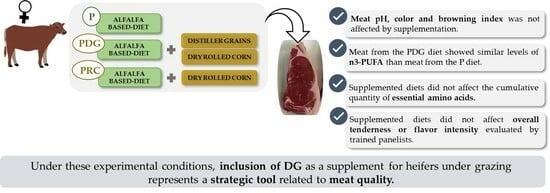
Meat Quality Traits in Beef from Heifers: Effect of including Distiller Grains in Finishing Pasture-Based Diets
Abstract
1. Introduction
2. Materials and Methods
2.1. Meat Samples
2.2. Instrumental Color and pH Determination
2.3. Myoglobin (Mb) Content and Browning Index Determination
2.4. Water Holding Capacity (WHC)
2.4.1. Thawing Loss
2.4.2. Cooking Loss
2.5. Warner-Bratzler Shear Force (WBSF)
2.6. Sensory Analysis
2.7. Fat Content and Fatty Acid Profile
2.8. Amino Acid Profile
2.9. Statistical Analysis
3. Results
3.1. Meat pH, WHC, Color and Texture (WBSF)
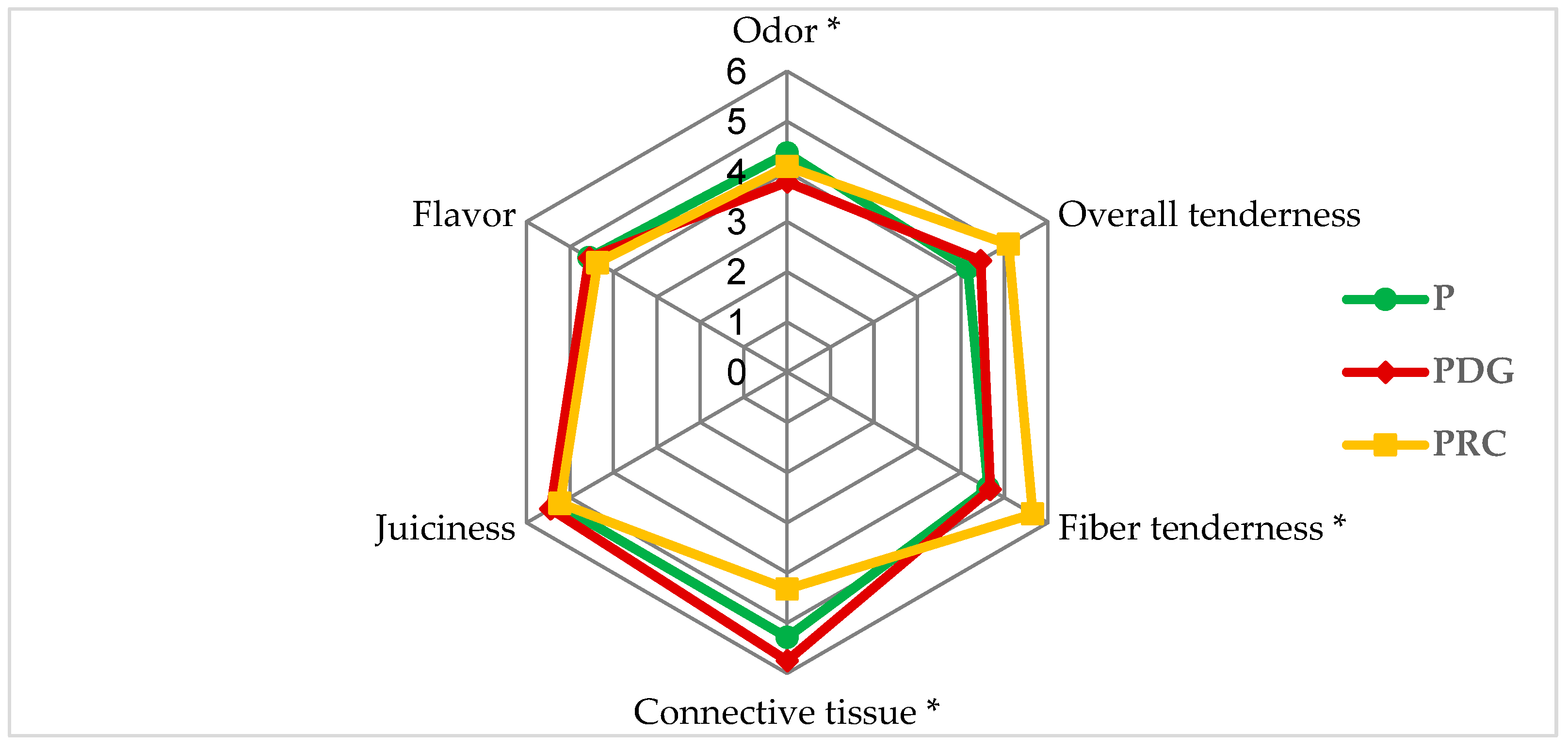
3.2. Sensory Analysis
3.3. Nutritional Quality
3.3.1. IMF and Fatty Acid Profile
3.3.2. Amino Acid Profile
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- OECD. OECD-FAO Agricultural Outlook 2020–2029. 2020. Available online: https://www.oecd-ilibrary.org/agriculture-and-food/oecd-fao-agricultural-outlook-2020-2029_1112c23b-en (accessed on 17 July 2023).
- Pulina, G.; Acciaro, M.; Atzori, A.S.; Battacone, G.; Crovetto, G.M.; Mele, M.; Pirlo, G.; Rassu, S.P.G. Animal board invited review—Beef for future: Technologies for a sustainable and profitable beef industry. Animal 2021, 15, 100358. [Google Scholar] [CrossRef]
- Rotta, P.P.; Prado, R.M.D.; Prado, I.N.D.; Valero, M.V.; Visentaine, J.V.; Silva, R.R. The Effects of Genetic Groups, Nutrition, Finishing Systems and Gender of Brazilian Cattle on Carcass Characteristics and Beef Composition and Appearance: A Review. Asian-Australas. J. Anim. Sci. 2009, 22, 1718–1734. [Google Scholar] [CrossRef]
- Venkata Reddy, B.; Sivakumar, A.S.; Jeong, D.W.; Woo, Y.-B.; Park, S.-J.; Lee, S.-Y.; Byun, J.-Y.; Kim, C.-H.; Cho, S.-H.; Hwang, I. Beef quality traits of heifer in comparison with steer, bull and cow at various feeding environments. Anim. Sci. J. 2015, 86, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.R.F.; Evans, P.R.; Nute, G.R.; Richardson, R.I.; Scollan, N.D. A comparison between red clover silage and grass silage feeding on fatty acid composition, meat stability and sensory quality of the M. Longissimus muscle of dairy cull cows. Meat Sci. 2009, 81, 738–744. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, T.; Hattori, A.; Takahashi, K. Structural changes in intramuscular connective tissue during the fattening of Japanese black cattle: Effect of marbling on beef tenderization. J. Anim. Sci. 1999, 77, 93–104. [Google Scholar] [CrossRef]
- Steiner, J.L.; Franzluebbers, A.J. Farming with grass—For people, for profit, for production, for protection. J. Soil Water Conserv. 2009, 64, 75A–80A. [Google Scholar] [CrossRef]
- O’Sullivan, A.; O’Sullivan, K.; Galvin, K.; Moloney, A.P.; Troy, D.J.; Kerry, J.P. Grass silage versus maize silage effects on retail packaged beef quality. J. Anim. Sci. 2002, 80, 1556–1563. [Google Scholar] [CrossRef][Green Version]
- USDA. Data Products. 2022. Available online: https://www.ers.usda.gov/data-products/chart-gallery/gallery/chart-detail/?chartId=104424 (accessed on 7 June 2023).
- Merayo, M.; Pighin, D.; Grigioni, G. Meat quality traits and feeding distillers grains to cattle: A review. Anim. Prod. Sci. 2020, 60, 1123–1135. [Google Scholar] [CrossRef]
- Pouzo, L.B.; Ceconi, I.; Davies, P.; Mendez, S.; Ortiz Miranda, S.; Testa, M. Pava Animal performance and meat quality characteristics from feedlot-finished steers fed increasing leveles of wet distillers grain. Meat Sci. 2023, 204, 143747. [Google Scholar] [CrossRef]
- Klopfenstein, T.J.; Erickson, G.E.; Bremer, V.R. Board-Invited Review: Use of distillers by-products in the beef cattle feeding industry. J. Anim. Sci. 2008, 86, 1223–1231. [Google Scholar] [CrossRef]
- Merayo, M.; Pighin, D.; Cunzolo, S.; Grigioni, G. Is beef quality affected by the inclusion of distiller grains in cattle diets? Anim. Sci. J. 2022, 93, e13761. [Google Scholar] [CrossRef] [PubMed]
- Harris, H.L.; Cupp, A.S.; Roberts, A.J.; Funston, R.N. Utilization of soybeans or corn milling by-products in beef heifer development diets. J. Anim. Sci. 2008, 86, 476–482. [Google Scholar] [CrossRef] [PubMed]
- Lawton Stewart, R.J.; Segers, J.; Hammond, K.; Cheely, T.; Ward, B.; Allen, B. Distillers Grains in Beef Cattle Diets. 2017. Available online: https://secure.caes.uga.edu/extension/publications/files/pdf/B1482_1.PDF (accessed on 10 July 2023).
- IPCVA. Informe de Faena y Producción. 2do Trimestre 2021. 2021. Available online: http://www.ipcva.com.ar/documentos/2389_1628273025_informedefaenayproduccin2trimestre2021.pdf (accessed on 10 July 2023).
- Drouillard, J.S. Current situation and future trends for beef production in the United States of America—A review. Asian-Australas. J. Anim. Sci. 2018, 31, 1007–1016. [Google Scholar] [CrossRef] [PubMed]
- Latimori, N.J.; Kloster, A.M.; García, P.T.; Carduza, F.J.; Grigioni, G.; Pensel, N.A. Diet and genotype effects on the quality index of beef produced in the Argentine Pampeana region. Meat Sci. 2008, 79, 463–469. [Google Scholar] [CrossRef] [PubMed]
- Pordomingo, A.J.; Grigioni, G.; Carduza, F.; Garcia, T.P.; Pordomingo, A.B.; Volpi Lagreca, G. Productividad y características de la carne de vaquillonas F1 del cruzamiento de Angus con Criollo, Hereford o Shorton en pasturas de alfalfa. Rev. Argent. Prod. Anim. 2012, 32, 47–61. [Google Scholar]
- MAGYP. Informe Estadísticos Comercio de Ganado y Carne Bovina 2018–2019. 2019. Available online: https://www.magyp.gob.ar/sitio/areas/gestion/_files/000000_estadisticas/_archivos//000000_Bovinos/000004_Otros%20Informes/000002_Informe%20Estad%C3%ADstico%20de%20Faena%20Comercial%20Bovina%202018-2019.pdf (accessed on 10 July 2023).
- Merayo, M. Granos de Destilería en Alimentación Animal: Su Incidencia en Calidad de Carne Bovina. Ph.D. Thesis, Universidad de Buenos Aires, Buenos Aires, Argentina, 2020. Available online: http://hdl.handle.net/20.500.12123/10199 (accessed on 9 July 2023).
- Pecka-Kiełb, E.; Zachwieja, A.; Miśta, D.; Zawadzki, W.; Zielak-Steciwko, A. Use of Corn Dried Distillers Grains (DDGS) in Feeding of Ruminants; Jacob-Lopes, E., Zepka, L.Q., Eds.; IntechOpen: Rijeka, Croatia, 2017; p. Ch. 25. ISBN 978-953-51-2892-2. [Google Scholar]
- Kloster, A.M.; Zurbriggen, G.A.; García Montaño, T.; Carduza, F.; Casseta, G.; Conde, M.B. Suplementación estival de vaquillonas cruza continental con grano de maíz y una mezcla (50:50) de burlanda y maíz sobre una pastura de alfalfa y gramíneas. In Revista Argentina de Producción Animal, Proceedings of the 42° Congreso Argentino de Producción Animal, Bahía Blanca, Argentina, 15–18 October 2019; RAPA: Balcarce, Artentina, 2019; Volume 39, p. 83. [Google Scholar]
- Merayo, M.; Rizzo, S.A.; Rossetti, L.; Pighin, D.; Grigioni, G. Effect of Aging and Retail Display Conditions on the Color and Oxidant/Antioxidant Status of Beef from Steers Finished with DG-Supplemented Diets. Foods 2022, 11, 884. [Google Scholar] [CrossRef]
- AMSA. Meat Color Measurements Guidelines; American Meat Science Association: Champaign, IL, USA, 2012; ISBN 8005172672. [Google Scholar]
- AMSA. Research Guidelines for Cookery, Sensory Evaluation, and Instrumental Tenderness Measurements of Meat, 2nd ed.; American Meat Science Association: Champaign, IL, USA, 2016. [Google Scholar]
- ISO 8586/2007; Sensory Analysis—General Guidance for the Design of Test Rooms. ISO: Geneva, Switzerland, 2019.
- Garcia, P.T.; Pensel, N.A.; Sancho, A.M.; Latimori, N.J.; Kloster, A.M.; Amigone, M.A.; Casal, J.J. Beef lipids in relation to animal breed and nutrition in Argentina. Meat Sci. 2008, 79, 500–508. [Google Scholar] [CrossRef]
- Park, P.W.; Goins, R.E. In situ preparation of fatty acid methyl esters for analysis of fatty acid composition in foods. J. Food Sci. 1994, 59, 1262–1266. [Google Scholar] [CrossRef]
- Ulbricht, T.L.V.; Southgate, D.A.T. Coronary heart disease: Seven dietary factors. Lancet 1991, 338, 985. [Google Scholar] [CrossRef]
- Spackman, D.H.; Stein, W.H.; Moore, S. Automatic Recording Apparatus for Use in Chromatography of Amino Acids. Anal. Chem. 1958, 30, 1190–1206. [Google Scholar] [CrossRef]
- Di Rienzo, J.; Casanoves, F.; Balzarini, M.; Gonzalez, L.; Tablada, M.; Robledo, C. InfoStat Versión 2020. 2020. Available online: www.infostat.com.ar (accessed on 29 June 2023).
- Apaoblaza, A.; Gerrard, S.D.; Matarneh, S.K.; Wicks, J.C.; Kirkpatrick, L.; England, E.M.; Scheffler, T.L.; Duckett, S.K.; Shi, H.; Silva, S.L.; et al. Muscle from grass- and grain-fed cattle differs energetically. Meat Sci. 2020, 161, 107996. [Google Scholar] [CrossRef] [PubMed]
- Hughes, J.M.; Oiseth, S.K.; Purslow, P.P.; Warner, R.D. A structural approach to understanding the interactions between colour, water-holding capacity and tenderness. Meat Sci. 2014, 98, 520–532. [Google Scholar] [CrossRef] [PubMed]
- Suman, S.P.; Mancini, R.A.; Faustman, C. Lipid-Oxidation-Induced Carboxymyoglobin Oxidation. J. Agric. Food Chem. 2006, 54, 9248–9253. [Google Scholar] [CrossRef] [PubMed]
- Hocquette, J.; Botreau, R.; Legrand, I.; Polkinghorne, R.; Pethick, D.; Lherm, M.; Picard, B.; Doreau, M.; Terlouw, E. Win–win strategies for high beef quality, consumer satisfaction, and farm efficiency, low environmental impacts and improved animal welfare. Anim. Prod. Sci. 2014, 54, 1537–1548. [Google Scholar] [CrossRef]
- Nuernberg, K.; Dannenberger, D.; Nuernberg, G.; Ender, K.; Voigt, J.; Scollan, N.D.; Wood, J.D.; Nute, G.R.; Richardson, R.I. Effect of a grass-based and a concentrate feeding system on meat quality characteristics and fatty acid composition of longissimus muscle in different cattle breeds. Livest. Prod. Sci. 2005, 94, 137–147. [Google Scholar] [CrossRef]
- Greenwood, P.L. Review: An overview of beef production from pasture and feedlot globally, as demand for beef and the need for sustainable practices increase. Animal 2021, 15, 100295. [Google Scholar] [CrossRef]
- Salami, S.A.; Luciano, G.; O’Grady, M.N.; Biondi, L.; Newbold, C.J.; Kerry, J.P.; Priolo, A. Sustainability of feeding plant by-products: A review of the implications for ruminant meat production. Anim. Feed Sci. Technol. 2019, 251, 37–55. [Google Scholar] [CrossRef]
- Blasina Máspoli, M.; Piñeyrúa Belo, A.; Renau Risso, M. Evaluation of Self-Consumption System for Winter Supplementation of Calves on Natural Pastures; Universidad de la República: Montevideo, Uruguay, 2010. [Google Scholar]
- Soulat, J.; Picard, B.; Bord, C.; Monteils, V. Characterization of Four Rearing Managements and Their Influence on Carcass and Meat Qualities in Charolais Heifers. Foods 2022, 11, 1262. [Google Scholar] [CrossRef]
- Hocquette, J.-F.; Botreau, R.; Picard, B.; Jacquet, A.; Pethick, D.W.; Scollan, N.D. Opportunities for predicting and manipulating beef quality. Meat Sci. 2012, 92, 197–209. [Google Scholar] [CrossRef]
- Scollan, N.D.; Dannenberger, D.; Nuernberg, K.; Richardson, I.; MacKintosh, S.; Hocquette, J.-F.; Moloney, A.P. Enhancing the nutritional and health value of beef lipids and their relationship with meat quality. Meat Sci. 2014, 97, 384–394. [Google Scholar] [CrossRef]
- Warner, R.D. The Eating Quality of Meat-IV Water-Holding Capacity and Juiciness. In Woodhead Publishing Series in Food Science, Technology and Nutrition, Lawrie’s Meat Science; Toldrá, F., Ed.; Woodhead Publishing: Cambridge, UK, 2017; pp. 419–459. ISBN 9780081006979. [Google Scholar]
- Lage, J.F.; Paulino, P.V.R.; Filho, S.C.V.; Souza, E.J.O.; Duarte, M.S.; Benedeti, P.D.B.; Souza, N.K.P.; Cox, R.B. Influence of genetic type and level of concentrate in the finishing diet on carcass and meat quality traits in beef heifers. Meat Sci. 2012, 90, 770–774. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Qi, J.; Li, J.; Zhang, Y.; Yang, X.; Xin, L.; Niu, L.; Xu, B.; Qian, Z.; Zhu, L.; et al. Effects of dietary resveratrol supplementation in cattle on the anti-oxidative capacity and meat quality of beef steaks under high-oxygen packaging. Meat Sci. 2023, 204, 109238. [Google Scholar] [CrossRef] [PubMed]
- García-Torres, S.; López-Gajardo, A.; Tejerina, D.; Prior, E.; Cabeza de Vaca, M.; Horcada, A. Effect of Two Organic Production Strategies and Ageing Time on Textural Characteristics of Beef from the Retinta Breed. Foods 2020, 9, 1417. [Google Scholar] [CrossRef] [PubMed]
- Cheng, K.; Yu, C.; Li, Z.; Li, S.; Yan, E.; Song, Z.; Zhang, H.; Zhang, L.; Wang, T. Resveratrol improves meat quality, muscular antioxidant capacity, lipid metabolism and fiber type composition of intrauterine growth retarded pigs. Meat Sci. 2020, 170, 108237. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Arner, A.; Puolanne, E.; Ertbjerg, P. On the water-holding of myofibrils: Effect of sarcoplasmic protein denaturation. Meat Sci. 2016, 119, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Tornberg, E. Effects of heat on meat proteins—Implications on structure and quality of meat products. Meat Sci. 2005, 70, 493–508. [Google Scholar] [CrossRef]
- Chao, M.D.; Domenech-Perez, K.I.; Senaratne-Lenagala, L.S.; Calkins, C.R. Feeding wet distillers grains plus solubles contributes to sarcoplasmic reticulum membrane instability. Anim. Prod. Sci. 2018, 58, 2215–2223. [Google Scholar] [CrossRef]
- Mancini, R.A.; Hunt, M.C. Current research in meat color. Meat Sci. 2005, 71, 100–121. [Google Scholar] [CrossRef]
- Gagaoua, M.; Hughes, J.; Terlouw, E.M.C.; Warner, R.D.; Purslow, P.P.; Lorenzo, J.M.; Picard, B. Proteomic biomarkers of beef colour. Trends Food Sci. Technol. 2020, 101, 234–252. [Google Scholar] [CrossRef]
- Holman, B.W.B.; van de Ven, R.J.; Mao, Y.; Coombs, C.E.O.; Hopkins, D.L. Using instrumental (CIE and reflectance) measures to predict consumers’ acceptance of beef colour. Meat Sci. 2017, 127, 57–62. [Google Scholar] [CrossRef]
- Coria, M.S.; Reineri, P.S.; Pighin, D.; Barrionuevo, M.G.; Carranza, P.G.; Grigioni, G.; Palma, G.A. Feeding strategies alter gene expression of the calpain system and meat quality in the longissimus muscle of Braford steers. Asian-Australas. J. Anim. Sci. 2020, 33, 753–762. [Google Scholar] [CrossRef] [PubMed]
- Roeber, D.L.; Gill, R.; DiCostanzo, A. Meat quality responses to feeding distiller’s grains to finishing Holstein steers. J. Anim. Sci. 2005, 83, 2455–2460. [Google Scholar] [CrossRef] [PubMed]
- Shin, E.-C.; Shurson, G.C.; Gallaher, D.D. Antioxidant capacity and phytochemical content of 16 sources of corn distillers dried grains with solubles (DDGS). Anim. Nutr. 2018, 4, 435–441. [Google Scholar] [CrossRef] [PubMed]
- Depenbusch, B.E.; Coleman, C.M.; Higgins, J.J.; Drouillard, J.S. Effects of increasing levels of dried corn distillers grains with solubles on growth performance, carcass characteristics, and meat quality of yearling heifers. J. Anim. Sci. 2009, 87, 2653–2663. [Google Scholar] [CrossRef]
- Salami, S.A.; O’Grady, M.N.; Luciano, G.; Priolo, A.; McGee, M.; Moloney, A.P.; Kerry, J.P. Fatty acid composition, shelf-life and eating quality of beef from steers fed corn or wheat dried distillers’ grains with solubles in a concentrate supplement to grass silage. Meat Sci. 2021, 173, 108381. [Google Scholar] [CrossRef]
- Dunne, P.G.; O’Mara, F.P.; Monahan, F.J.; Moloney, A.P. Changes in colour characteristics and pigmentation of subcutaneous adipose tissue and M. longissimus dorsi of heifers fed grass, grass silage or concentrate-based diets. Meat Sci. 2006, 74, 231–241. [Google Scholar] [CrossRef]
- Franco, D.; Bispo, E.; González, L.; Vázquez, J.A.; Moreno, T. Effect of finishing and ageing time on quality attributes of loin from the meat of Holstein-Fresian cull cows. Meat Sci. 2009, 83, 484–491. [Google Scholar] [CrossRef]
- Gil, M.; Serra, X.; Piedrafita, J.; Quintanilla, R.; Oliver, M.A. Fiber characterization of muscle Longissimus thoracis from bruna dels Pirineus cattle breed. In Proceedings of the 44th ICOMST, Barcelona, Spain, 30 August–4 September 1998; pp. 704–706. [Google Scholar]
- Miller, M.F.; Carr, M.A.; Ramsey, C.B.; Crockett, K.L.; Hoover, L.C. Consumer thresholds for establishing the value of beef tenderness. J. Anim. Sci. 2001, 79, 3062–3068. [Google Scholar] [CrossRef]
- Destefanis, G.; Brugiapaglia, A.; Barge, M.T.; Dal Molin, E. Relationship between beef consumer tenderness perception and Warner–Bratzler shear force. Meat Sci. 2008, 78, 153–156. [Google Scholar] [CrossRef]
- Hocquette, J.-F.; Van Wezemael, L.; Chriki, S.; Legrand, I.; Verbeke, W.; Farmer, L.; Scollan, N.D.; Polkinghorne, R.; Rødbotten, R.; Allen, P.; et al. Modelling of beef sensory quality for a better prediction of palatability. Meat Sci. 2014, 97, 316–322. [Google Scholar] [CrossRef]
- Picard, B.; Gagaoua, M.; Al Jammas, M.; Bonnet, M. Beef tenderness and intramuscular fat proteomic biomarkers: Effect of gender and rearing practices. J. Proteom. 2019, 200, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Aldai, N.; Aalhus, J.L.; Dugan, M.E.R.; Robertson, W.M.; McAllister, T.A.; Walter, L.J.; McKinnon, J.J. Comparison of wheat- versus corn-based dried distillers’ grains with solubles on meat quality of feedlot cattle. Meat Sci. 2010, 84, 569–577. [Google Scholar] [CrossRef] [PubMed]
- Platter, W.J.; Tatum, J.D.; Belk, K.E.; Chapman, P.L.; Scanga, J.A.; Smith, G.C. Relationships of consumer sensory ratings, marbling score, and shear force value to consumer acceptance of beef strip loin steaks. J. Anim. Sci. 2003, 81, 2741–2750. [Google Scholar] [CrossRef]
- Kinman, L.A.; Hilton, G.G.; Richards, C.J.; Morgan, J.B.; Krehbiel, C.R.; Hicks, R.B.; Dillwith, J.W.; VanOverbeke, D.L. Impact of feeding various amounts of wet and dry distillers grains to yearling steers on palatability, fatty acid profile, and retail case life of longissimus muscle. J. Anim. Sci. 2011, 89, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Poste, L.M.; Butler, G.; Mackie, D.; Agar, V.E.; Thompson, B.K.; Cliplef, R.L.; McKay, R.M. Correlations of sensory and instrumental meat tenderness values as affected by sampling techniques. Food Qual. Prefer. 1993, 4, 207–214. [Google Scholar] [CrossRef]
- Resconi, V.C.; Campo, M.M.; Font i Furnols, M.; Montossi, F.; Sañudo, C. Sensory quality of beef from different finishing diets. Meat Sci. 2010, 86, 865–869. [Google Scholar] [CrossRef]
- Pighin, D.; Pazos, A.; Chamorro, V.; Paschetta, F.; Cunzolo, S.A.; Godoy, F.; Messina, V.; Pordomingo, A.J.; Grigioni, G. A contribution of beef to human health: A review of the role of the animal production systems. Sci. World J. 2016, 2016, 8681491. [Google Scholar] [CrossRef]
- Nantapo, C.W.T.; Muchenje, V.; Nkukwana, T.T.; Hugo, A.; Descalzo, A.; Grigioni, G.; Hoffman, L.C. Socio-economic dynamics and innovative technologies affecting health-related lipid content in diets: Implications on global food and nutrition security. Food Res. Int. 2015, 76, 896–905. [Google Scholar] [CrossRef]
- Mapiye, C.; Vahmani, P.; Mlambo, V.; Muchenje, V.; Dzama, K.; Hoffman, L.C.; Dugan, M.E.R. The trans-octadecenoic fatty acid profile of beef: Implications for global food and nutrition security. Food Res. Int. 2015, 76, 992–1000. [Google Scholar] [CrossRef]
- de Mello, A.S.; Jenschke, B.E.; Senaratne, L.S.; Carr, T.P.; Erickson, G.E.; Calkins, C.R. Effects of finishing diets containing wet distillers grains plus solubles on beef quality attributes and fatty acid profile. Meat Sci. 2018, 136, 16–22. [Google Scholar] [CrossRef]
- Pordomingo, A.J.; García, T.P.; Volpi Lagreca, G. Effect of feeding treatment during the backgrounding phase of beef production from pasture on: II. Longissimus muscle proximate composition, cholesterol and fatty acids. Meat Sci. 2012, 90, 947–955. [Google Scholar] [CrossRef] [PubMed]
- Silva, R.G.D.; Prado, I.N.D.; Matsushita, M.; Souza, N.E. Dietary effects on muscle fatty acid composition of finished heifers. Pesqui. Agropecu. Bras. 2002, 37, 95–101. [Google Scholar] [CrossRef]
- Bhutta, Z. Protein: Digestibility and availability. In Encyclopedia of Human Nutrition; Sadler, M., Strain, J., Caballero, B., Eds.; Academic Press: San Diego, CA, USA, 1999; pp. 1646–1656. [Google Scholar]
- Lee, S.-H.; Kim, C.-N.; Ko, K.-B.; Park, S.-P.; Kim, H.-K.; Kim, J.-M.; Ryu, Y.-C. Comparisons of Beef Fatty Acid and Amino Acid Characteristics between Jeju Black Cattle, Hanwoo, and Wagyu Breeds. Food Sci. Anim. Resour. 2019, 39, 402–409. [Google Scholar] [CrossRef]
- Joint FAO/WHO/UNU Expert Consultation on Protein and Amino Acid Requirements in Human Nutrition (2002: Geneva, Switzerland), Food and Agriculture Organization of the United Nations, World Health Organization & United Nations University. Protein and Amino Acid Requirements in Human Nutrition: Report of a Joint FAO/WHO/UNU Expert Consultation; WHO technical report series; World Health Organization: Geneva, Switzerland, 2007; p. 935. Available online: https://apps.who.int/iris/handle/10665/43411 (accessed on 10 July 2023).
- Alekseeva, E.; Kolchina, V. Amino acid composition of beef obtained fromthe specialized meat cattle. IOP Conf. Ser. Earth Environ. Sci. 2019, 341, 012136. [Google Scholar] [CrossRef]
- Young, V.R.; Pellett, P.L. Amino acid composition in relation to protein nutritional quality of meat and poultry products. Am. J. Clin. Nutr. 1984, 40, 737–742. [Google Scholar] [CrossRef] [PubMed]
- Bergen, W.G. Amino Acids in Beef Cattle Nutrition and Production. In Amino Acids in Nutrition and Health: Advances in Experimental Medicine and Biology; Wu, G., Ed.; Springer International Publishing: Cham, Switzerland, 2021; pp. 29–42. ISBN 978-3-030-54462-1. [Google Scholar]
- Patel, M.; Sonesson, U.; Hessle, A. Upgrading plant amino acids through cattle to improve the nutritional value for humans: Effects of different production systems. Animal 2017, 11, 519–528. [Google Scholar] [CrossRef] [PubMed]
- Joint FAO/WHO/UNU Expert Consultation on Energy and Protein Requirements (1981: Rome, Italy), Food and Agriculture Organization of the United Nations, WHO, United Nations University. Energy and Protein Requirements: Report of a Joint FAO/WHO/UNU Expert Consultation [Held in Rome from 5 to 17 October 1981]; World Health Organization technical report series; World Health Organization: Geneva, Switzerland, 1985; p. 724. Available online: https://apps.who.int/iris/handle/10665/39527 (accessed on 10 July 2023).
- FASS. Guide for the Care and Use of Agricultural Animals in Research and Teaching, 3rd ed.; FASS Inc.: Champaign, IL, USA, 2010; Available online: www.fass.org (accessed on 15 July 2023).
| Parameter | Diets 1 | SEM 2 | p-Value | ||
|---|---|---|---|---|---|
| P | PDG | PRC | |||
| pH | 5.56 | 5.63 | 5.58 | 0.05 | 0.55 |
| Thawing loss, % | 4.48 | 3.15 | 3.38 | 0.29 | 0.10 |
| Cooking loss, % | 24.90 b | 26.25 a | 23.89 b | 0.42 | 0.005 |
| Meat color | |||||
| Lightness L* | 31.10 | 30.67 | 30.34 | 0.74 | 0.77 |
| Redness a* | 22.57 | 22.46 | 22.75 | 0.92 | 0.97 |
| Yellowness b* | 9.71 | 8.85 | 9.74 | 0.69 | 0.60 |
| Fat color | |||||
| Lightness L* | 68.32 | 67.36 | 70.22 | 1.34 | 0.33 |
| Yellowness b* | 18.72 a | 14.55 b | 14.82 b | 0.93 | 0.01 |
| Mb, mg/g meat | 3.36 a | 2.37 b | 3.24 ab | 0.24 | 0.02 |
| Browning index (A503/A581) | 0.51 | 0.56 | 0.61 | 0.07 | 0.23 |
| WBSF, N | 40.65 b | 46.86 a | 47.70 a | 1.92 | 0.039 |
| Toughness, N/mm s | 201.55 | 212.48 | 225.57 | 12.89 | 0.35 |
| Parameter 1 | Diets 2 | SEM 3 | p-Value | ||
|---|---|---|---|---|---|
| P | PDG | PRC | |||
| IMF (g/100 g muscle) | 2.21 | 2.48 | 2.00 | 0.42 | 0.73 |
| Fatty acid profile | |||||
| C 14:0, % | 2.76 | 2.83 | 2.71 | 0.25 | 0.94 |
| C 15:0, % | 0.71 | 0.73 | 0.72 | 0.08 | 0.99 |
| C 16:0, % | 27.12 | 27.14 | 26.53 | 0.67 | 0.77 |
| C 16:1 c-9, % | 3.28 | 3.37 | 3.50 | 0.23 | 0.81 |
| C 17:0, % | 0.75 a | 0.62 b | 0.64 a,b | 0.03 | 0.024 |
| C 18:0, % | 13.51 | 12.50 | 12.42 | 0.46 | 0.21 |
| C 18:1 t, % | 1.32 b | 2.14 a | 1.61 a,b | 0.21 | 0.038 |
| C 18:1 n-9 cis, % | 37.93 | 36.53 | 34.99 | 0.81 | 0.07 |
| C 18:1 c-11, % | 1.46 | 1.46 | 1.60 | 0.06 | 0.16 |
| C 18:1 c-12, % | 0.26 | 0.14 | 0.16 | 0.04 | 0.17 |
| C 18:2 n-6, % | 3.26 | 3.79 | 4.71 | 0.42 | 0.08 |
| C 18:3 n-3, % | 0.89 b | 0.88 b | 1.30 a | 0.09 | 0.007 |
| CLA, % | 0.28 | 0.31 | 0.35 | 0.02 | 0.10 |
| C 20:4 n-6, % | 0.73 | 1.22 | 1.66 | 0.26 | 0.07 ŧ |
| C 20:5 n-3, % | 0.26 | 0.39 | 0.54 | 0.08 | 0.07 |
| C 22:4 n-6, % | 0.10 b | 0.08 b | 0.17 a | 0.02 | 0.012 |
| C 22:6 n-3, % | 0.48 | 0.68 | 0.86 | 0.12 | 0.11 |
| SFA % | 43.40 | 42.46 | 41.67 | 0.89 | 0.41 |
| MUFA, % | 41.21 | 39.91 | 38.49 | 0.95 | 0.16 |
| PUFA, % | 5.74 b | 7.64 a,b | 9.22 a | 0.87 | 0.041 |
| n-3 PUFA, % | 1.81 | 1.94 | 2.69 | 0.27 | 0.07 |
| n-6 PUFA, % | 3.28 b | 3.88 a,b | 4.87 a | 0.42 | 0.05 |
| n-6 PUFA/n-3 PUFA | 2.09 | 2.10 | 1.83 | 0.12 | 0.22 |
| MUFA/SFA | 0.95 | 0.94 | 0.93 | 0.03 | 0.89 |
| PUFA/SFA | 0.14 b | 0.19 a,b | 0.23 a | 0.02 | 0.047 |
| TI | 1.58 | 1.52 | 1.39 | 0.07 | 0.18 |
| AI | 0.79 | 0.82 | 0.79 | 0.05 | 0.84 |
| Parameter | Diets 1 | SEM 2 | p-Value | ||
|---|---|---|---|---|---|
| P | PDG | PRC | |||
| Alanine (Ala), % | 6.37 a | 5.94 b | 6.07 a,b | 0.10 | 0.048 |
| Arginine (Arg), % | 0.35 b | 7.26 a | 7.30 a | 0.06 | 0.0001 |
| Aspartate (Asp), % | 10.49 | 8.68 | 10.24 | 0.44 | 0.05 |
| Cysteine (Cys), % | 0.94 | 0.98 | 0.88 | 0.06 | 0.598 |
| Glutamate (Glu), % | 14.73 a | 13.31 b | 13.46 a,b | 0.29 | 0.027 |
| Glycine (Gly), % | 4.94 a | 4.65 b | 4.70 b | 0.05 | 0.009 |
| Histidine (His), % | 6.60 | 6.99 | 6.60 | 0.15 | 0.186 |
| Isoleucine (Ile), % | 4.11 | 4.36 | 4.21 | 0.12 | 0.423 |
| Leucine (Leu), % | 8.12 a | 7.69 b | 7.60 b | 0.06 | 0.001 |
| Lysine (Lys), % | 9.10 a | 8.68 b | 8.75 a,b | 0.08 | 0.021 |
| Methionine (Met), % | 3.69 | 3.13 | 3.18 | 0.15 | 0.075 ŧ |
| Phenylalanine (Phe), % | 4.34 c | 4.59 a | 4.48 b | 0.02 | 0.001 |
| Proline (Pro), % | 6.72 a | 4.69 a,b | 2.17 b | 0.75 | 0.015 |
| Serine (Ser), % | 5.34 | 5.43 | 5.62 | 0.15 | 0.446 |
| Threonine (Thr), % | 4.70 | 4.67 | 4.85 | 0.10 | 0.457 |
| Tryptophan (Trp), % | 0.40 b | 0.22 c | 0.63 a | 0.02 | 0.001 |
| Tyrosine (Tyr), % | 4.05 | 4.25 | 4.22 | 0.12 | 0.518 |
| Valine (Val), % | 4.12 | 4.42 | 2.96 | 0.16 | 0.051 ŧ |
| Ratio Met/Trp | 9.35 b | 14.61 a | 5.06 b | 1.17 | 0.003 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Merayo, M.; Pighin, D.; Cunzolo, S.; Veggetti, M.; Soteras, T.; Chamorro, V.; Pazos, A.; Grigioni, G. Meat Quality Traits in Beef from Heifers: Effect of including Distiller Grains in Finishing Pasture-Based Diets. Agriculture 2023, 13, 1977. https://doi.org/10.3390/agriculture13101977
Merayo M, Pighin D, Cunzolo S, Veggetti M, Soteras T, Chamorro V, Pazos A, Grigioni G. Meat Quality Traits in Beef from Heifers: Effect of including Distiller Grains in Finishing Pasture-Based Diets. Agriculture. 2023; 13(10):1977. https://doi.org/10.3390/agriculture13101977
Chicago/Turabian StyleMerayo, Manuela, Dario Pighin, Sebastián Cunzolo, Mariela Veggetti, Trinidad Soteras, Verónica Chamorro, Adriana Pazos, and Gabriela Grigioni. 2023. "Meat Quality Traits in Beef from Heifers: Effect of including Distiller Grains in Finishing Pasture-Based Diets" Agriculture 13, no. 10: 1977. https://doi.org/10.3390/agriculture13101977
APA StyleMerayo, M., Pighin, D., Cunzolo, S., Veggetti, M., Soteras, T., Chamorro, V., Pazos, A., & Grigioni, G. (2023). Meat Quality Traits in Beef from Heifers: Effect of including Distiller Grains in Finishing Pasture-Based Diets. Agriculture, 13(10), 1977. https://doi.org/10.3390/agriculture13101977