Assessing Fungal Plant Pathogen Presence in Irrigation Water from the Rio Grande River in South Texas, USA
Abstract
1. Introduction
2. Materials and Methods
2.1. Collection of Irrigation Water Samples
2.2. DNA Extraction
2.3. High-Throughput Amplicon Sequencing
2.4. Data Processing
2.5. Bioinformatics Analysis
3. Results
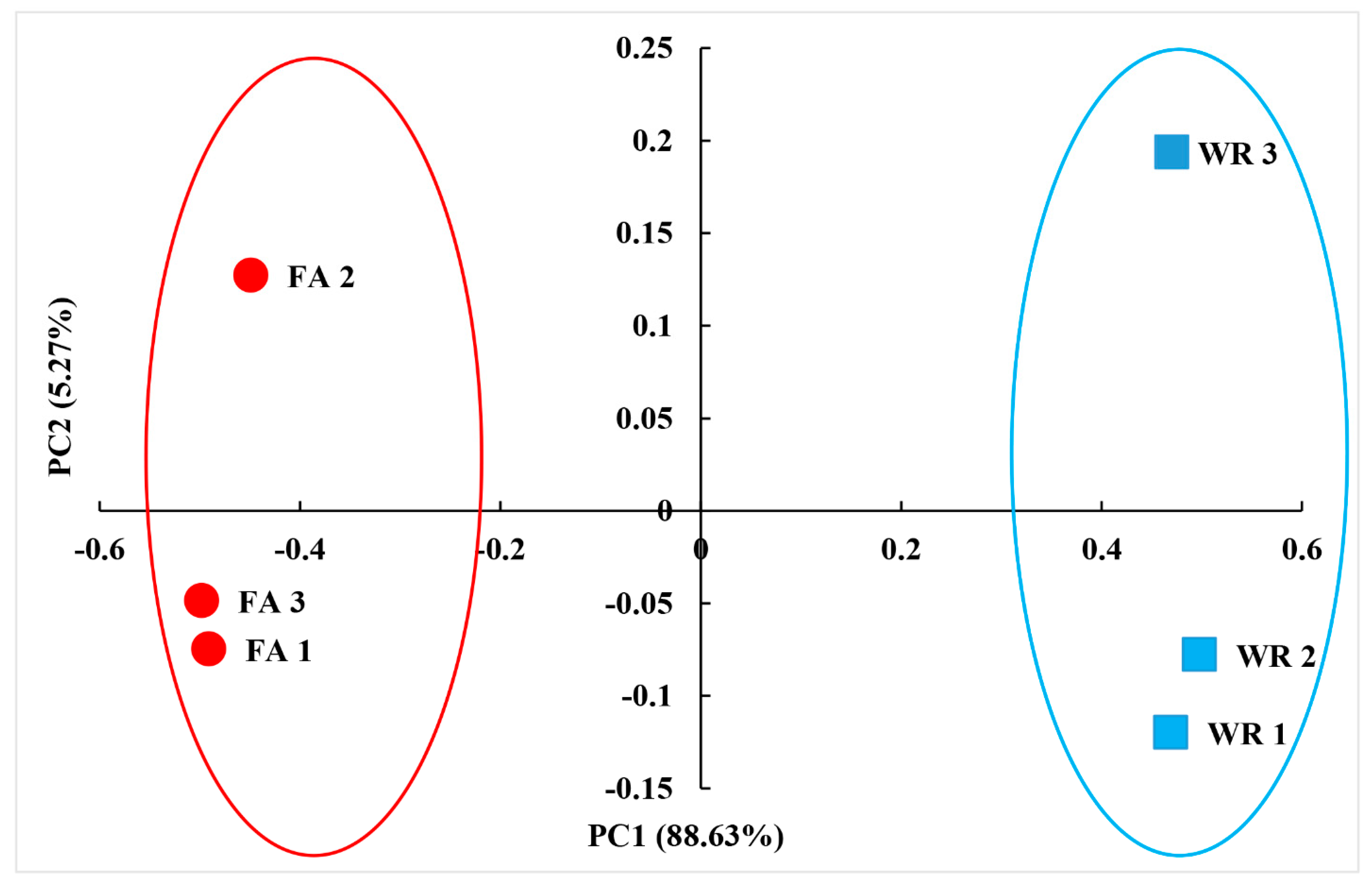
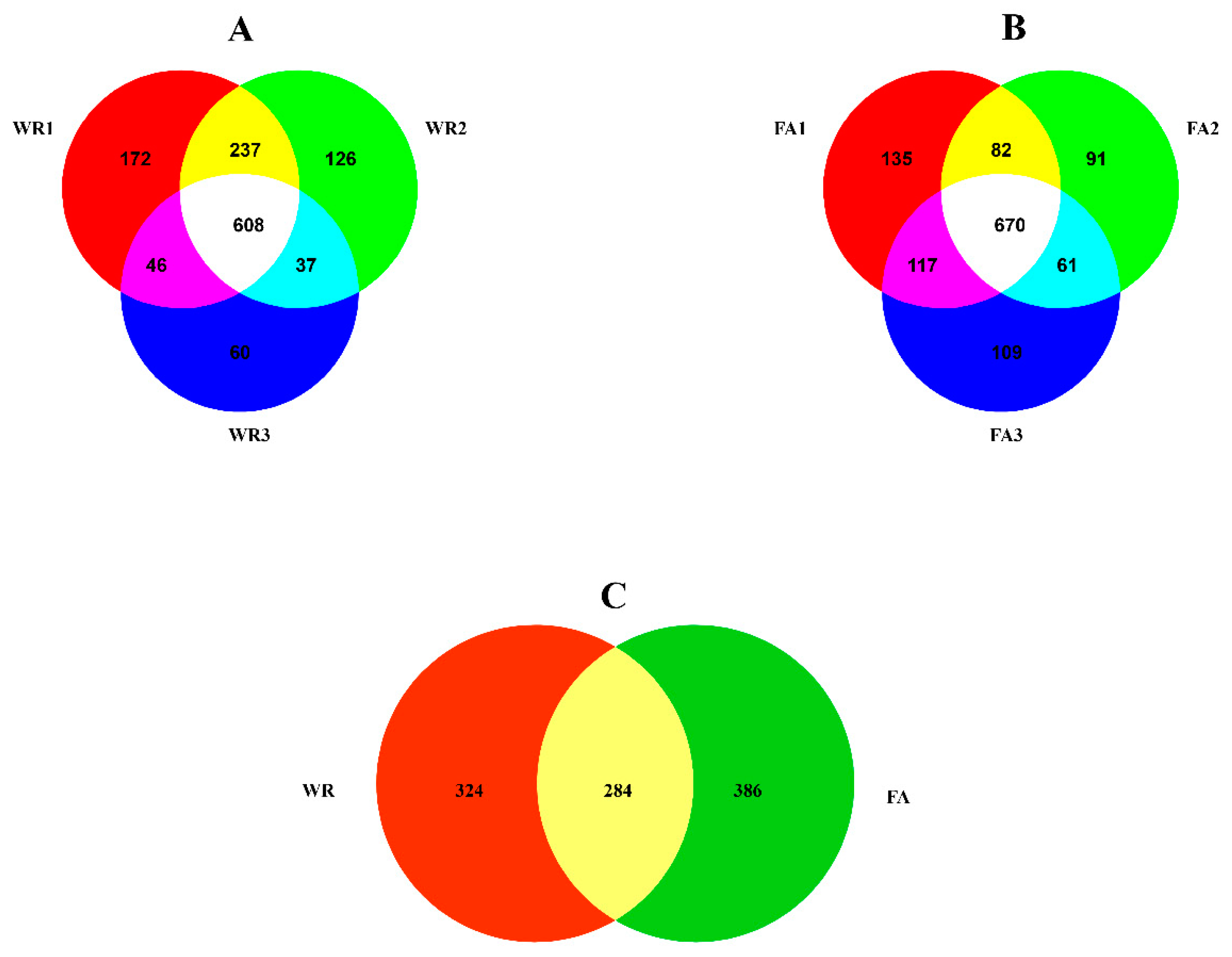
3.1. Diversity of Fungal Communities in Irrigation Water from Two Locations: Mercedes Irrigation District (WR) and South Farm (FA)
3.2. Differences in Composition and Structure between WR and FA
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Rister, M.E.; Sturdivant, A.W.; Lacewell, R.D.; Michelsen, A.M. Challenges and opportunities for water of the Rio Grande. J. Agric. Appl. Econ. 2011, 43, 367–378. [Google Scholar] [CrossRef]
- Malakar, A.; Snow, D.D.; Ray, C. Irrigation water quality—A contemporary perspective. Water 2019, 11, 1482. [Google Scholar] [CrossRef]
- De La Garza, M.N.; Ren, J.; Ancona, V. Spatiotemporal variations of hydrochemical characteristics of irrigation water: A case study of the Lower Rio Grande Valley, USA. Water Supply 2023, 23, 2001–2013. [Google Scholar] [CrossRef]
- Hong, C.; Moorman, G. Plant pathogens in irrigation water: Challenges and opportunities. Crit. Rev. Plant Sci. 2005, 24, 189–208. [Google Scholar] [CrossRef]
- Bewley, W.; Buddin, W. On the Fangus Flora of glass-house water supplies in relation to Plant Diseases. Ann. Appl. Biol. 1921, 8, 10. [Google Scholar] [CrossRef]
- Ali-Shtayeh, M.; MacDonald, J. Occurrence of Phytophthora species in irrigation water in the Nablus area (West Bank of Jordan). Phytopathol. Mediterr. 1991, 30, 143–150. [Google Scholar]
- McIntosh, D. The occurrence of Phytophthora spp. in irrigation systems in British Columbia. Can. J. Bot. 1966, 44, 1591–1596. [Google Scholar] [CrossRef]
- Pittis, J.; Colhoun, J. Isolation and identification of pythiaceous fungi from irrigation water and their pathogenicity to Antirrhinum, tomato and Chamaecyparis lawsoniana. J. Phytopathol. 1984, 110, 301–318. [Google Scholar] [CrossRef]
- Pottorff, L.P.; Panter, K.L. Survey of Pythium and Phytophthora spp. in irrigation water used by Colorado commercial greenhouses. HortTechnology 1997, 7, 153–155. [Google Scholar] [CrossRef]
- Shokes, F.; McCarter, A.M. Occurrence, dissemination, and survival of plant pathogens in surface irrigation ponds in southern Georgia. Phytopathology 1979, 69, 510–516. [Google Scholar] [CrossRef]
- Knight, L. A Field Guide to Irrigation in the Lower Rio Grande Valley; Texas Department of Transportation: Austin, TX, USA, 2009.
- Fernandez, L.; Robinson, J.R.; Lacewell, R.D.; Rister, M.E.; Ellis, J.R.; Sturdivant, A.W.; Stubbs, M.J. Evolution of Irrigation Districts and Operating Institutions: Texas, Lower Rio Grande Valley; Texas Water Resources Institute: College Station, TX, USA, 2003. [Google Scholar]
- Erwin, D.C.; Ribeiro, O.K. Phytophthora Diseases Worldwide; American Phytopathological Society Press: St. Paul, MN, USA, 1996. [Google Scholar]
- Notomi, T.; Okayama, H.; Masubuchi, H.; Yonekawa, T.; Watanabe, K.; Amino, N.; Hase, T. Loop-mediated isothermal amplification of DNA. Nucleic Acids Res. 2000, 28, e63. [Google Scholar] [CrossRef]
- Schena, L.; Ippolito, A.; Nigro, F. Real-Time PCR Detection and QUantification of Soilborne Fungal Pathogens: The Case of “Rosellinia necatrix”, “Phytophthora nicotianae”, “P. citrophthora” and “Verticillium dahlia”. Real Time PCR Detect. QUantification Soilborne Fungal Pathog. 2004, 43, 1000–1008. [Google Scholar]
- Duan, Y.; Zhou, L.; Hall, D.G.; Li, W.; Doddapaneni, H.; Lin, H.; Liu, L.; Vahling, C.M.; Gabriel, D.W.; Williams, K.P. Complete genome sequence of citrus huanglongbing bacterium,‘Candidatus Liberibacter asiaticus’ obtained through metagenomics. Mol. Plan Microbe Interact. 2009, 22, 1011–1020. [Google Scholar] [CrossRef] [PubMed]
- Franco Ortega, S.; Ferrocino, I.; Adams, I.; Silvestri, S.; Spadaro, D.; Gullino, M.L.; Boonham, N. Monitoring and surveillance of aerial mycobiota of rice paddy through DNA metabarcoding and qPCR. J. Fungi 2020, 6, 372. [Google Scholar] [CrossRef] [PubMed]
- Tremblay, É.D.; Duceppe, M.-O.; Bérubé, J.A.; Kimoto, T.; Lemieux, C.; Bilodeau, G.J. Screening for exotic forest pathogens to increase survey capacity using metagenomics. Phytopathology 2018, 108, 1509–1521. [Google Scholar] [CrossRef]
- Prigigallo, M.I.; Abdelfattah, A.; Cacciola, S.O.; Faedda, R.; Sanzani, S.M.; Cooke, D.E.; Schena, L. Metabarcoding analysis of Phytophthora diversity using genus-specific primers and 454 pyrosequencing. Phytopathology 2016, 106, 305–313. [Google Scholar] [CrossRef]
- Català, S.; Perez-Sierra, A.; Abad-Campos, P. The use of genus-specific amplicon pyrosequencing to assess Phytophthora species diversity using eDNA from soil and water in northern Spain. PLoS ONE 2015, 10, e0119311. [Google Scholar] [CrossRef]
- Redekar, N.R.; Eberhart, J.L.; Parke, J.L. Diversity of Phytophthora, Pythium, and Phytopythium species in recycled irrigation water in a container nursery. Phytobiomes J. 2019, 3, 31–45. [Google Scholar] [CrossRef]
- Gardes, M.; Bruns, T.D. ITS primers with enhanced specificity for basidiomycetes-application to the identification of mycorrhizae and rusts. Mol. Ecol. 1993, 2, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Zhang, D.; Cai, X.; Xia, L.; Luo, Y.; Cheng, X.; An, S. Significant alterations in soil fungal communities along a chronosequence of Spartina alterniflora invasion in a Chinese Yellow Sea coastal wetland. Sci. Total Environ. 2019, 693, 133548. [Google Scholar] [CrossRef]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Pena, A.G.; Goodrich, J.K.; Gordon, J.I. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335. [Google Scholar] [CrossRef] [PubMed]
- Gill, S.R.; Pop, M.; DeBoy, R.T.; Eckburg, P.B.; Turnbaugh, P.J.; Samuel, B.S.; Gordon, J.I.; Relman, D.A.; Fraser-Liggett, C.M.; Nelson, K.E. Metagenomic analysis of the human distal gut microbiome. Science 2006, 312, 1355–1359. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. Search and clustering orders of magnitude faster than BLAST. Bioinformatics 2010, 26, 2460–2461. [Google Scholar] [CrossRef] [PubMed]
- Kõljalg, U.; Nilsson, R.H.; Abarenkov, K.; Tedersoo, L.; Taylor, A.F.; Bahram, M.; Bates, S.T.; Bruns, T.D.; Bengtsson-Palme, J.; Callaghan, T.M. Towards a unified paradigm for sequence-based identification of fungi. Mol. Ecol. 2013, 22, 5271–5277. [Google Scholar] [CrossRef]
- Lozupone, C.A.; Hamady, M.; Kelley, S.T.; Knight, R. Quantitative and qualitative β diversity measures lead to different insights into factors that structure microbial communities. Appl. Environ. Microbiol. 2007, 73, 1576–1585. [Google Scholar] [CrossRef]
- Gubler, W.; Feliciano, A.; Bordas, A.; Civerolo, E.; Melvin, J.; Welch, N. First report of blossom blight of strawberry caused by Xanthomonas fragariae and Cladosporium cladosporioides in California. Plant Dis. 1999, 83, 400. [Google Scholar] [CrossRef]
- Briceño, E.X.; Latorre, B.A. Characterization of Cladosporium rot in grapevines, a problem of growing importance in Chile. Plant Dis. 2008, 92, 1635–1642. [Google Scholar] [CrossRef]
- Brunings, A.M.; Datnoff, L.E.; Palmateer, A.J.; Locke, J.C.; Krause, C.R. Exserohilum leaf spot on tiger grass. Plant Health Prog. 2009, 10, 1. [Google Scholar] [CrossRef]
- Luo, Z.; He, F.; Fan, H.; Wang, X.; Hua, M.; Hu, F.; Li, X.; Liu, Z.; Yu, N. First report of leaf spot disease caused by Exserohilum rostratum on pineapple in Hainan province, China. Plant Dis. 2012, 96, 458. [Google Scholar] [CrossRef]
- Palmateer, A.; McLean, K.; Van Santen, E.; Morgan-Jones, G. Occurrence of Nigrospora lint rot caused by Nigrospora oryzae on cotton in Alabama. Plant Dis. 2003, 87, 873. [Google Scholar] [CrossRef]
- Zhang, L.; Li, S.; Tan, G.; Shen, J.; He, T. First report of Nigrospora oryzae causing leaf spot of cotton in China. Plant Dis. 2012, 96, 1379. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Zhao, K.; Zhao, Y.; Zhang, Y.; Fu, Q.; Huang, S. Nigrospora oryzae causing panicle branch rot disease on Oryza sativa (rice). Plant Dis. 2021, 105, 2724. [Google Scholar] [CrossRef] [PubMed]
- Fakhrunnisa, M.H.; Ghaffar, A. Seed-borne mycoflora of wheat, sorghum and barley. Pak. J. Bot. 2006, 38, 185–192. [Google Scholar]
- Standen, J. Nigrospora oryzae (B. and Br.) Fetch on Maize. Phytopathology 1945, 35, 552–564. [Google Scholar]
- Xu, J.; Xu, X.-D.; Cao, Y.-Y.; Zhang, W.-M. First report of greenhouse tomato wilt caused by Plectosphaerella cucumerina in China. Plant Dis. 2014, 98, 158. [Google Scholar] [CrossRef]
- Carlucci, A.; Raimondo, M.; Santos, J.; Phillips, A. Plectosphaerella species associated with root and collar rots of horticultural crops in southern Italy. Pers. Mol. Phylogeny Evol. Fungi 2012, 28, 34–48. [Google Scholar] [CrossRef]
- Wei, T.; Luo, M.; Zhang, H.; Jia, W.; Zeng, Y.; Jiang, Y. Curvularia verruculosa as new causal pathogen of common bean leaf spot disease in China. Crop Prot. 2022, 162, 106091. [Google Scholar] [CrossRef]
- Rajput, N.A.; Huo, C.; Cao, J.; Atiq, M.; Atif, R.M.; Lodhi, A.M.; Syed, R.N.; Sarfraz, S.; Hameed, A.; Zhao, Z. First report of Curvularia verruculosa causing leaf spot disease of grape (Vitis vinifera) in Afghanistan. J. Plant Pathol. 2020, 102, 1337. [Google Scholar] [CrossRef]
- Shirsath, L.P.; Patil, S.P.; Patil, U.K. Incidence of leaf spot disease on cotton caused by Curvularia verruculosa and role of its hydrolytic enzymes in pathogenesis. Physiol. Mol. Biol. Plants 2018, 24, 711–714. [Google Scholar] [CrossRef]
- Ali, M.; Warren, H. Physiological races of Colletotrichum graminicola on sorghum. Plant Dis. 1987, 71, 402–404. [Google Scholar] [CrossRef]
- Chala, A.; Tronsmo, A.; Brurberg, M. Genetic differentiation and gene flow in Colletotrichum sublineolum in Ethiopia, the centre of origin and diversity of sorghum, as revealed by AFLP analysis. Plant Pathol. 2011, 60, 474–482. [Google Scholar] [CrossRef]
- Sanzani, S.M.; Li Destri Nicosia, M.G.; Faedda, R.; Cacciola, S.O.; Schena, L. Use of quantitative PCR detection methods to study biocontrol agents and phytopathogenic fungi and oomycetes in environmental samples. J. Phytopathol. 2014, 162, 1–13. [Google Scholar] [CrossRef]
- Schena, L.; Li Destri Nicosia, M.; Sanzani, S.; Faedda, R.; Ippolito, A.; Cacciola, S. Development of quantitative PCR detection methods for phytopathogenic fungi and oomycetes. J. Plant Pathol. 2013, 95, 7–24. [Google Scholar]
- Stinson, L.F.; Keelan, J.A.; Payne, M.S. Characterization of the bacterial microbiome in first-pass meconium using propidium monoazide (PMA) to exclude nonviable bacterial DNA. Lett. Appl. Microbiol. 2019, 68, 378–385. [Google Scholar] [CrossRef]
- Erkus, O.; de Jager, V.C.; Geene, R.T.; van Alen-Boerrigter, I.; Hazelwood, L.; van Hijum, S.A.; Kleerebezem, M.; Smid, E.J. Use of propidium monoazide for selective profiling of viable microbial cells during Gouda cheese ripening. Int. J. Food Microbiol. 2016, 228, 1–9. [Google Scholar] [CrossRef]
- Weaver, D.; Gago, S.; Bromley, M.; Bowyer, P. The human lung mycobiome in chronic respiratory disease: Limitations of methods and our current understanding. Curr. Fungal Infect. Rep. 2019, 13, 109–119. [Google Scholar] [CrossRef]
- Nilsson, R.H.; Ryberg, M.; Kristiansson, E.; Abarenkov, K.; Larsson, K.-H.; Kõljalg, U. Taxonomic reliability of DNA sequences in public sequence databases: A fungal perspective. PLoS ONE 2006, 1, e59. [Google Scholar] [CrossRef] [PubMed]
- Schloss, P.D.; Westcott, S.L.; Ryabin, T.; Hall, J.R.; Hartmann, M.; Hollister, E.B.; Lesniewski, R.A.; Oakley, B.B.; Parks, D.H.; Robinson, C.J. Introducing mothur: Open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl. Environ. Microbiol. 2009, 75, 7537–7541. [Google Scholar] [CrossRef]
- Blankenberg, D.; Gordon, A.; Von Kuster, G.; Coraor, N.; Taylor, J.; Nekrutenko, A.; Team, G. Manipulation of FASTQ data with Galaxy. Bioinformatics 2010, 26, 1783–1785. [Google Scholar] [CrossRef]
| Sample Name | OTU Number | Chao-1 | ACE | Shannon | Simpson |
|---|---|---|---|---|---|
| WR | 940.67 ± 166.54 a | 976.86 ± 189.91 a | 975.5 ± 191.78 a | 4.6 ± 0.043 a | 0.03 ± 0.003 b |
| FA | 955 ± 50.03 a | 1049.99 ± 52.37 a | 1049.99 ± 53.79 a | 3.63 ± 0.13 b | 0.11 ± 0.03 a |
| Phylum | Class | Relative Abundance (%) | p Value | |
|---|---|---|---|---|
| WR | FA | |||
| Ascomycota | Dothideomycetes | 0.2740% | 0.4626% | 0.3083 |
| Saccharomycetes | 0.0383% | 0.1041% | 0.3542 | |
| Basidiomycota | Tremellomycetes | 0.0408% | ||
| Chytridiomycota | Cladochytriomycetes | 0.0230% | ||
| Chytridiomycetes | 1.4648% | 1.7089% | 0.5811 | |
| GS13 | 0.0696% | 0.0192% | 0.0161 * | |
| Rhizophydiomycetes | 0.5661% | 0.1422% | 0.0373 * | |
| Monoblepharomycota | Sanchytriomycetes | 0.0348% | 0.0456% | 0.3009 |
| Sordariomycetes | 0.1054% | 0.1118% | 0.9255 | |
| Phylum | Class | Family | Relative Abundance (%) | p Value | |
|---|---|---|---|---|---|
| WR | FA | ||||
| Ascomycota | Dothideomycetes | Cladosporiaceae | 0.1541% | 0.2111% | 0.3989 |
| Dothideomycetes | Pleosporaceae | 0.0470% | 0.0948% | 0.4757 | |
| Sordariomycetes | Stachybotryaceae | 0.0155% | 0.0531% | 0.4837 | |
| Dothideomycetes | Lentitheciaceae | 0.0215% | |||
| Dothideomycetes | Plectosphaerellaceae | 0.0216% | |||
| Saccharomycetales | Saccharomycetales | 0.0256% | |||
| Sordariomycetes | Trichosphaeriaceae | 0.0288% | |||
| Dothideomycetes | Aureobasidiaceae | 0.0343% | |||
| Saccharomycetes | Metschnikowiaceae | 0.0929% | |||
| Sordariomycetes | Microdochiaceae | 0.0108% | |||
| Basidiomycota | Tremellomycetes | Bulleribasidiaceae | 0.0272% | ||
| Chytridiomycota | Chytridiomycetes | Chytridiaceae | 1.3873% | 1.6499% | 0.5338 |
| Cladochytriomycetes | Nowakowskiellaceae | 0.0230% | |||
| Rhizophydiomycetes | Aquamycetaceae | 0.0735% | |||
| Rhizophydiomycetes | Alphamycetaceae | 0.0186% | |||
| Monoblepharomycota | Sanchytriomycetes | Sanchytriaceae | 0.0348% | 0.0456% | 0.3011 |
| ID | Plant Pathogen | Host | WR | FA |
|---|---|---|---|---|
| OTU112 | Cladosporium cladosporioides | Strawberry and grapevine | √ | √ |
| OTU213 | Exserohilum rostratum | Tiger Grass and pineapple leaf | √ | √ |
| OTU456 | Nigrospora oryzae | Rice, wheat, sorghum, barley, maize and cotton | √ * | √ |
| OTU295, OTU229 | Curvularia_verruculosa | Bean, cotton, and grape | √ | |
| OTU609, OTU730 | Colletotrichum sublineola | Sorghum | √ | |
| OTU316 | Plectosphaerella cucumerina | Melon, pepper and tomato | √ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Calderon, M.; Yang, C.; Ancona, V. Assessing Fungal Plant Pathogen Presence in Irrigation Water from the Rio Grande River in South Texas, USA. Agriculture 2023, 13, 1401. https://doi.org/10.3390/agriculture13071401
Calderon M, Yang C, Ancona V. Assessing Fungal Plant Pathogen Presence in Irrigation Water from the Rio Grande River in South Texas, USA. Agriculture. 2023; 13(7):1401. https://doi.org/10.3390/agriculture13071401
Chicago/Turabian StyleCalderon, Miriam, Chuanyu Yang, and Veronica Ancona. 2023. "Assessing Fungal Plant Pathogen Presence in Irrigation Water from the Rio Grande River in South Texas, USA" Agriculture 13, no. 7: 1401. https://doi.org/10.3390/agriculture13071401
APA StyleCalderon, M., Yang, C., & Ancona, V. (2023). Assessing Fungal Plant Pathogen Presence in Irrigation Water from the Rio Grande River in South Texas, USA. Agriculture, 13(7), 1401. https://doi.org/10.3390/agriculture13071401







