Evaluation of Spring Wheat (20 Varieties) Adaptation to Soil Drought during Seedlings Growth Stage
Abstract
:1. Introduction
2. Results and Discussion
2.1. Results
2.1.1. Water Content and Electrolyte Leakage
| Parameters | Growth Conditions | Mean | CV | Minimum | Maximum | SI |
|---|---|---|---|---|---|---|
| Water Content and Electrolyte Leakage | ||||||
| water content (%) | control | 86.5 | 1.2 | 83.6 | 88.0 | |
| drought | 83.8 | 1.7 | 80.7 | 86.0 | 96.9 | |
| RWC (%) | control | 92.2 | 2.5 | 88.7 | 96.1 | |
| drought | 73.5 | 10.8 | 59.9 | 90.0 | 79.7 | |
| EL (%) | control | 3.3 | 29.7 | 1.8 | 5.7 | |
| drought | 4.6 | 18.1 | 3.2 | 6.0 | 138.8 | |
| Growth and Accumulation of Biomass | ||||||
| leaf area (cm2) | control | 30.0 | 8.8 | 25.5 | 34.5 | |
| drought | 21.0 | 13.0 | 15.9 | 25.0 | 69.9 | |
| dry weight of seedling (mg) | control | 92.2 | 11.1 | 73.5 | 108.2 | |
| drought | 75.1 | 13.3 | 48.2 | 90.8 | 81.4 | |
| RGRa (m2 m−2 day−1) × 102 | control | 9.0 | 11.0 | 6.9 | 11.0 | |
| drought | 5.4 | 27.4 | 2.4 | 7.6 | 59.6 | |
| NAR (g m−2 day−1) × 10 | control | 31.7 | 11.2 | 25.4 | 38.8 | |
| drought | 28.4 | 16.0 | 19.4 | 37.9 | 89.6 | |
| Gas Exchange | ||||||
| PN (μmol CO2 m−2 s−1) | control | 13.1 | 7.7 | 11.6 | 15.9 | |
| drought | 1.5 | 46.4 | 0.5 | 2.6 | 11.4 | |
| WUE (μmol CO2 × mmol H2O−1) | control | 5.6 | 21.6 | 4.2 | ||
| drought | 6.5 | 59.1 | 1.7 | 15.0 | 115.5 | |
| Leaf Pigment Content | ||||||
| chlorophyll (a + b) (mg m−2) | control | 415.5 | 11.3 | 348.7 | 507.2 | |
| drought | 367.0 | 10.2 | 324.7 | 469.5 | 88.3 | |
| carotenoids (mg m−2) | control | 32.3 | 8.4 | 28.9 | 40.0 | |
| drought | 29.8 | 6.0 | 27.8 | 34.7 | 92.2 | |
| Photochemical Efficiency | ||||||
| ABS/CS (ru) | control | 1946.6 | 3.6 | 1802.9 | 2085.9 | |
| drought | 1826.8 | 3.5 | 1698.7 | 1919.7 | 93.8 | |
| TRo/CS (ru) | control | 1492.1 | 4.6 | 1374.4 | 1642.5 | |
| drought | 1391.1 | 4.1 | 1265.5 | 1472.3 | 93.2 | |
| ETo/CS (ru) | control | 699.5 | 7.6 | 597.3 | 801.3 | |
| drought | 654.1 | 7.1 | 562.3 | 737.3 | 93.5 | |
| DIo/CS (ru) | control | 454.5 | 3.4 | 420.0 | 481.0 | |
| drought | 435.7 | 4.3 | 398.3 | 471.2 | 95.9 | |
| RC/CS (ru) | control | 675.2 | 7.0 | 587.0 | 773.6 | |
| drought | 626.6 | 5.9 | 544.5 | 686.7 | 92.8 | |
| Weight of Seeds | ||||||
| (g plant−1) | control | 17.4 | 21.2 | 13.0 | 27.6 | |
| drought | 14.3 | 22.9 | 10.2 | 25.9 | 82.5 | |
| Variety | Water Content | RWC | EL | Leaf Area | Dry Weight of Seedling | RGRa | NAR |
|---|---|---|---|---|---|---|---|
| Banti | 95.0 * | 72.4 * | 130.6 * | 54.5 * | 75.5 * | 38.5 * | 92.6 |
| Bombona | 95.8 * | 70.3 * | 248.0 * | 78.1 * | 91.8 | 65.0 * | 99.7 |
| Bryza | 99.5 | 93.8 | 198.9 * | 81.8 * | 78.7 * | 77.7 * | 77.2 * |
| Cytra | 98.5 | 87.9 * | 173.8 * | 73.5 * | 86.4 * | 66.8 * | 95.3 |
| Hewilla | 98.7 | 89.3 * | 246.9 * | 67.6 * | 79.9 * | 64.5 * | 92.1 |
| Jasna | 97.7 | 80.3 * | 105.0 | 82.4 * | 91.9 | 75.8 * | 98.3 |
| Katoda | 96.6 | 81.9 * | 109.8 | 76.7 * | 85.4 * | 70.0 * | 92.1 |
| Koksa | 98.3 | 80.9 * | 142.3 * | 74.4 * | 81.9 * | 67.9 * | 88.3 * |
| Korynta | 97.2 | 91.7 * | 228.4 * | 82.1 * | 80.3 * | 78.6 * | 80.3 * |
| Monsun | 96.2 * | 82.2 * | 142.5 * | 57.9 * | 65.6 * | 44.6 * | 68.3 * |
| Nawra | 93.3 * | 65.5 * | 118.6 * | 55.1 * | 74.9 * | 28.7 * | 86.3 * |
| Parabola | 95.9 * | 81.6 * | 107.8 | 67.1 * | 85.1 * | 60.1 * | 99.6 |
| Radunia | 96.5 | 84.6 * | 138.6 * | 83.4 * | 89.7 | 80.7 * | 95.5 |
| Raweta | 100.0 | 78.0 * | 135.5 * | 53.9 * | 77.4 * | 38.4 * | 95.0 |
| Torka | 96.5 | 77.3 * | 124.2 * | 71.5 * | 83.1 * | 61.0 * | 90.7 |
| Vinjet | 95.9 * | 69.2 * | 115.1 * | 72.8 * | 83.6 * | 54.2 * | 89.8 * |
| Waluta | 98.7 | 81.5 * | 132.8 * | 80.6 * | 83.1 * | 75.6 * | 85.3 * |
| Zadra | 96.9 | 79.2 * | 105.1 | 60.9 * | 72.1 * | 45.7 * | 79.1 * |
| Zebra | 95.3 * | 67.1 * | 156.9 * | 64.8 * | 84.3 * | 47.1 * | 97.8 |
| Żura | 96.4 | 79.1 * | 104.7 | 65.7 * | 77.5 * | 52.7 * | 86.1 * |
| F | 1.97 | 9.68 | 15.06 | 13.11 | 12.63 | 8.31 | 3.03 |
| p | 0.011 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
2.1.2. Growth and Accumulation of Biomass
2.1.3. Gas Exchange and Leaf Pigment Content
2.1.4. Photochemical Efficiency
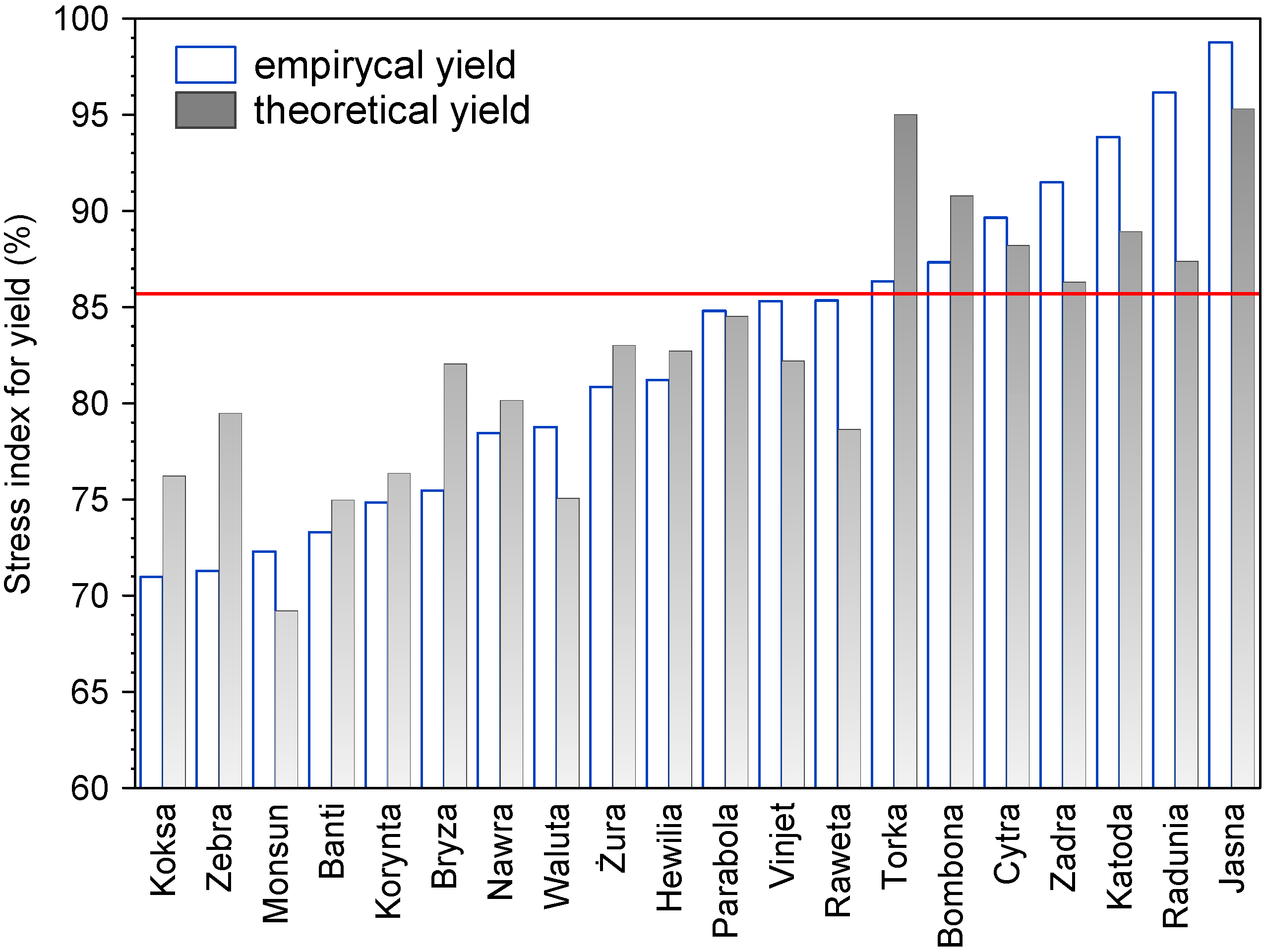
2.1.5. Consequent Effect on the Yield of Grain
| Odmiana | PN | WUE | Chl (a + b) | Carotenoids | ABS/CS | TRo/CS | ETo/CS | DIo/CS | RC/CS | Weight of Seeds |
|---|---|---|---|---|---|---|---|---|---|---|
| Banti | 13.3 * | 79.2 * | 77.3 * | 85.7 * | 93.9 * | 93.9 * | 94.9 | 93.7 * | 96.7 | 73.3 * |
| Bombona | 4.8 * | 54.8 * | 89.0 * | 93.6 | 94.8 * | 95.8 | 98.6 | 91.8 * | 95.4 | 87.3 * |
| Bryza | 15.8 * | 180.6 * | 92.0 | 95.3 | 93.7 * | 92.8 * | 91.0 * | 96.9 | 89.3 * | 75.5 * |
| Cytra | 11.7 * | 192.3 * | 97.3 | 98.3 | 97.8 | 96.1 | 92.0 * | 103.4 | 92.5 * | 89.7 * |
| Hewillia | 21.3 * | 121.3 * | 91.9 | 94.9 | 95.3 | 94.6 * | 97.9 | 97.3 | 92.7 * | 81.2 * |
| Jasna | 13.3 * | 76.4 * | 97.1 | 98.4 | 96.4 | 96.0 | 99.1 | 97.6 | 96.3 | 98.8 |
| Katoda | 15.7 * | 266.9 * | 80.8 * | 88.3 * | 97.6 | 98.1 | 101.6 | 95.7 * | 96.7 | 93.8 * |
| Koksa | 7.3 * | 260.4 * | 83.7 * | 89.5 * | 87.1 * | 84.8 * | 82.7 * | 95.2 | 83.1 * | 71.0 * |
| Korynta | 10.4 * | 85.8 * | 85.2 * | 90.9 * | 88.6 * | 86.3 * | 78.7 * | 96.3 | 83.2 * | 74.8 * |
| Monsun | 4.3 * | 48.7 * | 86.9 * | 91.2 | 92.7 * | 92.1 * | 92.8 | 94.5 * | 91.8 * | 72.3 * |
| Nawra | 6.3 * | 92.1 * | 93.7 | 86.8 * | 92.0 * | 92.0 * | 92.7 | 92.0 * | 92.2 * | 78.5 * |
| Parabola | 13.6 * | 124.6 * | 97.7 | 98.7 | 99.1 | 97.8 | 99.9 | 103.4 | 97.8 | 84.8 * |
| Radunia | 12.5 * | 240.9 * | 89.3 * | 93.4 | 89.9 * | 88.0 * | 84.3 * | 96.2 * | 86.1 * | 96.2 |
| Raweta | 11.5 * | 48.8 * | 93.2 | 95.6 | 98.6 | 98.8 | 100.7 | 98.1 | 102.6 | 85.3 * |
| Torka | 19.5 * | 118.1 * | 93.1 | 95.9 | 95.1 * | 94.8 * | 98.4 | 96.1 | 93.5 | 86.3 * |
| Vinjet | 4.5 * | 97.3 * | 88.8 * | 93.0 | 90.0 * | 90.4 * | 94.7 | 88.5 * | 92.4 * | 85.3 * |
| Waluta | 4.3 * | 35.5 * | 81.6* | 88.4 * | 93.2 * | 92.5 * | 92.1 | 95.6 * | 92.1 * | 78.8 * |
| Zadra | 19.5 * | 125.0 * | 79.8* | 87.1 * | 93.5 * | 93.0 * | 94.9 | 95.1 * | 91.0 * | 91.5 * |
| Zebra | 4.1 * | 512 * | 81.6* | 88.1 * | 91.9 * | 91.7 * | 94.1 | 92.5 * | 92.8 * | 71.3 * |
| Żura | 15.4 * | 79.6 * | 93.1 | 96.2 | 97.1 | 96.9 | 93.9 | 98.0 | 102.2 | 80.9 * |
| F | 14.91 | 13.62 | 1.94 | 1.85 | 2.94 | 3.07 | 3.18 | 4.78 | 3.52 | 11.02 |
| p | 0.000 | 0.000 | 0.013 | 0.019 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
2.1.6. Correlation between Seedlings Tolerance, Final Yield and Drought
2.2. Discussion
2.2.1. Water Management and Growth of Seedlings
| 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | |
| 1 | 0.701 | ns | ns | ns | 0.489 | ns | ns | ns | ns | ns | ns | ns | ns | 0.471 | ns | ns |
| 2 | 1.000 | ns | 0.476 | ns | 0.671 | ns | 0.475 | ns | ns | ns | ns | ns | ns | 0.631 | ns | ns |
| 3 | 1.000 | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns | |
| 4 | 1.000 | 0.705 | 0.958 | ns | ns | ns | ns | ns | ns | ns | ns | ns | −0454 | ns | ||
| 5 | 1.000 | 0.627 | 0.783 | ns | ns | ns | ns | ns | ns | ns | ns | ns | 0.546 | |||
| 6 | 1.000 | ns | ns | 0.444 | ns | ns | ns | ns | ns | ns | ns | ns | ||||
| 7 | 1.000 | ns | ns | ns | ns | ns | ns | ns | ns | ns | 0.444 | |||||
| 8 | 1.000 | ns | ns | ns | ns | ns | ns | 0.478 | ns | ns | ||||||
| 9 | 1.000 | ns | ns | ns | ns | ns | ns | ns | ns | |||||||
| 10 | 1.000 | 0.868 | 0.459 | ns | ns | 0.521 | ns | ns | ||||||||
| 11 | 1.000 | 0.519 | ns | ns | 0.628 | ns | ns | |||||||||
| 12 | 1.000 | 0.975 | 0.801 | 0.598 | 0.839 | ns | ||||||||||
| 13 | 1.000 | 0.890 | ns | 0.902 | ns | |||||||||||
| 14 | 1.000 | ns | 0.833 | ns | ||||||||||||
| 15 | 1.000 | ns | ns | |||||||||||||
| 16 | 1.000 | ns |
| Step | Trait | R2 | F | Standard Deviation |
|---|---|---|---|---|
| 1 | dry weight of seedling | 0.546 | 7.63 | 7.28 |
| 2 | photosynthesis (PN) | 0.682 | 7.40 | 6.53 |
| 3 | leaf area | 0.688 | 7.38 | 6.42 |
| 4 | NAR | 0.813 | 7.33 | 5.54 |
| Wilkins’s lambda | Partial lambda | F Removal | p | |
|---|---|---|---|---|
| dry weight of seedling | 0.686 | 0.433 | 19.60 | 0.000 |
| leaf area | 0.602 | 0.494 | 15.38 | 0.001 |
| NAR | 0.628 | 0.474 | 16.67 | 0.001 |
| photosynthesis (PN) | 0.656 | 0.454 | 18.07 | 0.001 |
2.2.2. Gas Exchange
2.2.3. PSII Photochemical Activity and the Final Yield
2.2.4. The Relationship between Seedlings Traits under Drought and the Final Yield
| Variety * | Sensitivity to Stress | p = 0.65 Sensitive | p = 0.35 Tolerant |
|---|---|---|---|
| Koksa | Sensitive | 148 | 1512 |
| Zebra | Sensitive | 286 | 1255 |
| Monsun | Sensitive | 883 | 2752 |
| Banti | Sensitive | 547 | 2492 |
| Korynta | Sensitive | 371 | 1532 |
| Bryza | Sensitive | 618 | 763 |
| Nawra | Sensitive | 492 | 980 |
| Waluta | Sensitive | 360 | 1745 |
| Żura | Sensitive | 205 | 460 |
| Hewilla | Sensitive | 504 | 1234 |
| Parabola | Sensitive | 293 | 683 |
| Vinjet | Sensitive | 343 | 519 |
| Raweta | Sensitive | 314 | 1583 |
| Torka | Tolerant | 2026 | 295 |
| Bombona | Tolerant | 1458 | 360 |
| Cytra | Tolerant | 596 | 068 |
| Zadra | Tolerant | 1065 | 570 |
| Katoda | Tolerant | 702 | 061 |
| Radunia | Tolerant | 607 | 277 |
| Jasna | Tolerant | 1921 | 205 |
3. Experimental Section
3.1. Plant Material
3.2. Plant Growth Condition
3.3. Plant Measurements
3.3.1. The Rates of Growth and Accumulation of Biomass
3.3.2. The Integrity of Plasma Membrane and Water Balance
3.3.3. Gas Exchange
3.3.4. Photochemical Efficiency
- (a)
- The energy fluxes (per active cross section of leaf, CS) for absorption, ABS (ABS/CS), trapping (TRo/CS), electron transport (ETo/CS) and dissipation (DIo/CS):
- (b)
- The amount of active PSII reaction centers per CS (RC/CS):where:RC/CS = ABS/CS × φPo × (VJ/Mo)Mo = 4 × (F300μs−Fo)/(Fm−Fo)
3.3.5. Leaf Pigment Content
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Morgan, J.M. Growth and yield of wheat lines with differing osmoregulative capacity at high soil water deficit in seasons of varying evaporative demand. Field Crops Res. 1995, 40, 143–152. [Google Scholar] [CrossRef]
- Hasegawa, M.; Bressan, R.; Pardo, J.M. The dawn of plant salt tolerance genetics. Trends Plant Sci. 2000, 5, 317–319. [Google Scholar] [CrossRef]
- Moussa, H.R.; Abdel-Aziz, S.M. Comparative response of drought tolerant and drought sensitive maize genotypes to water stress. Aust. J. Crop Sci. 2008, 1, 31–36. [Google Scholar]
- Lawlor, D.W.; Cornic, G. Photosynthetic carbon assimilation and associated metabolism in relation to water deficits in higher plants. Plant Cell Environ. 2002, 25, 275–294. [Google Scholar] [CrossRef]
- Guo, P.; Baum, M.; Varshney, R.K.; Graner, A.; Grando, S.; Ceccarelli, S. QTLs for chlorophyll and chlorophyll fluorescence parameters in barley under post-flowering drought. Euphytica 2008, 163, 203–214. [Google Scholar] [CrossRef]
- Robredo, A.; Pérez-López, U.; Sainz de la Maza, H.; González-Moro, B.; Lacuesta, M.; Mena-Petite, A.; Muñoz-Rueda, A. Elevated CO2 alleviates the impact of drought on barley improving water status by lowering stomatal conductance and delaying its effect on photosynthesis. Environ. Exp. Bot. 2007, 59, 252–263. [Google Scholar]
- Araus, J.L.; Amaro, T.; Voltas, J.; Nakkoul, H.; Nachit, M.M. Chlorophyll fluorescence as a selection criterion for grain yield in durum wheat under Mediterranean conditions. Field Crop Res. 1998, 55, 209–223. [Google Scholar] [CrossRef]
- Lu, C.; Zhang, J. Effects of water stress on photosystem II photochemistry and its thermostability in wheat plants. J. Exp. Bot. 1999, 50, 1199–1206. [Google Scholar] [CrossRef]
- Kocheva, K.V.; Busheva, M.C.; Georgiev, G.I.; Lambrev, P.H.; Goldsev, V.N. Influence of short-term osmotic stress on the photosynthetic activity of barley seedlings. Biol. Plant. 2005, 49, 145–148. [Google Scholar] [CrossRef]
- Oukarroum, A.; El Madidi, S.; Schansker, G.; Strasser, R.J. Probing the responses of barley cultivars (Hordeum vulgare L.) by chlorophyll a fluorescence OLKJIP under drought stress and re-watering. Environ. Exp. Bot. 2007, 60, 438–446. [Google Scholar] [CrossRef]
- Jedmowski, C.; Ashoub, A.; Brüggemann, W. Reactions of Egyptian landraces of Hordeum vulgare and Sorghum bicolor to drought stress, evaluated by the OJIP fluorescence transient analysis. Acta Physiol. Plant. 2013, 35, 345–354. [Google Scholar] [CrossRef]
- Chen, G.; Sagi, M.; Weining, S.; Krugman, T.; Fahima, T.; Korol, A.B.; Nevo, E. Wild barley eibi1 mutation identifies a gene essential for leaf water conservation. Planta 2004, 219, 684–693. [Google Scholar]
- Bencze, S.; Bamberger, Z.; Janda, T.; Balla, K.; Bedő, Z.; Veisz, O. Drought tolerance in cereals in terms of water retention, photosynthesis and antioxidant enzyme activities. Cent. Eur. J. Biol. 2011, 6, 376–387. [Google Scholar] [CrossRef]
- Babu, R.C.; Zhang, J.; Blum, A.; Ho, T.-H.; Wu, R.; Nguyen, H.T. HVA1, a LEA gene from barley confers dehydration tolerance in transgenic rice (Oryza sativa L.) via cell membrane protection. Plant Sci. 2004, 166, 855–862. [Google Scholar] [CrossRef]
- Geravandia, M.; Farshadfara, E.; Kahrizia, D. Evaluation of some physiological traits as indicators of drought tolerance in bread wheat genotypes. Russ. J. Plant Physiol. 2011, 58, 69–75. [Google Scholar] [CrossRef]
- Shao, H.B.; Liang, Z.S.; Shao, M.A. Changes of anti-oxidative enzymes and MDA content under soil water deficits among 10 wheat (Triticum aestivum L) genotypes at maturation stage. Coll. Surf. B Biointerfaces 2005, 45, 7–13. [Google Scholar] [CrossRef]
- Huseynova, I.; Rustanamova, S.M. Screening for drought stress tolerance in wheat genotypes using molecular markers. Proc. ANAS Biol. Sci. 2010, 65, 132–139. [Google Scholar]
- Pantuwan, G.; Fukai, S.; Cooper, M.; Rajatasereekul, S.; O’Toole, J.C. Yield response of rice (Oryza sativa L) genotypes to drought under rained lowlands. II. Selection of drought resistant genotypes. Field Crop Res. 2002, 73, 169–180. [Google Scholar] [CrossRef]
- Passioura, J. The drought environment physical, biological and agricultural perspectives. J. Exp. Bot. 2007, 5, 113–117. [Google Scholar]
- Bajji, M.; Kinet, J.M.; Lutts, S. The use of the electrolyte leakage method for assessing cell membrane stability as a water stress tolerance test in durum wheat. Plant Growth Regul. 2002, 36, 61–70. [Google Scholar] [CrossRef]
- Robredo, A.; Pérez-López, U.; Lacuesta, M.; Mena-Petite, A.; Muñoz-Rueda, A. Influence of water stress on photosynthetic characteristics in barley plants under ambient and elevated CO2 concentrations. Biol. Plant. 2010, 54, 285–292. [Google Scholar] [CrossRef]
- Lawson, T.; Oxborough, K.; Morison, J.I.L.; Baker, N.R. The responses of guard and mesophyll cell photosynthesis to CO2, O2, light and water stress in a range of species are similar. J. Exp. Bot. 2003, 54, 1743–1752. [Google Scholar] [CrossRef]
- Lu, C.M.; Zhang, J.H. Effect of water stress on photosynthesis, chlorophyll fluorescence and photoinhibition in wheat plants. Aust. J. Plant Physiol. 1998, 25, 883–892. [Google Scholar] [CrossRef]
- Acar, O.; Türkan, I.; Özdemir, F. Superoxide dismutase and peroxidase activities in drought sensitive and resistant barley. Acta Physiol Plant. 2001, 23, 351–356. [Google Scholar] [CrossRef]
- Farrat, J.M.; Bailly, C.; Leymarie, J.; Hamman, B.; Come, D.; Corbineau, F. Wheat seedlings as a model to understand the desiccation tolerance and sensitivity. Physiol Plant. 2004, 120, 563–574. [Google Scholar] [CrossRef]
- Mihailović, N.; Jelić, G.; Filipović, R.; Durdević, M.; Dżeletović, Ż. Effect of nitrogen form on maize response to drought stress. Plant Soil 1992, 144, 191–197. [Google Scholar] [CrossRef]
- Cornic, G.; Briantais, J.M. Partioning of photosynthetic electron flow between CO2 and O2 reduction in a C3 leaf (Phaseolus vulgaris L) at different CO2 concentrations during drought stress. Planta 1991, 183, 178–184. [Google Scholar]
- Liang, J.; Zhang, J.; Wong, M. Can stomatal closure caused by xylem ABA explain the inhibition of leaf photosynthesis under soil drying? Photosynth. Res. 1997, 51, 149–159. [Google Scholar] [CrossRef]
- Berkowitz, G.A.; Gibbs, M. Reduced osmotic potential effects on photosynthesis Identification of stromal acidification as a mediating factor. Plant Physiol. 1983, 71, 905–911. [Google Scholar] [CrossRef]
- Condon, A.G.; Richards, R.A.; Rebetzke, G.J.; Farquhar, G.D. Improving intrinsic water-use efficiency and crop yield. Crop Sci. 2002, 42, 122–131. [Google Scholar] [CrossRef]
- Quarrie, S.A.; Stojanovic, J.; Pekic, S. Improving drought tolerant in small-grained cereals, a case study, progress and prospects. Plant Growth Regul. 1999, 29, 1–21. [Google Scholar] [CrossRef]
- Ekoziem. Available online: http://www.ekoziem.com.pl/?ziemia-uniwersalna,19 (accessed on 2 April 2014).
- Hoagland, D.R.; Arnon, D.I. The water-culture method for plants without soil. Univ. Calif. Agric. Exp. Sta. Circ. 1938, 347, 29–32. [Google Scholar]
- Bouslama, M.; Schapaugh, W.T., Jr. Stress tolerance in soybeans. I. Evaluation of three screening techniques for heat and drought tolerance. Crop Sci. 1984, 24, 933–937. [Google Scholar] [CrossRef]
- Květ, J.; Ondok, J.P.; Nečas, J.; Jarvis, P.G. Methods of Growth Analysis. In Plant. Photosynthetic Production Manual of Methods; Šesták, Z., Čatský, Z., Jarvis, P.G., Eds.; Springer: Hague, The Netherlands, 1971; pp. 343–391. [Google Scholar]
- Markowski, A.; Skrudlik, G. Electrolyte leakage, ATP content in leaves and intensity of net photosynthesis in maize seedlings at permanent of different daily exposure to low. J. Agron. Crop Sci. 1995, 175, 109–117. [Google Scholar] [CrossRef]
- Songstad, D.D.; Duncan, D.R.; Widholm, J.M. Proline and polyamine involvement in chilling tolerance of maize suspension cultures. J. Exp. Bot. 1990, 41, 289–294. [Google Scholar] [CrossRef]
- Barrs, H.D. Determination of Water Deficits in Plant Tissues. In Water Deficits and Plant Growth; Kozlowski, T.T., Ed.; Academic Press: New York, NY, USA, 1968; pp. 236–259. [Google Scholar]
- Strasser, R.J.; Tsimilli-Michael, M. Stress in plants, from daily rhythm to global changes, detected and quantified by the JIP-test. Chem. Nouv. 2001, 75, 3321–3326. [Google Scholar]
- Arnon, D.I. Copper enzymes in isolated chloroplasts Polyphenol oxidase in Beta vulgaris. Plant Physiol. 1949, 24, 1–15. [Google Scholar] [CrossRef]
- Jaspars, E.M.J. Pigmentation of tobacco crown-gall tissues cultured in vitro in dependence of the composition of the medium. Physiol. Plant. 1965, 18, 933–940. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Biesaga-Kościelniak, J.; Ostrowska, A.; Filek, M.; Dziurka, M.; Waligórski, P.; Mirek, M.; Kościelniak, J. Evaluation of Spring Wheat (20 Varieties) Adaptation to Soil Drought during Seedlings Growth Stage. Agriculture 2014, 4, 96-112. https://doi.org/10.3390/agriculture4020096
Biesaga-Kościelniak J, Ostrowska A, Filek M, Dziurka M, Waligórski P, Mirek M, Kościelniak J. Evaluation of Spring Wheat (20 Varieties) Adaptation to Soil Drought during Seedlings Growth Stage. Agriculture. 2014; 4(2):96-112. https://doi.org/10.3390/agriculture4020096
Chicago/Turabian StyleBiesaga-Kościelniak, Jolanta, Agnieszka Ostrowska, Maria Filek, Michał Dziurka, Piotr Waligórski, Magdalena Mirek, and Janusz Kościelniak. 2014. "Evaluation of Spring Wheat (20 Varieties) Adaptation to Soil Drought during Seedlings Growth Stage" Agriculture 4, no. 2: 96-112. https://doi.org/10.3390/agriculture4020096
APA StyleBiesaga-Kościelniak, J., Ostrowska, A., Filek, M., Dziurka, M., Waligórski, P., Mirek, M., & Kościelniak, J. (2014). Evaluation of Spring Wheat (20 Varieties) Adaptation to Soil Drought during Seedlings Growth Stage. Agriculture, 4(2), 96-112. https://doi.org/10.3390/agriculture4020096






