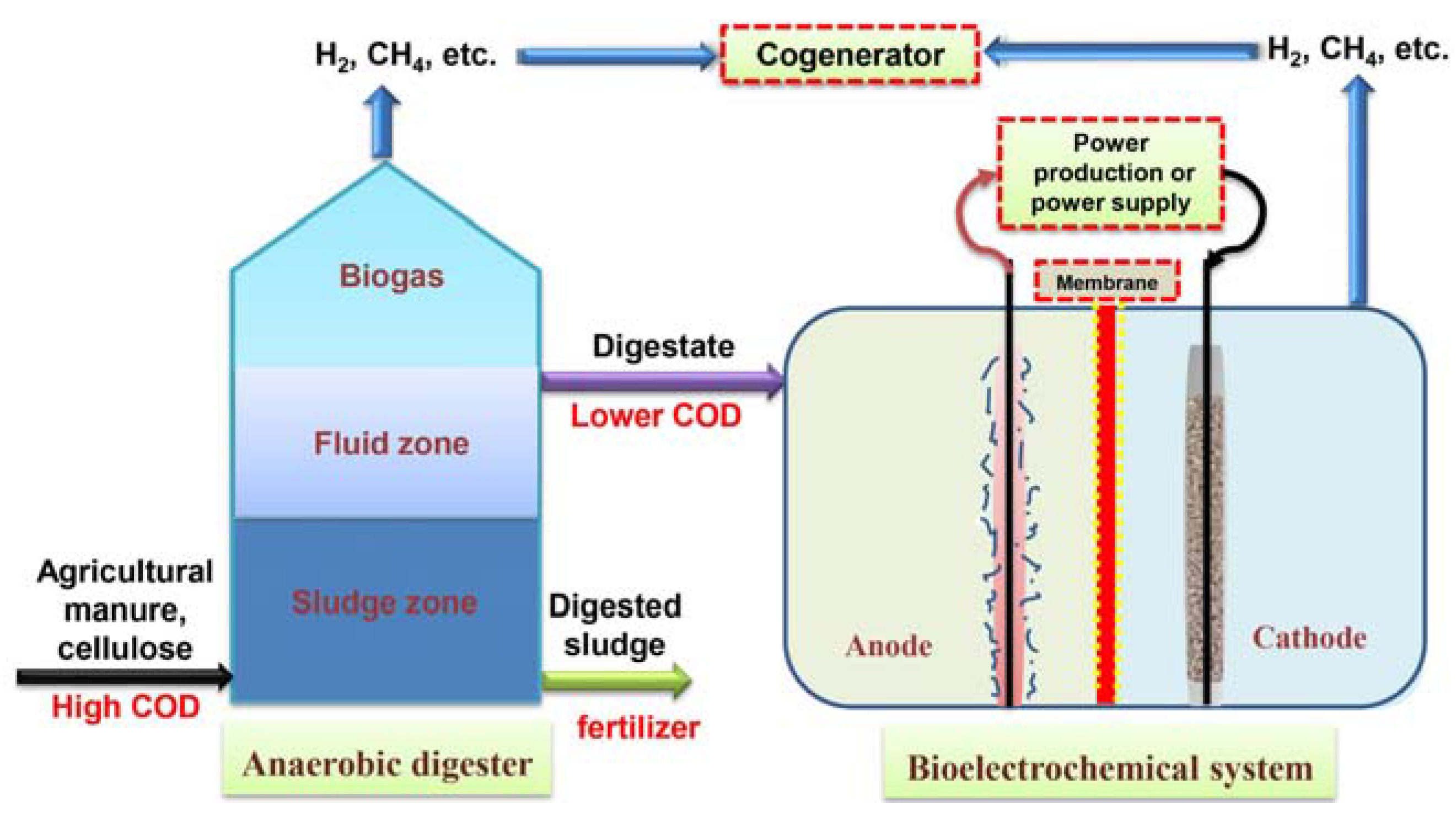
3. BES for Freshwater Supply to Agriculture
3.1. MDCs for Saline Water Desalination
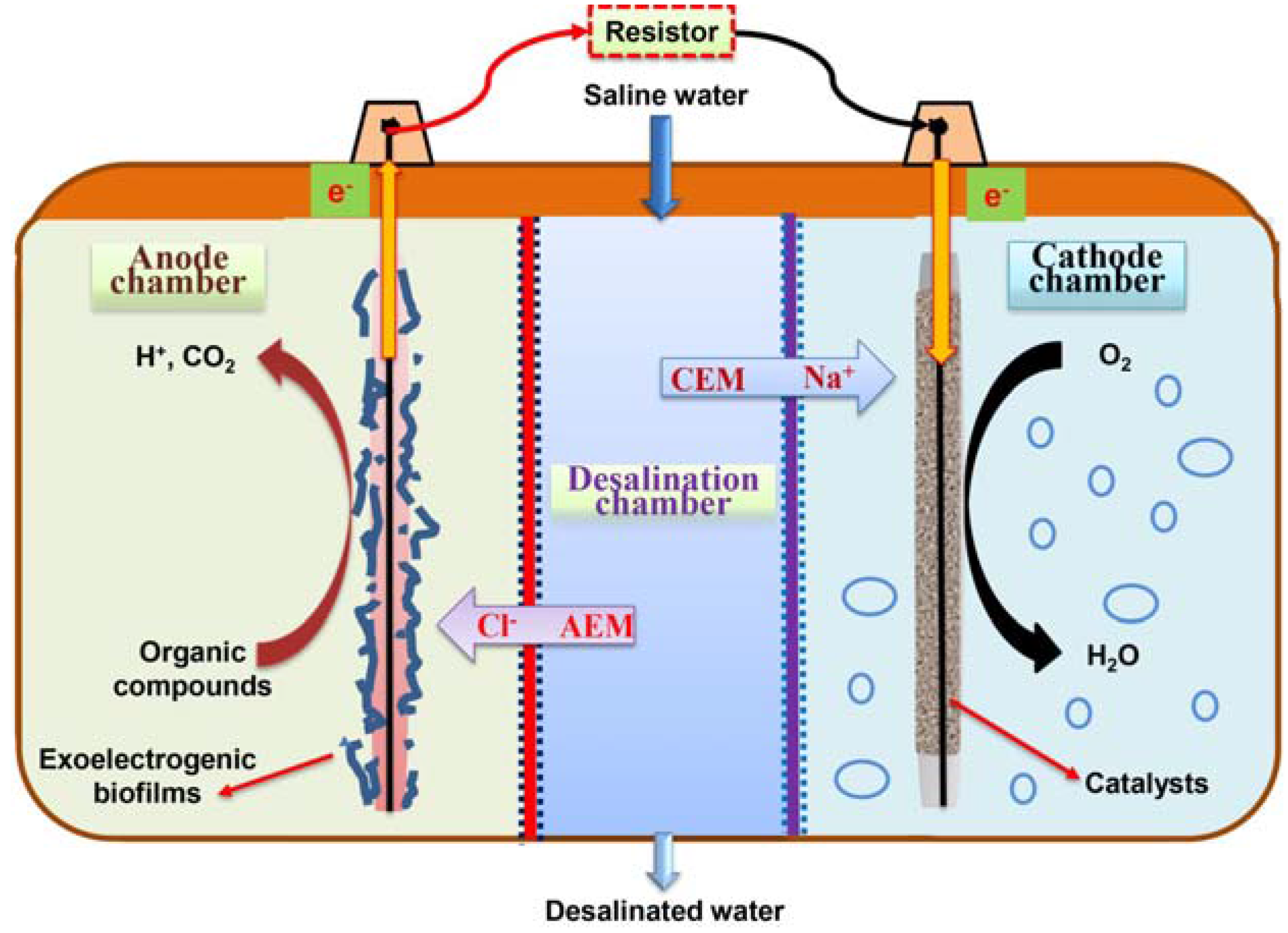
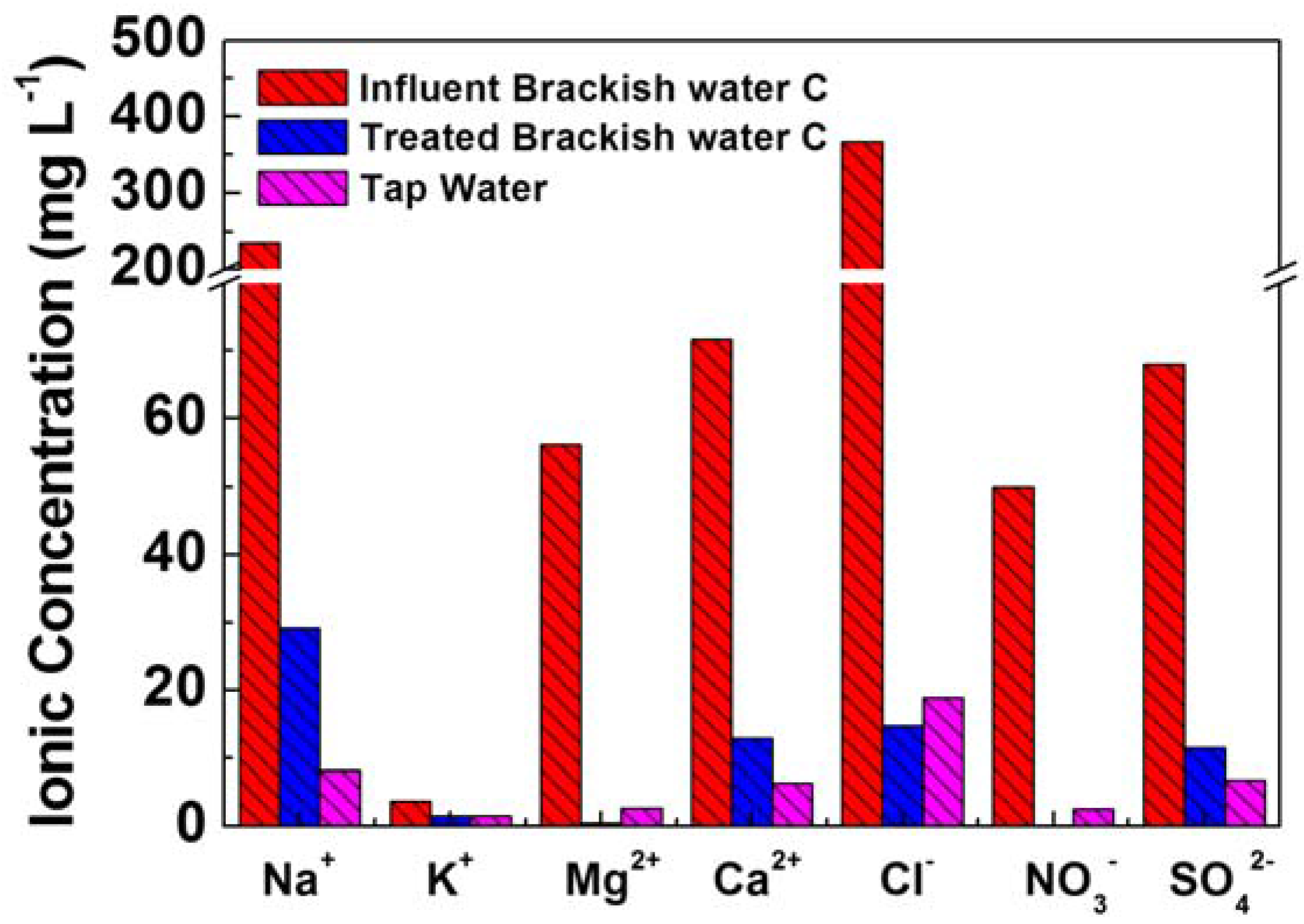
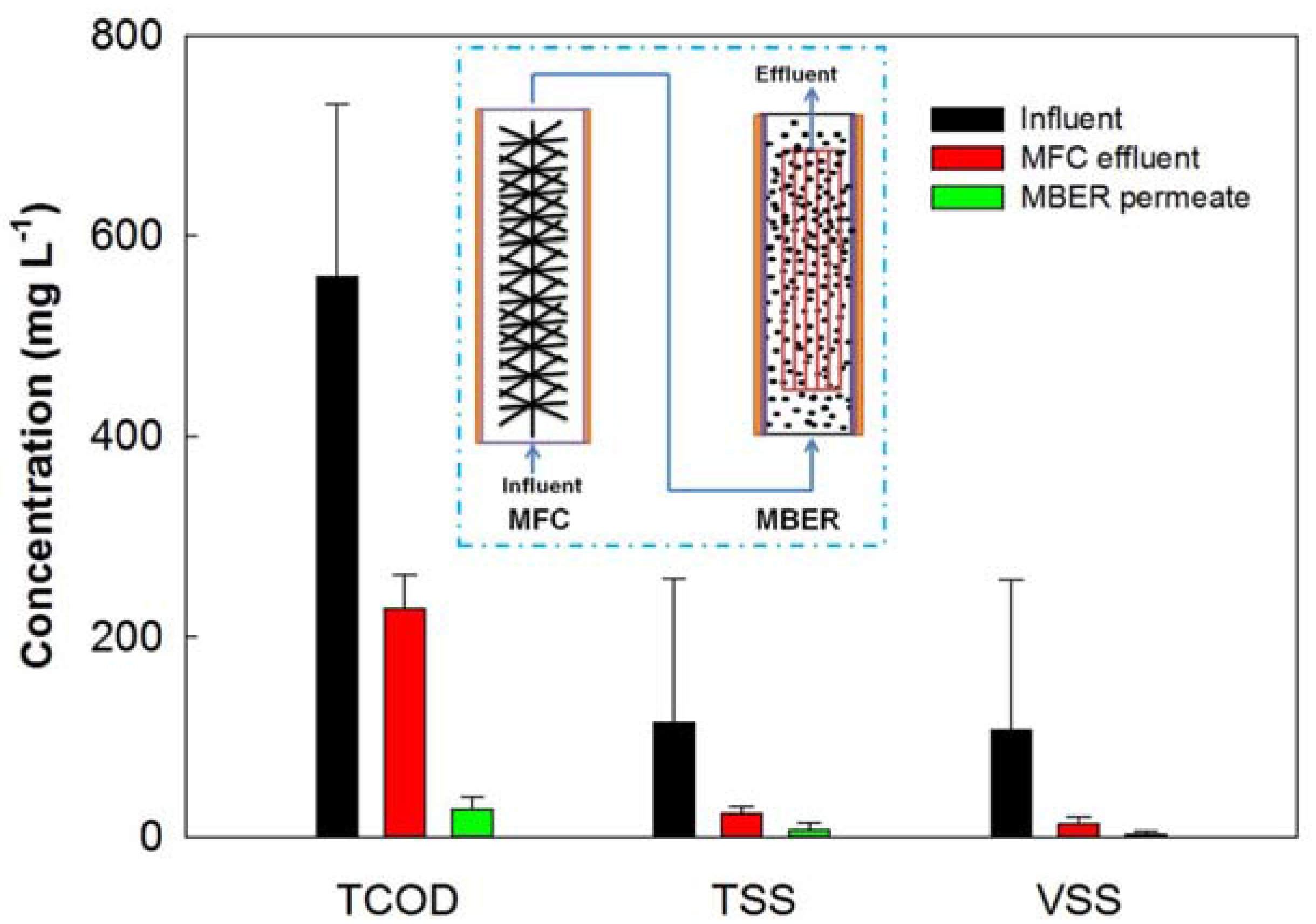
3.2. BES Integrated with Membrane Filtration for Wastewater Reclamations
| Agricultural Reuse Description | Treatment | Reclaimed Water Quality |
|---|---|---|
| • Non-processed food crops • Any crop consumed raw by human | Secondary Filtration Disinfection | • pH = 6–9 • ≤ 10 mg·L−1 BOD • ≤ 2 NTU • No detectable fecal coliforms/100 mL • ≥ 1 mg·L−1 residual chlorine * |
| • Processed food crops • Crops which are non-edible by humans, such as fodder, fiber, pasture, etc. | Secondary Disinfection | • pH = 6–9 • ≤ 30 mg·L−1 BOD • ≤ 30 mg·L−1 TSS • ≤ 200 fecal coliforms/100 mL • ≥ 1 mg·L−1 residual chlorine * |
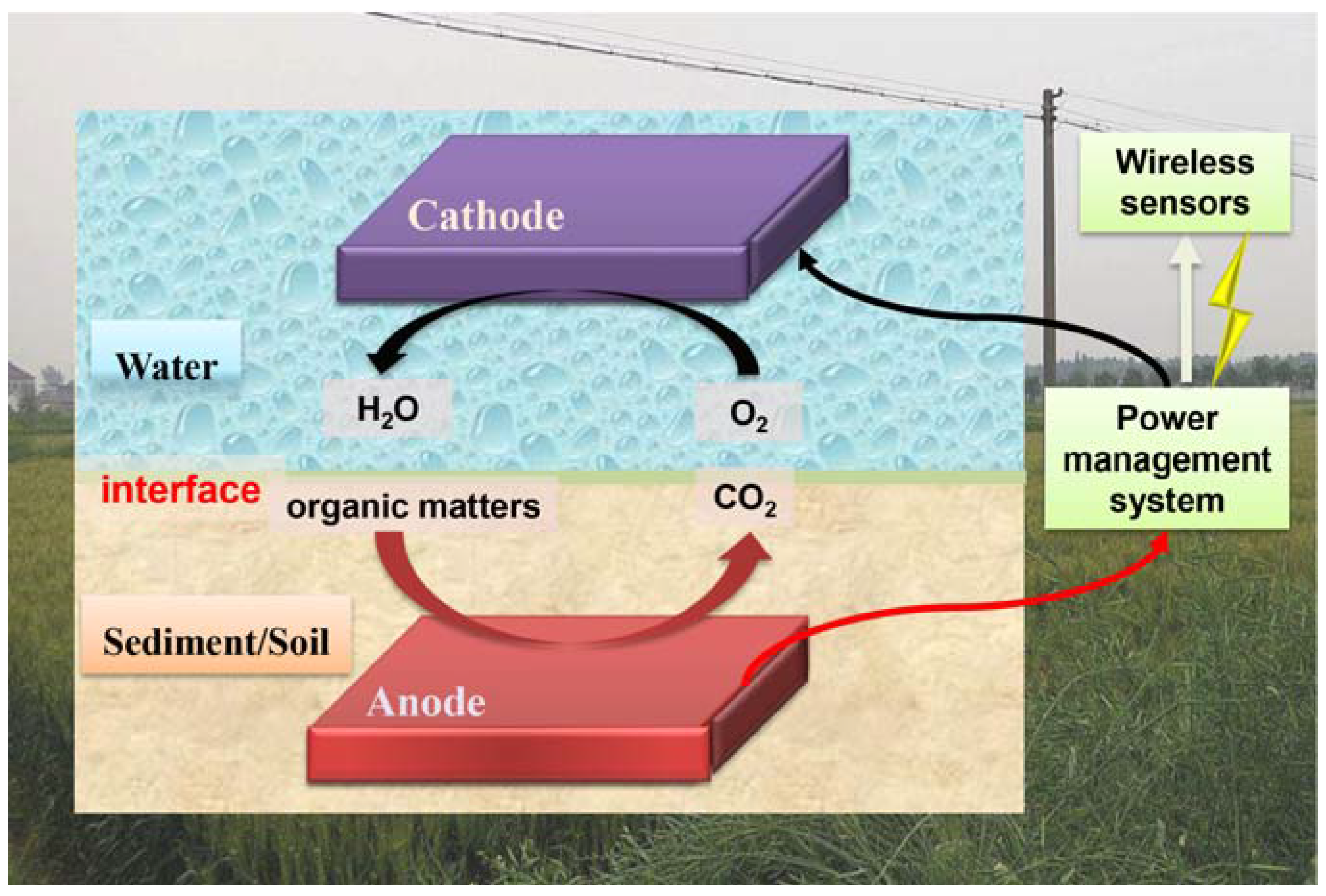
4. BES for Agricultural Monitoring
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Lu, Z.; Chang, D.; Ma, J.; Huang, G.; Cai, L.; Zhang, L. Behavior of metal ions in bioelectrochemical systems: A review. J. Power Sources 2015, 275, 243–260. [Google Scholar] [CrossRef]
- Logan, B.E. Exoelectrogenic bacteria that power microbial fuel cells. Nat. Rev. Microbiol. 2009, 7, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Torres, C.I.; Marcus, A.K.; Rittmann, B.E. Kinetics of consumption of fermentation products by anode-respiring bacteria. Appl. Microbiol. Biotechnol. 2007, 77, 689–697. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Xu, M.; Guo, J.; Sun, G. Bacterial extracellular electron transfer in bioelectrochemical systems. Process Biochem. 2012, 47, 1707–1714. [Google Scholar] [CrossRef]
- Liu, H.; Logan, B.E. Electricity generation using an air-cathode single chamber microbial fuel cell in the presence and absence of a proton exchange membrane. Environ. Sci. Technol. 2004, 38, 4040–4046. [Google Scholar] [CrossRef] [PubMed]
- Call, D.; Logan, B.E. Hydrogen production in a single chamber microbial electrolysis cell lacking a membrane. Environ. Sci. Technol. 2008, 42, 3401–3406. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Huang, X.; Liang, P.; Xiao, K.; Zhou, Y.; Zhang, X.; Logan, B.E. A new method for water desalination using microbial desalination cells. Environ. Sci. Technol. 2009, 43, 7148–7152. [Google Scholar] [CrossRef] [PubMed]
- Rabaey, K.; Rozendal, R.A. Microbial electrosynthesis—Revisiting the electrical route for microbial production. Nat. Rev. Microbiol. 2010, 8, 706–716. [Google Scholar] [CrossRef] [PubMed]
- Logan, B.E.; Call, D.; Cheng, S.; Hamelers, H.V.M.; Sleutels, T.; Jeremiasse, A.W.; Rozendal, R.A. Microbial electrolysis cells for high yield hydrogen gas production from organic matter. Environ. Sci. Technol. 2008, 42, 8630–8640. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.A.; Xing, D.F.; Call, D.F.; Logan, B.E. Direct biological conversion of electrical current into methane by electromethanogenesis. Environ. Sci. Technol. 2009, 43, 3953–3958. [Google Scholar] [CrossRef] [PubMed]
- Logan, B.E.; Rabaey, K. Conversion of wastes into bioelectricity and chemicals by using microbial electrochemical technologies. Science 2012, 337, 686–690. [Google Scholar] [CrossRef] [PubMed]
- Tender, L.M.; Gray, S.A.; Groveman, E.; Lowy, D.A.; Kauffman, P.; Melhado, J.; Tyce, R.C.; Flynn, D.; Petrecca, R.; Dobarro, J. The first demonstration of a microbial fuel cell as a viable power supply: Powering a meteorological buoy. J. Power Sources 2008, 179, 571–575. [Google Scholar] [CrossRef]
- Li, W.W.; Yu, H.Q.; He, Z. Towards sustainable wastewater treatment by using microbial fuel cells-centered technologies. Energy Environ. Sci. 2014, 7, 911–924. [Google Scholar] [CrossRef]
- Pant, D.; Singh, A.; van Bogaert, G.; Olsen, S.I.; Nigam, P.S.; Diels, L.; Vanbroekhoven, K. Bioelectrochemical systems (BES) for sustainable energy production and product recovery from organic wastes and industrial wastewaters. Rsc Adv. 2012, 2, 1248–1263. [Google Scholar] [CrossRef]
- Min, B.; Kim, J.; Oh, S.; Regan, J.M.; Logan, B.E. Electricity generation from swine wastewater using microbial fuel cells. Water Res. 2005, 39, 4961–4968. [Google Scholar] [CrossRef] [PubMed]
- Hanson, J.; Liebig, M.; Merrill, S.; Tanaka, D.; Krupinsky, J.; Stott, D. Dynamic cropping systems. Agron. J. 2007, 99, 939–943. [Google Scholar] [CrossRef]
- Öborn, I.; Edwards, A.; Witter, E.; Oenema, O.; Ivarsson, K.; Withers, P.; Nilsson, S.; Stinzing, A.R. Element balances as a tool for sustainable nutrient management: A critical appraisal of their merits and limitations within an agronomic and environmental context. Eur. J. Agron. 2003, 20, 211–225. [Google Scholar] [CrossRef]
- Dordas, C. Role of nutrients in controlling plant diseases in sustainable agriculture. A review. Agron. Sustain. Dev. 2008, 28, 33–46. [Google Scholar] [CrossRef]
- Cherry, D.C.; Huggins, B.; Gilmore, K. Children’s health in the rural environment. Pediatr. Clin. N. Am. 2007, 54, 121–133. [Google Scholar] [CrossRef] [PubMed]
- Chadwick, D. Emissions of ammonia, nitrous oxide and methane from cattle manure heaps: Effect of compaction and covering. Atmos. Environ. 2005, 39, 787–799. [Google Scholar] [CrossRef]
- Lim, S.J.; Park, W.; Kim, T.-H.; Shin, I.H. Swine wastewater treatment using a unique sequence of ion exchange membranes and bioelectrochemical system. Bioresour. Technol. 2012, 118, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Ieropoulos, I.; Greenman, J.; Melhuish, C. Microbial fuel cells based on carbon veil electrodes: Stack configuration and scalability. Int. J. Energy Res. 2008, 32, 1228–1240. [Google Scholar] [CrossRef]
- Ringeisen, B.R.; Henderson, E.; Wu, P.K.; Pietron, J.; Ray, R.; Little, B.; Biffinger, J.C.; Jones-Meehan, J.M. High power density from a miniature microbial fuel cell using shewanella oneidensis dsp10. Environ. Sci. Technol. 2006, 40, 2629–2634. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Nirmalakhandan, N. Electricity production in membrane-less microbial fuel cell fed with livestock organic solid waste. Bioresour. Technol. 2011, 102, 5831–5835. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Tang, J.; Cui, J.; Liu, Q.; Giesy, J.P.; Hecker, M. Synergy of electricity generation and waste disposal in solid-state microbial fuel cell (MFC) of cow manure composting. Int. J. Electrochem. Sci. 2014, 9, 3144–3157. [Google Scholar]
- Yokoyama, H.; Ohmori, H.; Ishida, M.; Waki, M.; Tanaka, Y. Treatment of cow-waste slurry by a microbial fuel cell and the properties of the treated slurry as a liquid manure. Anim. Sci. J. 2006, 77, 634–638. [Google Scholar] [CrossRef]
- Scott, K.; Murano, C. A study of a microbial fuel cell battery using manure sludge waste. J. Chem. Technol. Biotechnol. 2007, 82, 809–817. [Google Scholar] [CrossRef]
- Kiely, P.D.; Cusick, R.; Call, D.F.; Selembo, P.A.; Regan, J.M.; Logan, B.E. Anode microbial communities produced by changing from microbial fuel cell to microbial electrolysis cell operation using two different wastewaters. Bioresour. Technol. 2011, 102, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.R.; Dec, J.; Bruns, M.A.; Logan, B.E. Removal of odors from swine wastewater by using microbial fuel cells. Appl. Environ. Microbiol. 2008, 74, 2540–2543. [Google Scholar] [CrossRef] [PubMed]
- Ryu, J.H.; Lee, H.L.; Lee, Y.P.; Kim, T.S.; Kim, M.K.; Anh, D.T.N.; Tran, H.T.; Ahn, D.H. Simultaneous carbon and nitrogen removal from piggery wastewater using loop configuration microbial fuel cell. Process Biochem. 2013, 48, 1080–1085. [Google Scholar] [CrossRef]
- Nam, J.-Y.; Kim, H.-W.; Shin, H.-S. Ammonia inhibition of electricity generation in single-chambered microbial fuel cells. J. Power Sources 2010, 195, 6428–6433. [Google Scholar] [CrossRef]
- Wagner, R.C.; Regan, J.M.; Oh, S.-E.; Zuo, Y.; Logan, B.E. Hydrogen and methane production from swine wastewater using microbial electrolysis cells. Water Res. 2009, 43, 1480–1488. [Google Scholar] [CrossRef] [PubMed]
- Ichihashi, O.; Hirooka, K. Removal and recovery of phosphorus as struvite from swine wastewater using microbial fuel cell. Bioresour. Technol. 2012, 114, 303–307. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.R.; Zuo, Y.; Regan, J.M.; Logan, B.E. Analysis of ammonia loss mechanisms in microbial fuel cells treating animal wastewater. Biotechnol. Bioeng. 2008, 99, 1120–1127. [Google Scholar] [CrossRef] [PubMed]
- Rittmann, B.E.; McCarty, P.L. Environmental Biotechnology: Principles and Applications; McGraw-Hill Education: Boston, MA, USA, 2001. [Google Scholar]
- Cheng, S.A.; Kiely, P.; Logan, B.E. Pre-acclimation of a wastewater inoculum to cellulose in an aqueous-cathode mec improves power generation in air-cathode mfcs. Bioresour. Technol. 2011, 102, 367–371. [Google Scholar] [CrossRef] [PubMed]
- Bridgwater, T. Biomass for energy. J. Sci. Food Agric. 2006, 86, 1755–1768. [Google Scholar] [CrossRef]
- Lynd, L.R. Overview and evaluation of fuel ethanol from cellulosic biomass: Technology, economics, the environment, and policy. Annu. Rev. Energy Environ. 1996, 21, 403–465. [Google Scholar] [CrossRef]
- Rezaei, F.; Xing, D.; Wagner, R.; Regan, J.M.; Richard, T.L.; Logan, B.E. Simultaneous cellulose degradation and electricity production by enterobacter cloacae in a microbial fuel cell. Appl. Environ. Microbiol. 2009, 75, 3673–3678. [Google Scholar] [CrossRef] [PubMed]
- Rezaei, F.; Richard, T.L.; Logan, B.E. Enzymatic hydrolysis of cellulose coupled with electricity generation in a microbial fuel cell. Biotechnol. Bioeng. 2008, 101, 1163–1169. [Google Scholar] [CrossRef] [PubMed]
- Niessen, J.; Schröder, U.; Harnisch, F.; Scholz, F. Gaining electricity from in situ oxidation of hydrogen produced by fermentative cellulose degradation. Lett. Appl. Microbiol. 2005, 41, 286–290. [Google Scholar] [CrossRef] [PubMed]
- Ren, Z.; Ward, T.E.; Regan, J.M. Electricity production from cellulose in a microbial fuel cell using a defined binary culture. Environ. Sci. Technol. 2007, 41, 4781–4786. [Google Scholar] [CrossRef] [PubMed]
- Hassan, S.H.; Kim, Y.S.; Oh, S.-E. Power generation from cellulose using mixed and pure cultures of cellulose-degrading bacteria in a microbial fuel cell. Enzym. Microb. Technol. 2012, 51, 269–273. [Google Scholar] [CrossRef] [PubMed]
- Krause, D.O.; Denman, S.E.; Mackie, R.I.; Morrison, M.; Rae, A.L.; Attwood, G.T.; McSweeney, C.S. Opportunities to improve fiber degradation in the rumen: Microbiology, ecology, and genomics. FEMS Microbiol. Rev. 2003, 27, 663–693. [Google Scholar] [CrossRef]
- Rismani-Yazdi, H.; Christy, A.D.; Dehority, B.A.; Morrison, M.; Yu, Z.; Tuovinen, O.H. Electricity generation from cellulose by rumen microorganisms in microbial fuel cells. Biotechnol. Bioeng. 2007, 97, 1398–1407. [Google Scholar] [CrossRef] [PubMed]
- Hassan, S.H.; El-Rab, S.M.G.; Rahimnejad, M.; Ghasemi, M.; Joo, J.-H.; Sik-Ok, Y.; Kim, I.S.; Oh, S.-E. Electricity generation from rice straw using a microbial fuel cell. Int. J. Hydrog. Energy 2014, 39, 9490–9496. [Google Scholar] [CrossRef]
- Reese, E. Polysaccharases and the hydrolysis of insoluble substrates. In Biological Transformation of Wood by Microorganisms; Springer-Verlag: Berlin/Heidelberg, Germany, 1975; pp. 165–181. [Google Scholar]
- Takahashi, M.; Takahashi, H.; Nakano, Y.; Konishi, T.; Terauchi, R.; Takeda, T. Characterization of a cellobiohydrolase (MoCel6A) produced by magnaporthe oryzae. Appl. Environ. Microbiol. 2010, 76, 6583–6590. [Google Scholar] [CrossRef] [PubMed]
- Sørensen, A.; Lübeck, M.; Lübeck, P.S.; Ahring, B.K. Fungal beta-glucosidases: A bottleneck in industrial use of lignocellulosic materials. Biomolecules 2013, 3, 612–631. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Cheng, J. Hydrolysis of lignocellulosic materials for ethanol production: A review. Bioresour. Technol. 2002, 83, 1–11. [Google Scholar] [CrossRef]
- Lalaurette, E.; Thammannagowda, S.; Mohagheghi, A.; Maness, P.C.; Logan, B.E. Hydrogen production from cellulose in a two-stage process combining fermentation and electrohydrogenesis. Int. J. Hydrog. Energy 2009, 34, 6201–6210. [Google Scholar] [CrossRef]
- Wang, A.J.; Sun, D.; Cao, G.L.; Wang, H.Y.; Ren, N.Q.; Wu, W.M.; Logan, B.E. Integrated hydrogen production process from cellulose by combining dark fermentation, microbial fuel cells, and a microbial electrolysis cell. Bioresour. Technol. 2011, 102, 4137–4143. [Google Scholar] [CrossRef] [PubMed]
- Angenent, L.T.; Karim, K.; Al-Dahhan, M.H.; Wrenn, B.A.; Domíguez-Espinosa, R. Production of bioenergy and biochemicals from industrial and agricultural wastewater. Trends Biotechnol. 2004, 22, 477–485. [Google Scholar] [CrossRef] [PubMed]
- Weld, R.J.; Singh, R. Functional stability of a hybrid anaerobic digester/microbial fuel cell system treating municipal wastewater. Bioresour. Technol. 2011, 102, 842–847. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.R.; Premier, G.C.; Hawkes, F.R.; Rodríguez, J.; Dinsdale, R.M.; Guwy, A.J. Modular tubular microbial fuel cells for energy recovery during sucrose wastewater treatment at low organic loading rate. Bioresour. Technol. 2010, 101, 1190–1198. [Google Scholar] [CrossRef] [PubMed]
- Rozendal, R.A.; Leone, E.; Keller, J.; Rabaey, K. Efficient hydrogen peroxide generation from organic matter in a bioelectrochemical system. Electrochem. Commun. 2009, 11, 1752–1755. [Google Scholar] [CrossRef]
- Rabaey, K.; Butzer, S.; Brown, S.; Keller, J.; Rozendal, R.A. High current generation coupled to caustic production using a lamellar bioelectrochemical system. Environ. Sci. Technol. 2010, 44, 4315–4321. [Google Scholar] [CrossRef] [PubMed]
- Verstraete, W.; Morgan-Sagastume, F.; Aiyuk, S.; Waweru, M.; Rabaey, K.; Lissens, G. Anaerobic digestion as a core technology in sustainable management of organic matter. Water Sci. Technol. 2005, 52, 59–66. [Google Scholar] [PubMed]
- Pham, T.; Rabaey, K.; Aelterman, P.; Clauwaert, P.; de Schamphelaire, L.; Boon, N.; Verstraete, W. Microbial fuel cells in relation to conventional anaerobic digestion technology. Eng. Life Sci. 2006, 6, 285–292. [Google Scholar] [CrossRef]
- Sung, S.; Liu, T. Ammonia inhibition on thermophilic anaerobic digestion. Chemosphere 2003, 53, 43–52. [Google Scholar] [CrossRef]
- Cord-Ruwisch, R.; Law, Y.; Cheng, K.Y. Ammonium as a sustainable proton shuttle in bioelectrochemical systems. Bioresour. Technol. 2011, 102, 9691–9696. [Google Scholar] [CrossRef] [PubMed]
- Desloover, J.; Abate Woldeyohannis, A.; Verstraete, W.; Boon, N.; Rabaey, K. Electrochemical resource recovery from digestate to prevent ammonia toxicity during anaerobic digestion. Environ. Sci. Technol. 2012, 46, 12209–12216. [Google Scholar] [CrossRef] [PubMed]
- Desloover, J.; de Vrieze, J.; de Vijver, M.V.; Mortelmans, J.; Rozendal, R.; Rabaey, K. Electrochemical nutrient recovery enables ammonia toxicity control and biogas desulfurization in anaerobic digestion. Environ. Sci. Technol. 2015, 49, 948–955. [Google Scholar] [CrossRef] [PubMed]
- Rabaey, K.; van de Sompel, K.; Maignien, L.; Boon, N.; Aelterman, P.; Clauwaert, P.; de Schamphelaire, L.; Pham, H.T.; Vermeulen, J.; Verhaege, M. Microbial fuel cells for sulfide removal. Environ. Sci. Technol. 2006, 40, 5218–5224. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Mu, Z.-X.; Chen, Y.-P.; Sheng, G.-P.; Liu, X.-W.; Chen, Y.-Z.; Zhao, Y.; Wang, H.-L.; Yu, H.-Q.; Wei, L.; et al. Microbe-assisted sulfide oxidation in the anode of a microbial fuel cell. Environ. Sci. Technol. 2009, 43, 3372–3377. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Tong, Z.-H.; Sheng, G.-P.; Chen, Y.-Z.; Zhang, F.; Mu, Z.-X.; Wang, H.-L.; Zeng, R.J.; Liu, X.-W.; Yu, H.-Q.; et al. Microbial communities involved in electricity generation from sulfide oxidation in a microbial fuel cell. Biosens. Bioelectron. 2010, 26, 470–476. [Google Scholar] [CrossRef] [PubMed]
- Ieropoulos, I.; Greenman, J.; Melhuish, C.; Hart, J. Energy accumulation and improved performance in microbial fuel cells. J. Power Sources 2005, 145, 253–256. [Google Scholar] [CrossRef]
- Zhang, Y.; Angelidaki, I. Submersible microbial desalination cell for simultaneous ammonia recovery and electricity production from anaerobic reactors containing high levels of ammonia. Bioresour. Technol. 2015, 177, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Angelidaki, I. Counteracting ammonia inhibition during anaerobic digestion by recovery using submersible microbial desalination cell. Biotechnol. Bioeng. 2015, 112, 1478–1482. [Google Scholar] [CrossRef] [PubMed]
- Ghermandi, A.; Messalem, R. The advantages of nf desalination of brackish water for sustainable irrigation: The case of the arava valley in israel. Desalin. Water Treat. 2009, 10, 101–107. [Google Scholar] [CrossRef]
- Assessment, M.E. Ecosystems and Human Well-Being: Current State and Trends; Island Press: Washington, DC, USA, 2005. [Google Scholar]
- UNESCO. The 3rd United Nations World Water Development Report: Water in a Changing World; United Nations Educational, Scientific, and Cultural Organization: Paris, France, 2009. [Google Scholar]
- Shannon, M.A.; Bohn, P.W.; Elimelech, M.; Georgiadis, J.G.; Mariñas, B.J.; Mayes, A.M. Science and technology for water purification in the coming decades. Nature 2008, 452, 301–310. [Google Scholar] [CrossRef] [PubMed]
- Miller, G.W. Integrated concepts in water reuse: Managing global water needs. Desalination 2006, 187, 65–75. [Google Scholar] [CrossRef]
- Elimelech, M.; Phillip, W.A. The future of seawater desalination: Energy, technology, and the environment. Science 2011, 333, 712–717. [Google Scholar] [CrossRef] [PubMed]
- Redondo, J. Brackish-, sea-and wastewater desalination. Desalination 2001, 138, 29–40. [Google Scholar] [CrossRef]
- Mathioulakis, E.; Belessiotis, V.; Delyannis, E. Desalination by using alternative energy: Review and state-of-the-art. Desalination 2007, 203, 346–365. [Google Scholar] [CrossRef]
- Zhang, B.; He, Z. Energy production, use and saving in a bioelectrochemical desalination system. Rsc Adv. 2012, 2, 10673–10679. [Google Scholar] [CrossRef]
- Jacobson, K.S.; Drew, D.M.; He, Z. Use of a liter-scale microbial desalination cell as a platform to study bioelectrochemical desalination with salt solution or artificial seawater. Environ. Sci. Technol. 2011, 45, 4652–4657. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Ren, Z.J. A comprehensive review of microbial electrochemical systems as a platform technology. Biotechnol. Adv. 2013, 31, 1796–1807. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; He, Z. Improving water desalination by hydraulically coupling an osmotic microbial fuel cell with a microbial desalination cell. J. Membr. Sci. 2013, 441, 18–24. [Google Scholar] [CrossRef]
- Jacobson, K.S.; Drew, D.M.; He, Z. Efficient salt removal in a continuously operated upflow microbial desalination cell with an air cathode. Bioresour. Technol. 2011, 102, 376–380. [Google Scholar] [CrossRef] [PubMed]
- Brastad, K.S.; He, Z. Water softening using microbial desalination cell technology. Desalination 2013, 309, 32–37. [Google Scholar] [CrossRef]
- Chen, X.; Xia, X.; Liang, P.; Cao, X.; Sun, H.; Huang, X. Stacked microbial desalination cells to enhance water desalination efficiency. Environ. Sci. Technol. 2011, 45, 2465–2470. [Google Scholar] [CrossRef] [PubMed]
- Mehanna, M.; Saito, T.; Yan, J.; Hickner, M.; Cao, X.; Huang, X.; Logan, B.E. Using microbial desalination cells to reduce water salinity prior to reverse osmosis. Energy Environ. Sci. 2010, 3, 1114–1120. [Google Scholar] [CrossRef]
- Ping, Q.; Huang, Z.; Dosoretz, C.; He, Z. Integrated experimental investigation and mathematical modeling of brackish water desalination and wastewater treatment in microbial desalination cells. Water Res. 2015, 77, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Pang, H.-C.; Li, Y.-Y.; Yang, J.-S.; Liang, Y.-S. Effect of brackish water irrigation and straw mulching on soil salinity and crop yields under monsoonal climatic conditions. Agric. Water Manag. 2010, 97, 1971–1977. [Google Scholar] [CrossRef]
- Ayers, R.S.; Westcot, D.W. Water Quality for Agriculture; Fao irrigation and drainage paper 29 rev. 1; Food and Agricultural Organization: Rome, Italy, 1985. [Google Scholar]
- Scianna, J.; Pick, T.; Logar, R. Plant materials technical note number MT-62: Determining the suitability of salt-affected water and soil for tree and shrub plantings. Available online: http://www.nrcs.usda.gov/wps/portal/nrcs/detail/mt/homeowner/?cid=nrcs2144p2012_057692 (accessed on 28 April 2015).
- Gardiner, D.T.; Miller, R.W. Soils in our Environment; Pearson/Prentice Hall: Upper Saddle River, New Jersey, 2008. [Google Scholar]
- Pedersen, J.A.; Yeager, M.A.; Suffet, I. Xenobiotic organic compounds in runoff from fields irrigated with treated wastewater. J. Agric. Food Chem. 2003, 51, 1360–1372. [Google Scholar] [CrossRef] [PubMed]
- Levine, A.D.; Asano, T. Peer reviewed: Recovering sustainable water from wastewater. Environ. Sci. Technol. 2004, 38, 201A–208A. [Google Scholar] [CrossRef] [PubMed]
- Angelakis, A.; Do Monte, M.M.; Bontoux, L.; Asano, T. The status of wastewater reuse practice in the mediterranean basin: Need for guidelines. Water Res. 1999, 33, 2201–2217. [Google Scholar] [CrossRef]
- Chen, W.; Lu, S.; Peng, C.; Jiao, W.; Wang, M. Accumulation of cd in agricultural soil under long-term reclaimed water irrigation. Environ. Pollut. 2013, 178, 294–299. [Google Scholar] [CrossRef] [PubMed]
- Oron, G.; Goemans, M.; Manor, Y.; Feyen, J. Poliovirus distribution in the soil-plant system under reuse of secondary wastewater. Water Res. 1995, 29, 1069–1078. [Google Scholar] [CrossRef]
- Xu, J.; Wu, L.; Chang, A.C.; Zhang, Y. Impact of long-term reclaimed wastewater irrigation on agricultural soils: A preliminary assessment. J. Hazard. Mater. 2010, 183, 780–786. [Google Scholar] [CrossRef] [PubMed]
- USEPA. Guidelines for Water Reuse, 2012; U.S. Environmental Protection Agency: Washington, DC, USA, 2012. Available online: http://nepis.epa.gov/Adobe/PDF/P2100FS2017K.pdf (accessed on 28 April 2015).
- Howell, J.A. Future of membranes and membrane reactors in green technologies and for water reuse. Desalination 2004, 162, 1–11. [Google Scholar] [CrossRef]
- Quist-Jensen, C.A.; Macedonio, F.; Drioli, E. Membrane technology for water production in agriculture: Desalination and wastewater reuse. Desalination 2015, 364, 17–32. [Google Scholar] [CrossRef]
- Melin, T.; Jefferson, B.; Bixio, D.; Thoeye, C.; de Wilde, W.; de Koning, J.; van der Graaf, J.; Wintgens, T. Membrane bioreactor technology for wastewater treatment and reuse. Desalination 2006, 187, 271–282. [Google Scholar] [CrossRef]
- Yuan, H.; He, Z. Integrating membrane filtration into bioelectrochemical systems as next generation energy-efficient wastewater treatment technologies for water reclamation: A review. Bioresour. Technol. 2015, in press. [Google Scholar]
- Wang, Y.-P.; Liu, X.-W.; Li, W.-W.; Li, F.; Wang, Y.-K.; Sheng, G.-P.; Zeng, R.J.; Yu, H.-Q. A microbial fuel cell–membrane bioreactor integrated system for cost-effective wastewater treatment. Appl. Energy 2012, 98, 230–235. [Google Scholar] [CrossRef]
- Ge, Z.; Ping, Q.Y.; He, Z. Hollow-fiber membrane bioelectrochemical reactor for domestic wastewater treatment. J. Chem. Technol. Biotechnol. 2013, 88, 1584–1590. [Google Scholar] [CrossRef]
- Li, J.; Ge, Z.; He, Z. A fluidized bed membrane bioelectrochemical reactor for energy-efficient wastewater treatment. Bioresour. Technol. 2014, 167, 310–315. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Ge, Z.; He, Z. Advancing membrane bioelectrochemical reactor (MBER) with hollow-fiber membranes installed in the cathode compartment. J. Chem. Technol. Biotechnol. 2014, 89, 1330–1336. [Google Scholar] [CrossRef]
- Hoover, L.A.; Phillip, W.A.; Tiraferri, A.; Yip, N.Y.; Elimelech, M. Forward with osmosis: Emerging applications for greater sustainability. Environ. Sci. Technol. 2011, 45, 9824–9830. [Google Scholar] [CrossRef] [PubMed]
- Coates, R.W.; Delwiche, M.J.; Broad, A.; Holler, M. Wireless sensor network with irrigation valve control. Comput. Electron. Agric. 2013, 96, 13–22. [Google Scholar] [CrossRef]
- Zhang, R.-B.; Guo, J.-J.; Zhang, L.; Zhang, Y.-C.; Wang, L.-H.; Wang, Q. A calibration method of detecting soil water content based on the information-sharing in wireless sensor network. Comput. Electron. Agric. 2011, 76, 161–168. [Google Scholar] [CrossRef]
- Dong, X.; Vuran, M.C.; Irmak, S. Autonomous precision agriculture through integration of wireless underground sensor networks with center pivot irrigation systems. Ad Hoc Netw. 2013, 11, 1975–1987. [Google Scholar] [CrossRef]
- Srbinovska, M.; Gavrovski, C.; Dimcev, V.; Krkoleva, A.; Borozan, V. Environmental parameters monitoring in precision agriculture using wireless sensor networks. J. Clean. Prod. 2015, 88, 297–307. [Google Scholar] [CrossRef]
- Akbari, S. Energy Harvesting for Wireless Sensor Networks Review. Proceedings of the 2014 Federated Conference on Computer Science and Information Systems (FedCSIS), Warsaw, Poland, 7–10 September 2014; Available online: http://ieeexplore.ieee.org/xpl/articleDetails.jsp?reload=true&arnumber=6933124 (accessed on 29 April 2015).
- Zhang, F.; Tian, L.; He, Z. Powering a wireless temperature sensor using sediment microbial fuel cells with vertical arrangement of electrodes. J. Power Sources 2011, 196, 9568–9573. [Google Scholar] [CrossRef]
- De Schamphelaire, L.; Rabaey, K.; Boeckx, P.; Boon, N.; Verstraete, W. Outlook for benefits of sediment microbial fuel cells with two bio-electrodes. Microb. Biotechnol. 2008, 1, 446–462. [Google Scholar] [CrossRef] [PubMed]
- Bond, D.R.; Holmes, D.E.; Tender, L.M.; Lovley, D.R. Electrode-reducing microorganisms that harvest energy from marine sediments. Science 2002, 295, 483–485. [Google Scholar] [CrossRef] [PubMed]
- Tender, L.M.; Reimers, C.E.; Stecher, H.A.; Holmes, D.E.; Bond, D.R.; Lowy, D.A.; Pilobello, K.; Fertig, S.J.; Lovley, D.R. Harnessing microbially generated power on the seafloor. Nat. Biotechnol. 2002, 20, 821–825. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Ge, Z.; He, Z. Sediment microbial fuel cells for wastewater treatment: Challenges and opportunities. Environ. Sci.: Water Res. Technol. 2015, 1, 279–284. [Google Scholar] [CrossRef]
- He, Z.; Shao, H.B.; Angenent, L.T. Increased power production from a sediment microbial fuel cell with a rotating cathode. Biosens. Bioelectron. 2007, 22, 3252–3255. [Google Scholar] [CrossRef] [PubMed]
- Reimers, C.; Girguis, P.; Stecher, H.; Tender, L.; Ryckelynck, N.; Whaling, P. Microbial fuel cell energy from an ocean cold seep. Geobiology 2006, 4, 123–136. [Google Scholar] [CrossRef]
- Shantaram, A.; Beyenal, H.; Raajan, R.; Veluchamy, A.; Lewandowski, Z. Wireless sensors powered by microbial fuel cells. Environ. Sci. Technol. 2005, 39, 5037–5042. [Google Scholar] [CrossRef] [PubMed]
- Donovan, C.; Dewan, A.; Heo, D.; Beyenal, H. Batteryless, wireless sensor powered by a sediment microbial fuel cell. Environ. Sci. Technol. 2008, 42, 8591–8596. [Google Scholar] [CrossRef] [PubMed]
- Ieropoulos, I.; Greenman, J.; Melhuish, C. Imitating metabolism: Energy autonomy in biologically inspired robots. In Proceedings of the 2nd International Symposium on Imitation of Animals and Artifacts, Aberystwyth, UK, 7–11 April 2003; pp. 191–194.
- Ieropoulos, I.; Greenman, J.; Melhuish, C.; Horsfield, I. Ecobot-III-a Robot with Guts. Proceedings of the Alife XII Conference, Odense, Denmark, 19–23 August 2010; pp. 733–740. Available online: https://mitpress.mit.edu/sites/default/files/titles/alife/0262290758chap131.pdf (accessed on 29 April 2015).
- Ieropoulos, I.; Melhuish, C.; Greenman, J.; Horsfield, I. Ecobot-ii: An artificial agent with a natural metabolism. J. Adv. Robot. Syst. 2005, 2, 295–300. [Google Scholar] [CrossRef]
- Wilkinson, S. “Gastrobots”—Benefits and challenges of microbial fuel cells in foodpowered robot applications. Auton. Robot. 2000, 9, 99–111. [Google Scholar] [CrossRef]
- Donovan, C.; Dewan, A.; Peng, H.; Heo, D.; Beyenal, H. Power management system for a 2.5 w remote sensor powered by a sediment microbial fuel cell. J. Power Sources 2011, 196, 1171–1177. [Google Scholar] [CrossRef]
- Selavo, L.; Wood, A.; Cao, Q.; Sookoor, T.; Liu, H.; Srinivasan, A.; Wu, Y.; Kang, W.; Stankovic, J.; Young, D. Luster: Wireless Sensor Network for Environmental Research. In Proceedings of the 5th International Conference on Embedded Networked Sensor Systems, 6–9 November 2007; ACM: Sydney, Australia; pp. 103–116.
- Baggio, A. Wireless Sensor Networks in Precision Agriculture. In Proceedings of the ACM Workshop on Real-World Wireless Sensor Networks (REALWSN 2005), Stockholm, Sweden, 20–21 June 2005.
- Vellidis, G.; Tucker, M.; Perry, C.; Kvien, C.; Bednarz, C. A real-time wireless smart sensor array for scheduling irrigation. Comput. Electron. Agric. 2008, 61, 44–50. [Google Scholar] [CrossRef]
- Hokazono, S.; Hayashi, K. Variability in environmental impacts during conversion from conventional to organic farming: A comparison among three rice production systems in japan. J. Clean. Prod. 2012, 28, 101–112. [Google Scholar] [CrossRef]
- Huang, D.-Y.; Zhou, S.-G.; Chen, Q.; Zhao, B.; Yuan, Y.; Zhuang, L. Enhanced anaerobic degradation of organic pollutants in a soil microbial fuel cell. Chem. Eng. J. 2011, 172, 647–653. [Google Scholar] [CrossRef]
- Li, X.; Wang, X.; Zhang, Y.; Cheng, L.; Liu, J.; Li, F.; Gao, B.; Zhou, Q. Extended petroleum hydrocarbon bioremediation in saline soil using pt-free multianodes microbial fuel cells. RSC Adv. 2014, 4, 59803–59808. [Google Scholar] [CrossRef]
- Rodrigo, J.; Boltes, K.; Esteve-Nuñez, A. Microbial-electrochemical bioremediation and detoxification of dibenzothiophene-polluted soil. Chemosphere 2014, 101, 61–65. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Wang, C.; Zheng, Z.-Y.; Che, J.; Xiao, Y.; Yang, Z.-H.; Zhao, F. Ameliorating acidic soil using bioelectrochemistry systems. RSC Adv. 2014, 4, 62544–62549. [Google Scholar] [CrossRef]
- Moqsud, M.A.; Yoshitake, J.; Bushra, Q.S.; Hyodo, M.; Omine, K.; Strik, D. Compost in plant microbial fuel cell for bioelectricity generation. Waste Manag. (Oxf.) 2015, 36, 63–69. [Google Scholar] [CrossRef] [PubMed]
- De Schamphelaire, L.; Van den Bossche, L.; Dang, H.S.; Höfte, M.; Boon, N.; Rabaey, K.; Verstraete, W. Microbial fuel cells generating electricity from rhizodeposits of rice plants. Environ. Sci. Technol. 2008, 42, 3053–3058. [Google Scholar] [CrossRef] [PubMed]
- Kaku, N.; Yonezawa, N.; Kodama, Y.; Watanabe, K. Plant/microbe cooperation for electricity generation in a rice paddy field. Appl. Microbiol. Biotechnol. 2008, 79, 43–49. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, X.; Abu-Reesh, I.M.; He, Z. Development of Bioelectrochemical Systems to Promote Sustainable Agriculture. Agriculture 2015, 5, 367-388. https://doi.org/10.3390/agriculture5030367
Li X, Abu-Reesh IM, He Z. Development of Bioelectrochemical Systems to Promote Sustainable Agriculture. Agriculture. 2015; 5(3):367-388. https://doi.org/10.3390/agriculture5030367
Chicago/Turabian StyleLi, Xiaojin, Ibrahim M. Abu-Reesh, and Zhen He. 2015. "Development of Bioelectrochemical Systems to Promote Sustainable Agriculture" Agriculture 5, no. 3: 367-388. https://doi.org/10.3390/agriculture5030367
APA StyleLi, X., Abu-Reesh, I. M., & He, Z. (2015). Development of Bioelectrochemical Systems to Promote Sustainable Agriculture. Agriculture, 5(3), 367-388. https://doi.org/10.3390/agriculture5030367







