Analysis of Heavy Metal Impacts on Cereal Crop Growth and Development in Contaminated Soils
Abstract
:1. Introduction
1.1. The Key Role of Cereal Crops in Ensuring World Food Resilience
1.2. The Challenge of Heavy Metal Contamination in Soil and Its Potential Impact on Crop Production
2. Unravelling the Intricate Relationship between Heavy Metals and Cereals
2.1. Uptake and Translocation of Heavy Metals in Cereals
2.2. Metal Tolerance and Accumulation
2.3. Impact of Heavy Metals on Plant Physiology
2.4. Effects of Heavy Metals on Cereal Crop Yield and Quality
2.5. Bioavailability and Bioaccumulation of Heavy Metals
2.6. Metal Interaction and Competition
2.7. Strategies to Mitigate Heavy Metal Uptake in Cereal Crops
3. Mechanisms of Heavy Metal-induced Growth Impairment in Plants: Favourable Conditions for Metal Toxicity
3.1. Disruption of Enzymatic Activities
3.2. Reactive Oxygen Species Production
3.3. Nutrient Imbalances
3.4. Disruption of Membrane Integrity
4. Symptoms and Manifestations of Heavy Metal Toxicity on Cereal Plants
4.1. Chlorosis
4.2. Stunted Growth
4.3. Reduced Root Growth
4.4. Leaf Deformities
4.5. Reduced Flowering and Fruit Development in Cereals
4.6. Necrosis
4.7. Water Stress
5. Factors Predisposing Cereal Crops to Heavy Metal Toxicity
5.1. Soil pH and Heavy Metal Bioavailability in Cereal Crops
5.2. Soil Organic Matter and Its Role in Mitigating Heavy Metal Toxicity in Cereal Crops
5.3. Soil Texture and Its Influence on Heavy Metal Availability in Cereal Crops
5.4. Plant Species and Varieties: Key Players in Heavy Metal Tolerance
5.5. Metal Concentration in Soil: A Determining Factor in Heavy Metal Toxicity to Cereal Crops
5.6. Influence of Environmental Factors on the Toxicity of Heavy Metals to Cereal Crops
6. Cereal Defence Strategies against Heavy Metal Toxicity: Detoxification Mechanisms and Adaptation Strategies
6.1. Metal Transporters: Orchestrating Heavy Metal Transferring in Cereal Plants
- -
- NRAMP (Natural Resistance-associated Macrophage Protein)
- NRAMP proteins are involved in the transport of divalent cations such as iron (Fe2+), manganese (Mn2+), zinc (Zn2+), and cadmium (Cd2+);
- They play a crucial role in metal uptake from the soil into the roots.
- -
- ZIP (Zrt/Irt-like Proteins)
- ZIP transporters are responsible for the uptake of essential divalent metals like zinc (Zn2+), iron (Fe2+), and manganese (Mn2+);
- They are involved in the movement of metals from the roots to the above-ground parts of the plant.
- -
- HMA (Heavy Metal ATPase)
- HMA transporters are ATPases that are responsible for the transport of various heavy metals, including copper (Cu2+), zinc (Zn2+), and cadmium (Cd2+);
- They are involved in metal compartmentalization and detoxification within plant cells.
- -
- IRT (Iron-regulated Transporter)
- IRT transporters are specific to iron (Fe2+). They play a decisive role in iron uptake from the soil;
- These transporters are particularly important for plants growing in iron-deficient soils.
- -
- YSL (Yellow Stripe-like Transporters)
- YSL transporters are involved in the uptake of metal–chelate complexes, particularly iron (Fe3+) bound to phytosiderophores;
- They play a role in iron uptake in graminaceous plants like cereals.
- -
- Ferrous Iron Transporter (IRT1)
- IRT1 is a specific transporter for ferrous iron (Fe2+). It plays a key role in iron uptake from the soil.
- -
- COPT (Copper Transporter)
- COPT transporters are involved in the uptake of copper (Cu2+);
- They play a role in copper homeostasis and distribution within the plant.
- -
- MTP (Metal Tolerance Protein)
- MTP transporters are involved in the transport of various heavy metals, including zinc (Zn2+), cobalt (Co2+), and cadmium (Cd2+);
- They are implicated in metal detoxification and sequestration.
- -
- MerT and MerP Proteins
- In some bacteria, like those with the mer operon, MerT, and MerP proteins are involved in the transport of mercury (Hg2+);
- These proteins work in conjunction with other components of the mer operon to facilitate mercury uptake and detoxification.
- -
- NiCoT Transporters (Nickel and Cobalt Transporters)
- These transporters are responsible for the uptake of nickel (Ni2+) in plants;
- They play a role in regulating the cellular concentration of nickel and are essential for nickel-dependent enzymes.
6.2. Metal Sequestration: A Key Defence Mechanism in Cereal Crops
6.3. Metal Chelation in Cereal Crop Defence against Heavy Metal Toxicity
6.4. Enhanced Efflux: Empowering Cereal Crops against Heavy Metal Stress
6.5. Siderophores as Nature’s Metal Detoxifiers: Protecting Cereals from Heavy Metal Toxicity
6.6. Antioxidant Production: Shielding Cereal Crops from Heavy Metal-Induced Oxidative Stress
6.7. Mycorrhizal Symbiosis: Fungal Allies in Shielding Cereal Crops from Heavy Metal Uptake
6.8. Harnessing Plant Growth-Promoting Bacteria for Cereal Defence against Heavy Metal Toxicity
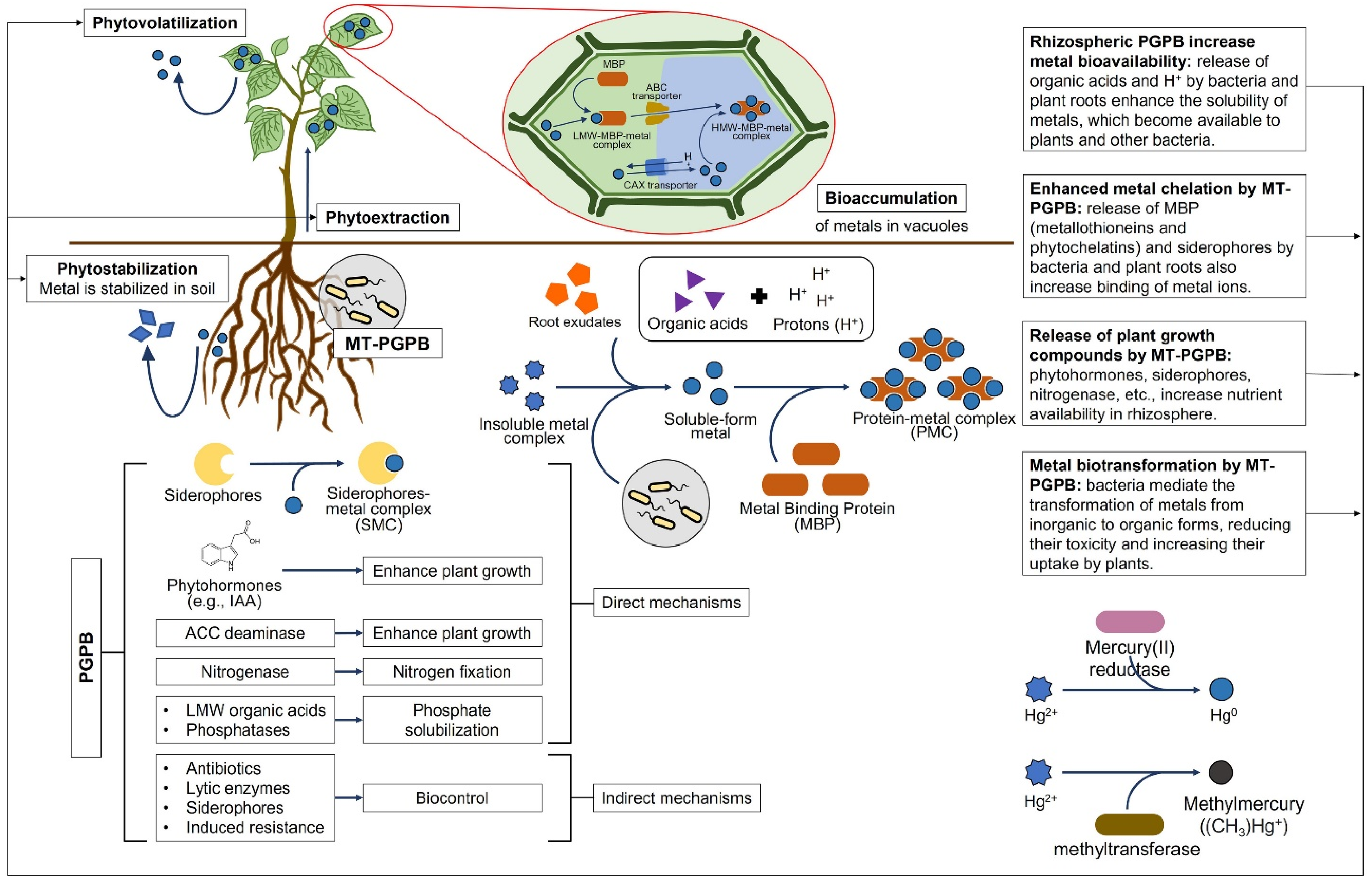
7. Exploring New Frontiers: Unravelling the Impact of Heavy Metal Presence in Soil on Cereals for Promoting Innovation beyond the State of the Art
8. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Golia, E.E. The impact of heavy metal contamination on soil quality and plant nutrition. Sustainable management of moderate contaminated agricultural and urban soils, using low cost materials and promoting circular economy. Sustain. Chem. Pharm. 2023, 33, 101046. [Google Scholar] [CrossRef]
- Mng’ong’o, M.; Munishi, L.K.; Ndakidemi, P.A.; Blake, W.; Comber, S.; Hutchinson, T.H. Toxic metals in East African agro-ecosystems: Key risks for sustainable food production. J. Environ. Manag. 2021, 294, 112973. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Tang, Z.; Sun, J.; Xin, X.; Zhang, M.; Cheng, J. Heavy metals in soil contaminated through e-waste processing activities in a recycling area: Implications for risk management. Process Saf. Environ. Prot. 2019, 125, 189–196. [Google Scholar] [CrossRef]
- Tian, W.; Zhang, M.; Zong, D.; Li, W.; Li, X.; Wang, Z.; Zhang, Y.; Niu, Y.; Xiang, P. Are high-risk heavy metal(loid)s contaminated vegetables detrimental to human health? A study of incorporating bioaccessibility and toxicity into accurate health risk assessment. Sci. Total Environ. 2023, 897, 165514. [Google Scholar] [CrossRef]
- Zakaria, Z.; Zulkafflee, N.S.; Mohd Redzuan, N.A.; Selamat, J.; Ismail, M.R.; Praveena, S.M.; Tóth, G.; Ahmad Faizal Abdull Razis, A.F. Understanding potential heavy metal contamination, absorption, translocation and accumulation in rice and human health risks. Plants 2021, 10, 1070. [Google Scholar] [CrossRef] [PubMed]
- Javaid, S.; Ashraf, K.; Sultan, K.; Siddiqui, M.H.; Ali, H.M.; Chen, Y.; Zaman, Q. Risk assessment of potentially toxic metals and metalloids in soil, water and plant continuum of fragrant rice. Agronomy 2022, 12, 2480. [Google Scholar] [CrossRef]
- Rai, P.K.; Lee, S.S.; Zhang, M.; Tsang, Y.F.; Kim, K.H. Heavy metals in food crops: Health risks, fate, mechanisms, and management. Environ. Int. 2019, 125, 365–385. [Google Scholar] [CrossRef]
- Ohiagu, F.O.; Lele, K.C.; Chikezie, P.C.; Verla, A.W.; Enyoh, C.E. Bioaccumulation and health risk assessment of heavy metals in Musa paradisiaca, Zea mays, Cucumeropsis manii and Manihot esculenta cultivated in Onne, Rivers State, Nigeria. Environ. Anal. Health Toxicol. 2020, 35, e2020011. [Google Scholar] [CrossRef]
- Achterbosch, T.; van Berkum, S.; Meijerink, G.; Asbreuk, H.; Oudendag, D. Cash Crops and Food Security: Contributions to Income, Livelihood Risk and Agricultural Innovation; LEI Wageningen UR: Wageningen, The Netherlands, 2014. [Google Scholar]
- Serna-Saldivar, S.O. Cereal Grains: Properties, Processing, and Nutritional Attributes; CRC Press: Boca Raton, FL, USA, 2016. [Google Scholar]
- Raheem, D.; Dayoub, M.; Birech, R.; Nakiyemba, A. The contribution of cereal grains to food security and sustainability in Africa: Potential application of UAV in Ghana, Nigeria, Uganda, and Namibia. Urban Sci. 2021, 5, 8. [Google Scholar] [CrossRef]
- FAO. The Future of Food and Agriculture: Trends and Challenges; Food and Agriculture Organization of the United Nations: Rome, Italy, 2017. [Google Scholar]
- Grote, U.; Fasse, A.; Nguyen, T.T.; Erenstein, O. Food security and the dynamics of wheat and maize value chains in Africa and Asia. Front. Sustain. Food Syst. 2021, 4, 617009. [Google Scholar] [CrossRef]
- Fatima, Z.; Ahmed, M.; Hussain, M.; Abbas, G.; Ul-Allah, S.; Ahmad, S.; Ahmed, N.; Ali, M.A.; Sarwar, G.; Haque, E.; et al. The fingerprints of climate warming on cereal crops phenology and adaptation options. Sci. Rep. 2020, 10, 18013. [Google Scholar] [CrossRef]
- Bala, B.K. Drying and Storage of Cereal Grains; John Wiley & Sons: New York, NY, USA, 2016. [Google Scholar]
- Richards, R.A.; Rebetzke, G.J.; Condon, A.G.; van Herwaarden, A.F. Breeding opportunities for increasing the efficiency of water use and crop yield in temperate cereals. Crop Sci. 2002, 42, 111–121. [Google Scholar] [CrossRef] [PubMed]
- Hussain, N.; Sun, D.-W.; Pu, H. Classical and emerging non-destructive technologies for safety and quality evaluation of cereals: A review of recent applications. Trends Food Sci. Technol. 2019, 91, 598–608. [Google Scholar] [CrossRef]
- Kruseman, G.; Mottaleb, K.A.; Tesfaye, K.; Bairagi, S.; Robertson, R.; Mandiaye, D.; Frija, A.; Gbegbelegbe, S.; Alene, A.; Prager, S. Rural transformation and the future of cereal-based agri-food systems. Glob. Food Sec. 2020, 26, 100441. [Google Scholar] [CrossRef]
- Battat, M.; Lampietti, J. The grain chain: Trade and food security in Arab countries. In Trade Policy and Food Security: Improving Access to Food in Developing Countries in the Wake of High World Prices; Gillson, I., Fouad, A., Eds.; The World Bank Group: Washington, DC, USA, 2015; pp. 189–214. [Google Scholar]
- Capone, R.; El Bilali, H.; Debs, P.; Cardone, G.; Driouech, N. Food system sustainability and food security: Connecting the dots. J. Food Sec. 2014, 2, 13–22. [Google Scholar]
- Allee, A.; Lynd, L.R.; Vaze, V. Cross-national analysis of food security drivers: Comparing results based on the food insecurity experience scale and global food security index. Food Sec. 2021, 13, 1245–1261. [Google Scholar] [CrossRef]
- Cai, J.; Ma, E.; Lin, J.; Liao, L.; Han, Y. Exploring global food security pattern from the perspective of spatio-temporal evolution. J. Geogr. Sci. 2020, 30, 179–196. [Google Scholar] [CrossRef]
- Legass, A.; Assen, M.; Tesfaye, M. Predicting soil erosion by water: RUSLE application for soil conservation planning in central rift valley of Ethiopia. Environ. Eng. Manag. J. 2021, 20, 1521–1534. [Google Scholar]
- Zhang, B.; Hou, H.; Huang, Z.; Zhao, L. Estimation of heavy metal soil contamination distribution, hazard probability, and population at risk by machine learning prediction modeling in Guangxi, China. Environ. Pollut. 2023, 330, 121607. [Google Scholar] [CrossRef]
- Wang, F.; Guan, Q.; Tian, J.; Lin, J.; Yang, Y.; Yang, L.; Pan, N. Contamination characteristics, source apportionment, and health risk assessment of heavy metals in agricultural soil in the Hexi Corridor. Catena 2020, 191, 104573. [Google Scholar] [CrossRef]
- Tóth, G.; Hermann, T.; Da Silva, M.R.; Montanarella, L. Heavy metals in agricultural soils of the European Union with implications for food safety. Environ. Int. 2016, 88, 299–309. [Google Scholar] [CrossRef]
- Tóth, G.; Hermann, T.; Szatmári, G.; Pásztor, L. Maps of heavy metals in the soils of the European Union and proposed priority areas for detailed assessment. Sci. Total Environ. 2016, 565, 1054–1062. [Google Scholar] [CrossRef] [PubMed]
- Ding, Q.; Cheng, G.; Wang, Y.; Zhuang, D. Effects of natural factors on the spatial distribution of heavy metals in soils surrounding mining regions. Sci. Total Environ. 2016, 578, 577–585. [Google Scholar] [CrossRef] [PubMed]
- Tepanosyan, G.; Sahakyan, L.; Belyaeva, O.; Asmaryan, S.; Saghatelyan, A. Continuous impact of mining activities on soil heavy metals levels and human health. Sci. Total Environ. 2018, 639, 900–909. [Google Scholar] [CrossRef] [PubMed]
- Afonne, O.J.; Ifediba, E.C. Heavy metals risks in plant foods—Need to step up precautionary measures. Curr. Opin. Toxicol. 2020, 22, 1–6. [Google Scholar] [CrossRef]
- Pehoiu, G.; Murarescu, O.; Radulescu, C.; Dulama, I.D.; Teodorescu, S.; Stirbescu, R.M.; Bucurica, I.A.; Stanescu, S.G. Heavy metals accumulation and translocation in native plants grown on tailing dumps and human health risk. Plant Soil 2020, 456, 405–424. [Google Scholar] [CrossRef]
- Ismael, M.A.; Elyamine, A.M.; Moussa, M.G.; Cai, M.; Zhao, M.; Hu, C. Cadmium in plants: Uptake, toxicity, and its interactions with selenium fertilizers. Metallomics 2019, 11, 255–277. [Google Scholar] [CrossRef] [PubMed]
- Shahid, M.; Dumat, C.; Khalid, S.; Niazi, N.K.; Antunes, P.M.C. Cadmium bioavailability, uptake, toxicity and detoxification in soil-plant system. In Reviews of Environmental Contamination and Toxicology; de Voogt, P., Ed.; Springer: Cham, Switzerland, 2016; Volume 241, pp. 73–137. [Google Scholar]
- Clemens, S.; Ma, J.F. Toxic heavy metal and metalloid accumulation in crop plants and foods. Annu. Rev. Plant Biol. 2016, 67, 489–512. [Google Scholar] [CrossRef]
- Deng, X.; Chen, X.; Yang, Y.; Lu, L.; Yuan, X.; Zeng, X.; Zeng, Q. Cadmium accumulation in rice (Oryza sativa L.) alleviated by basal alkaline fertilizers followed by topdressing of manganese fertilizer. Environ. Pollut. 2020, 262, 114289. [Google Scholar] [CrossRef]
- Hafeez, A.; Rasheed, R.; Ashraf, M.A.; Qureshi, F.F.; Hussain, I.; Iqbal, H. Effect of heavy metals on growth, physiological and biochemical responses of plants. In Plants and Their Interaction to Environmental Pollution. Damage Detection, Adaptation, Tolerance, Physiological and Molecular Responses; Husen, A., Ed.; Elsevier: Amsterdam, The Netherlands, 2023; pp. 139–159. [Google Scholar]
- Jamla, M.; Khare, T.; Joshi, S.; Patil, S.; Penna, S.; Kumar, S. Omics approaches for understanding heavy metal responses and tolerance in plants. Curr. Plant Biol. 2021, 27, 100213. [Google Scholar] [CrossRef]
- Ansari, M.S.; Tauseef, A.; Haris, M.; Hussain, T.; Khan, A.A. Effects of heavy metals present in sewage sludge, their impact on soil fertility, soil microbial activity, and environment. In Development in Waste Water Treatment Research and Processes: Treatment and Reuse of Sewage Sludge: An Innovative Approach for Wastewater Treatment; Shah, M.P., Shah, N., Rodriguez-Couto, S., Banerjee, R., Eds.; Elsevier: Amsterdam, The Netherlands, 2022; pp. 197–214. [Google Scholar]
- Qin, S.; Liu, H.; Nie, Z.; Rengel, Z.; Gao, W.; Li, C.; Zhao, P. Toxicity of cadmium and its competition with mineral nutrients for uptake by plants: A review. Pedosphere 2020, 30, 168–180. [Google Scholar] [CrossRef]
- Diaconu, M.; Pavel, L.V.; Hlihor, R.M.; Rosca, M.; Fertu, D.I.; Lenz, M.; Corvini, P.X.; Gavrilescu, M. Characterization of heavy metal toxicity in some plants and microorganisms—A preliminary approach for environmental bioremediation. New Biotechnol. 2020, 56, 130–139. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, M.; Ma, Y. Mitigation of heavy metal stress in the soil through optimized interaction between plants and microbes. J. Environ. Manag. 2023, 345, 118732. [Google Scholar] [CrossRef] [PubMed]
- Okewale, I.A.; Grobler, H. Assessment of heavy metals in tailings and their implications on human health. Geosyst. Geoenviron. 2023, 2, 100203. [Google Scholar] [CrossRef]
- Wong, C.; Roberts, S.M.; Saab, I.N. Review of regulatory reference values and background levels for heavy metals in the human diet. Regul. Toxicol. Pharmacol. 2022, 130, 105122. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Regulation (EC) No. 1881/2006. Commission Regulation (EC) No 1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in foodstuffs (Text with EEA relevance). OJ 2006, L 364, 5–24. [Google Scholar]
- Alexander, J.; Benford, D.; Cockburn, A.; Cravedi, J.-P.; Dogliotti, E.; Di Domenico, A.; Maria Luisa Férnandez-Cruz, M.-L.; Fürst, R.; Fink-Gremmels, J.; Galli, C.L.; et al. Cadmium in food. Scientific opinion of the panel on contaminants in the food chain. EFSA J. 2009, 980, 1–139. [Google Scholar]
- Awino, F.B.; Maher, W.; Lynch, A.J.J.; Asanga Fai, P.B.; Otim, O. Comparison of metal bioaccumulation in crop types and consumable parts between two growth periods. Integr. Environ. Assess. Manag. 2022, 18, 1056–1071. [Google Scholar] [CrossRef] [PubMed]
- Shahid, M.; Dumat, C.; Khalid, S.; Schreck, E.; Xiong, T.; Nabeel Khan Niazi, N.K. Foliar heavy metal uptake, toxicity and detoxification in plants: A comparison of foliar and root metal uptake. J. Hazard. Mater. 2017, 325, 36–58. [Google Scholar] [CrossRef]
- Cataldo, D.A.; Wildung, R.E. Soil and plant factors influencing the accumulation of heavy metals by plants. Environ. Health Perspect. 1978, 27, 149–159. [Google Scholar] [CrossRef]
- Abedi, T.; Gavanji, S.; Mojiri, A. Lead and zinc uptake and toxicity in maize and their management. Plants 2022, 11, 1922. [Google Scholar] [CrossRef] [PubMed]
- Khan, I.U.; Qi, S.-S.; Gul, F.; Manan, S.; Rono, J.K.; Naz, M.; Shi, X.-N.; Zhang, H.; Dai, Z.-C.; Du, D.-L. A green approach used for heavy metals ‘phytoremediation’ via invasive plant species to mitigate environmental pollution: A review. Plants 2023, 12, 725. [Google Scholar] [CrossRef] [PubMed]
- Thomas, M.A. comparative study of the factors affecting uptake and distribution of Cd with Ni in barley. Plant Physiol. Biochem. 2021, 162, 730–736. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.J.; Wang, P. Arsenic and cadmium accumulation in rice and mitigation strategies. Plant Soil 2020, 446, 1–21. [Google Scholar] [CrossRef]
- Carrijo, D.R.; LaHue, G.T.; Parikh, S.J.; Chaney, R.L.; Linquist, B.A. Mitigating the accumulation of arsenic and ca dmium in rice grain: A quantitative review of the role of water management. Sci. Total Environ. 2020, 839, 156245. [Google Scholar] [CrossRef]
- Zhou, M.; Zheng, S. Multi-omics uncover the mechanism of wheat under heavy metal stress. Int. J. Mol. Sci. 2022, 23, 15968. [Google Scholar] [CrossRef] [PubMed]
- Rizvi, A.; Zaidi, A.; Ameen, F.; Ahmed, B.; AlKahtani, M.D.F.; Khan, M.S. Heavy metal induced stress on wheat: Phytotoxicity and microbiological management. RSC Adv. 2020, 10, 38379–38403. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.F.; Shen, R.F.; Shao, J.F. Transport of cadmium from soil to grain in cereal crops: A review. Pedosphere 2021, 31, 3–10. [Google Scholar] [CrossRef]
- Zulkafflee, N.S.; Redzuan, N.A.M.; Nematbakhsh, S.; Selamat, J.; Ismail, M.R.; Praveena, S.M.; Lee, S.Y.; Abdull Razis, A.F. Heavy metal contamination in Oryza sativa L. at the eastern region of Malaysia and its risk assessment. Int. J. Environ. Res. Public Health 2022, 19, 739. [Google Scholar] [CrossRef]
- Antisari, L.V.; Orsini, F.; Marchetti, L.; Vianello, G.; Gianquinto, G. Heavy metal accumulation in vegetables grown in urban gardens. Agron. Sustain. Dev. 2015, 35, 1139–1147. [Google Scholar] [CrossRef]
- Zhou, H.; Yang, W.-T.; Zhou, X.; Liu, L.; Gu, J.-F.; Wang, W.-L.; Zou, J.-L.; Tian, T.; Peng, P.Q.; Liao, B.-H. Accumulation of heavy metals in vegetable species planted in contaminated soils and the health risk assessment. Int. J. Environ. Res. Public Health 2016, 13, 289. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Ma, C.; Chen, H.; Zhang, J.; White, J.C.; Chen, G.; Xing, B. Xylem-based long-distance transport and phloem remobilization of copper in Salix integra Thunb. J. Hazard. Mater. 2020, 392, 122428. [Google Scholar] [CrossRef] [PubMed]
- Zulfiqar, U.; Farook, M.; Hussain, F.; Maqsood, M.; Hussain, M.; Ishfaq, M.; Ahmad, M.; Anjum, M.Z. Lead toxicity in plants: Impacts and remediation. J. Environ. Manag. 2019, 250, 109557. [Google Scholar] [CrossRef] [PubMed]
- Pasricha, S.; Mathur, V.; Garg, A.; Lenka, S.; Verma, K.; Agarwal, S. Molecular mechanisms underlying heavy metal uptake, translocation and tolerance in hyperaccumulators-an analysis. Heavy metal tolerance in hyperaccumulators. Environ. Chall. 2021, 4, 100197. [Google Scholar] [CrossRef]
- Yan, A.; Wang, Y.; Tan, S.N.; Mohd Yusof, M.L.; Ghosh, S.; Chen, Z. Phytoremediation: A promising approach for revegetation of heavy metal-polluted land. Front. Plant Sci. 2020, 11, 359. [Google Scholar] [CrossRef] [PubMed]
- Maiti, D.; Saha, D.; Kumar, A. Metal bioaccumulation in native plants from a coal fly ash dump in an abandoned opencast coal mine. Environ. Eng. Manag. J. 2022, 21, 2019–2029. [Google Scholar] [CrossRef]
- Shumaker, K.L.; Begonia, G. Heavy metal uptake, translocation, and bioaccumulation studies of Triticum aestivum cultivated in contaminated dredged materials. Int. J. Environ. Res. Public Health 2005, 2, 293–298. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Roychoudhury, A. Omics tools to understand abiotic stress response and adaptation in rye, oat and barley. In Omics Approach to Manage Abiotic Stress in Cereals; Roychoudhury, A., Aftab, T., Acharya, K., Eds.; Springer: Singapore, 2022; pp. 513–529. [Google Scholar]
- Fukusaki, E.; Kobayashi, A. Plant metabolomics: Potential for practical operation. J. Biosci. Bioeng. 2005, 100, 347–354. [Google Scholar] [CrossRef]
- Jeyasri, R.; Muthuramalingam, P.; Satish, L.; Pandian, S.K.; Chen, J.-T.; Ahmar, S.; Wang, X.; Mora-Poblete, F.; Ramesh, M. An overview of abiotic stress in cereal crops: Negative impacts, regulation, biotechnology and integrated omics. Plants 2021, 10, 1472. [Google Scholar] [CrossRef]
- González, Á.; del Mar Gil-Diaz, M.; del Carmen Lobo, M. Metal tolerance in barley and wheat cultivars: Physiological screening methods and application in phytoremediation. J. Soils Sediments 2017, 17, 1403–1412. [Google Scholar] [CrossRef]
- Haddad, M.; Nassar, D.; Shtaya, M. Heavy metals accumulation in soil and uptake by barley (Hordeum vulgare) irrigated with contaminated water. Sci. Rep. 2023, 13, 4121. [Google Scholar] [CrossRef] [PubMed]
- Riyazuddin, R.; Nisha, N.; Ejaz, B.; Khan, M.I.R.; Kumar, M.; Ramteke, P.W.; Gupta, R.A. Comprehensive review on the heavy metal toxicity and sequestration in plants. Biomolecules 2022, 12, 43. [Google Scholar] [CrossRef]
- Lwalaba, J.L.W.; Zvobgo, G.; Mwamba, T.M.; Louis, L.T.; Fu, L.; Kirika, B.A.; Tshibangu, A.K.; Adil, M.F.; Sehar, S.; Mukobo, R.P. High accumulation of phenolics and amino acids confers tolerance to the combined stress of cobalt and copper in barley (Hordeum vulagare). Plant Physiol. Biochem. 2020, 155, 927–937. [Google Scholar] [CrossRef] [PubMed]
- Szewinska, J.; Rozanska, E.; Papierowska, E.; Labudda, M. Proteolytic and structural changes in rye and triticale roots under aluminum stress. Cells 2021, 10, 3046. [Google Scholar] [CrossRef] [PubMed]
- Li, X.F.; Ma, J.F.; Matsumoto, H. Pattern of aluminum-induced secretion of organic acids differs between rye and wheat. Plant Physiol. 2000, 123, 1537–1544. [Google Scholar] [CrossRef] [PubMed]
- Priya, A.K.; Muruganandam, M.; Ali, S.S.; Kornaros, M. Clean-up of heavy metals from contaminated soil by phytoremediation: A multidisciplinary and eco-friendly approach. Toxics 2023, 11, 422. [Google Scholar] [CrossRef]
- Batlles-delaFuente, A.; Abad-Segura, E.; González-Zamar, M.-D.; Cortés-García, F.J. An evolutionary approach on the framework of circular economy applied to agriculture. Agronomy 2022, 12, 620. [Google Scholar] [CrossRef]
- Abhilash, P.C.; Tripathi, V.; Edrisi, S.A.; Dubey, R.K.; Bakshi, M.; Dubey, P.K.; Singh, H.B.; Ebbs, S.D. Sustainability of crop production from polluted lands. Energ. Ecol. Environ. 2016, 1, 54–65. [Google Scholar] [CrossRef]
- Ghori, N.H.; Ghori, T.; Hayat, M.Q.; Imadi, S.R.; Gul, A.; Altay, V.; Ozturk, M. Heavy metal stress and responses in plants. Int. J. Environ. Sci. Technol. 2019, 16, 1807–1828. [Google Scholar] [CrossRef]
- Haider, F.U.; Liqun, C.; Coulter, J.A.; Cheema, S.A.; Wu, J.; Zhang, R.; Wenjun, M.; Farooq, M. Cadmium toxicity in plants: Impacts and remediation strategies. Ecotoxicol. Environ. Saf. 2021, 211, 111887. [Google Scholar] [CrossRef]
- Dias, M.C.; Monteiro, C.; Moutinho-Pereira, J.; Correia, C.; Gonçalves, B.; Santos, C. Cadmium toxicity affects photosynthesis and plant growth at different levels. Acta Physiol. Plant 2013, 35, 1281–1289. [Google Scholar] [CrossRef]
- Wang, H.; Zhao, S.; Liu, R.; Zhou, W.; Jin, J.Y. Changes of photosynthetic activities of maize (Zea mays L.) seedlings in response to cadmium stress. Photosynthetica 2009, 47, 277–283. [Google Scholar] [CrossRef]
- Aslam, M.; Aslam, A.; Sheraz, M.; Ali, B.; Ulhassan, Z.; Najeeb, U.; Zhou, W.; Gill, R.A. Lead toxicity in cereals: Mechanistic insight into toxicity, mode of action, and management. Front. Plant Sci. 2021, 11, 587785. [Google Scholar] [CrossRef] [PubMed]
- Khanum, S.; Al Tawaha, A.R.M.; Al-Tawaha, A.R.; Abusalem, M.; Rauf, A.; Karnwal, A.; Dey, A.; Shatnawi, M.; Thangadurai, D.; Sangeetha, J.; et al. Cereal physiology, flowering, and grain yield under abiotic stress imposed by different heavy metals. In Omics Approach to Manage Abiotic Stress in Cereals; Roychoudhury, A., Aftab, T., Acharya, K., Eds.; Springer: Singapore, 2022; pp. 37–46. [Google Scholar]
- Ahmad, I.; Tahir, M.; Daraz, U.; Ditta, A.; Hussain, M.B.; Khan, Z.U.H. Responses and tolerance of cereal crops to metal and metalloid toxicity. In Agronomic Crops; Hasanuzzaman, M., Ed.; Springer: Singapore, 2020; pp. 235–264. [Google Scholar]
- Giri, S.; Mahato, M.K.; Bhattacharjee, S.; Singh, A.K. Development of a new noncarcinogenic heavy metal pollution index for quality ranking of vegetable, rice, and milk. Ecol. Indic. 2020, 113, 106214. [Google Scholar] [CrossRef]
- Sohail, M.I.; Rehman, M.Z.; Aziz, T.; Akmal, F.; Azhar, M.; Nadeem, F.; Aslam, M.; Siddiqui, A.; Khalid, M.A. Iron bio-fortification and heavy metal/(loid)s contamination in cereals: Successes, issues, and challenges. Crop Pasture Sci. 2022, 73, 877–895. [Google Scholar] [CrossRef]
- Pandey, R.; Vengavasi, K.; Hawkesford, M.J. Plant adaptation to nutrient stress. Plant Physiol. Rep. 2021, 26, 583–586. [Google Scholar] [CrossRef]
- Li, Y.; Rahman, S.U.; Qiu, Z.; Shahzad, S.M.; Nawaz, M.F.; Huang, J.; Naveed, S.; Li, L.; Wang, X.; Cheng, H. Toxic effects of cadmium on the physiological and biochemical attributes of plants, and phytoremediation strategies: A review. Environ. Pollut. 2023, 325, 121433. [Google Scholar] [CrossRef] [PubMed]
- Raza, A.; Tabassum, J.; Zahid, Z.; Charagh, S.; Bashir, S.; Barmukh, R.; Khan, R.S.A.; Barbosa, F., Jr.; Zhang, C.; Chen, H.; et al. Advances in “omics” approaches for improving toxic metals/metalloids tolerance in plants. Front. Plant Sci. 2022, 12, 794373. [Google Scholar] [CrossRef] [PubMed]
- Hussain, B.; Umer, M.J.; Li, J.; Ma, Y.; Abbas, Y.; Ashraf, M.N.; Tahir, N.; Ullah, A.; Gogoi, N.; Farook, M. Strategies for reducing cadmium accumulation in rice grains. J. Clean. Prod. 2021, 286, 125557. [Google Scholar] [CrossRef]
- Hussain, I.; Afzal, S.; Ashraf, M.A.; Rasheed, R.; Saleem, M.H.; Alatawi, A.; Ameen, F.; Fahad, S. Effect of metals or trace elements on wheat growth and its remediation in contaminated soil. J. Plant Growth Regul. 2023, 42, 2258–2282. [Google Scholar] [CrossRef]
- Mourad, A.M.I.; Eltaher, S.; Börner, A.; Sallam, A. Unlocking the genetic control of spring wheat kernel traits under normal and heavy metals stress conditions. Plant Soil 2023, 484, 257–278. [Google Scholar] [CrossRef]
- Kumar, S.; Shah, S.H.; Vimala, Y.; Jatav, H.S.; Ahmad, P.; Chen, Y.; Siddique, K.H.M. Abscisic acid: Metabolism, transport, crosstalk with other plant growth regulators, and its role in heavy metal stress mitigation. Front. Plant Sci. 2022, 13, 972856. [Google Scholar] [CrossRef] [PubMed]
- Farook, M.U.; Ishaaq, I.; Barutcular, C.; Skalicky, M.; Maqbool, R.; Rastogi, A.; Hussain, S.; Allakhverdiev, S.I.; Zhu, J. Mitigation effects of selenium on accumulation of cadmium and morpho-physiological properties in rice varieties. Plant Physiol. Biochem. 2022, 170, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Saha, B.; Chowardhara, B.; Kar, S.; Devi, S.S.; Awasthi, J.P.; Moulick, D.; Tanti, B.; Panda, S.K. Advances in heavy metal-induced stress alleviation with respect to exogenous amendments in crop plants. In Priming and Pretreatment of Seeds and Seedlings; Hasanuzzaman, M., Fotopoulos, V., Eds.; Springer: Singapore, 2019; pp. 313–332. [Google Scholar]
- Rahman, S.U.; Nawaz, M.F.; Gul, S.; Yasin, G.; Hussain, B.; Li, Y.; Cheng, H. State-of-the-art OMICS strategies against toxic effects of heavy metals in plants: A review. Ecotoxicol. Environ. Saf. 2022, 242, 113952. [Google Scholar] [CrossRef] [PubMed]
- Feng, Z.; Ji, S.; Ping, J.; Cui, D. Recent advances in metabolomics for studying heavy metal stress in plants. TrAC Trends Anal. Chem. 2021, 143, 116402. [Google Scholar] [CrossRef]
- Charles, R.; Jolliet, O.; Gaillard, G.; Pellet, D. Environmental analysis of intensity level in wheat crop production using life cycle assessment. Agric. Ecosyst. Environ. 2006, 113, 216–225. [Google Scholar] [CrossRef]
- Arif, N.; Sharma, N.C.; Yadav, V.; Ramawat, N.; Dubey, N.K.; Tripathi, D.K.; Chauhan, D.K.; Sahi, S. Understanding heavy metal stress in a rice crop: Toxicity, tolerance mechanisms, and amelioration strategies. J. Plant Biol. 2019, 62, 239–253. [Google Scholar] [CrossRef]
- Ramzani, P.M.A.; Khan, W.D.; Iqbal, M.; Kausar, S.; Ali, S.; Rizwan, M.; Virk, Z.A. Effect of different amendments on rice (Oryza sativa L.) growth, yield, nutrient uptake and grain quality in Ni-contaminated soil. Environ. Sci. Pollut. Res. 2016, 23, 18585–18595. [Google Scholar] [CrossRef]
- Hagemeyer, J. Ecophysiology of plant growth under heavy metal stress. In Heavy Metal Stress in Plants; Prasad, M.N.V., Hagemeyer, J., Eds.; Springer: Berlin/Heidelberg, Germany, 1999; pp. 157–181. [Google Scholar]
- Boudjabi, S.; Chenchouni, H. Comparative effectiveness of exogenous organic amendments on soil fertility, growth, photosynthesis and heavy metal accumulation in cereal crops. Heliyon 2023, 9, e14615. [Google Scholar] [CrossRef]
- Farrag, K.; Senesi, N.; Nigro, F.; Petrozza, A.; Palma, A.; Shaarawi, S.; Brunetti, G. Growth responses of crop and weed species to heavy metals in pot and field experiments. Environ. Sci. Pollut. Res. 2012, 19, 3636–3644. [Google Scholar] [CrossRef]
- Ahmad, I.; Akhtar, M.J.; Zahir, Z.A.; Mitter, B. Organic amendments: Effects on cereals growth and cadmium remediation. Int. J. Environ. Sci. Technol. 2015, 12, 2919–2928. [Google Scholar] [CrossRef]
- Wang, F.; Wang, Z.; Kou, C.; Ma, Z.; Zhao, D. Responses of wheat yield, macro- and micro-nutrients, and heavy metals in soil and wheat following the application of manure compost on the North China Plain. PLoS ONE 2016, 11, e0146453. [Google Scholar] [CrossRef] [PubMed]
- Huamain, C.; Chunrong, Z.; Cong, T.; Yongguan, Z. Heavy metal pollution in soils in China: Status and countermeasures. Ambio 1999, 28, 130–134. [Google Scholar]
- Rizvi, A.; Ahmed, B.; Khan, M.S.; Rajput, V.D.; Umar, S.; Minkina, T.; Lee, J. Maize associated bacterial microbiome linked mitigation of heavy metal stress: A multidimensional detoxification approach. Environ. Exp. Bot. 2022, 200, 104911. [Google Scholar] [CrossRef]
- Tavarez, M.; Grusak, M.A.; Sankaran, R.R. Effects of zinc fertilization on grain cadmium accumulation, gene expression, and essential mineral partitioning in rice. Agronomy 2022, 12, 2182. [Google Scholar] [CrossRef]
- Bhardwaj, I.; Garg, N. Cereals and phytohormones under heavy metal stress. In Sustainable Remedies for Abiotic Stress in Cereals; Abdel Latef, A.A.H., Ed.; Springer: Singapore, 2022; pp. 369–393. [Google Scholar]
- Das, R.; Biswas, S. Influence of abiotic stresses on seed production and quality. In Seed Biology Updates; Jimenez-Lopez, J.C., Ed.; IntechOpen: Rijeka, Croatia, 2022; pp. 1–24. [Google Scholar]
- Halford, N.G.; Curtis, T.Y.; Chen, Z.; Huang, J. Effects of abiotic stress and crop management on cereal grain composition: Implications for food quality and safety. J. Exp. Bot. 2015, 66, 1145–1156. [Google Scholar] [CrossRef] [PubMed]
- Hou, D.; O’Connor, D.; Igalavithana, A.D.; Alessi, D.S.; Luo, J.; Tsang, D.C.W.; Sparks, D.L.; Yamauchi, Y.; Rinklebe, J.; Ok, J.S. Metal contamination and bioremediation of agricultural soils for food safety and sustainability. Nat. Rev. Earth Environ. 2020, 1, 366–381. [Google Scholar] [CrossRef]
- Wei, J.; Cen, K. Contamination and health risk assessment of heavy metals in cereals, legumes, and their products: A case study based on the dietary structure of the residents of Beijing, China. J. Clean. Prod. 2020, 260, 121001. [Google Scholar] [CrossRef]
- Nugent, A.P.; Thielecke, F. Wholegrains and health: Many benefits but do contaminants pose any risk? Nutr. Bull. 2019, 44, 107–115. [Google Scholar] [CrossRef]
- Tong, C.; Hill, C.B.; Zhou, G.; Zhang, X.-Q.; Jia, Y.; Li, C. Opportunities for improving waterlogging tolerance in cereal crops—Physiological traits and genetic mechanisms. Plants 2021, 10, 1560. [Google Scholar] [CrossRef]
- Gholizadeh, A.; Borůvka, L.; Saberioon, M.M.; Kozák, J.; Vašát, R.; Němeček, K. Comparing different data preprocessing methods for monitoring soil heavy metals based on soil spectral features. Soil Water Res. 2015, 10, 218–227. [Google Scholar] [CrossRef]
- Liu, B.; Ai, S.; Zhang, W.; Huang, D.; Zhang, Y. Assessment of the bioavailability, bioaccessibility and transfer of heavy metals in the soil-grain-human systems near a mining and smelting area in NW China. Sci. Total Environ. 2017, 609, 822–829. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.-h.; Zhu, G.-y.; Li, H.-l.; Han, X.-m.; Li, J.-m. Accumulation and bioavailability of heavy metals in a soil-wheat/maize system with long-term sewage sludge amendments. J. Integr. Agric. 2017, 17, 1861–1870. [Google Scholar] [CrossRef]
- Kim, R.Y.; Yoon, J.K.; Kim, T.S.; Yang, J.E.; Owens, G.; Kim, K.-R. Bioavailability of heavy metals in soils: Definitions and practical implementation—A critical review. Environ. Geochem. Health 2015, 37, 1041–1061. [Google Scholar] [CrossRef] [PubMed]
- Hlihor, R.M.; Apostol, L.C.; Smaranda, C.; Pavel, L.V.; Caliman, F.A.; Robu, B.M.; Gavrilescu, M. Bioavailability processes for contaminants in soils and their use in risk assessment. Environ. Eng. Manag. J. 2009, 8, 1199–1206. [Google Scholar]
- Feszterová, M.; Porubcová, L.; Tirpáková, A. The monitoring of selected heavy metals content and bioavailability in the soil-plant system and its impact on sustainability in agribusiness food chains. Sustainability 2021, 13, 7021. [Google Scholar] [CrossRef]
- Aziz, R.; Rafiq, M.T.; Li, T.; Liu, D.; He, Z.; Stoffella, P.J.; Sun, K.; Xiaoe, Y. Uptake of cadmium by rice grown on contaminated soils and its bioavailability/toxicity in human cell lines (Caco-2/HL-7702). J. Agric. Food Chem. 2015, 63, 3599–3608. [Google Scholar] [CrossRef]
- Aslam, M.M.; Okal, E.J.; Waseem, M. Cadmium toxicity impacts plant growth and plant remediation strategies. Plant Growth Regul. 2023, 99, 397–412. [Google Scholar] [CrossRef]
- Murtaza, G.; Usman, Y.; Niazi, N.K.; Usman, M.; Hussain, T. Bioaccumulation of potentially toxic elements in cereal and legume crops: A review. Clean 2017, 45, 1700548. [Google Scholar] [CrossRef]
- Thielecke, F.; Nugent, A.P. Contaminants in grain—A major risk for whole grain safety? Nutrients 2018, 10, 1213. [Google Scholar] [CrossRef]
- Chandravanshi, L.; Shiv, K.; Kumar, S. Developmental toxicity of cadmium in infants and children: A review. Environ. Anal. Health Toxicol. 2021, 36, e2021003. [Google Scholar] [CrossRef] [PubMed]
- Matovic, V.; Buha, A.; Dukic-Cosic, D.; Bulat, Z. Insight into the oxidative stress induced by lead and/or cadmium in blood, liver and kidneys. Food Chem. Toxicol. 2015, 78, 130–140. [Google Scholar] [CrossRef] [PubMed]
- Rana, M.N.; Tangpong, J.; Rahman, M.M. Toxicodynamics of lead, cadmium, mercury and arsenic- induced kidney toxicity and treatment strategy: A mini review. Toxicol. Rep. 2018, 5, 704–713. [Google Scholar] [CrossRef] [PubMed]
- Suhani, I.; Sahab, S.; Srivastava, V.; Singh, R.P. Impact of cadmium pollution on food safety and human health. Curr. Opin. Toxicol. 2021, 27, 1–7. [Google Scholar] [CrossRef]
- Ramon, F.; Lull, C. Legal measures to prevent and manage soil contamination and to increase food safety for consumer health: The case of Spain. Environ. Pollut. 2019, 250, 883–891. [Google Scholar] [CrossRef] [PubMed]
- Song, T.; Das, D.; Hu, Q.; Yang, F.; Zhang, J. Alternate wetting and drying irrigation and phosphorus rates affect grain yield and quality and heavy metal accumulation in rice. Sci. Total Environ. 2021, 752, 141862. [Google Scholar] [CrossRef] [PubMed]
- Gharibzahedi, S.M.T.; Jafari, S.M. The importance of minerals in human nutrition: Bioavailability, food fortification, processing effects and nanoencapsulation. Trends Food Sci. Technol. 2017, 62, 119–132. [Google Scholar] [CrossRef]
- Murtaza, M.; Javed, W.; Hussain, A.; Qadir, M.; Aslam, M. Soil-applied zinc and copper suppress cadmium uptake and improve the performance of cereals and legumes. Int. J. Phytoremediat. 2017, 19, 199–206. [Google Scholar] [CrossRef]
- Zhao, F.J.; Tang, Z.; Song, J.J.; Huang, X.Y.; Wang, P. Toxic metals and metalloids: Uptake, transport, detoxification, phytoremediation, and crop improvement for safer food. Mol. Plant 2022, 15, 27–44. [Google Scholar] [CrossRef]
- Yang, Y.; Li, Y.; Chen, W.; Wang, M.; Wang, T.; Dai, Y. Dynamic interactions between soil cadmium and zinc affect cadmium phytoavailability to rice and wheat: Regional investigation and risk modeling. Environ. Pollut. 2020, 267, 115613. [Google Scholar] [CrossRef]
- Chaney, R.L. How does contamination of rice soils with Cd and Zn cause high incidence of human Cd disease in subsistence rice farmers. Curr. Pollut. Rep. 2015, 1, 13–22. [Google Scholar] [CrossRef]
- Du, J.; Zeng, J.; Ming, X.; He, Q.; Tao, Q.; Jiang, M.; Gao, S.; Li, X.; Lei, T.; Pan, Y.; et al. The presence of zinc reduced cadmium uptake and translocation in Cosmos bipinnatus seedlings under cadmium/zinc combined stress. Plant Physiol. Biochem. 2020, 151, 223–232. [Google Scholar] [CrossRef] [PubMed]
- Prasad, M.N.V. Heavy Metal Stress in Plants: From Biomolecules to Ecosystems, 2nd ed.; Springer: Berlin/Heidelberg, Germany, 2004. [Google Scholar]
- Hamid, Y.; Tang, L.; Sohail, M.I.; Cao, X.; Hussain, B.; Aziz, M.Z.; Usman, M.; He, Z.-l.; Yang, X. An explanation of soil amendments to reduce cadmium phytoavailability and transfer to food chain. Sci. Total Environ. 2019, 660, 80–96. [Google Scholar] [CrossRef] [PubMed]
- Diacono, M.; Montemurro, F. Long-term effects of organic amendments on soil fertility. In Sustainable Agriculture; Lichtfouse, E., Hamelin, M., Navarrete, M., Debaeke, P., Eds.; Springer: Dordrecht, Germany, 2011; Volume 2, pp. 761–786. [Google Scholar]
- Diacono, M.; Montemurro, F. Olive pomace compost in organic emmer crop: Yield, soil properties, and heavy metals’ fate in plant and soil. J. Soil Sci. Plant Nutr. 2019, 19, 63–70. [Google Scholar] [CrossRef]
- Kärenlampi, S.; Schat, H.; Vangronsveld, J.; Verkleij, J.A.C.; van der Lelie, D.; Mergeay, M.; Tervahauta, A.I. Genetic engineering in the improvement of plants for phytoremediation of metal polluted soils. Environ. Pollut. 2000, 107, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Saeed, M.; Quiraishi, U.M.; Malik, R.N. Advancement in mitigating the effects of heavy metal toxicity in wheat. In Abiotic Stresses in Wheat. Unfolding the Challenges; Khan, M.K., Pandey, A., Hamurcu, M., Gupta, O.P., Gezin, S., Eds.; Elsevier-Academic Press: London, UK, 2023; pp. 313–327. [Google Scholar]
- Sharma, J.K.; Kumar, N.; Singh, N.P.; Santal, A.R. Phytoremediation technologies and their mechanism for removal of heavy metal from contaminated soil: An approach for a sustainable environment. Front. Plant Sci. 2023, 14, 1076876. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Wang, W.; Carswell, A.; Misselbrook, T.; Shen, J.; Han, J. Fate and transfer of heavy metals following repeated biogas slurry application in a rice-wheat crop rotation. J. Environ. Manag. 2020, 270, 110938. [Google Scholar] [CrossRef] [PubMed]
- Tang, L.; Luo, W.J.; Chen, W.K.; He, Z.L.; Gurajala, H.K.; Hamid, Y.; Deng, M.H.; Yang, X.E. Field crops (Ipomoea aquatica Forsk. and Brassica chinensis L.) for phytoremediation of cadmium and nitrate co-contaminated soils via rotation with Sedum alfredii Hance. Environ. Sci. Pollut. Res. 2017, 24, 19293–19305. [Google Scholar] [CrossRef]
- Rehman, A.; Farooq, M.; Ozturk, L.; Asif, M.; Siddique, K.H.M. Zinc nutrition in wheat-based cropping systems. Plant Soil 2018, 422, 283–315. [Google Scholar] [CrossRef]
- Sánchez-Navarro, A.; de Carmenl Salas-Sanjuan, M.; Blanco-Bernardeau, M.A.; Sánchez-Romero, J.A.; Delgado-Iniesta, M.J. Medium-term effect of organic amendments on the chemical properties of a soil used for vegetable cultivation with cereal and legume rotation in a semiarid climate. Land 2023, 12, 897. [Google Scholar] [CrossRef]
- Witkowska, D.; Słowik, J.; Chilicka, K. Heavy metals and human health: Possible exposure pathways and the competition for protein binding sites. Molecules 2021, 26, 6060. [Google Scholar] [CrossRef] [PubMed]
- Balali-Mood, M.; Naseri, K.; Tahergorabi, Z.; Khazdair, M.R.; Sadeghi, M. Toxic mechanisms of five heavy metals: Mercury, Lead, Chromium, Cadmium, and Arsenic. Front. Pharmacol. 2021, 12, 643972. [Google Scholar] [CrossRef] [PubMed]
- Yadav, S.; Modi, P.; Dave, A.; Vijapura, A.; Patel, D.; Patel, M. Effect of abiotic stress on crops. In Sustainable Crop Production; Hasanuzzaman, M., Fujita, M., Filho, M.C.M.T., Nogueira, T.A.R., Galindo, F.S., Eds.; IntechOpen: Rijeka, Croatia, 2020; pp. 1–21. [Google Scholar]
- Swami, P.; Munjal, R.; Deswal, K. Targeting photosynthesis under abiotic stress. J. Cereal Res. 2021, 14, 53–66. [Google Scholar] [CrossRef]
- Maurya, A.M.; Sinha, D.; Kamzakshi; Mukherjee, S. Plant response to heavy metals (at the cellular level). In Heavy Metals in Plants: Physiological to Molecular Approach; Kumar, J., Gaur, S., Srivastava, P.K., Mishra, R.K., Prasad, S.M., Chauhan, D.K., Eds.; CRC Press: Boca Raton, FL, USA, 2022; pp. 125–148. [Google Scholar]
- Sharma, R.K.; Agrawal, M. Biological effects of heavy metals: An overview. J. Environ. Biol. 2005, 26, 301–313. [Google Scholar] [PubMed]
- Khan, A.; Khan, S.; Khan, M.A.; Qamar, Z.; Waqas, M. The uptake and bioaccumulation of heavy metals by food plants, their effects on plants nutrients, and associated health risk: A review. Environ. Sci. Pollut. Res. 2015, 22, 13772–13799. [Google Scholar] [CrossRef] [PubMed]
- Qaswar, M.; Hussain, S.; Rengel, Z. Zinc fertilisation increases grain zinc and reduces grain lead and cadmium concentrations more in zinc-biofortified than standard wheat cultivar. Sci. Total Environ. 2017, 605–606, 454–460. [Google Scholar] [CrossRef] [PubMed]
- Sperdouli, I. Heavy metal toxicity effects on plants. Toxics 2022, 10, 715. [Google Scholar] [CrossRef]
- Ejaz, U.; Khan, S.M.; Khalid, N.; Ahmad, Z.; Jehangir, S.; Fatima Rizvi, Z.; Lho, L.H.; Han, H.; Raposo, A. Detoxifying theheavy metals: A multipronged study of tolerance strategies against heavy metals toxicity in plants. Front. Plant Sci. 2023, 14, 1154571. [Google Scholar] [CrossRef]
- Gupta, D.K.; Pena, L.B.; Romero-Puertas, M.C.; Hernández, A.; Inouhe, M.; Sandalio, L.M. NADPH oxidases differentially regulate ROS metabolism and nutrient uptake under cadmium toxicity. Plant Cell Environ. 2016, 40, 509–526. [Google Scholar] [CrossRef]
- Mei, S.; Lin, K.; Williams, D.V.; Liu, Y.; Dai, H.; Cao, F. Cadmium accumulation in cereal crops and tobacco: A review. Agronomy 2022, 12, 1952. [Google Scholar] [CrossRef]
- Tabassum; Jeena, A.S.; Pandey, D. Metal induced genotoxicity and oxidative stress in plants, assessment methods, and role of various factors in genotoxicity regulation. In Induced Genotoxicity and Oxidative Stress in Plants; Khan, Z., Ansari, M.Y.K., Shahwar, D., Eds.; Springer: Singapore, 2021; pp. 133–149. [Google Scholar]
- Jena, A.B.; Samal, R.R.; Bhol, N.K.; Duttaroy, A.K. Cellular Red-Ox system in health and disease: The latest update. Biomed. Pharmacother. 2013, 162, 114606. [Google Scholar] [CrossRef] [PubMed]
- Page, V.; Feller, U. Heavy metals in crop plants: Transport and redistribution processes on the whole plant level. Agronomy 2015, 5, 447–463. [Google Scholar] [CrossRef]
- Asad, S.A.; Farooq, M.; Afzal, A.; West, H. Integrated phytobial heavy metal remediation strategies for a sustainable clean environment—A review. Chemosphere 2019, 217, 925–941. [Google Scholar] [CrossRef] [PubMed]
- Elango, D.; Devi, K.D.; Jeyabalakrishnan, H.K.; Rajendran, K.; Haridass, V.K.T.; Dharmaraj, D.; Charuchandran, C.V.; Charuchandran, C.V.; Wang, W.; Fakude, M.; et al. Agronomic, breeding, and biotechnological interventions to mitigate heavy metal toxicity problems in agriculture. Agric. Food Res. 2022, 10, 100374. [Google Scholar] [CrossRef]
- Chandwani, S.; Amaresan, N. Role of ACC deaminase producing bacteria for abiotic stress management and sustainable agriculture production. Environ. Sci. Pollut. Res. 2022, 29, 22843–22859. [Google Scholar] [CrossRef] [PubMed]
- Hasanuzzaman, M.; Nahar, K.; Rahman, A.; Al Mahmud, J.; Alharby, H.F.; Fujita, M. Exogenous glutathione attenuates lead-induced oxidative stress in wheat by improving antioxidant defense and physiological mechanisms. J. Plant Interact. 2018, 13, 203–212. [Google Scholar] [CrossRef]
- Sies, H. Oxidative stress: Concept and some practical aspects. Antioxidants 2020, 9, 852. [Google Scholar] [CrossRef] [PubMed]
- Hejna, M.; Gottardo, D.; Baldi, A.; Dell’Orto, V.; Cheli, F.; Zaninelli, M.; Rossi, L. Review: Nutritional ecology of heavy metals. Animal 2018, 12, 2156–2170. [Google Scholar] [CrossRef]
- Yaashikaa, P.R.; Kumar, P.S.; Jeevanantham, S.; Saravanan, R. A review on bioremediation approach for heavy metal detoxification and accumulation in plants. Environ. Pollut. 2022, 301, 119035. [Google Scholar] [CrossRef]
- Roosta, H.R.; Estaji, A.; Niknam, F. Effect of iron, zinc and manganese shortage-induced change on photosynthetic pigments, some osmoregulators and chlorophyll fluorescence parameters in lettuce. Photosynthetica 2018, 56, 606–615. [Google Scholar] [CrossRef]
- Halmemies-Beauchet-Filleau, A.; Rinne, M.; Lamminen, M.; Mapato, C.; Ampapon, T. Review: Alternative and novel feeds for ruminants: Nutritive value, product quality and environmental aspects. Animal 2018, 12, 295–309. [Google Scholar] [CrossRef]
- Giri, V.P.; Shukla, P.; Tripathi, A.; Verma, P.; Kumar, N.; Pandey, S.; Dimkpa, C.O.; Mishra, A. A review of sustainable use of biogenic nanoscale agro-materials to enhance stress tolerance and nutritional value of plants. Plants 2023, 12, 815. [Google Scholar] [CrossRef] [PubMed]
- Ercal, N.; Gurer-Orhan, H.; Aykin-Burns, N. Toxic metals and oxidative stress part I: Mechanisms involved in metal-induced oxidative damage. Curr. Top. Med. Chem. 2001, 1, 529–539. [Google Scholar] [CrossRef] [PubMed]
- Roychoudhury, A.; Chakraborty, S. Cellular and molecular phytotoxicity of lead and mercury. In Cellular and Molecular Phytotoxicity of Heavy Metals. Nanotechnology in the Life Sciences; Faisal, M., Saquib, Q., Alatar, A.A., Al-Khedhairy, A.A., Eds.; Springer: Cham, The Netherlands, 2020; pp. 373–387. [Google Scholar]
- Al Mahmud, J.; Bhuyan, M.H.M.B.; Anee, T.I.; Nahar, K.; Fujita, M.; Hasanuzzaman, M. Reactive oxygen species metabolism and antioxidant defense in plants under metal/metalloid stress. In Plant Abiotic Stress Tolerance; Hasanuzzaman, M., Hakeem, K., Nahar, K., Alharby, H., Eds.; Springer: Cham, Switzerland, 2019; pp. 221–257. [Google Scholar]
- Pais, I.P.; Moreira, R.; Semedo, J.N.; Ramalho, J.C.; Lidon, F.C.; Coutinho, J.; Maçãs, B.; Scotti-Campos, P. Wheat crop under waterlogging: Potential soil and plant effects. Plants 2023, 12, 149. [Google Scholar] [CrossRef] [PubMed]
- Nadeem, F.; Farooq, M. Application of micronutrients in rice-wheat cropping system of South Asia. Rice Sci. 2019, 26, 356–371. [Google Scholar] [CrossRef]
- Huang, B.; Liao, Q.; Fu, H.; Ye, Z.; Mao, Y.; Luo, J.; Wang, Y.; Yuan, H.; Xin, J. Effect of potassium intake on cadmium transporters and root cell wall biosynthesis in sweet potato. Ecotoxicol. Environ. Saf. 2023, 250, 114501. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Duan, S.; Wu, Q.; Yu, M.; Shabala, S. Reducing cadmium accumulation in plants: Structure–function relations and tissue-specific operation of transporters in the spotlight. Plants 2020, 9, 223. [Google Scholar] [CrossRef] [PubMed]
- Hoque, M.N.; Tahjib-Ul-Arif, M.; Hannan, A.; Sultana, N.; Akhter, S.; Hasanuzzaman, M.; Akter, F.; Hossain, M.S.; Sayed, M.A.; Hasan, M.T.; et al. Melatonin modulates plant tolerance to heavy metal stress: Morphological responses to molecular mechanisms. Int. J. Mol. Sci. 2021, 22, 11445. [Google Scholar] [CrossRef] [PubMed]
- Goncharuk, E.A.; Zagoskina, N.V. Heavy Metals, Their phytotoxicity, and the role of phenolic antioxidants in plant stress responses with focus on cadmium: Review. Molecules 2023, 28, 3921. [Google Scholar] [CrossRef]
- Yoneyama, T. Iron delivery to the growing leaves associated with leaf chlorosis in mugineic acid family phytosiderophores-generating graminaceous crops. Soil Sci. Plant Nutr. 2021, 67, 415–426. [Google Scholar] [CrossRef]
- Asopa, P.P.; Bhatt, R.; Sihag, S.; Lothary, S.G.; Kachhwaha, S. Effect of cadmium on physiological parameters of cereal and millet plants—A comparative study. Int. J. Phytoremediat. 2017, 19, 225–230. [Google Scholar] [CrossRef] [PubMed]
- Chandwani, S.; Kayasth, R.; Naik, H.; Amaresan, N. Current status and future prospect of managing lead (Pb) stress through microbes for sustainable agriculture. Environ. Monit. Assess. 2023, 195, 479. [Google Scholar] [CrossRef] [PubMed]
- Bechtaoui, N.; Rabiu, M.K.; Raklami, A.; Oufdou, K.; Hafidi, M.; Jemo, M. Phosphate-dependent regulation of growth and stresses management in plants. Front. Plant Sci. 2021, 12, 679916. [Google Scholar] [CrossRef] [PubMed]
- Emamverdian, A.; Ding, Y.; Mokhberdoran, F.; Xie, Y. Heavy metal stress and some mechanisms of plant defense response. Sci. World J. 2015, 2015, 756120. [Google Scholar] [CrossRef] [PubMed]
- Ganguly, R.; Sarkar, A.; Dasgupta, D.; Acharya, K.; Keswani, C.; Popova, V.; Minkina, T.; Maksimov, A.Y.; Chakraborty, N. Unravelling the efficient applications of zinc and selenium for mitigation of abiotic stresses in plants. Agriculture 2022, 12, 1551. [Google Scholar] [CrossRef]
- Armienta, M.A.; Beltrán, M.; Martínez, S.; Labastida, I. Heavy metal assimilation in maize (Zea mays L.) plants growing near mine tailings. Environ. Geochem. Health 2020, 42, 2361–2375. [Google Scholar] [CrossRef] [PubMed]
- Gill, R.A.; Kanwar, M.K.; dos Reis, A.R.; Ali, B. Editorial: Heavy metal toxicity in plants: Recent insights on physiological and molecular aspects. Front. Plant Sci. 2022, 12, 830682. [Google Scholar] [CrossRef] [PubMed]
- Khoudi, H. Significance of vacuolar proton pumps and metal/H+ antiporters in plant heavy metal tolerance. Physiol. Plant. 2021, 173, 384–393. [Google Scholar] [CrossRef]
- Bücker-Neto, L.; Sobral Paiva, A.L.; Machado, R.D.; Arenhart, R.A.; Margis-Pinheiro, M. Interactions between plant hormones and heavy metals responses. Genet. Mol. Biol. 2017, 40, 373–386. [Google Scholar] [CrossRef]
- Westfall, C.S.; Muehler, A.M.; Jez, J.M. Enzyme action in the regulation of plant hormone responses. J. Biol. Chem. 2013, 288, 19304–19311. [Google Scholar] [CrossRef]
- Angulo-Bejarano, P.I.; Puente-Rivera, J.; Cruz-Ortega, R. Metal and metalloid toxicity in plants: An overview on molecular aspects. Plants 2021, 10, 635. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, T.; Islam, K.R.; Muhammad, S. Toxic effects of heavy metals on early growth and tolerance of cereal crops. Pak. J. Bot. 2007, 39, 451–462. [Google Scholar]
- Bengough, A.G.; McKenzie, B.M.; Hallett, P.D.; Valentine, T.A. Root elongation, water stress, and mechanical impedance: A review of limiting stresses and beneficial root tip traits. J. Exp. Bot. 2011, 62, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Caracciolo, A.B.; Terenzi, V. Rhizosphere microbial communities and heavy metals. Microorganisms 2021, 9, 462. [Google Scholar] [CrossRef]
- Singh, S.; Parihar, P.; Singh, R.; Singh, V.P.; Prasad, S.M. Heavy metal tolerance in plants: Role of transcriptomics, proteomics, metabolomics, and ionomics. Front. Plant Sci. 2015, 6, 1143. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.U.; Nawaz, M.; Mahmood, A.; Shah, A.A.; Shah, A.N.; Muhammad, F.; Batool, M.; Rasheed, A.; Jaremko, M.; Abdelsalam, N.R.; et al. The role of zinc to mitigate heavy metals toxicity in crops. Front. Environ. Sci. 2022, 10, 990223. [Google Scholar] [CrossRef]
- Hacisalihoglu, G.; Kochian, L.V. How do some plants tolerate low levels of soil zinc? Mechanisms of zinc efficiency in crop plants. New Phytol. 2003, 159, 341–350. [Google Scholar] [CrossRef]
- Hu, J.; Tao, R.; Cao, C.; Xie, J.; Gao, Y.; Hu, H.; Ma, Z.; Ma, Y. Effect of Leaf Surface Regulation of Zinc Fertilizer on Absorption of Cadmium, Plumbum and Zinc in Rice (Oryza sativa L.). Sustainability 2023, 15, 1877. [Google Scholar] [CrossRef]
- Tank, Y.; Zhang, J.; Wang, L.; Wang, H.; Long, H.; Yang, L.; Li, G.; Guo, J.; Wang, Y.; Li, Y.; et al. Water deficit aggravated the inhibition of photosynthetic performance of maize under mercury stress but is alleviated by brassinosteroids. J. Hazard. Mater. 2023, 443, 130365. [Google Scholar] [CrossRef]
- Siposova, K.; Labancova, E.; Hackulicova, D.; Kollarova, K.; Vivodova, Z. The changes in the maize root cell walls after exogenous application of auxin in the presence of cadmium. Environ. Sci. Pollut. Res. 2023, 30, 87102–87117. [Google Scholar] [CrossRef]
- Flood, J. The importance of plant health to food security. Food Sec. 2010, 2, 215–231. [Google Scholar] [CrossRef]
- Singh, A.; Prasad, S.M. Reduction of heavy metal load in food chain: Technology assessment. Rev. Environ. Sci. Biotechnol. 2011, 10, 199–214. [Google Scholar] [CrossRef]
- Shahid, M.; Khalid, S.; Abbas, G.; Shahid, N.; Nadeem, M.; Sabir, M.; Aslam, M.; Dumat, C. Heavy metal stress and crop productivity. In Crop Production and Global Environmental Issues; Hakeem, K., Ed.; Springer: Cham, Switzerland, 2015; pp. 1–25. [Google Scholar]
- Ashraf, U.; Tang, X. Yield and quality responses, plant metabolism and metal distribution pattern in aromatic rice under lead (Pb) toxicity. Chemosphere 2017, 176, 141–155. [Google Scholar] [CrossRef] [PubMed]
- Shah, V.; Daverey, A. Phytoremediation: A multidisciplinary approach to clean up heavy metal contaminated soil. Environ. Technol. Innov. 2020, 18, 100774. [Google Scholar] [CrossRef]
- Kosakivska, I.V.; Vedenicheva, N.P.; Babenko, L.M.; Voytenko, L.V.; Romanenko, K.O.; Vasyuk, V.A. Exogenous phytohormones in the regulation of growth and development of cereals under abiotic stresses. Mol. Biol. Rep. 2022, 49, 617–628. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Chen, J.; Ma, Y.; Huang, M.; Qiu, T.; Bian, H.; Han, N.; Wang, J. Function, Mechanism, and application of plant melatonin: An update with a focus on the cereal crop, barley (Hordeum vulgare L.). Antioxidants 2022, 11, 634. [Google Scholar] [CrossRef] [PubMed]
- Fahad, S.; Nie, L.; Chen, Y.; Wu, C.; Xiong, D.; Saud, S.; Hongyan, L.; Cui, L.; Huang, J. Crop plant hormones and environmental stress. In Sustainable Agriculture Reviews; Lichtfouse, E., Ed.; Springer: Cham, Switzerland, 2015; Volume 15, pp. 371–400. [Google Scholar]
- Groszmann, M.; Chandler, P.M.; Ross, J.J.; Swain, S.M. Manipulating gibberellin control over growth and fertility as a possible target for managing wild radish weed populations in cropping systems. Front. Plant Sci. 2020, 11, 190. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Kapoor, D.; Gautam, S.; Landi, M.; Kandhol, N.; Araniti, F.; Ramakrishnan, M.; Satish, L.; Singh, V.P.; Sharma, P.; et al. Heavy metal induced regulation of plant biology: Recent insights. Physiol. Plant. 2022, 174, e13688. [Google Scholar] [CrossRef]
- Abhinandan, K.; Skori, L.; Stanic, M.; Hickerson, N.M.N.; Jamshed, M.; Samuel, M.A. Abiotic stress signaling in wheat—An inclusive overview of hormonal interactions during abiotic stress responses in wheat. Front. Plant Sci. 2018, 9, 734. [Google Scholar] [CrossRef]
- Upreti, K.K.; Sharma, M. Role of plant growth regulators in abiotic stress tolerance. In Abiotic Stress Physiology of Horticultural Crops; Rao, N., Shivashankara, K., Laxman, R., Eds.; Springer: New Delhi, India, 2016; pp. 19–46. [Google Scholar]
- Varma, S.; Ekta; Jangra, M. Heavy metals stress and defense strategies in plants: An overview. J. Pharmacogn. Phytochem. 2021, 10, 608–614. [Google Scholar]
- Rai, A.; Belkacem, M.; Assadi, I.; Bollinger, J.-C.; Elfalleh, W.; Assadi, A.A.; Amrane, A.; Mouni, L. Bacteria in soil: Promising bioremediation agents in arid and semi-arid environments for cereal growth enhancement. Appl. Sci. 2022, 12, 11567. [Google Scholar] [CrossRef]
- Marles, J.T. Mineral nutrient composition of vegetables, fruits and grains: The context of reports of apparent historical declines. J. Food Compost. Anal. 2017, 56, 93–103. [Google Scholar] [CrossRef]
- Fleury, F.; Mignotte, B.; Vayssière, J.-L. Mitochondrial reactive oxygen species in cell death signaling. Biochimie 2002, 84, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Dutta, S.; Mitra, M.; Agarwal, P.; Mahapatra, K.; De, S.; Sett, U.; Roy, S. Oxidative and genotoxic damages in plants in response to heavy metal stress and maintenance of genome stability. Plant Signal. Behav. 2018, 13, e1460048. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Jha, A.B.; Dubey, R.S.; Pessarakli, M. Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J. Bot. 2012, 2012, 217037. [Google Scholar] [CrossRef]
- Jaishankar, M.; Tseten, T.; Anbalagan, N.; Mathew, B.B.; Beeregowda, K.N. Toxicity, mechanism and health effects of some heavy metals. Interdiscip Toxicol. 2014, 7, 60–72. [Google Scholar] [CrossRef] [PubMed]
- Rucińska-Sobkowiak, R. Water relations in plants subjected to heavy metal stresses. Acta Physiol. Plant 2016, 38, 257. [Google Scholar] [CrossRef]
- Islam, M.; Sandhi, A. Heavy Metal and Drought Stress in Plants: The Role of Microbes—A Review. Gesunde Pflanz. 2023, 75, 695–708. [Google Scholar] [CrossRef]
- Guo, Z.; Gao, Y.; Yuan, X.; Yuan, M.; Huang, L.; Wang, S.; Liu, C.; Duan, C. Effects of heavy metals on stomata in plants: A review. Int. J. Mol. Sci. 2023, 24, 9302. [Google Scholar] [CrossRef]
- Gallego, S.M.; Pena, L.B.; Barcia, R.A.; Azpilicueta, C.E.; Iannone, M.F.; Rosales, E.P.; Zawoznik, M.S.; Groppa, M.D.; Benavides, M.P. Unravelling cadmium toxicity and tolerance in plants: Insight into regulatory mechanisms. Environ. Exp. Bot. 2012, 83, 33–46. [Google Scholar] [CrossRef]
- De Silva, N.D.G.; Cholewa, E.; Ryser, P. Effects of combined drought and heavy metal stresses on xylem structure and hydraulic conductivity in red maple (Acer rubrum L.). J. Exp. Bot. 2012, 63, 5957–5966. [Google Scholar] [CrossRef] [PubMed]
- Jung, M.C. Heavy metal concentrations in soils and factors affecting metal uptake by plants in the vicinity of a Korean Cu-W mine. Sensors 2008, 8, 2413–2423. [Google Scholar] [CrossRef] [PubMed]
- Rashid, A.; Schutte, B.J.; Ulery, A.; Deyholos, M.K.; Sanogo, S.; Lehnhoff, E.A.; Beck, L. Heavy metal contamination in agricultural soil: Environmental pollutants affecting crop health. Agronomy 2023, 13, 1521. [Google Scholar] [CrossRef]
- Hou, S.; Zheng, N.; Tang, L.; Ji, X.; Li, Y. Effect of soil pH and organic matter content on heavy metals availability in maize (Zea mays L.) rhizospheric soil of non-ferrous metals smelting area. Environ. Monit. Assess. 2019, 191, 634. [Google Scholar] [CrossRef] [PubMed]
- Chojnacka, K.; Chojnacki, A.; Gorecka, H.; Gorecki, H. Bioavailability of heavy metals from polluted soils to plants. Sci. Total Environ. 2005, 337, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Zheng, F.; Ali, S.; Zhang, H.; Ouyang, Y.; Qiu, B.; Wu, F.; Zhang, G. The influence of pH and organic matter content in paddy soil on heavy metal availability and their uptake by rice plants. Environ. Pollut. 2011, 159, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Chen, X.; Qin, X.; Xu, C.; Yan, X. Effects of soil pH on the growth and cadmium accumulation in Polygonum hydropiper (L.) in low and moderately cadmium-contaminated paddy soil. Land 2023, 12, 652. [Google Scholar] [CrossRef]
- Neenan, M. The effects of soil acidity on the growth of cereals with particular reference to the differential reaction of varieties thereto. Plant Soil 1960, 12, 324–338. [Google Scholar] [CrossRef]
- Li, S.; Sun, X.; Li, S.; Liu, X.; Ma, Q.; Zhou, W. Effects of amendments on the bioavailability, transformation and accumulation of heavy metals by pakchoi cabbage in a multi-element contaminated soil. RSC Adv. 2021, 11, 4395–4405. [Google Scholar] [CrossRef]
- Hong, C.O.; Owens, V.N.; Kim, Y.G.; Lee, S.M.; Park, H.C.; Kim, K.K.; Son, H.J.; Suh, J.M.; Kim, P.J. Soil pH effect on phosphate induced cadmium precipitation in arable soil. Bull. Environ. Contam. Toxicol. 2014, 93, 101–105. [Google Scholar] [CrossRef]
- Impa, S.M.; Johnson-Beebout, S.E. Mitigating zinc deficiency and achieving high grain Zn in rice through integration of soil chemistry and plant physiology research. Plant Soil 2012, 361, 3–41. [Google Scholar] [CrossRef]
- Stefanowicz, A.M.; Kapusta, P.; Zubek, S.; Stanek, M.; Woch, M.W. Soil organic matter prevails over heavy metal pollution and vegetation as a factor shaping soil microbial communities at historical Zn–Pb mining sites. Chemosphere 2020, 240, 124922. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, H.; Fekri, M.; Farpoor, M.H.; Mahmoodabadi, M. Phosphorus sorption-desorption in soil as influenced by organic matter, carbonates and Fe-Al oxides. Environ. Eng. Manag. J. 2021, 20, 1435–1444. [Google Scholar] [CrossRef]
- Rong, Q.; Zhong, K.; Huang, H.; Li, C.; Zhang, C.; Nong, X. Humic acid reduces the available cadmium, copper, lead, and zinc in soil and their uptake by tobacco. Appl. Sci. 2020, 10, 1077. [Google Scholar] [CrossRef]
- Bot, A.; Benites, J. The Importance of Soil Organic Matter: Key to Drought-Resistant Soil and Sustained Food and Production; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2005. [Google Scholar]
- Yavitt, J.B.; Pipes, G.T.; Olmos, E.C.; Zhang, J.; Shapleigh, J.P. Soil organic matter, soil structure, and bacterial community structure in a post-agricultural landscape. Front. Earth Sci. 2021, 9, 590103. [Google Scholar] [CrossRef]
- Bronick, C.J.; Lal, R. Soil structure and management: A review. Geoderma 2005, 124, 3–22. [Google Scholar] [CrossRef]
- Ortiz, A.; Sansinenea, E. The role of beneficial microorganisms in soil quality and plant health. Sustainability 2022, 14, 5358. [Google Scholar] [CrossRef]
- Riaz, M.; Kamran, M.; Fang, Y.; Wang, Q.; Cao, H.; Yang, G.; Deng, L.; Wang, Y.; Zhou, Y.; Anastopulos, I. Arbuscular mycorrhizal fungi-induced mitigation of heavy metal phytotoxicity in metal contaminated soils: A critical review. J. Hazard. Mater. 2021, 402, 123919. [Google Scholar] [CrossRef]
- Dietterich, L.H.; Gonneau, C.; Casper, B.B. Arbuscular mycorrhizal colonization has little consequence for plant heavy metal uptake in contaminated field soils. Ecol. Appl. 2017, 27, 1862–1875. [Google Scholar] [CrossRef]
- Usman, M.; Zia-ur-Rehman, M.; Rizwan, M.; Abbas, T.; Ayub, M.A.; Naeem, A.; Alharby, H.F.; Alabdallah, N.; Alharbi, B.M.; Qamar, M.J.; et al. Effect of soil texture and zinc oxide nanoparticles on growth and accumulation of cadmium by wheat: A life cycle study. Environ. Res. 2023, 216, 114397. [Google Scholar] [CrossRef]
- Orhue, E.R.; Frank, U.O. Fate of some heavy metals in soils: A review. J. Appl. Nat. Sci. 2011, 3, 131–138. [Google Scholar] [CrossRef]
- Zhou, W.; Ren, L.; Zhu, L. Reducement of cadmium adsorption on clay minerals by the presence of dissolved organic matter from animal manure. Environ. Pollut. 2017, 223, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Pikula, D.; Stepien, W. Effect of the degree of soil contamination with heavy metals on their mobility in the soil profile in a microplot experiment. Agronomy 2021, 11, 878. [Google Scholar] [CrossRef]
- Pedroli, G.B.M.; Maasdam, W.A.C.; Verstraten, J.M. Zinc in poor sandy soils and associated groundwater. A case study. Sci. Total Environ. 1990, 91, 59–77. [Google Scholar] [CrossRef]
- Zhang, M.K.; He, Z.L.; Calvert, D.V.; Stofella, P.J. Extractability and mobility of copper and zinc accumulated in sandy soils. Pedosphere 2006, 16, 43–49. [Google Scholar] [CrossRef]
- Otunola, B.O.; Ololade, O.O. A review on the application of clay minerals as heavy metal adsorbents for remediation purposes. Environ. Technol. Innov. 2020, 18, 100692. [Google Scholar] [CrossRef]
- Hoque, M.M.; Islam, A.; Islam, A.R.M.T.; Pal, S.C.; Mahammad, S.; Alam, E. Assessment of soil heavy metal pollution and associated ecological risk of agriculture dominated mid-channel bars in a subtropical river basin. Sci. Rep. 2023, 13, 11104. [Google Scholar] [CrossRef] [PubMed]
- Pokorska-Niewiada, K.; Rajkowska-Myśliwiec, M.; Protasowicki, M. Acute lethal toxicity of heavy metals to the seeds of plants of high importance to humans. Bull. Environ. Contam. Toxicol. 2018, 101, 222–228. [Google Scholar] [CrossRef]
- Muszynska, A.; Guendel, A.; Melzer, M.; Moya, Y.A.T.; Röder, M.S.; Rolletschek, H.; Rutten, T.; Munz, E.; Melz, G.; Ortleb, S.; et al. A mechanistic view on lodging resistance in rye and wheat: A multiscale comparative study. Plant Biotechnol. J. 2021, 19, 2646–2661. [Google Scholar] [CrossRef]
- Bukhari, S.A.H.; Peerzada, A.M.; Javed, M.H.; Dawood, M.; Hussain, N.; Ahmad, S. Growth and development dynamics in agronomic crops under environmental stress. In Agronomic Crops; Hasanuzzaman, M., Ed.; Springer: Singapore, 2019; pp. 83–114. [Google Scholar]
- Yu, E.; Wang, W.; Yamaji, N.; Fukuoka, S.; Che, J.; Ueno, D.; Ando, T.; Deng, F.; Hori, K.; Yano, M.; et al. Duplication of a manganese/cadmium transporter gene reduces cadmium accumulation in rice grain. Nat. Food 2022, 3, 597–607. [Google Scholar] [CrossRef]
- Li, K.; Yu, H.; Li, T.; Huang, F. Cadmium accumulation characteristics of low-cadmium rice (Oryza sativa L.) line and F1 hybrids grown in cadmium-contaminated soils. Environ. Sci. Pollut. Res. 2017, 24, 17566–17576. [Google Scholar] [CrossRef] [PubMed]
- Bali, A.S.; Sidhu, G.P.S.; Kumar, V. Root exudates ameliorate cadmium tolerance in plants: A review. Environ. Chem. Lett. 2020, 18, 1243–1275. [Google Scholar] [CrossRef]
- Gavrilescu, M. Enhancing phytoremediation of soils polluted with heavy metals. Curr. Opin. Biotechnol. 2022, 74, 21–31. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, B.A.; Ellis, N.; Kim, C.S.; Bi, X. The role of tailored biochar in increasing plant growth, and reducing bioavailability, phytotoxicity, and uptake of heavy metals in contaminated soil. Environ. Pollut. 2017, 230, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Mickelbart, M.; Hasegawa, P.; Bailey-Serres, J. Genetic mechanisms of abiotic stress tolerance that translate to crop yield stability. Nat. Rev. Genet. 2015, 16, 237–251. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Sharifi, R.; Khan, M.S.S.; Islam, F.; Bhat, J.A.; Kui, L.; Majeed, A. Wheat microbiome: Structure, dynamics, and role in improving performance under stress environments. Front. Microbiol. 2022, 12, 821546. [Google Scholar] [CrossRef] [PubMed]
- Thakur, M.; Praveen, S.; Divte, P.R.; Mitra, R.; Kumar, M.; Gupta, C.K.; Kalidindi, U.; Bansal, R.; Roy, S.; Anand, A.; et al. Metal tolerance in plants: Molecular and physicochemical interface determines the “not so heavy effect” of heavy metals. Chemosphere 2022, 287, 131957. [Google Scholar] [CrossRef]
- Yang, L.; Meng, F.; Ma, C.; Hou, D. Elucidating the spatial determinants of heavy metals pollution in different agricultural soils using geographically weighted regression. Sci. Total Environ. 2022, 853, 158628. [Google Scholar] [CrossRef]
- Sychta, K.; Słomka, A.; Kuta, E. Insights into plant programmed cell death induced by heavy metals—Discovering a Terra Incognita. Cells 2021, 10, 65. [Google Scholar] [CrossRef]
- Mahar, A.; Wang, P.; Ali, A.; Awasthi, M.K.; Lahori, A.H.; Wang, Q.; Li, R.; Zhang, Z. Challenges and opportunities in the phytoremediation of heavy metals contaminated soils: A review. Ecotoxicol. Environ. Saf. 2016, 126, 111–121. [Google Scholar] [CrossRef]
- Hlihor, R.M.; Cozma, P.; Gavrilescu, M. Removal of heavy metals from the environment by phytoremediation and microbial remediation. In Sustainable Solutions for Environmental Pollution: Air, Water and Soil Reclamation. Volume 2: Air, Water and Soil Reclamation; El-Gendy, N.S., Ed.; Wiley-Scrivener Publishing: Hoboken, NJ, USA; Beverly, MA, USA, 2022; pp. 95–146. [Google Scholar]
- Ali, H.; Khan, E.; Ilahi, I. Environmental chemistry and ecotoxicology of hazardous heavy metals: Environmental persistence, toxicity, and bioaccumulation. J. Chem. 2019, 2019, 730305. [Google Scholar] [CrossRef]
- Intawongse, M.; Dean, J.R. Uptake of heavy metals by vegetable plants grown on contaminated soil and their bioavailability in the human gastrointestinal tract. Food Addit. Contam. 2006, 23, 36–68. [Google Scholar] [CrossRef] [PubMed]
- Khan, Z.S.; Rizwan, M.; Hafeez, M.; Ali, S.; Adrees, M.; Qayyum, M.F.; Khalid, S.; Rehman, M.Z.; Sarwar, M.A. Effects of silicon nanoparticles on growth and physiology of wheat in cadmium contaminated soil under different soil moisture levels. Environ. Sci. Pollut. Res. 2020, 27, 4958–4968. [Google Scholar] [CrossRef] [PubMed]
- Kormoker, T.; Proshad, R.; Islam, M.S.; Shamsuzzoha, M.; Akter, A.; Tusher, T. Concentrations, source apportionment and potential health risk of toxic metals in foodstuffs of Bangladesh. Toxin Rev. 2020, 40, 1447–1460. [Google Scholar] [CrossRef]
- Minhas, P.S.; Saha, J.K.; Dotaniya, M.L.; Sarkar, A.; Saha, M. Wastewater irrigation in India: Current status, impacts and response options. Sci. Total Environ. 2022, 808, 152001. [Google Scholar] [CrossRef] [PubMed]
- Hameed, A.; Rasool, S.; Azooz, M.M.; Hossain, M.A.; Ahanger, M.A.; Ahmad, P. Heavy metal stress: Plant responses and signaling. In Plant Metal Interaction: Emerging Remediation Techniques; Ahmad, P., Ed.; Elsevier: Amsterdam, The Netherlands, 2015; pp. 557–583. [Google Scholar]
- Jogawat, A.; Yadav, B.; Chhaya; Narayan, O.P. Metal transporters in organelles and their roles in heavy metal transportation and sequestration mechanisms in plants. Physiol. Plant. 2021, 173, 259–275. [Google Scholar] [CrossRef] [PubMed]
- Raychaudhuri, S.S.; Pramanick, P.; Talukder, P.; Basak, A. Polyamines, metallothioneins, and phytochelatins—Natural defense of plants to mitigate heavy metals. In Studies in Natural Products Chemistry; Atta-ur-Rahman, A.-u., Ed.; Elsevier: Amsterdam, The Netherland, 2021; Volume 69, pp. 227–261. [Google Scholar]
- Chatterjee, S.; Kumari, S.; Rath, S.; Priyadarshanee, M.; Das, D. Diversity, structure and regulation of microbial metallothionein: Metal resistance and possible applications in sequestration of toxic metals. Metallomics 2020, 12, 1637–1655. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.Y.; Chen, X.W.; Huang, W.X.; Wu, L.; Yu, Z.S.; Xiang, L.; Mo, C.H.; Li, Y.W.; Cai, Q.Y.; Wong, M.H.; et al. Cell wall modification induced by an arbuscular mycorrhizal fungus enhanced cadmium fixation in rice root. J. Hazard. Mater. 2021, 416, 125894. [Google Scholar] [CrossRef]
- Pál, M.; Janda, T.; Szalai, G. Interactions between plant hormones and thiol-related heavy metal chelators. Plant Growth Regul. 2018, 85, 173–185. [Google Scholar] [CrossRef]
- Chen, Z.C.; Liao, H. Organic acid anions: An effective defensive weapon for plants against aluminum toxicity and phosphorus deficiency in acidic soils. J. Genet. Genom. 2016, 43, 631–638. [Google Scholar] [CrossRef]
- Chen, Y.T.; Wang, Y.; Yeh, K.C. Role of root exudates in metal acquisition and tolerance. Curr. Opin. Plant Biol. 2017, 39, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Parwez, R.; Afab, T.; Khan, M.M.A.; Naeem, M. Exogenous abscisic acid fine-tunes heavy metal accumulation and plant’s antioxidant defence mechanism to optimize crop performance and secondary metabolite production in Trigonella foenum-graecum L. under nickel stress. Plant Sci. 2023, 332, 111703. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Yuan, X.; Li, L.; Zeng, M.; Yang, J.; Tang, H.; Duan, C. Genome-wide analysis of the ATP-Binding Cassette (ABC) transporter family in Zea mays L. and its response to heavy metal stresses. Int. J. Mol. Sci. 2022, 23, 2109. [Google Scholar] [CrossRef] [PubMed]
- Dahuja, A.; Kumar, R.R.; Sakhare, A.; Watts, A.; Singh, B.; Goswami, S.; Sachdev, A.; Praveen, S. Role of ATP-binding cassette transporters in maintaining plant homeostasis under abiotic and biotic stresses. Physiol. Plant. 2020, 171, 785–801. [Google Scholar] [CrossRef] [PubMed]
- Kolaj-Robin, O.; Russel, D.; Hayes, K.A.; Pembroke, J.T.; Soulimane, T. Cation diffusion facilitator family: Structure and function. FEBS Lett. 2015, 589, 1283–1295. [Google Scholar] [CrossRef] [PubMed]
- El-Sappah, A.H.; Abbas, M.; Rather, S.A.; Wani, S.H.; Soaud, N.; Noor, Z.; Qiulan, H.; Eldomiaty, A.H.; Mir, R.R.; Li, J. Genome-wide identification and expression analysis of metal tolerance protein (MTP) gene family in soybean (Glycine max) under heavy metal stress. Mol. Biol. Rep. 2023, 50, 2975–2990. [Google Scholar] [CrossRef] [PubMed]
- Mondal, S.; Pramanik, K.; Pal, P.; Mitra, S.; Ghosh, S.S.; Mondal, T.; Soren, T.; Maiti, T.K. Multifaceted roles of root exudates in light of plant-microbe interaction. In Unravelling Plant-Microbe Synergy. Developments in Applied Microbiology and Biotechnology; Chandra, D., Bhatt, P., Eds.; Elsevier: Amsterdam, The Netherlands, 2023; pp. 49–76. [Google Scholar]
- Orhan, F.; Demirci, A.; Bozari, S. Advantage of halophilic-halotolerant bacteria under salt stress. Environ. Eng. Manag. J. 2022, 21, 1741–1749. [Google Scholar] [CrossRef]
- Roskova, Z.; Skarohlid, R.; McGachy, L. Siderophores: An alternative bioremediation strategy? Sci. Total Environ. 2022, 819, 153144. [Google Scholar] [CrossRef]
- Rizvi, A.; Khan, M.S. Heavy metal-mediated toxicity to maize: Oxidative damage, antioxidant defence response and metal distribution in plant organs. Int. J. Environ. Sci. Technol. 2019, 16, 4873–4886. [Google Scholar] [CrossRef]
- Phour, M.; Sindhu, S.S. Mitigating abiotic stress: Microbiome engineering for improving agricultural production and environmental sustainability. Planta 2022, 256, 85. [Google Scholar] [CrossRef]
- Zulfiqar, F.; Ashraf, M. Proline alleviates abiotic stress induced oxidative stress in plants. J. Plant Growth Regul. 2023, 42, 4629–4651. [Google Scholar] [CrossRef]
- Zandi, P.; Schnug, E. Reactive oxygen species, antioxidant responses and implications from a microbial modulation perspective. Biology 2022, 11, 155. [Google Scholar] [CrossRef] [PubMed]
- Mishra, P.; Sharma, P. Superoxide Dismutases (SODs) and their role in regulating abiotic stress induced oxidative stress in plants. In Reactive Oxygen, Nitrogen and Sulfur Species in Plants: Production, Metabolism, Signaling and Defense Mechanisms; Hasanuzzaman, M., Fotopoulos, V., Nahar, K., Fujita, M., Eds.; John Wiley & Sons Ltd.: New York, NY, USA, 2019; pp. 53–88. [Google Scholar]
- Zhao, H.; Zhang, R.; Yan, X.; Fan, K. Superoxide dismutase nanozymes: An emerging star for anti-oxidation. J. Mater. Chem. B 2021, 9, 6939–6957. [Google Scholar] [CrossRef] [PubMed]
- Jan, A.T.; Azam, M.; Siddiqui, K.; Ali, A.; Choi, I.; Haq, O.M.R. Heavy metals and human health: Mechanistic insight into toxicity and counter defense system of antioxidants. J. Mol. Sci. 2015, 16, 29592–29630. [Google Scholar] [CrossRef] [PubMed]
- Fakhar, A.; Gul, B.; Rafique, M.; Ortas, I. Use of Biostimulants to increase heavy metal tolerance in cereals. In Sustainable Remedies for Abiotic Stress in Cereals; Abdel Latef, A.A.H., Ed.; Springer: Singapore, 2022; pp. 575–598. [Google Scholar]
- Liu, L.; Huang, L.; Lin, X.; Sun, C. Hydrogen peroxide alleviates salinity-induced damage through enhancing proline accumulation in wheat seedlings. Plant Cell Rep. 2020, 39, 567–575. [Google Scholar] [CrossRef] [PubMed]
- Khan, Y.; Shah, S.; Tian, H. The roles of arbuscular mycorrhizal fungi in influencing plant nutrients, photosynthesis, and metabolites of cereal crops—A review. Agronomy 2022, 12, 2191. [Google Scholar] [CrossRef]
- Mbodj, D.; Effa-Effa, B.; Kane, A.; Manneh, B.; Gantet, P.; Laplaze, L.; Diedhiou, A.G.; Grondin, A. Arbuscular mycorrhizal symbiosis in rice: Establishment, environmental control and impact on plant growth and resistance to abiotic stresses. Rhizosphere 2018, 8, 12–16. [Google Scholar] [CrossRef]
- Kuyper, T.W.; Jansa, J. Arbuscular mycorrhiza: Advances and retreats in our understanding of the ecological functioning of the mother of all root symbioses. Plant Soil 2023, 489, 41–88. [Google Scholar] [CrossRef]
- Dhalaria, J.; Kumar, D.; Kumar, H.; Nepovimova, E.; Kuca, K.; Islam, M.T.; Verma, R. Arbuscular mycorrhizal fungi as potential agents in ameliorating heavy metal stress in plants. Agronomy 2020, 10, 815. [Google Scholar] [CrossRef]
- Göhre, V.; Paszkowski, U. Contribution of the arbuscular mycorrhizal symbiosis to heavy metal phytoremediation. Planta 2006, 223, 1115–1122. [Google Scholar] [CrossRef]
- Li, H.; Wang, H.; Zhao, J.; Zhang, L.; Li, Y.; Wang, H.; Teng, H.; Yuan, Z.; Yuan, Z. Physio-biochemical and transcriptomic features of arbuscular mycorrhizal fungi relieving cadmium stress in wheat. Antioxidants 2022, 11, 2390. [Google Scholar] [CrossRef] [PubMed]
- Bano, S.A.; Ashfaq, D. Role of mycorrhiza to reduce heavy metal stress. Nat. Sci. 2013, 5, 16–20. [Google Scholar] [CrossRef]
- Upadhyaya, S.; Panda, S.K.; Bhattacharjee, M.K.; Dutta, S. Role of arbuscular mycorrhiza in h eavy metal tolerance in plants: Prospects for phytoremediation. J. Phytol. 2010, 2, 16–27. [Google Scholar]
- Souza, R.; Ambrosini, A.; Passaglia, L.M.P. Plant growth-promoting bacteria as inoculants in agricultural soils. Genet. Mol. Biol. 2015, 38, 401–419. [Google Scholar] [CrossRef]
- Kashyap, S.; Barman, R.; Nath, M.; Agarwala, N. Microbial symbionts for alleviation of heavy metal toxicity in crop plants. In Biostimulants in Alleviation of Metal Toxicity in Plants: Emerging Trends and Opportunities. Biostimulants and Protective Biochemical Agents; Gill, S.S., Tuteja, N., Khan, N.A., Gill, R., Eds.; Elsevier: Amsterdam, The Netherlands, 2023; pp. 371–400. [Google Scholar]
- Becze, A.; Vincze, E.B.; Varga, H.M.; Gyongyver, M. Effect of plant growth promoting rhizobacteria on Zea mays development and growth under heavy metal and salt stress condition. Environ. Eng. Manag. J. 2021, 20, 547–557. [Google Scholar]
- Gavrilescu, M. Water, soil, and plants interactions in a threatened environment. Water 2021, 13, 2746. [Google Scholar] [CrossRef]
- Alves, A.R.A.; Yin, Q.; Oliveira, R.S.; Silva, E.F.; Novo, L.A.B. Plant growth-promoting bacteria in phytoremediation of metal-polluted soils: Current knowledge and future directions. Sci. Total Environ. 2022, 838, 156435. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhao, S.; Liu, S.; Peng, J.; Zhang, H.; Zhao, Q.; Zheng, L.; Chen, Y.; Shen, Z.; Xu, X.; et al. Enhancing the phytoremediation of heavy metals by combining hyperaccumulator and heavy metal-resistant plant growth-promoting bacteria. Front. Plant Sci. 2022, 13, 912350. [Google Scholar] [CrossRef]
- Tak, H.I.; Ahmad, F.; Babalola, O.O. Advances in the application of plant growth-promoting rhizobacteria in phytoremediation of heavy metals. Rev. Environ. Contam. Toxicol. 2013, 223, 33–52. [Google Scholar]
- Sobariu, D.L.; Tudorache Fertu, D.I.; Diaconu, M.; Pavel, L.V.; Hlihor, R.M.; Drăgoi, E.N.; Curteanu, S.; Lenz, M.; Corvini, P.F.X. Rhizobacteria and plant symbiosis in heavy metal uptake and its implications for soil bioremediation. New Biotechnol. 2017, 39 Pt A, 125–134. [Google Scholar] [CrossRef]
- Gladkov, E.A.; Tereshonok, D.V.; Stepanova, A.Y.; Gladkova, O.V. Plant–microbe interactions under the action of heavy metals and under the conditions of flooding. Diversity 2023, 15, 175. [Google Scholar] [CrossRef]
- Li, Q.; Wang, Y.; Li, Y.; Li, L.; Tang, M.; Hu, W.; Chen, L.; Ai, S. Speciation of heavy metals in soils and their immobilization at micro-scale interfaces among diverse soil components. Sci. Total Environ. 2022, 825, 153862. [Google Scholar] [CrossRef]
- Shaaf, S.; Bretani, G.; Biswas, A.; Fontana, I.M.; Rossini, L. Genetics of barley tiller and leaf development. J. Integr. Plant Biol. 2019, 61, 226–256. [Google Scholar] [CrossRef] [PubMed]
- San Martín, Y.B.; León, H.F.T.; Rodríguez, A.A.; Marqués, A.M.; López, M.I.S. Rhamnolipids application for the removal of vanadium from contaminated sediment. Curr. Microbiol. 2021, 78, 1949–1960. [Google Scholar] [CrossRef]
- Huang, H.J.; Jia, Q.Y.; Jing, W.X.; Dahms, H.U.; Wang, L. Screening strains for microbial biosorption technology of cadmium. Chemosphere 2020, 251, 126428. [Google Scholar] [CrossRef]
- Wani, P.A.; Khan, M.S.; Zaidi, A. Chromium-reducing and plant growth-promoting Mesorhizobium improves chickpea growth in chromium-amended soil. Biotechnol. Lett. 2008, 30, 159–163. [Google Scholar] [CrossRef]
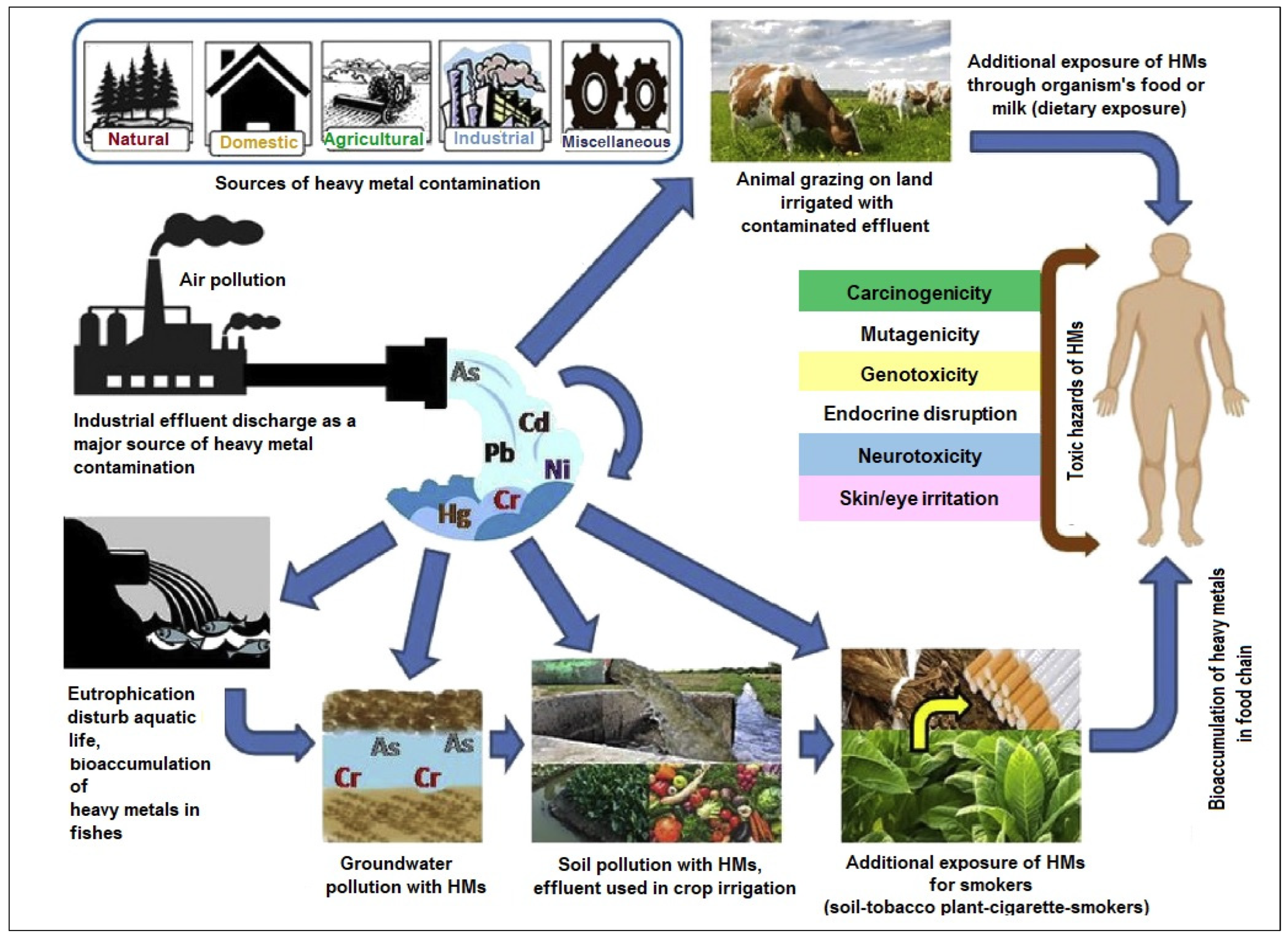
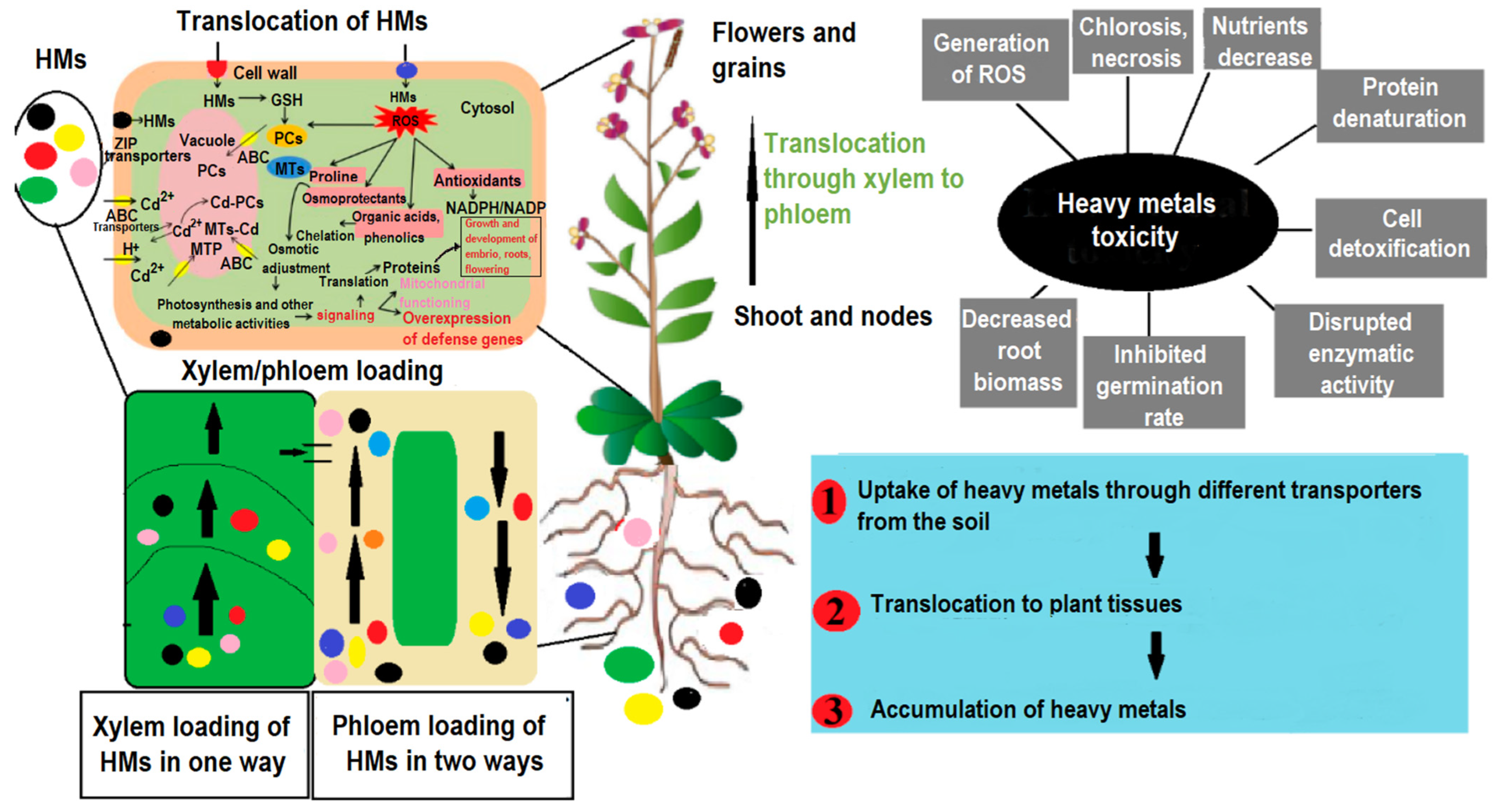
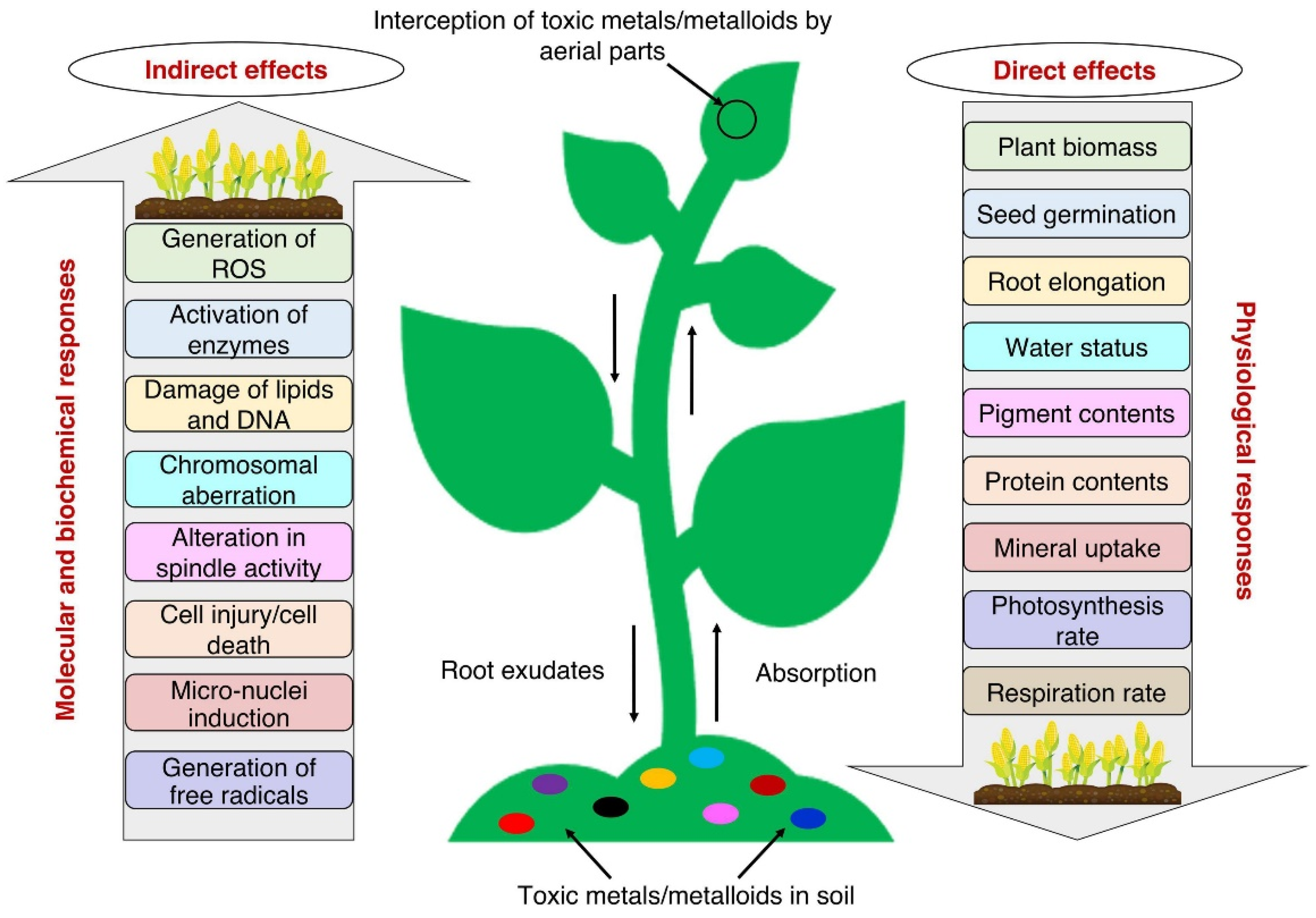
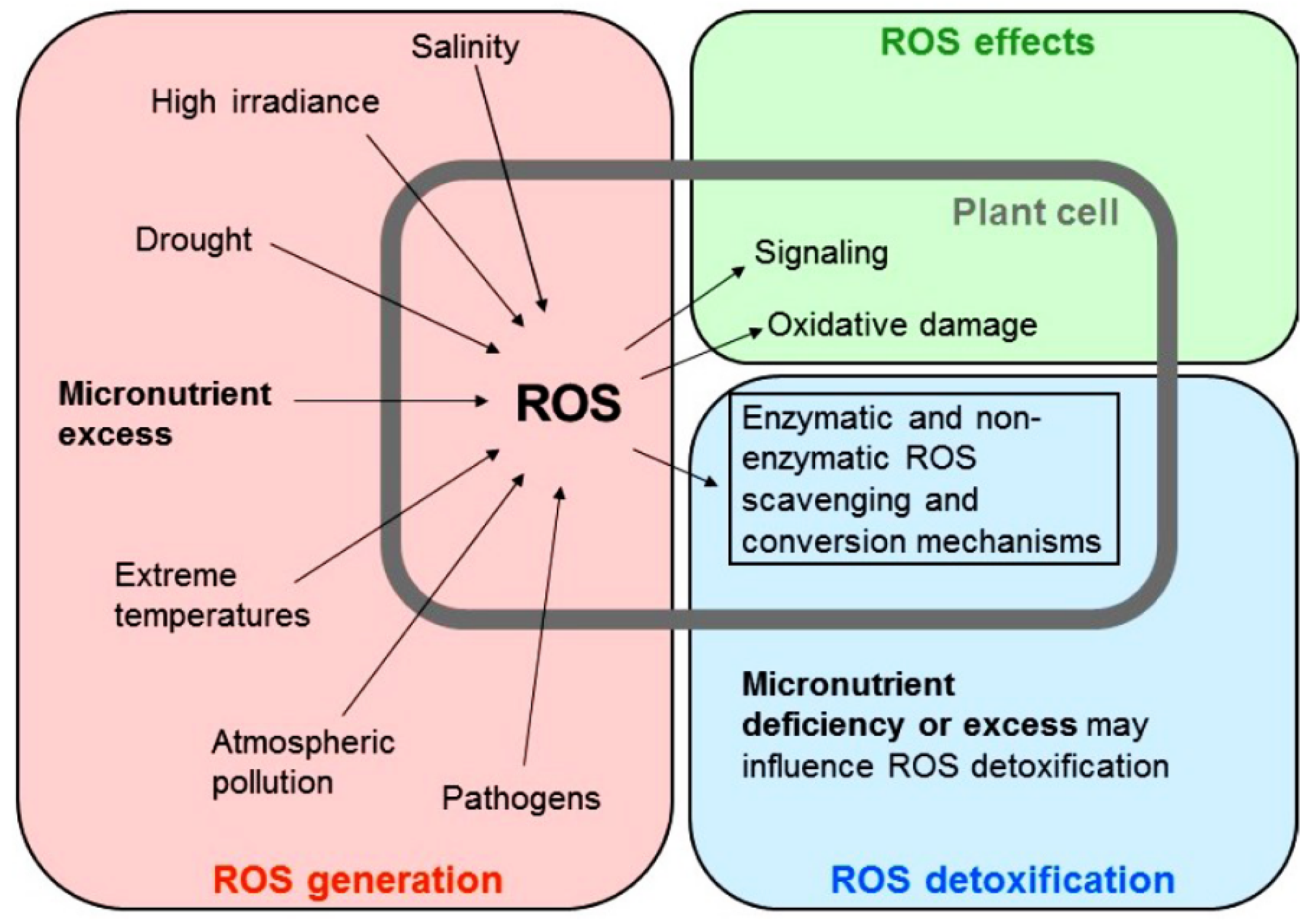
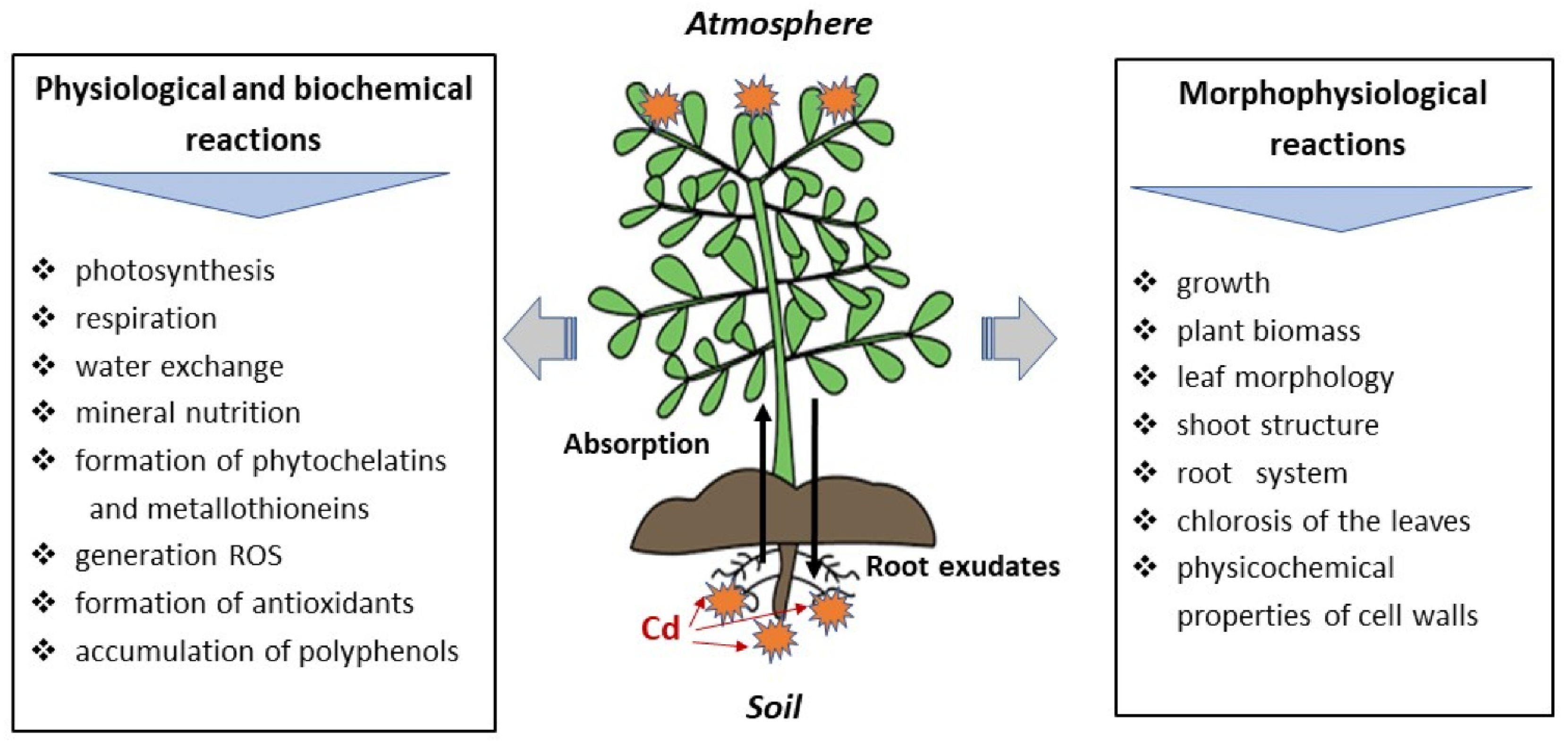
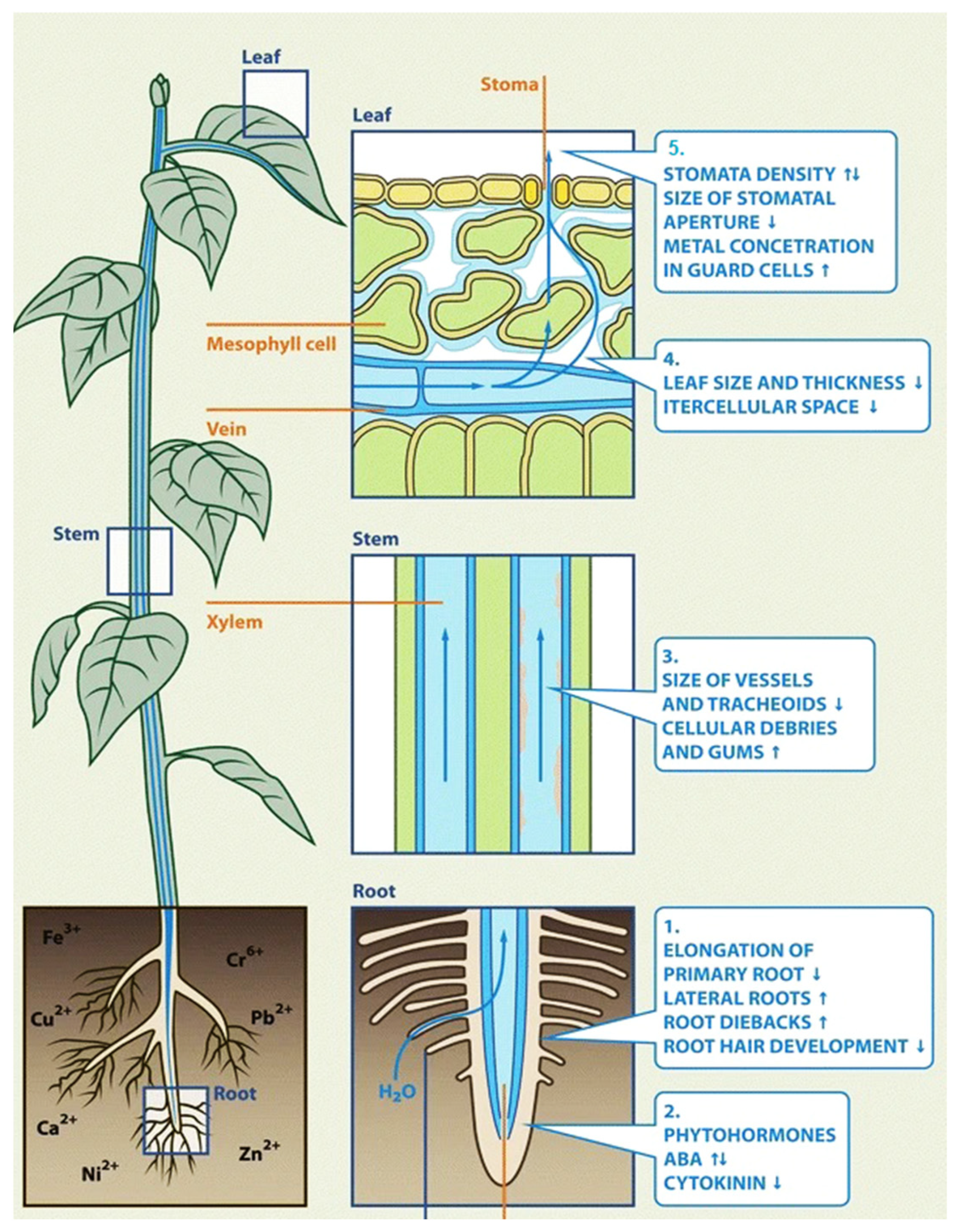
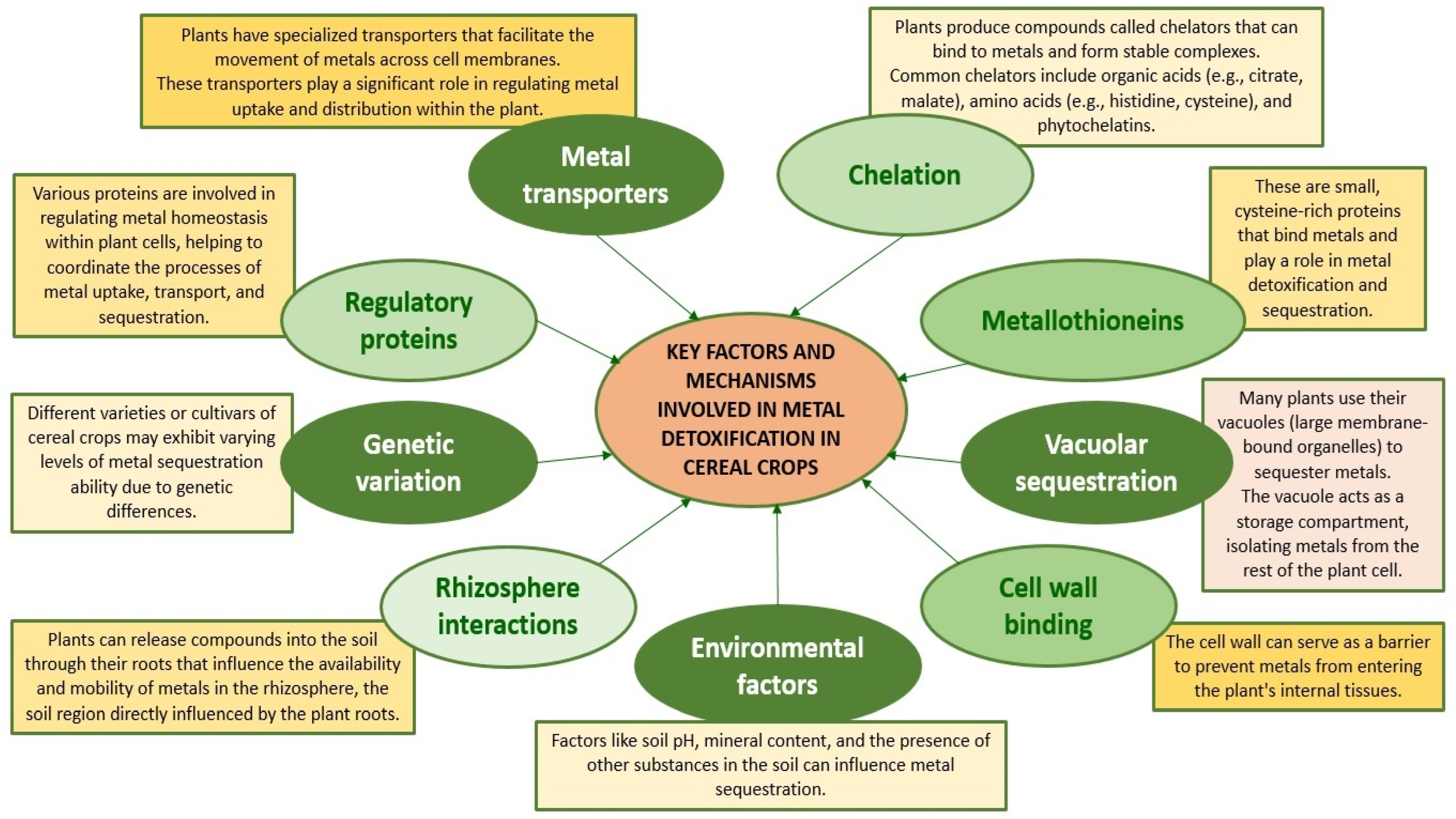
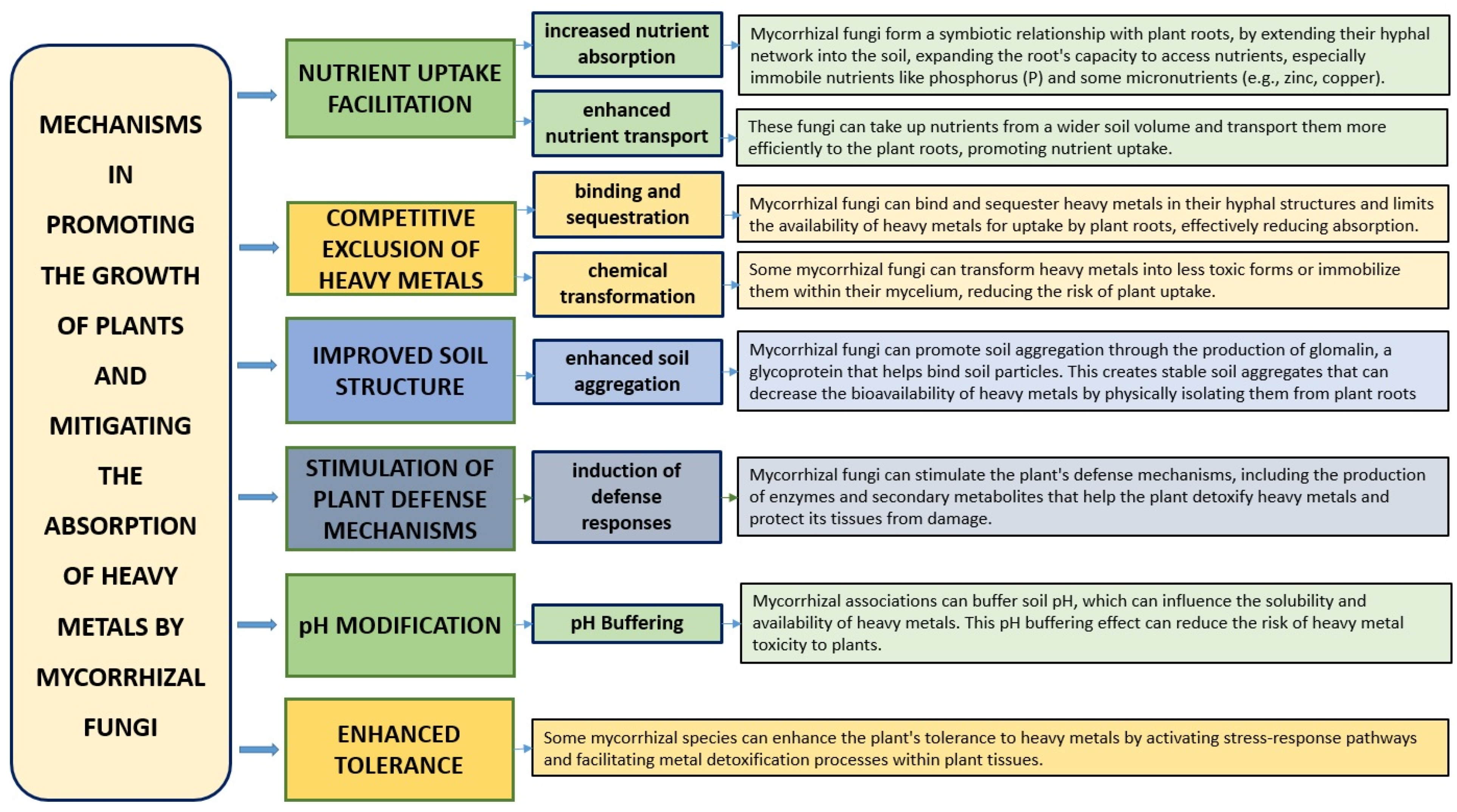
| Cereals Importance | Motivation |
|---|---|
| Essential food for millions of people |
|
| Global production and consumption |
|
| Caloric and nutritional safety |
|
| Food safety for vulnerable populations |
|
| Livelihoods and rural development |
|
| Role in food trade |
|
| Animal feed and livestock production |
|
| Climate resilience |
|
| Heavy Metal | Toxicity in Cereals |
|---|---|
| Cadmium (Cd) |
|
| Arsenic (As) |
|
| Mercury (Hg) |
|
| Lead (Pb) |
|
| Nickel (Ni) |
|
| Copper (Cu) |
|
| Zinc (Zn) |
|
| Chromium (Cr) |
|
| Growth Indicators in Cereal Plants | Impact of Heavy Metals |
|---|---|
| Plant height | Stunted growth, reduced height compared to healthy plants |
| Root length and density | Inhibited root elongation and reduced density, affecting water and nutrient absorption |
| Shoot and root biomass | Decreased biomass, resulting in reduced plant size and production |
| Leaf area | Reduced area, limiting photosynthesis and carbohydrate production |
| Chlorophyll content | Decline in content, leading to chlorosis and reduced photosynthesis efficiency |
| Photosynthesis rate | Decreased rate due to impaired chloroplasts and enzymes |
| Water use efficiency | Disrupted water uptake and efficiency, causing wilting |
| Nutrient uptake | Interference with essential nutrient uptake, causing imbalances |
| Flowering and fruiting | Negative impact on flowering and fruit development, reducing crop yield |
| Seed germination and seedling emergence | Delayed germination and emergence, affecting early growth |
| Reproductive development | Disruption in reproductive development, reducing seed production and quality |
| Yield and grain quality | Lower crop yield and compromised grain quality, affecting economic value |
| Key Aspect to be Investigated | Focus |
|---|---|
| Metal-specific uptake and translocation |
|
| Genomic and genetic studies |
|
| Metal interactions and synergies |
|
| Soil–plant-microbe interactions |
|
| Agronomic practices |
|
| Phytoremediation and phytoextraction |
|
| Food safety assessment |
|
| Modelling and predictive tools |
|
| Climate change effects |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vasilachi, I.C.; Stoleru, V.; Gavrilescu, M. Analysis of Heavy Metal Impacts on Cereal Crop Growth and Development in Contaminated Soils. Agriculture 2023, 13, 1983. https://doi.org/10.3390/agriculture13101983
Vasilachi IC, Stoleru V, Gavrilescu M. Analysis of Heavy Metal Impacts on Cereal Crop Growth and Development in Contaminated Soils. Agriculture. 2023; 13(10):1983. https://doi.org/10.3390/agriculture13101983
Chicago/Turabian StyleVasilachi, Ionela Cătălina, Vasile Stoleru, and Maria Gavrilescu. 2023. "Analysis of Heavy Metal Impacts on Cereal Crop Growth and Development in Contaminated Soils" Agriculture 13, no. 10: 1983. https://doi.org/10.3390/agriculture13101983
APA StyleVasilachi, I. C., Stoleru, V., & Gavrilescu, M. (2023). Analysis of Heavy Metal Impacts on Cereal Crop Growth and Development in Contaminated Soils. Agriculture, 13(10), 1983. https://doi.org/10.3390/agriculture13101983








