Low Soil Phosphorus Availability Increases Acid Phosphatases Activities and Affects P Partitioning in Nodules, Seeds and Rhizosphere of Phaseolus vulgaris
Abstract
:1. Introduction
2. Results
2.1. Growth and Nodulation

2.2. Soil Available P

2.3. Acid Phosphatase and Phytase Activities in Rhizospheric Soils

2.4. Acid Phosphatase and Phytase Activities in Nodules and Seeds
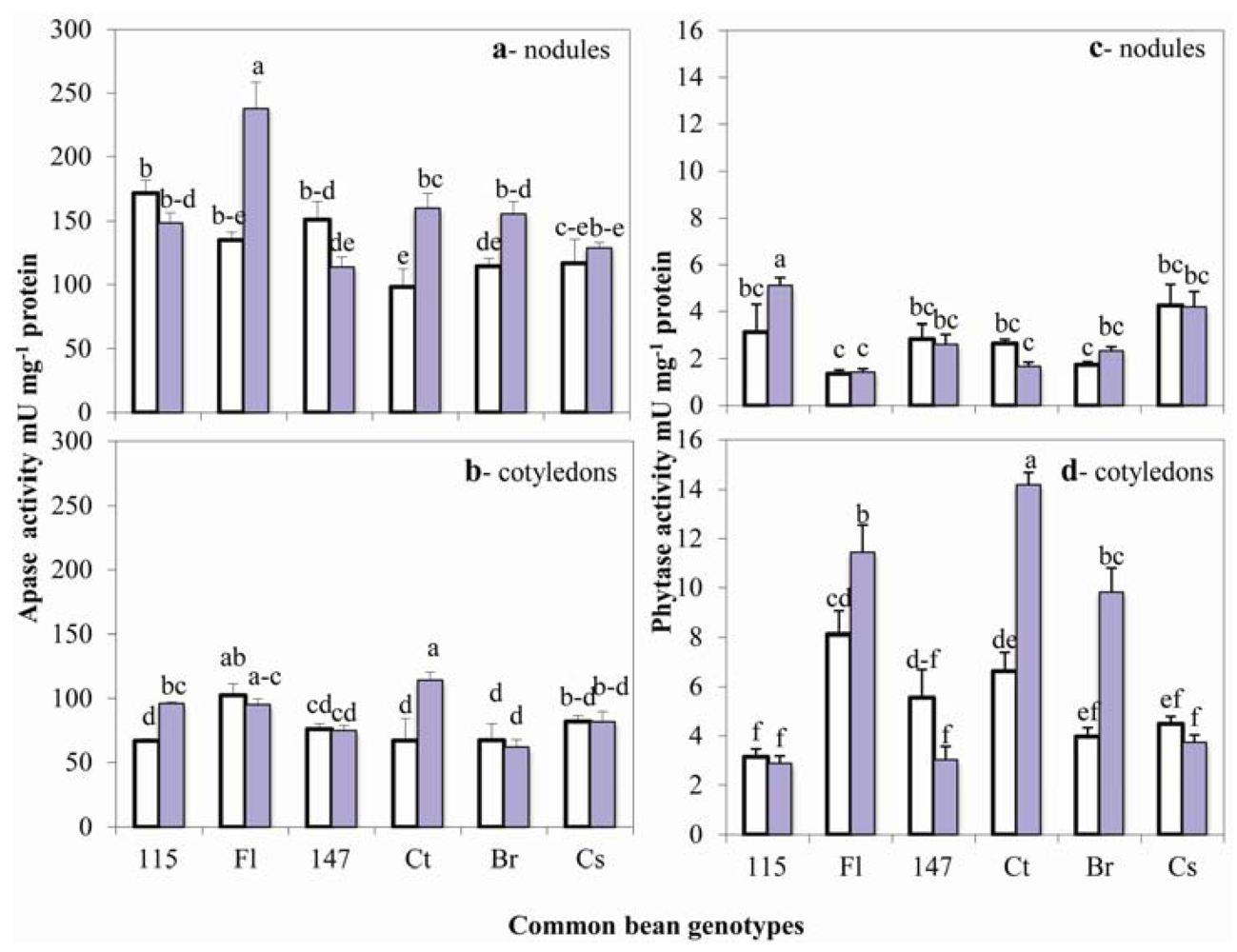
2.5. Phosphorus and Phytate Distribution in Nodules and Seeds
| Genotypes | Nodule P (mg g−1) | Seed P (mg g−1) | Nodule Phy (mg g−1) | Seed Phy (mg g−1) | ||||
| S1 | S2 | S1 | S2 | S1 | S2 | S1 | S2 | |
| L115 | 4.95 ± 0.5e | 7.8 ± 0.9a–d | 9.5 ± 0.64a | 5.9 ± 0.25ef | 4.8 ± 0.1cd | 5.9 ± 0.6bc | 6.2 ± 0.2ab | 4.9 ± 0.1c–f |
| Fl | 9.3 ± 0.1a–c | 5.96 ± 1.3de | 6.2 ± 0.3d–f | 6.3 ± 0.3d–f | 6.3 ± 0.16b | 4.42 ± 0.3d | 5.7 ± 0.3a–d | 4.7 ± 0.2ef |
| L147 | 9.6 ± 0.3ab | 7.8 ± 0.2a–d | 8.2 ± 0.6b | 7.24 ± 0.3ab | 6 ± 0.6bc | 5.9 ± 0.3bc | 5.8 ± 0.2a–c | 4.9 ± 0.2c–f |
| Ct | 10.45 ± 0.55a | 9.2 ± 0.27a–c | 7.1 ± 0.2cd | 7.1 ± 0.3cd | 9.3 ± 0.4a | 6.4 ± 0.7b | 6.2 ± 0.3ab | 4.5 ± 0.3ef |
| Br | 6.23 ± 1.4c–e | 5.91 ± 0.53de | 7.1 ± 0.2c–e | 5.51 ± 0.4f | 5.7 ± 0.2b–d | 4.8 ± 0.3cd | 6.6 ± 0.3a | 5.4 ± 0.3b–e |
| Cs | 6.4 ± 1.1b–e | 5.96 ± 1.3de | 5.96 ± 0.2ef | 5.35 ± 0.3f | 6 ± 0.3bc | 5.4 ± 0.3b–d | 4.2 ± 0.1f | 4.9 ± 0.3d–f |
2.6. P Uptake, Use Efficiency and Absorption Efficiency by Root

2.7. Relationship between Nodulation, Shoot Growth, P and N Contents
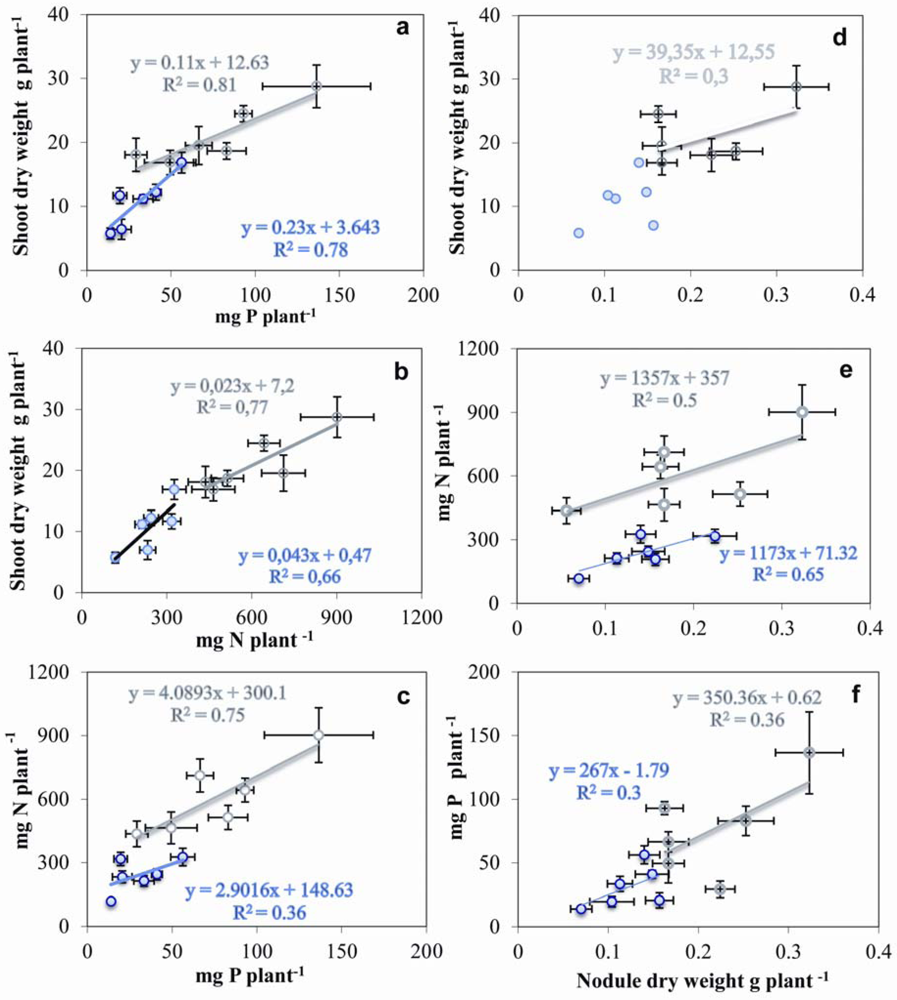
3. Discussion
4. Experimental Section
4.1. Plant Material and Field Conditions
4.2. Soils Analyses
| Characteristics | S1 | S2 |
|---|---|---|
| Clay (%) | 18.93 | 17.32 |
| Sand (%) | 49.86 | 66.22 |
| Silt (%) | 33.75 | 15.75 |
| pH | 8.2 | 8.09 |
| Organic matter (%) | 1.61 | 1.39 |
| CaCO3 total (%) | 15 | 16.3 |
| CaCO3 active (%) | 21 | 33 |
| Ptotal (g Kg−1) | 1.6 | 0.54 |
| Polsen (g Kg−1) | 0.0167 | 0.0043 |
| Nitrogen (g Kg−1) | 1.21 | 1.09 |
| K+ (g Kg−1) | 0.29 | 0.149 |
4.3. Harvest and Measurement of Plant, Nodule and Yield Components
4.4. APase and Phytase Activities Assays in Rhizosphere Soils
4.5. APase and Phytase Activities Assays in Nodules and Seeds
4.6. Determination of P, N, Phytate and Statistical Analyses
5. Conclusions
Acknowledgements
References
- Broughton, W.J.; Hernander, G.; Blair, B.; Beebe, S.; Gepts, P.; Vanderleyden, J. Beans (Phaseolus spp.)–model food legumes. Plant Soil 2003, 252, 55–128. [Google Scholar] [CrossRef]
- Graham, P.H.; Vance, C.P. Legumes: Importance and constraints to greater use. Plant Physiol. 2003, 3, 872–877. [Google Scholar] [CrossRef]
- Vance, C.P. Symbiotic nitrogen fixation and phosphorus acquisition. Plant nutrition in a world of declining renewable resources. Plant Physiol. 2001, 127, 390–397. [Google Scholar] [CrossRef]
- Shenoy, V.V.; Kalagudi, G.M. Enhancing plant phosphorus use efficiency for sustainable cropping. Biotechnol. Adv. 2005, 23, 501–513. [Google Scholar] [CrossRef]
- Larue, T.A.; Patterson, R. How much nitrogen do legumes fix? Adv. Agron. 1981, 34, 15–38. [Google Scholar] [CrossRef]
- Hartwig, U.A.; Nosberger, J. What triggers the regulation of nitrogenase activity in forage legume nodules after defoliation? Plant Soil 1994, 161, 109–114. [Google Scholar] [CrossRef]
- Hellsten, A.; Huss-Danell, K. Interaction effects on nitrogen and phosphorus on nodulation in red clover (Trifolium patens L.). Acta Agric. Scand. 2001, 50, 135–142. [Google Scholar]
- Al-Niemi, T.S.; Kahn, M.L.; Mc Dermott, T.R. P metabolism in the bean Rhizobium tropici symbiosis. Plant Physiol. 1997, 113, 1233–1242. [Google Scholar]
- Raghothama, K.G. Phosphate acquisition. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1999, 50, 665–693. [Google Scholar] [CrossRef]
- Duff, S.M.G.; Sarath, G.; Plaxton, W.C. The role of acid phosphatases in plant phosphorus metabolism. Physiol. Plant. 1994, 90, 791–800. [Google Scholar] [CrossRef]
- Richardson, A.E.; Hadobas, P.A.; Hayes, J.E. Extracellular secretion of Aspergillus phytase from Arabidopsis roots enables plants to obtain phosphorus from phytate. Plant J. 2001, 25, 641–649. [Google Scholar]
- Tarafdar, J.C.; Yadav, R.S.; Niwas, R.J. Relative efficiency of fungal intra- and extracellular phosphatases and phytase. Plant Nutr. Soil Sci. 2002, 165, 17–19. [Google Scholar] [CrossRef]
- Yadav, B.K.; Tarafdar, J.C. Phytase activity in the rhizosphere of crops, trees and grasses under arid environment. J. Arid Environ. 2004, 58, 285–293. [Google Scholar] [CrossRef]
- Araújo, A.P.; Plassard, C.; Drevon, J.J. Phosphatase and phytase activities in nodules of common bean genotypes at different levels of phosphorus supply. Plant Soil 2008, 312, 129–138. [Google Scholar] [CrossRef]
- Li, L.; Tang, C.; rengel, Z.; Zhang, F. Chickpea facilitates phosphorus uptake by intercropped wheat by an organic phosphorus source. Plant Soil 2003, 248, 297–303. [Google Scholar] [CrossRef]
- Radersma, S.; Grierson, P.F. Phosphorus mobilization in agroforestry: Organic anions, phosphatase activity and phosphorus fractions in the rhizosphere. Plant Soil 2004, 259, 209–219. [Google Scholar] [CrossRef]
- Kouas, S.; Alkama, N.; Abdelly, C.; Drevon, J.J. Proton release by nodulated roots varies among common bean genotypes (Phaseolus vulgaris) under phosphorus deficiency. Plant Nutr. Soil Sci. 2008, 171, 242–248. [Google Scholar] [CrossRef]
- Bargaz, A.; Ghoulam, C.; Amenc, L.; Lazali, M.; Faghire, M.; Abadie, J.; Drevon, J.-J. A phosphoenol pyruvate phosphatase gene transcript is induced in the root nodule cortex of Phaseolus vulgaris under P deficiency. J. Exp. Bot. 2012, in press.. [Google Scholar]
- Li, Y.-F.; Luo, A.-C.; Wei, X.-H.; Yao, X.-G. Changes in phosphorus fractions, pH and phosphatase activity in rhizosphere of two rice genotypes. Pedosphere 2008, 18, 785–794. [Google Scholar] [CrossRef]
- Bowen, G.D.; Rovira, A.D. The rhizosphere and its management to improve plant growth. Adv. Agron. 1999, 66, 1–102. [Google Scholar] [CrossRef]
- Bargaz, A.; Ghoulam, C.; Faghire, M.; Aslan Attar, H.; Drevon, J.J. The nodule conductance to the O2 diffusion increases with high phosphorus content in the Phaseolus vulgaris-rhizobia symbiosis. Symbiosis 2011, 53, 157–164. [Google Scholar] [CrossRef]
- Schulze, J.; Drevon, J.J. P-deficiency increases the O2 uptake per N2 reduced in alfalfa. J. Exp. Bot. 2005, 56, 1779–1784. [Google Scholar] [CrossRef]
- Yan, X.; Lynch, J.P.; Beebe, S.E. Genetic variation for phosphorus efficiency of common bean in contrasting soil types: II. Yield response. Crop Sci. 1995, 35, 1094–1099. [Google Scholar] [CrossRef]
- Rotaru, V.; Sinclair, T.R. Interactive influence of phosphorus and iron on nitrogen fixation by soybean. Environ. Exp. Bot. 2009, 66, 94–99. [Google Scholar] [CrossRef]
- Tong, X.J.; Lu, Y.G.; Yan, X. Studies on the characteristics of phosphorus efficiency of native soybean (Glycine Max L. Merr.) germplasm: Differences in characteristics of phosphorus efficiency of shoot and root among soybean genotypes and correlation analysis. Chin. J. Oil Crop Sci. 2000, 22, 48–53. [Google Scholar]
- Ortiz-Monasterio, R.J.I.; Sayre, K.D.; Rajaram, S.; Mc Mahon, M. Genetic progress in wheat yield and nitrogen use efficiency under four nitrogen rates. Crop Sci. 1997, 37, 898–904. [Google Scholar] [CrossRef]
- Zhang, J.H.; Zhang, J.Y.; Yang, X.H.; Jin, H.A. Study on genetic relationship of main maize inbred lines in Yunnan by SSR markers. J. Maize Sci. 2007, 15, 30–35. [Google Scholar]
- Drevon, J.J.; Alkama, N.; Araujo, A.; Beebe, B.; Aslan Attar, H.; Benoit, J.; Lopez, A.; Martinez-Romero, E.; Rodino, P.; Tajini, F.; Zaman-Allah, M. Nodular diagnosis for ecological engineering of the symbiotic nitrogen fixation with legumes. Proc. Environ. Sci. 2010, 9, 40–46. [Google Scholar]
- Olsen, S.R.; Cole, C.V.; Watanabe, F.S.; Dean, L.A. Estimation of Available Phosphorus in Soil by Extraction with Sodium Bicarbonate; Circular 939; USDA: Washington, DC, USA, 1954; p. 19. [Google Scholar]
- Murphy, J.; Riley, J.P. A modified single solution method for the determination of phosphate in natural waters. Acta Anal. Chim. 1962, 27, 31–36. [Google Scholar] [CrossRef]
- Tabatabai, M.A. Soil enzymes. In Methods of Soil Analysis, Part 2, Microbiological and Biochemical Properties; Soil Science Society of America: Madison, WI, USA, 1994; pp. 775–833. [Google Scholar]
- Ozturk, L.; Eker, S.; Bulent, T.; Cakmak, I. Variation in P efficiency among 73 bread and durum wheat genotypes grown in a P-deficient calcareous soil. Plant Soil 2005, 269, 69–80. [Google Scholar] [CrossRef]
- Pan, X.-W.; Li, W.-B.; Zhang, Q.-Y.; Li, Y.-H.; Liu, M.-S. Assessment on phosphorus efficiency characteristics of soybean genotypes in phosphorus-deficient Soils. Agric. Sci. 2008, 7, 958–969. [Google Scholar]
- Vaintraub, I.A.; Lapteva, N.A. Colorimetric determination of phytate in unpurified extracts of seeds and the products of their processing. Ann. Biochem. 1988, 175, 227–230. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bargaz, A.; Faghire, M.; Abdi, N.; Farissi, M.; Sifi, B.; Drevon, J.-J.; Cherkaoui Ikbal, M.; Ghoulam, C. Low Soil Phosphorus Availability Increases Acid Phosphatases Activities and Affects P Partitioning in Nodules, Seeds and Rhizosphere of Phaseolus vulgaris. Agriculture 2012, 2, 139-153. https://doi.org/10.3390/agriculture2020139
Bargaz A, Faghire M, Abdi N, Farissi M, Sifi B, Drevon J-J, Cherkaoui Ikbal M, Ghoulam C. Low Soil Phosphorus Availability Increases Acid Phosphatases Activities and Affects P Partitioning in Nodules, Seeds and Rhizosphere of Phaseolus vulgaris. Agriculture. 2012; 2(2):139-153. https://doi.org/10.3390/agriculture2020139
Chicago/Turabian StyleBargaz, Adnane, Mustapha Faghire, Neila Abdi, Mohamed Farissi, Bouaziz Sifi, Jean-Jacques Drevon, Mohamed Cherkaoui Ikbal, and Cherki Ghoulam. 2012. "Low Soil Phosphorus Availability Increases Acid Phosphatases Activities and Affects P Partitioning in Nodules, Seeds and Rhizosphere of Phaseolus vulgaris" Agriculture 2, no. 2: 139-153. https://doi.org/10.3390/agriculture2020139
APA StyleBargaz, A., Faghire, M., Abdi, N., Farissi, M., Sifi, B., Drevon, J.-J., Cherkaoui Ikbal, M., & Ghoulam, C. (2012). Low Soil Phosphorus Availability Increases Acid Phosphatases Activities and Affects P Partitioning in Nodules, Seeds and Rhizosphere of Phaseolus vulgaris. Agriculture, 2(2), 139-153. https://doi.org/10.3390/agriculture2020139





