Rice for Food Security: Revisiting Its Production, Diversity, Rice Milling Process and Nutrient Content
Abstract
1. Introduction
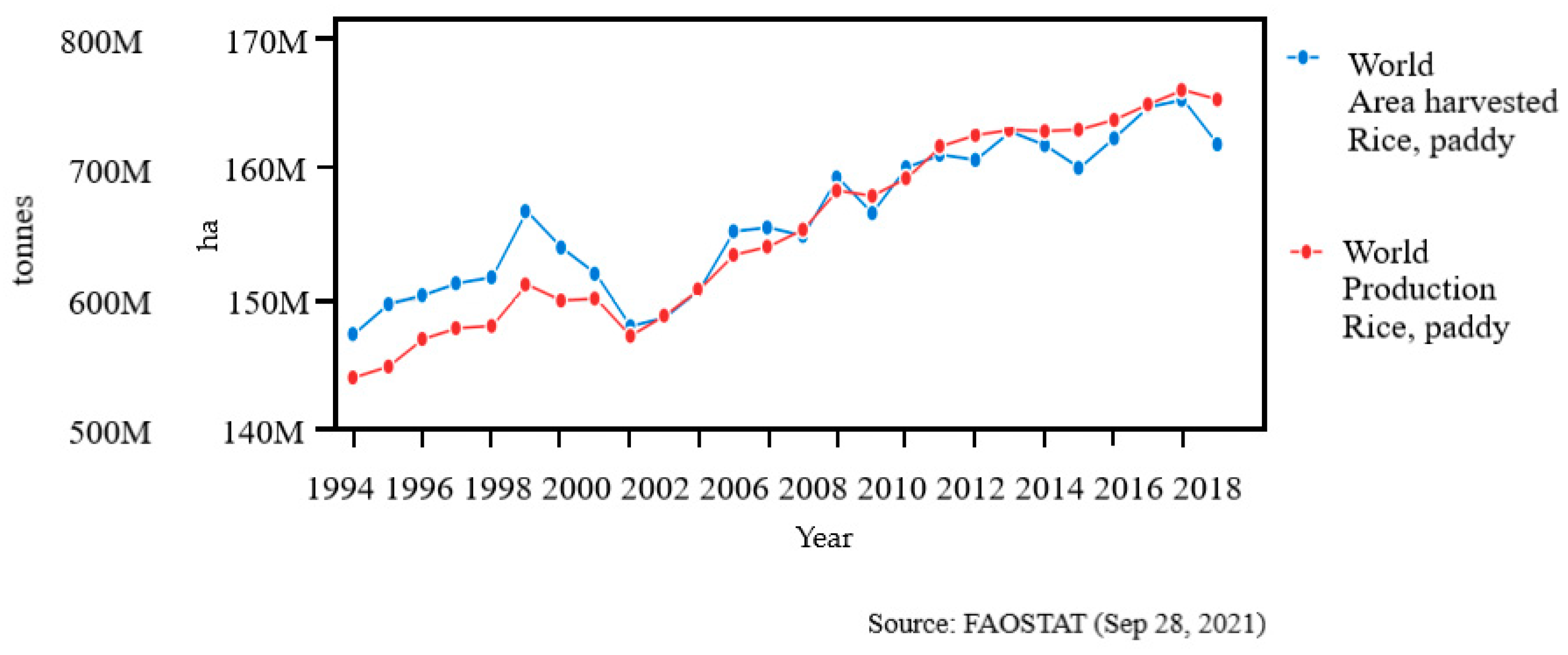
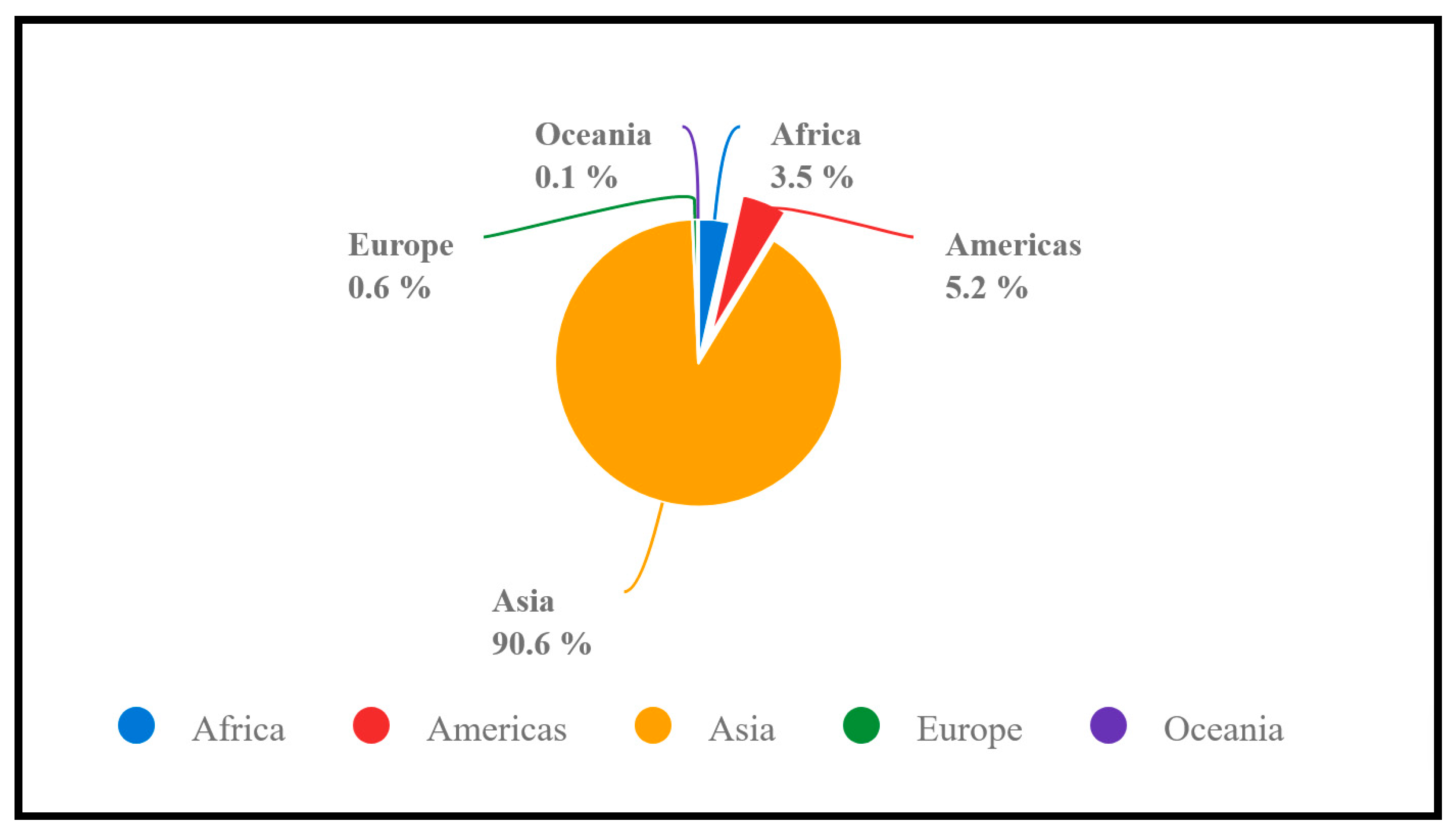
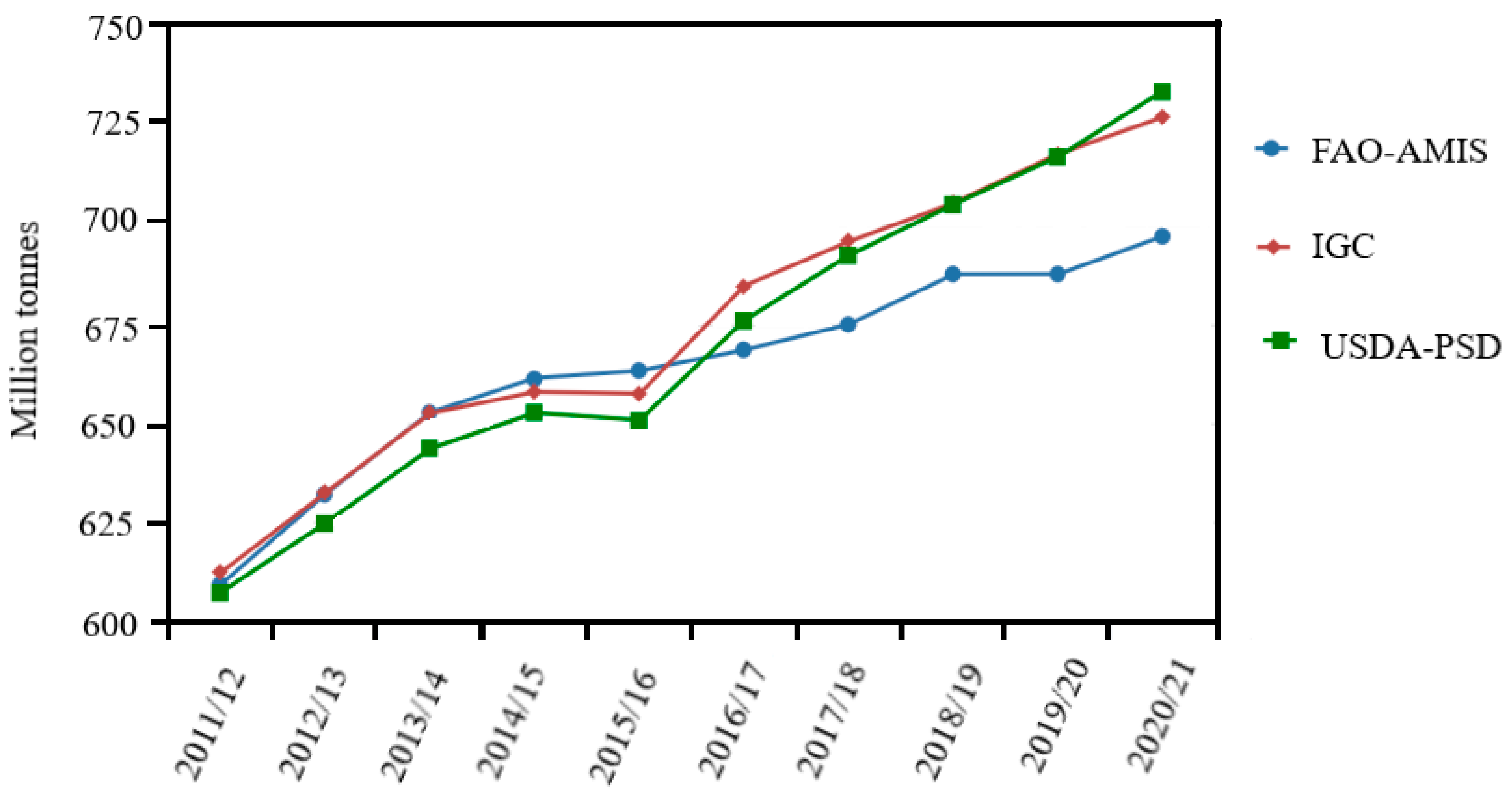
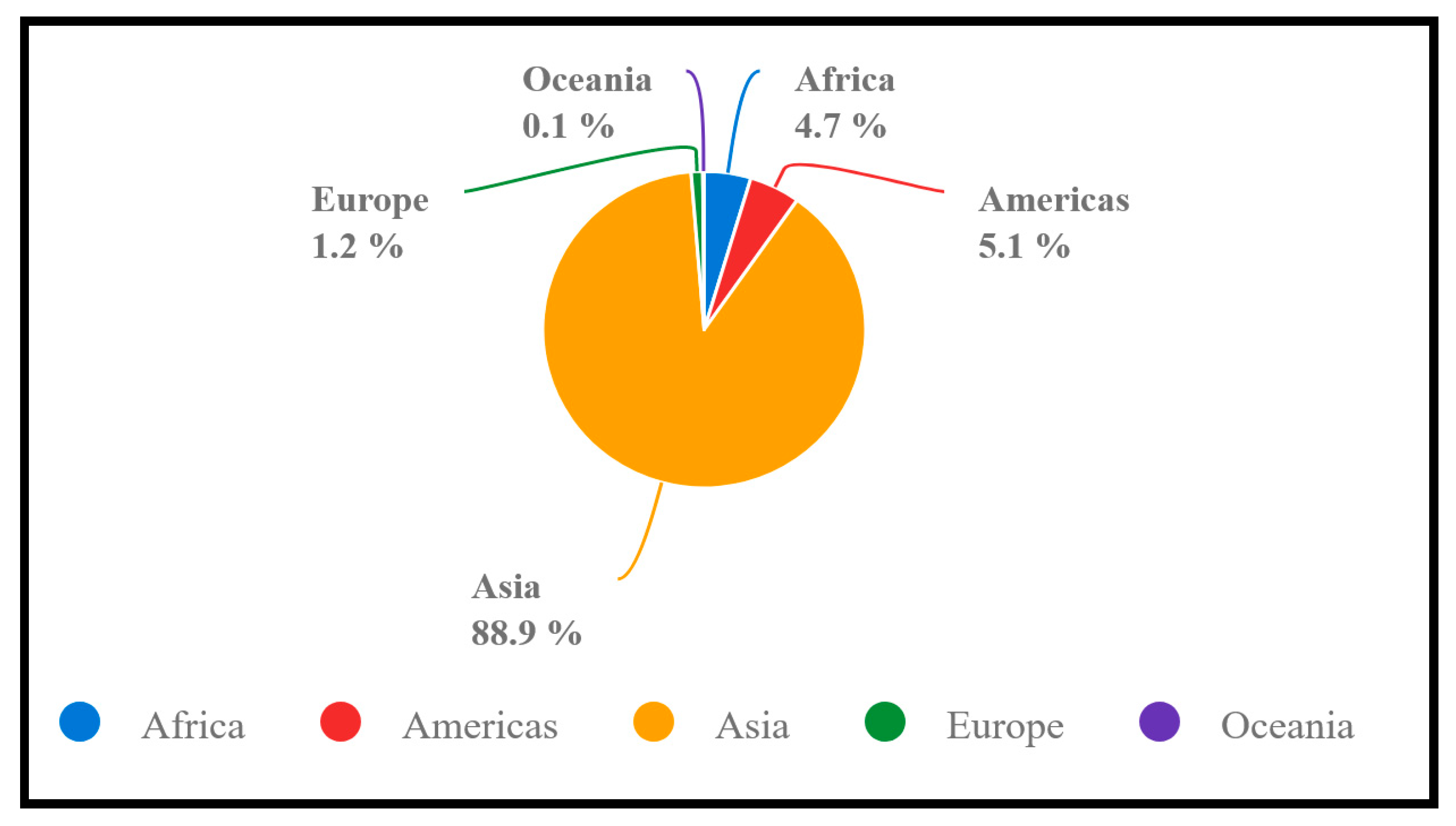
2. Global Rice Production, Consumption, and Ending Stock
3. Variety of Rice
3.1. White Rice
3.2. Brown Rice
3.3. Fragrant Rice
3.4. Basmati Rice
3.5. Ponni Rice
3.6. Glutinous or Waxy Rice
3.7. Red Rice
3.8. Japonica Rice
3.9. Weedy or Red Rice
3.10. Golden Rice
4. Composition of Rice Grain
4.1. Rice Anatomy
4.2. Starch
4.3. Protein
4.4. Lipids
4.5. Non-Starch Polysaccharides
4.6. Phenolic Compound
4.6.1. Phenolic Acids
4.6.2. Flavonoids
4.6.3. Proanthocyanidins and Anthocyanins
Proanthocyanidins
Anthocyanins
4.7. Volatile Components
5. Nutritional Value of Rice
6. Rice Processing
6.1. Storage of Paddy
6.2. Paddy Drying
6.3. Cleaning and Destoning
6.4. Dehusking and Separation of Husk
6.5. Whitening and Polishing
6.6. Rice Grading
7. Environmental Impacts of Rice Cultivation
8. Challenges to Overcoming Barriers and Current Policy Directions
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhao, M.; Lin, Y.; Chen, H. Improving nutritional quality of rice for human health. Theor. Appl. Genet. 2020, 133, 1397–1413. [Google Scholar] [CrossRef] [PubMed]
- FAOSTAT. Production/Yield Quantities of Rice, Paddy in World + (Total). Food and Agriculture Organization of the United Nations, 2021. Available online: https://www.fao.org/faostat/en/#data/QCL/visualize (accessed on 1 September 2021).
- USDA. World Rice Production 2021/2022. 2022. Available online: http://www.worldagriculturalproduction.com/crops/rice.aspx/ (accessed on 1 September 2021).
- Bandumula, N. Rice Production in Asia: Key to Global Food Security. Proc. Natl. Acad. Sci. India Sect. B Biol. Sci. 2018, 88, 1323–1328. [Google Scholar] [CrossRef]
- OECD/FAO. OECD-FAO Agricultural Outlook 2021–2030; OECD Publishing: Paris, France, 2021; pp. 7–8. [Google Scholar] [CrossRef]
- Van Oort, P.A.J.; Saito, K.; Tanaka, A.; Amovin-Assagba, E.; Van Bussel, L.G.J.; Van Wart, J.; de Groot, H.; van Ittersum, M.K.; Cassman, K.G.; Wopereis, M.C.S. Assessment of rice self-sufficiency in 2025 in eight African countries. Glob. Food Secur. 2015, 5, 39–49. [Google Scholar] [CrossRef]
- AMIS. AMIS MARKET MONITOR No. 94—December 2021. 2021. Available online: http://www.amis-outlook.org/fileadmin/user_upload/amis/docs/Market_monitor/AMIS_Market_Monitor_Issue_94.pdf (accessed on 1 October 2021).
- FAO. Crop Prospects and Food Situation—Quarterly Global Report No. 2, July 2021. Rome. 2021. Available online: https://doi.org/10.4060/cb5603en (accessed on 1 September 2021).
- von Goh, E.; Azam-Ali, S.; McCullough, F.; Mitra, S.R. The nutrition transition in Malaysia; Key drivers and recommendations for improved health outcomes. BMC Nutr. 2020, 6, 32. [Google Scholar] [CrossRef]
- Statista. Rice Consumption Worldwide in 2021/2022, by Country (in 1000 metric tons). 2021. Available online: https://www.statista.com/statistics/255971/top-countries-based-on-rice-consumption-2012-2013/ (accessed on 1 September 2021).
- International Rice Research Institute. Rice Consumption with Center Pivots and Linears; International Rice Research Institute: Los Baños, Philippines, 2013; Volume 12. [Google Scholar]
- Schreinemachers, P.; Simmons, E.B.; Wopereis, M.C.S. Tapping the economic and nutritional power of vegetables. Glob. Food Secur. 2018, 16, 36–45. [Google Scholar] [CrossRef]
- Samal, P.; Babu, S. The shape of rice agriculture towards 2050. In Proceedings of the 30th International Conference of Agricultural Economists, Vancouver, BC, Canada, 28 July–2 August 2018. [Google Scholar]
- Reardon, T.; Tschirley, D.; Liverpool-Tasie, L.S.O.; Awokuse, T.; Fanzo, J.; Minten, B.; Vos, R.; Dolislager, M.; Sauer, C.; Dhar, R.; et al. The processed food revolution in African food systems and the double burden of malnutrition. Glob. Food Secur. 2021, 28, 100466. [Google Scholar] [CrossRef]
- Fukagawa, N.K.; Ziska, L.H. Rice: Importance for global nutrition. J. Nutr. Sci. Vitaminol. 2019, 65, S2–S3. [Google Scholar] [CrossRef]
- Chauhan, B.S.; Jabran, K.; Mahajan, G. Rice Production Worldwide; Springer: Cham, Switzerland, 2017; pp. 1–563. [Google Scholar] [CrossRef]
- Priya, T.S.R.; Nelson, A.R.L.E.; Ravichandran, K.; Antony, U. Nutritional and functional properties of coloured rice varieties of South India: A review. J. Ethn. Foods 2019, 6, 11. [Google Scholar] [CrossRef]
- Calingacion, M.; Laborte, A.; Nelson, A.; Resurreccion, A.; Concepcion, J.C.; Daygon, V.D.; Mumm, R.; Reinke, R.; Dipti, S.; Bassinello, P.Z.; et al. Diversity of global rice markets and the science required for consumer-targeted rice breeding. PLoS ONE 2014, 9, e85106. [Google Scholar] [CrossRef]
- Qiu, X.; Pang, Y.; Yuan, Z.; Xing, D.; Xu, J.; Dingkuhn, M.; Li, Z.; Ye, G. Genome-wide association study of grain appearance and milling quality in a worldwide collection of Indica rice germplasm. PLoS ONE 2015, 10, e145577. [Google Scholar] [CrossRef]
- Weerawatanakorn, M.; Wu, J.C.; Pan, M.H.; Ho, C.T. Reactivity and stability of selected flavor compounds. J. Food Drug Anal. 2015, 23, 176–190. [Google Scholar] [CrossRef] [PubMed]
- BERNAS. Rice Type in Malaysia. 2022. Available online: https://www.bernas.com.my/bernas/index.php/ricepedia/rice-type-in-malaysia (accessed on 1 January 2022).
- Cho, D.H.; Lim, S.T. Germinated brown rice and its bio-functional compounds. Food Chem. 2016, 196, 259–271. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.; Li, Y.; Li, Y.; Ma, L.; Ashraf, U.; Tang, X.; Pan, S.; Tian, H.; Mo, Z. Application of γ-aminobutyric acid under low light conditions: Effects on yield, aroma, element status, and physiological attributes of fragrant rice. Ecotoxicol. Environ. Saf. 2021, 213, 111941. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.; Singh, A.K.; Mohapatra, T.; Krishnan, S.G.; Ellur, R.K. Pusa Basmati 1121—A rice variety with exceptional kernel elongation and volume expansion after cooking. Rice 2018, 11, 19. [Google Scholar] [CrossRef]
- Bera, A. Basmati rice: A new hope for farmers. Agric. Food e-Newslett. 2020, 2, 819–821. [Google Scholar] [CrossRef]
- Prodhan, Z.H.; Qingyao, S. Rice Aroma: A Natural Gift Comes with Price and the Way Forward. Rice Sci. 2020, 27, 86–100. [Google Scholar] [CrossRef]
- Ramasamy, S.; Robin, S.I.; Muthurajan, R. Developing improved versions of a popular rice variety (Improved White Ponni) through marker assisted backcross breeding. Green Farming 2020, 8, 506–511. [Google Scholar]
- Ramchander, S.; Ushakumari, R.; Arumugam, M.P. Quality characteristics of rice mutants generated through gamma radiation in white ponni quality characteristics of rice mutants generated through gamma radiation in white ponni. Int. J. Agric. Sci. 2015, 7, 719–723. [Google Scholar]
- Nawaz, M.A. Processing and Quality of Glutinous Rice; The University of Queensland: St. Lucia, QLD, Australia, 2018. [Google Scholar]
- Qiu, S.; Abbaspourrad, A.; Padilla-Zakour, O.I. Changes in the Glutinous Rice Grain and Physicochemical Electric Field. Foods 2021, 10, 395. [Google Scholar] [CrossRef]
- Agustin, A.T.; Safitri, A.; Fatchiyah, F. Java red rice (Oryza sativa L.) nutritional value and anthocyanin profiles and its potential role as antioxidant and anti-diabetic. Indones. J. Chem. 2021, 21, 968–978. [Google Scholar] [CrossRef]
- Gong, M.; Zhou, Z.; Liu, S.; Zhu, S.; Li, G.; Zhong, F.; Mao, J. Dynamic changes in physico-chemical attributes and volatile compounds during fermentation of Zhenjiang vinegars made with glutinous and non-glutinous japonica rice. J. Cereal Sci. 2021, 100, 103246. [Google Scholar] [CrossRef]
- Ratnasekera, D. Weedy rice: A threat to rice production in Sri Lanka. J. Univ. Ruhuna 2015, 3, 2. [Google Scholar] [CrossRef]
- Dimitrovski, T.; Andreevska, D.; Andov, D. Morphological and grain characterisation of Macedonian weedy rice (Oryza sativa L.). Maced. J. Ecol. Environ. 2018, 20, 5–17. [Google Scholar]
- de Steur, H.; Stein, A.J.; Demont, M. From Golden Rice to Golden Diets: How to turn its recent approval into practice. Glob. Food Secur. 2022, 32, 100596. [Google Scholar] [CrossRef] [PubMed]
- Dubock, A. The present status of Golden Rice. J. Huazhong Agric. 2014, 33, 69–84. [Google Scholar]
- Rodríguez, A.V.; Rodríguez-Oramas, C.; Velázquez, E.S.; de la Torre, A.H.; Armendáriz, C.R.; Iruzubieta, C.C. Myths and Realities about Genetically Modified Food: A Risk-Benefit Analysis. Appl. Sci. 2022, 12, 2861. [Google Scholar] [CrossRef]
- Zhu, Q.; Zeng, D.; Yu, S.; Cui, C.; Li, J.; Li, H.; Chen, J.; Zhang, R.; Zhao, X.; Chen, L.; et al. From Golden Rice to aSTARice: Bioengineering Astaxanthin Biosynthesis in Rice Endosperm. Mol. Plant 2018, 11, 1440–1448. [Google Scholar] [CrossRef]
- Swamy, B.M.; Samia, M.; Boncodin, R.; Marundan, S.; Rebong, D.B.; Ordonio, R.L.; Miranda, R.T.; Rebong, A.T.O.; Alibuyog, A.Y.; Adeva, C.C.; et al. Compositional Analysis of Genetically Engineered GR2E ‘golden Rice’ in Comparison to That of Conventional Rice. J. Agric. Food Chem. 2019, 67, 7986–7994. [Google Scholar] [CrossRef]
- Mallikarjuna Swamy, B.P.; Marundan, S.; Samia, M.; Ordonio, R.L.; Rebong, D.B.; Miranda, R.; Alibuyog, A.; Rebong, A.T.; Tabil, M.A.; Suralta, R.R.; et al. Development and characterization of GR2E Golden rice introgression lines. Sci. Rep. 2021, 11, 2496. [Google Scholar] [CrossRef]
- Hoogenkamp, H.; Kumagai, H.; Wanasundara, J.P.D. Rice Protein and Rice Protein Products; Elsevier Inc.: Amsterdam, The Netherlands, 2017. [Google Scholar] [CrossRef]
- Cornejo-Ramírez, Y.I.; Martínez-Cruz, O.; del Toro-Sánchez, C.L.; Wong-Corral, F.J.; Borboa-Flores, J.; Cinco-Moroyoqui, F.J. The structural characteristics of starches and their functional properties. CYTA-J. Food 2018, 16, 1003–1017. [Google Scholar] [CrossRef]
- Zhang, H.; Jang, S.G.; Lar, S.M.; Lee, A.R.; Cao, F.Y.; Seo, J.; Kwon, S.W. Genome-Wide Identification and Genetic Variations of the Starch Synthase Gene Family in Rice. Plants 2021, 10, 1154. [Google Scholar] [CrossRef] [PubMed]
- Belitz, H.-D.; Rosch, W.; Schieberle, P. Food Chemistry; Elsevier: Amsterdam, The Netherlands, 2009. [Google Scholar] [CrossRef]
- Bertoft, E. Understanding starch structure: Recent progress. Agronomy 2017, 7, 56. [Google Scholar] [CrossRef]
- Maung, T.Z.; Yoo, J.M.; Chu, S.H.; Kim, K.W.; Chung, I.M.; Park, Y.J. Haplotype Variations and Evolutionary Analysis of the Granule-Bound Starch Synthase I Gene in the Korean World Rice Collection. Front. Plant Sci. 2021, 12, 707237. [Google Scholar] [CrossRef] [PubMed]
- Madhusankha, G.D.M.; Thilakarathna, R.C. Meat tenderization mechanism and the impact of plant exogenous proteases: A review. Arab. J. Chem. 2020, 14, 102967. [Google Scholar] [CrossRef]
- Alcázar-Alay, S.C.; Meireles, M.A.A. Physicochemical properties, modifications and applications of starches from different botanical sources. Food Sci. Technol. 2015, 35, 215–236. [Google Scholar] [CrossRef]
- Vlad-Oros, B.; Preda, G.; Dudas, Z.; Dragomirescu, M.; Chiriac, A. Entrapment of glucoamylase by sol-gel technique in PhTES/TEOS hybrid matrixes. Process. Appl. Ceram. 2007, 1, 63–67. [Google Scholar] [CrossRef]
- Peng, X. Advanced Characterization of Glucan Particulates: Small-Granule Starches, Retention of Small Molecules and Local … no. December. 2018. Available online: http://pstorage-purdue-258596361474.s3.amazonaws.com/13701095/XingyunPengsDissertation_1201182.pdf (accessed on 1 September 2021).
- Cornejo, F.; Rosell, C.M. Physicochemical properties of long rice grain varieties in relation to gluten free bread quality. LWT-Food Sci. Technol. 2015, 62, 1203–1210. [Google Scholar] [CrossRef]
- Uraipong, C. Investigation into the Biological Functions of Rice Bran Protein Hydrolysates. Ph.D. Thesis, The University of New South Wales, Sydney, NSW, Australia, 2016; pp. 1–219. [Google Scholar]
- Pedrazzini, E.; Mainieri, D.; Marrano, C.A.; Vitale, A. Where do protein bodies of cereal seeds come from? Front. Plant Sci. 2016, 7, 1139. [Google Scholar] [CrossRef]
- Roustan, V.; Hilscher, J.; Weidinger, M.; Reipert, S.; Shabrangy, A.; Gebert, C.; Dietrich, B.; Dermendjiev, G.; Schnurer, M.; Roustan, P.-J.; et al. Protein sorting into protein bodies during barley endosperm development is putatively regulated by cytoskeleton members, MVBs and the HvSNF7s. Sci. Rep. 2020, 10, 1864. [Google Scholar] [CrossRef]
- Mohd Fairulnizal, M.N.; Norhayati, M.K.; Zaiton, A.; Norliza, A.H.; Rusidah, S.; Aswir, A.R.; Suraiami, M.; Mohd Naeem, M.N.; Jo-Lyn, A.; Mohd Azerulazree, J.; et al. Nutrient content in selected commercial rice in Malaysia: An update of Malaysian food composition database. Int. Food Res. J. 2015, 22, 768–776. [Google Scholar]
- Masumura, T.; Shigemitsu, T.; Morita, S.; Satoh, S. Identification of the region of rice 13 KDa prolamin essential for the formation of ER-derived protein bodies using a heterologous expression system. Biosci. Biotechnol. Biochem. 2015, 79, 566–573. [Google Scholar] [CrossRef] [PubMed]
- Saito, Y.; Shigemitsu, T.; Yamasaki, R.; Sasou, A.; Goto, F.; Kishida, K.; Kuroda, M.; Tanaka, K.; Morita, S.; Satoh, S.; et al. Formation mechanism of the internal structure of type I protein bodies in rice endosperm: Relationship between the localization of prolamin species and the expression of individual genes. Plant J. 2012, 70, 1043–1055. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.-Y.; Kim, J.-H.; Rico, C.-W.; Nam, S.-H. A comparative study on the physicochemical characteristics of black rice varieties. Int. J. Food Prop. 2011, 14, 1241–1254. [Google Scholar] [CrossRef][Green Version]
- Yoon, M.R.; Rico, C.W.; Koh, H.J.; Kang, M.Y. A study on the lipid components of rice in relation to palatability and storage. J. Korean Soc. Appl. Biol. Chem. 2012, 55, 515–521. [Google Scholar] [CrossRef]
- Zhang, X.; Yi, S.; Ning, Z.; Jinsong, B.; Wu, D.; Xiaoli, S. The effects of internal endosperm lipids on starch properties: Evidence from rice mutant starches. J. Cereal Sci. 2019, 89, 102804. [Google Scholar] [CrossRef]
- Lu, H.; Jiang, H.; Chen, Q. Determination of fatty acid content of rice during storage based on feature fusion of olfactory visualization sensor data and near-infrared spectra. Sensors 2021, 21, 3266. [Google Scholar] [CrossRef]
- Orsavova, J.; Misurcova, L.; Ambrozova, J.V.; Vicha, R.; Mlcek, J. Fatty acids composition of vegetable oils and its contribution to dietary energy intake and dependence of cardiovascular mortality on dietary intake of fatty acids. Int. J. Mol. Sci. 2015, 16, 12871–12890. [Google Scholar] [CrossRef]
- Devi, A.; Khatkar, B.S. Effects of fatty acids composition and microstructure properties of fats and oils on textural properties of dough and cookie quality. J. Food Sci. Technol. 2018, 55, 321–330. [Google Scholar] [CrossRef]
- Mohamed, I.O. Effects of processing and additives on starch physicochemical and digestibility properties. Carbohydr. Polym. Technol. Appl. 2021, 2, 100039. [Google Scholar] [CrossRef]
- Bao, J. Rice Starch; Elsevier Inc. in Cooperation with AACC International: Amsterdam, The Netherlands, 2018. [Google Scholar] [CrossRef]
- Wattanavanitchakorn, S.; Wansuksri, R.; Chaichoompu, E. Biochemical and molecular assessment of cooking quality and nutritional value of pigmented and non-pigmented whole grain rice. Preprints 2021. [Google Scholar] [CrossRef]
- Chawla, R.; Patil, G.R. Fiber. Compr. Rev. Food Sci. Food Safe 2010, 9, 178–196. [Google Scholar] [CrossRef]
- Sethy, K.; Mishra, S.K.; Mohanty, P.P.; Agarawal, J.; Meher, P.; Satapathy, D.; Sahoo, J.K.; Panda, S.; Nayak, S.M. An overview of Non Starch Polysaccharide. J. Anim. Nutr. Physiol. 2015, 1, 17–22. [Google Scholar]
- Sinha, A.K.; Kumar, V.; Makkar, H.P.S.; de Boeck, G.; Becker, K. Non-starch polysaccharides and their role in fish nutrition—A review. Food Chem. 2011, 127, 1409–1426. [Google Scholar] [CrossRef]
- Hartini, S.; Choct, M. The effects of diets containing different level of non-starch polysaccharides on performance and cannibalism in laying hens. J. Indones. Trop. Anim. Agric. 2010, 35, 145–150. [Google Scholar] [CrossRef][Green Version]
- Harholt, J.; Suttangkakul, A.; Scheller, H.V. Biosynthesis of pectin. Plant Physiol. 2010, 153, 384–395. [Google Scholar] [CrossRef]
- van Dam, J.E.G.; van den Broek, L.A.M.; Boeriu, C.G. Polysaccharides in human health care. Nat. Prod. Commun. 2017, 12, 821–830. [Google Scholar] [CrossRef]
- WCRF/AICR. Recommendations and public health and policy implications. In Continous Update Project; WCRF/AICR: London, UK, 2018. [Google Scholar]
- Uthumporn, U.; Nadiah, I.; Izzuddin, I.; Cheng, L.H.; Aida, H. Physicochemical characteristics of non-starch polysaccharides extracted from cassava tubers. Sains Malays. 2017, 46, 223–229. [Google Scholar] [CrossRef]
- Krishnanunni, K.; Senthilvel, P.; Ramaiah, S.; Anbarasu, A. Study of chemical composition and volatile compounds along with in-vitro assay of antioxidant activity of two medicinal rice varieties: Karungkuravai and Mappilai samba. J. Food Sci. Technol. 2015, 52, 2572–2584. [Google Scholar] [CrossRef]
- Ashokkumar, K.; Govindaraj, M.; Vellaikumar, S.; Shobhana, V.G.; Karthikeyan, A.; Akilan, M.; Sathishkumar, J. Comparative Profiling of Volatile Compounds in Popular South Indian Traditional and Modern Rice Varieties by Gas Chromatography–Mass Spectrometry Analysis. Front. Nutr. 2020, 7, 260. [Google Scholar] [CrossRef]
- Cory, H.; Passarelli, S.; Szeto, J.; Tamez, M.; Mattei, J. The Role of Polyphenols in Human Health and Food Systems: A Mini-Review. Front. Nutr. 2018, 5, 87. [Google Scholar] [CrossRef]
- Altemimi, A.; Lakhssassi, N.; Baharlouei, A.; Watson, D.G.; Lightfoot, D.A. Phytochemicals: Extraction, isolation, and identification of bioactive compounds from plant extracts. Plants 2017, 6, 42. [Google Scholar] [CrossRef]
- Ciulu, M.; de la Luz Cádiz-Gurrea, M.; Segura-Carretero, A. Extraction and analysis of phenolic compounds in rice: A review. Molecules 2018, 23, 2890. [Google Scholar] [CrossRef]
- Shao, Y.; Hu, Z.; Yu, Y.; Mou, R.; Zhu, Z.; Beta, T. Phenolic acids, anthocyanins, proanthocyanidins, antioxidant activity, minerals and their correlations in non-pigmented, red, and black rice. Food Chem. 2018, 239, 733–741. [Google Scholar] [CrossRef]
- Wisetkomolmat, J.; Arjin, C.; Satsook, A.; Seel-Audom, M.; Ruksiriwanich, W.; Prom-u-Thai, C.; Sringarm, K. Comparative Analysis of Nutritional Components and Phytochemical Attributes of Selected Thai Rice Bran. Front. Nutr. 2022, 9, 833730. [Google Scholar] [CrossRef]
- Podgórska, A.; Burian, M.; Gieczewska, K. Altered Cell Wall Plasticity Can Restrict Plant Growth under Ammonium Nutrition. Front. Plant Sci. 2017, 8, 1344. [Google Scholar] [CrossRef]
- Speranza, S.; Knechtl, R.; Witlaczil, R.; Schönlechner, R. Reversed-Phase HPLC Characterization and Quantification and Antioxidant Capacity of the Phenolic Acids and Flavonoids Extracted from Eight Varieties of Sorghum Grown in Austria. Front. Plant Sci. 2021, 12, 769151. [Google Scholar] [CrossRef]
- Singla, R.K.; Dubey, A.K.; Garg, A.; Sharma, R.K.; Fiorino, M.; Ameen, S.M.; Haddad, M.A.; Al-Hiary, M. Natural Polyphenols: Chemical Classification, Definition of Classes, Subcategories, and Structures. J. AOAC Int. 2019, 102, 1397–1400. [Google Scholar] [CrossRef]
- Kumar, N.; Goel, N. Phenolic acids: Natural versatile molecules with promising therapeutic applications. Biotechnol. Rep. 2019, 24, e00370. [Google Scholar] [CrossRef]
- Ansari, M.-U.-R. Recent Advances in Rice Research; IntechOpen: London, UK, 2021. [Google Scholar] [CrossRef]
- Bhat, F.M.; Sommano, S.R.; Riar, C.S.; Seesuriyachan, P.; Chaiyaso, T.; Prom-U-thai, C. Status of bioactive compounds from bran of pigmented traditional rice varieties and their scope in production of medicinal food with nutraceutical importance. Agronomy 2020, 10, 1817. [Google Scholar] [CrossRef]
- Huang, Y.P.; Lai, H.M. Bioactive compounds and antioxidative activity of colored rice bran. J. Food Drug Anal. 2016, 24, 564–574. [Google Scholar] [CrossRef]
- Verma, D.K.; Srivastav, P.P. Bioactive compounds of rice (Oryza sativa L.): Review on paradigm and its potential benefit in human health. Trends Food Sci. Technol. 2020, 97, 355–365. [Google Scholar] [CrossRef]
- Rauf, A.; Imran, M.; Abu-izneid, T.; Patel, S. Proanthocyanidins: A comprehensive review. Biomed. Pharmacother. 2019, 116, 108999. [Google Scholar] [CrossRef]
- Gunaratne, A.; Wu, K.; Li, D.; Bentota, A.; Corke, H.; Cai, Y.Z. Antioxidant activity and nutritional quality of traditional red-grained rice varieties containing proanthocyanidins. Food Chem. 2013, 138, 1153–1161. [Google Scholar] [CrossRef]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef]
- Khoo, H.E. Anthocyanidins and anthocyanins: Colored pigments as food, pharmaceutical ingredients, and the potential health benefits. Food Nutr. Res. 2017, 61, 1361779. [Google Scholar] [CrossRef]
- Daiponmak, W.; Theerakulpisut, P.; Thanonkeo, P.; Vanavichit, A. Changes of anthocyanin cyanidin-3-glucoside content and antioxidant activity in Thai rice varieties under salinity stress. Sci. Asia 2010, 36, 286–291. [Google Scholar] [CrossRef]
- Bharti, S.; Anand, S. Fragrance and Aroma in Rice. Food Sci. Rep. 2020, 1. [Google Scholar] [CrossRef]
- Hinge, V.R.; Patil, H.B.; Nadaf, A.B. Aroma volatile analyses and 2AP characterization at various developmental stages in Basmati and Non-Basmati scented rice (Oryza sativa L.) cultivars. Rice 2016, 9, 38. [Google Scholar] [CrossRef]
- Verma, S.; Srivastava, S.; Tiwari, N. Comparative study on nutritional and sensory quality of barnyard and foxtail millet food products with traditional rice products. J. Food Sci. Technol. 2015, 52, 5147–5155. [Google Scholar] [CrossRef]
- Yin, W.; Hewson, L.; Linforth, R.; Taylor, M.; Fisk, I.D. Effects of aroma and taste, independently or in combination, on appetite sensation and subsequent food intake. Appetite 2017, 114, 265–274. [Google Scholar] [CrossRef]
- Liyanaarachchi, G.D.; Kottearachchi, N.S.; Samarasekera, R. Volatile profiles of traditional aromatic rice varieties in Sri Lanka. J. Natl. Sci. Found. Sri Lanka 2014, 42, 87–93. [Google Scholar] [CrossRef]
- Ramtekey, V.; Cherukuri, S.; Modha, K.G.; Kumar, A.; Kethineni, U.B.; Pal, G.; Singh, A.N.; Kumar, S. Extraction, characterization, quantification, and application of volatile aromatic compounds from Asian rice cultivars. Rev. Anal. Chem. 2021, 40, 272–292. [Google Scholar] [CrossRef]
- Kasote, D.; Singh, V.K.; Bollinedi, H.; Singh, A.K.; Sreenivasulu, N. Profiling of 2-Acetyl-1-Pyrroline and Other Volatile Compounds in Raw and Cooked Rice of Traditional and Improved Varieties. Foods 2021, 10, 1917. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Hui, T.; Fang, F.; Ma, Q.; Li, S.; Zhang, D. Characterization and Discrimination of Key Aroma Compounds in Pre- and Postrigor Roasted Mutton by GC-O-MS, GC E-Nose and Aroma Recombination Experiments. Foods 2021, 10, 2387. [Google Scholar] [CrossRef]
- Müller-Wirtz, L.M.; Kiefer, D.; Ruffing, S.; Brausch, T.; Hüppe, T.; Sessler, D.I.; Volk, T.; Fink, T.; Kreuer, S.; Maurer, F. Quantification of Volatile Aldehydes Deriving from In Vitro Lipid Peroxidation in the Breath of Ventilated Patients. Molecules 2021, 26, 3089. [Google Scholar] [CrossRef]
- Lin, J.-Y.; Fan, W.; Gao, Y.-N.; Wu, S.-F.; Wang, S.-X. Study on volatile compounds in rice by HS-SPME and GC-MS. In Proceedings of the 10th International Working Conference on Stored Product Protection, Estoril, Portugal, 27 June–2 July 2010; pp. 125–134. [Google Scholar] [CrossRef]
- Rasane, P.; Jha, A.; Sabikhi, L.; Kumar, A.; Unnikrishnan, V.S. Nutritional advantages of oats and opportunities for its processing as value added foods—A review. J. Food Sci. Technol. 2015, 52, 662–675. [Google Scholar] [CrossRef]
- Carcea, M. Value of wholegrain rice in a healthy human nutrition. Agriculture 2021, 11, 720. [Google Scholar] [CrossRef]
- Kapoor, B.; Kapoor, D.; Gautam, S.; Singh, R.; Bhardwaj, S. Dietary Polyunsaturated Fatty Acids (PUFAs): Uses and Potential Health Benefits. Curr. Nutr. Rep. 2021, 10, 232–242. [Google Scholar] [CrossRef]
- Nguyen, H.D.; Jo, W.H.; Hong, N.; Hoang, M.; Kim, M. International Immunopharmacology Anti-inflammatory effects of B vitamins protect against tau hyperphosphorylation and cognitive impairment induced by 1, 2 diacetyl benzene: An in vitro and in silico study. Int. Immunopharmacol. 2022, 108, 108736. [Google Scholar] [CrossRef]
- Lattimer, J.M.; Haub, M.D. Effects of dietary fiber and its components on metabolic health. Nutrients 2010, 2, 1266–1289. [Google Scholar] [CrossRef]
- Ma, Z.-Q.; Yi, C.-P.; Wu, N.-N.; Ban, T. Reduction of phenolic profiles, dietary fiber, and antioxidant activities of rice after treatment with different milling processes. Cereal Chem. 2020, 97, 1158–1171. [Google Scholar] [CrossRef]
- Paul, H.; Nath, B.C.; Bhuiyan, M.G.K.; Paul, S. Effect of Degree of Milling on Rice Grain Quality. J. Agric. Eng. 2020, 42, 69–76. [Google Scholar]
- Kim, S.H.; Yang, Y.J.; Chung, I.M. The effect of degree of milling on the nutraceutical content in ecofriendly and conventional rice (Oryza sativa L.). Foods 2020, 9, 1297. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Guan, X.; Li, C. Effects of degree of milling on the starch digestibility of cooked rice during (in vitro) small intestine digestion. Int. J. Biol. Macromol. 2021, 188, 774–782. [Google Scholar] [CrossRef] [PubMed]
- van der Kamp, J.W.; Poutanen, K.; Seal, C.J.; Richardson, D.P. The HEALTHGRAIN definition of ‘whole grain’. Food Nutr. Res. 2014, 58, 1–8. [Google Scholar] [CrossRef]
- Develaraja, S.; Reddy, A.; Yadav, M.; Jain, S.; Yadav, H. Whole Grains in Amelioration of Metabolic Derangements. J. Nutr. Health Food Sci. 2016, 4, 1–11. [Google Scholar]
- Hollænder, P.L.B.; Ross, A.B.; Kristensen, M. Whole-grain and blood lipid changes in apparently healthy adults: A systematic review and meta-analysis of randomized controlled studies1-3. Am. J. Clin. Nutr. 2015, 102, 556–572. [Google Scholar] [CrossRef]
- Aune, D.; Keum, N.; Giovannucci, E.; Fadnes, L.T.; Boffetta, P.; Greenwood, D.C.; Tonstad, S.; Vatten, L.J.; Riboli, E.; Norat, T. Whole grain consumption and risk of cardiovascular disease, cancer, and all cause and cause specific mortality: Systematic review and dose-response meta-analysis of prospective studies. BMJ 2016, 353, i2716. [Google Scholar] [CrossRef]
- Nokkoul, R. Organic upland rice seed production. Adv. J. Food Sci. Technol. 2014, 6, 1313–1317. [Google Scholar] [CrossRef]
- Gul, K.; Yousuf, B.; Singh, A.K.; Singh, P.; Wani, A.A. Rice bran: Nutritional values and its emerging potential for development of functional food—A review. Bioact. Carbohydr. Diet. Fibre 2015, 6, 24–30. [Google Scholar] [CrossRef]
- Kumar, A.; Priyadarshinee, R.; Roy, A.; Dasgupta, D.; Mandal, T. Current techniques in rice mill effluent treatment: Emerging opportunities for waste reuse and waste-to-energy conversion. Chemosphere 2016, 164, 404–412. [Google Scholar] [CrossRef] [PubMed]
- Olayanju, A.T.; Okonkwo, C.E.; Ojediran, J.O.; Hussain, S.Z.; Dottie, E.P.; Ayoola, A.S. Interactive effects and modeling of some processing parameters on milling, cooking, and sensory properties for Nigerian rice using a one-step rice milling machine. Heliyon 2021, 7, e06739. [Google Scholar] [CrossRef] [PubMed]
- Bodie, A.R.; Micciche, A.C.; Atungulu, G.G.; Rothrock, M.J.; Ricke, S.C. Current Trends of Rice Milling Byproducts for Agricultural Applications and Alternative Food Production Systems. Front. Sustain. Food Syst. 2019, 3, 47. [Google Scholar] [CrossRef]
- Manandhar, A.; Milindi, P.; Shah, A. An overview of the post-harvest grain storage practices of smallholder farmers in developing countries. Agriculture 2018, 8, 57. [Google Scholar] [CrossRef]
- Mobolade, A.J.; Bunindro, N.; Sahoo, D.; Rajashekar, Y. Traditional methods of food grains preservation and storage in Nigeria and India. Ann. Agric. Sci. 2019, 64, 196–205. [Google Scholar] [CrossRef]
- Baributsa, D.; Ignacio, M.C.C. Developments in the Use of Hermetic Bags for Grain Storage; Burleigh Dodds Science Publishing: Cambridge, UK, 2020; pp. 171–198. [Google Scholar] [CrossRef]
- Covele, G.; Gulube, A.; Tivana, L.; Ribeiro-Barros, A.I.; Carvalho, M.O.; Ndayiragije, A.; Nguenha, R. Effectiveness of hermetic containers in controlling paddy rice (Oryza sativa L.) storage insect pests. J. Stored Prod. Res. 2020, 89, 101710. [Google Scholar] [CrossRef]
- Hossain, M.A.; Awal, A.; Alam, M.; Ali, R.; Azmal, F. Do Hermetic Storage Technology Significantly Abate Losses of Rice over Time? An Economic Evaluation. Int. J. Manag. Account. 2021, 3, 52–59. [Google Scholar] [CrossRef]
- Lane, B.; Woloshuk, C. Impact of storage environment on the efficacy of hermetic storage bags. J. Stored Prod. Res. 2017, 72, 83–89. [Google Scholar] [CrossRef]
- Awal, M.A.; Ali, M.R.; Hossain, M.A.; Alam, M.M.; Kalita, P.K.; Harvey, J. Hermetic bag an effective and economic rice storage technology in Bangladesh. In Proceedings of the 2017 ASABE Annual International Meeting, Washington, DC, USA, 16–19 July 2017; pp. 1–10. [Google Scholar] [CrossRef]
- Baributsa, D.; Baoua, I.B.; Bakoye, O.N.; Amadou, L.; Murdock, L.L. PICS bags safely store unshelled and shelled groundnuts in Niger. J. Stored Prod. Res. 2017, 72, 54–58. [Google Scholar] [CrossRef]
- Kanta, R.A. Paddy Quality during Storage in Different Storage Technologies. Master Thesis, Bangladesh Agricultural University, Mymensingh, Bangladesh, 2016. [Google Scholar]
- Prasetyo, T.; Riska, L.; Arlanta, R.; Sumardiono, S. Experimental Study of Paddy Grain Drying in Continuous Recirculation System Pneumatic Conveyor. MATEC Web Conf. 2018, 156, 05022. [Google Scholar] [CrossRef][Green Version]
- Sahari, Y.; Wahid, R.A.; Mhd Adnan, A.S.; Sairi, M.; Hosni, H.; Engku Abdullah, E.H.; Alwi, S.; Mohd Amin Tawakkal, M.H.; Zainol Abidin, M.Z.; Aris, Z. Study on the drying performance and milling quality of dried paddy using inclined bed dryers in two different paddy mills located in MADA and IADA KETARA. Int. Food Res. J. 2018, 25, 2572–2578. [Google Scholar]
- Nwilene, F.E.; Oikeh, S.O.; Agunbiade, T.A.; Oladimeji, O.; Ajayi, O.; Sié, M.; Gregorio, G.B.; Togola, A.; Touré, A.D. Growing Lowland Rice: A Production Handbook; Africe Rice Center (WARDA) E-Publishing: Cotonou, Benin, 2011. [Google Scholar]
- Romuli, S.; Schock, S.; Somda, M.K.; Müller, J. Drying performance and aflatoxin content of paddy rice applying an inflatable solar dryer in Burkina Faso. Appl. Sci. 2020, 10, 3533. [Google Scholar] [CrossRef]
- Salvatierra-Rojas, A.; Nagle, M.; Gummert, M.; de Bruin, T.; Müller, J. Development of an inflatable solar dryer for improved postharvest handling of paddy rice in humid climates. Int. J. Agric. Biol. Eng. 2017, 10, 269–282. [Google Scholar] [CrossRef]
- Wang, T.; Khir, R.; Pan, Z.; Yuan, Q. Simultaneous rough rice drying and rice bran stabilization using infrared radiation heating. LWT-Food Sci. Technol. 2017, 78, 281–288. [Google Scholar] [CrossRef]
- Dibagar, N.; Chayjan, R.A.; Kowalski, S.J.; Peyman, S.H. Deep bed rough rice air-drying assisted with airborne ultrasound set at 21 kHz frequency: A physicochemical investigation and optimization. Ultrason. Sonochem. 2019, 53, 25–43. [Google Scholar] [CrossRef]
- Jittanit, W.; Srzednicki, G.; Driscoll, R.H. Comparison between Fluidized Bed and Spouted Bed Drying for Seeds. Dry. Technol. 2013, 31, 52–56. [Google Scholar] [CrossRef]
- Tumpanuvatr, T.; Jittanit, W.; Surojanametakul, V. Effects of drying conditions in hybrid dryer on the GABA rice properties. J. Stored Prod. Res. 2018, 77, 177–188. [Google Scholar] [CrossRef]
- Mutungi, C.; Gaspar, A.; Abass, A. Postharvest Operations and Quality Specifications for Rice: A Trainer’s Manual for Smallholder Farmers in Tanzania; IITA: Ibadan, Nigeria, 2020. [Google Scholar]
- Sims, R.; Flammini, A.; Puri, M.; Bracco, S. Opportunities for Agri-Food Chains to Become Energy-Smart; FAO: Rome, Italy, 2016; Volume 43. [Google Scholar]
- Kahandage, P.D.; Weerasooriya, G.V.T.V.; Ranasinghe, V.P.; Kosgollegedara, E.J.; Piyathissa, S.D.S. Design, Development and Performance Evaluation of a Seed Paddy Cleaning Machine. Sri Lankan J. Agric. Ecosyst. 2021, 3, 41. [Google Scholar] [CrossRef]
- Adetola, O.A.; Akindahunsi, D.L. A Review on Performance of Rice De-stoning Machines. J. Eng. Res. Rep. 2020, 1–11. [Google Scholar] [CrossRef]
- Baker, A.; Dwyer-Joyce, R.S.; Briggs, C.; Brockfeld, M. Effect of different rubber materials on husking dynamics of paddy rice. Proc. Inst. Mech. Eng. Part J J. Eng. Tribol. 2012, 226, 516–528. [Google Scholar] [CrossRef]
- Dhankhar, P. Rice Milling. IOSR J. Eng. 2014, 4, 34–42. [Google Scholar] [CrossRef]
- Manickavasagan, A.; Chandini, S.K.; Venkatachalapathy, N. Brown Rice; Springer: Berlin, Germany, 2017; pp. 1–290. [Google Scholar] [CrossRef]
- Fouda, T. Study on the whitening characteristics of some laboratory rice milling machines. In Proceedings of the 13th Annual Conference Misr Society Agr. Eng., Cairo, Egypt, 14–15 December 2005; Volume 195, pp. 192–205. Available online: https://www.researchgate.net/publication/277814862%0AStudy (accessed on 1 September 2021).
- Das, M.; Gupta, S.; Kapoor, V.; Banerjee, R.; Bal, S. Enzymatic polishing of rice—A new processing technology. LWT-Food Sci. Technol. 2008, 41, 2079–2084. [Google Scholar] [CrossRef]
- Xu, Z.; Xu, Y.; Zhang, L.; Li, H.; Sui, Z.; Corkede, H. Polishing conditions in rice milling differentially affect the physicochemical properties of waxy, low- and high-amylose rice starch. J. Cereal Sci. 2021, 99, 103183. [Google Scholar] [CrossRef]
- Custodio, M.C.; Cuevas, R.P.; Ynion, J.; Laborte, A.G.; Velasco, M.L.; Demont, M. Rice quality: How is it defined by consumers, industry, food scientists, and geneticists? Trends Food Sci. Technol. 2019, 92, 122–137. [Google Scholar] [CrossRef] [PubMed]
- Chepa, N.; Yusoff, N.; Ahmad, N. Designing a Neural Network Model in Grading Malaysian Rice. Int. J. Eng. Technol. 2018, 7, 790–793. [Google Scholar]
- Ghatkamble, R. Grading and Classification of Rice Grain Using PNN Technique in Digital Image Processing. Int. J. Comput. Sci. Trends Technol. 2013, 6, 59–63. [Google Scholar]
- Temniranrat, P.; Sinthupinyo, W.; Prempree, P.; Chaitavon, K.; Porntheeraphat, S.; Prasertsak, A. Development of Paddy Rice Seed Classification Process using Machine Learning Techniques for Automatic Grading Machine. J. Sens. 2020, 2020, 7041310. [Google Scholar] [CrossRef]
- Tanck, P.; Kaushal, B. A New Technique of Quality Analysis for Rice Grading for Agmark Standards. Int. J. Innov. Technol. Explor. Eng. 2014, 3, 83–85. [Google Scholar]
- Ibrahim, S.; Zulkifli, N.A.; Sabri, N.; Shari, A.A.; Noordin, M.R.M. Rice grain classification using multi-class support vector machine (SVM). IAES Int. J. Artif. Intell. 2019, 8, 215–220. [Google Scholar] [CrossRef]
- Chepa, N.; Yusoff, N.; Ahmad, N. Exploring the determinants for grading malaysian rice. Int. J. Innov. Technol. Explor. Eng. 2019, 8, 199–203. [Google Scholar]
- Shiddiq, D.M.F.; Nazaruddin, Y.Y.; Muchtadi, F.I.; Raharja, S. Estimation of rice milling degree using image processing and Adaptive Network Based Fuzzy Inference System (ANFIS). In Proceedings of the 2011 2nd International Conference on Instrumentation Control and Automation, Bandung, Indonesia, 15–17 November 2011; pp. 98–103. [Google Scholar] [CrossRef]
- Liu, W.; Huang, Y.; Ye, Z.; Cai, W.; Yang, S.; Cheng, X.; Frank, I. Renyi’s entropy based multilevel thresholding using a novel meta-heuristics algorithm. Appl. Sci. 2020, 10, 3225. [Google Scholar] [CrossRef]
- Yao, Y.; Wu, W.; Yang, T.; Liu, T.; Chen, W.; Chen, C.; Li, R.; Zhou, T.; Sun, C.; Zhou, Y.; et al. Head rice rate measurement based on concave point matching. Sci. Rep. 2017, 7, 41353. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.S.; Sonnadara, U. Classification of Rice Grains Using Neural Networks. Proc. Tech. Sess. 2013, 29, 9–14. [Google Scholar]
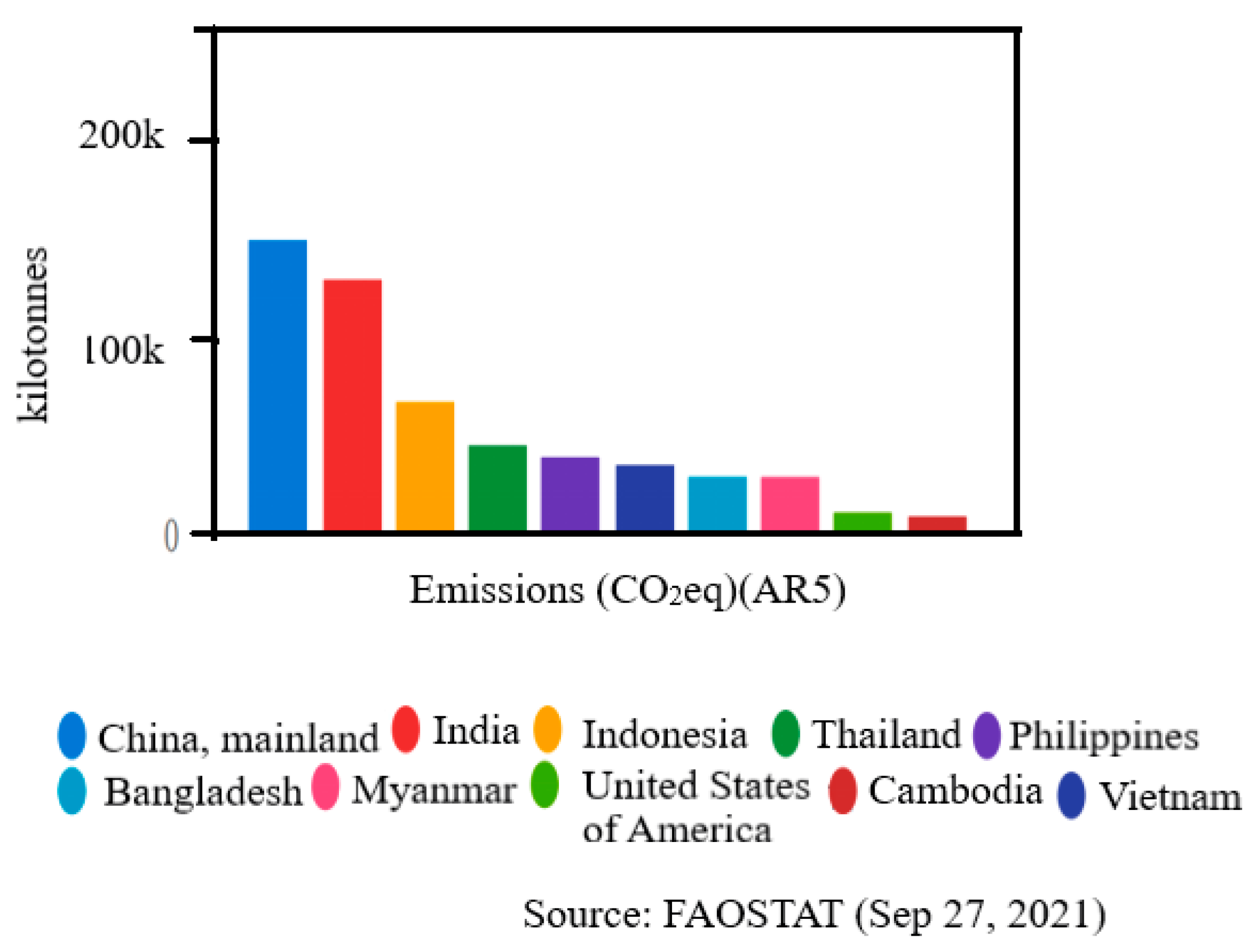
- FAOSTAT. Emissions Totals. 2021. Available online: https://www.fao.org/faostat/en/#data/GT/visualize (accessed on 1 September 2021).
- Wichelns, D. Managing water and soils to achieve adaptation and reduce methane emissions and arsenic contamination in Asian rice production. Water 2016, 8, 141. [Google Scholar] [CrossRef]
- Qin, X.; Li, Y.E.; Wang, H.; Li, J.; Wan, Y.; Gao, Q.; Liao, Y.; Fan, M. Effect of rice cultivars on yield-scaled methane emissions in a double rice field in South China. J. Integr. Environ. Sci. 2015, 12, 47–66. [Google Scholar] [CrossRef]
- Martínez-Eixarch, M.; Alcaraz, C.; Viñas, M.; Noguerol, J.; Aranda, X.; Prenafeta-Boldú, F.X.; Català-Forner, M.; Fennessy, M.S.; Ibáñez, C. The main drivers of methane emissions differ in the growing and flooded fallow seasons in Mediterranean rice fields. Plant Soil 2021, 460, 211–227. [Google Scholar] [CrossRef]
- Jiang, Y.; Zhang, H.; He, J.; Huan, P. Study on Methane Emission Factor of Paddy Fields in Hubei Province. IOP Conf. Ser. Earth Environ. Sci. 2021, 651, 042031. [Google Scholar] [CrossRef]
- Sass, R. CH4 emissions from rice agriculture. In Good Practice Guidance and Uncertainty Management in National Greenhouse Gas Inventories; Institute for Global Environmental Strategies (IGES): Hayama, Japan, 2003; pp. 399–417. [Google Scholar]
- Prangbang, P.; Yagi, K.; Aunario, J.K.S.; Sander, B.O.; Wassmann, R.; Jäkel, T.; Buddaboon, C.; Chidthaisong, A.; Towprayoon, S. Climate-Based Suitability Assessment for Methane Mitigation by Water Saving Technology in Paddy Fields of the Central Plain of Thailand. Front. Sustain. Food Syst. 2020, 4, 575823. [Google Scholar] [CrossRef]
- Epule, E.T.; Peng, C.; Mafany, N.M. Methane Emissions from Paddy Rice Fields: Strategies towards Achieving a Win-Win Sustainability Scenario between Rice Production and Methane Emission Reduction. J. Sustain. Dev. 2011, 4. [Google Scholar] [CrossRef]
- Koizumi, T.; Hubertus, S.; Furuhashi, G. Reviewing Indica and Japonica Rice Market Developments. OECD Publications. 2021. Available online: https://www.oecd.org/publications/reviewing-indica-and-japonica-rice-market-developments-0c500e05-en.htm (accessed on 1 October 2021).
- Firdaus, R.B.R.; Tan, M.L.; Rahmat, S.R.; Gunaratne, M.S. Paddy, rice and food security in Malaysia: A review of climate change impacts. Cogent Soc. Sci. 2020, 6, 1818373. [Google Scholar] [CrossRef]
- Alam, M.M.; Siwar, C.; Murad, M.W.; Toriman, M.E. Impacts of climate change on agriculture and food security issues in Malaysia: An empirical study on farm level assessment. World Appl. Sci. J. 2011, 14, 431–442. [Google Scholar]
- Müller, C.; Elliott, J.; Chryssanthacopoulos, J.; Deryng, D.; Folberth, C.; Pugh, T.A.M.; Schmid, E. Implications of climate mitigation for future agricultural production. Environ. Res. Lett. 2015, 10, 125004. [Google Scholar] [CrossRef]
- Nawaz, A.; Shafi, T.; Khaliq, A.; Mukhtar, H.; ul Haq, I. Tyrosinase: Sources, Structure and Applications. Int. J. Biotechnol. Bioeng. 2017, 3, 135–141. [Google Scholar] [CrossRef]
- Lin, H.; Yu, Y.; Wen, F.; Liu, P. Status of Food Security in East and Southeast Asia and Challenges of Climate Change. Climate 2022, 10, 40. [Google Scholar] [CrossRef]
- Wang, L.; Guo, Y.; Zhu, Y.; Li, Y.; Qu, Y.; Rong, C.; Ma, X.; Wang, Z. A new route for preparation of hydrochars from rice husk. Bioresour. Technol. 2010, 101, 9807–9810. [Google Scholar] [CrossRef]
- Mohanta, K.; Kumar, D.; Parkash, O. Properties and Industrial Applications of Rice husk: A review. Int. J. Emerg. Technol. Adv. Eng. 2012, 2, 86–90. [Google Scholar]
- Soltani, N.; Bahrami, A.; Pech-Canul, M.I.; González, L.A. Review on the physicochemical treatments of rice husk for production of advanced materials. Chem. Eng. J. 2015, 264, 899–935. [Google Scholar] [CrossRef]
- Bakar, R.A.; Yahya, R.; Gan, S.N. Production of High Purity Amorphous Silica from Rice Husk. Procedia Chem. 2016, 19, 189–195. [Google Scholar] [CrossRef]
- Zawawi, W.N.I.W.M.; Mansor, A.F.; Othman, N.S.; Mohidem, N.A.; Malek, N.A.N.N.; Mat, H. Synthesis and characterization of immobilized white-rot fungus Trametes versicolor in sol–gel ceramics. J. Sol-Gel Sci. Technol. 2016, 77, 28–38. [Google Scholar] [CrossRef]
- Suleman, M.; Zafar, M.; Ahmed, A.; Rashid, M.U.; Hussain, S.; Razzaq, A.; Mohidem, N.A.; Fazal, T.; Haider, B.; Park, Y.-K. Castor leaves-based biochar for adsorption of safranin from textile wastewater. Sustainability 2021, 13, 6926. [Google Scholar] [CrossRef]
- Shamsuri, N.A.A.; Mohidem, N.A.; Mansor, A.F.; Mat, H. Biodegradation of Dye Using Free Laccase and Sol-gel Laccase. Jurnal Teknologi 2012, 59, 39–42. [Google Scholar] [CrossRef][Green Version]
- Kumar, A.; Roy, A.; Priyadarshinee, R.; Sengupta, B.; Malaviya, A.; Dasguptamandal, D.; Mandal, T. Economic and sustainable management of wastes from rice industry: Combating the potential threats. Environ. Sci. Pollut. Res. 2017, 24, 26279–26296. [Google Scholar] [CrossRef] [PubMed]
- FAO. Crop Prospects and Food Situation—Quarterly Global Report No. 4, December 2020; FAO: Rome, Italy, 2020. [Google Scholar] [CrossRef]
- Teck, T.B.; Shan, F.P.; Firdaus, R.B.R.; Leong, T.M.; Senevi, G.M. Impact of climate change on rice yield in malaysia: A panel data analysis. Agriculture 2021, 11, 569. [Google Scholar] [CrossRef]
- Sulaiman, N.; Yeatman, H.; Russell, J.; Law, L.S. A Food Insecurity Systematic Review: Experience from Malaysia. Nutrients 2021, 13, 945. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.; Chhokar, R.S.; Meena, R.; Gill, S.C.; Tripathi, S.C.; Gupta, O.P.; Mangrauthia, S.K.; Sundaram, R.M.; Sawant, C.P.; Gupta, A.; et al. Challenges and opportunities in productivity and sustainability of rice cultivation system: A critical review in Indian perspective. Cereal Res. Commun. 2021. [Google Scholar] [CrossRef] [PubMed]
- Dhanda, S.; Yadav, A.; Yadav, D.B.; Chauhan, B.S. Emerging Issues and Potential Opportunities in the Rice—Wheat Cropping System of North-Western. Front. Plant Sci. 2022, 13, 832683. [Google Scholar] [CrossRef]
- Sarena, C.O.; Ashraf, S.; Aiysyah, T.S. The Status of the Paddy and Rice Industry in Malaysia. 2019. Available online: http://www.krinstitute.org/assets/contentMS/img/template/editor/20190409_RiceReport_FullReport_Final.pdf (accessed on 1 September 2021).
- FAO. Commodity Policy Developments. 2022. Available online: https://www.fao.org/economic/est/estcommodities/commoditypolicyarchive/en/?groupANDcommodity=rice (accessed on 1 October 2021).



| Country | 2018–2020 | 2030 | Growth Rate (% p.a.) |
|---|---|---|---|
| Africa | 27.4 | 31.5 | 1.2 |
| Oceania | 13.5 | 14.2 | 0.44 |
| North America | 6.3 | 6.6 | 0.42 |
| Europe | 20.7 | 25.6 | −0.08 |
| Latin America and Caribbean | 28.0 | 28.1 | −0.14 |
| Asia | 77.2 | 77.5 | −0.15 |
| Country | Total Rice Production (Tonnes) | Country | Total Rice Production (Tonnes) |
|---|---|---|---|
| Afghanistan | 444,452.15 | Democratic Republic of the Congo | 557,205.58 |
| Albania | 0 | Denmark | 0 |
| Algeria | 438.12 | Dominican Republic | 721,733.42 |
| Angola | 15,556.69 | Ecuador | 1,426,523.23 |
| Argentina | 1,193,141.73 | Chile | 133,629.04 |
| Australia | 772,382 | China | 195,488,413.9 |
| Azerbaijan | 8675.65 | China, Hong Kong SAR | 0 |
| Bangladesh | 42,178,999.19 | Egypt | 5,555,313.96 |
| Belgium | 0 | El Salvador | 38,195.92 |
| Belize | 14,115.96 | Estonia | 0 |
| Benin | 142,182.08 | Eswatini | 605.81 |
| Bhutan | 62,148.04 | Ethiopia | 60,023.92 |
| Bolivia (Plurinational State of) | 390,846.77 | Fiji | 11,576.42 |
| Brazil | 11,225,080.35 | Finland | 0 |
| Brunei Darussalam | 947.15 | France | 106,261.54 |
| Bulgaria | 34,938.38 | French Guyana | 24,066.54 |
| Burkina Faso | 189,713.31 | Gabon | 1281.96 |
| Burundi | 66,475.62 | Gambia | 36,824.31 |
| Cambodia | 6,585,414.31 | Germany | 0 |
| Cameroon | 126,887.38 | Ghana | 393,680.42 |
| Central African Republic | 24,122.92 | Greece | 204,750.77 |
| Chad | 169,578.62 | Guatemala | 29,947.27 |
| Chile | 133,629.04 | Guinea | 1,507,327.77 |
| China | 195,488,413.9 | Guinea-Bissau | 137,708.42 |
| China, Hong Kong SAR | 0 | Guyana | 580,718.54 |
| China, mainland | 193,772,091.5 | Haiti | 130,727.5 |
| China, Taiwan Province of | 1,716,322.38 | Honduras | 40,117.42 |
| Colombia | 2,263,214.04 | Hungary | 9842.65 |
| Comoros | 22,560.42 | India | 142,410,885.6 |
| Congo | 1232.73 | Indonesia | 54,883,555.81 |
| Costa Rica | 213,232.54 | Iran (Islamic Republic of) | 2,384,846.77 |
| Côte d’Ivoire | 1,090,520.46 | Iraq | 258,179.85 |
| Croatia | 0 | Ireland | 0 |
| Cuba | 513,772.5 | Italy | 1,447,809.31 |
| Cyprus | 0 | Jamaica | 70.31 |
| Czechia | 0 | Japan | 11,337,059.62 |
| Democratic People’s Republic of Korea | 2,327,908.92 | Kazakhstan | 314,242.15 |
| Kenya | 72,811.35 | ||
| Kyrgyzstan | 21,005.46 | Saudi Arabia | 0 |
| Lao People’s Democratic Republic | 2,773,653.88 | Senegal | 439,096.38 |
| Latvia | 0 | Sierra Leone | 702,817.46 |
| Liberia | 203,410 | Slovakia | 0 |
| Lithuania | 0 | Slovenia | 0 |
| Luxembourg | 0 | Solomon Islands | 2665.62 |
| Madagascar | 3,383,336.15 | Somalia | 5770.42 |
| Malawi | 93,924.08 | South Africa | 3070.85 |
| Malaysia | 2,345,032.54 | Spain | 791,654.73 |
| Mali | 1,438,024.58 | Sri Lanka | 3,341,954.46 |
| Malta | 0 | Sudan | 27,487.5 |
| Mauritania | 137,731.69 | Sudan (former) | 14,913.78 |
| Mauritius | 161.08 | Suriname | 218,101.96 |
| Mexico | 288,067.85 | Sweden | 0 |
| Micronesia (Federated States of) | 129.92 | Syrian Arab Republic | 149.33 |
| Morocco | 41,000.62 | Tajikistan | 62,629.96 |
| Mozambique | 163,375.15 | Thailand | 29,621,346.69 |
| Myanmar | 24,700,326.04 | Timor-Leste | 66,275.96 |
| Nepal | 4,294,742.65 | Togo | 103,441.65 |
| Netherlands | 0 | Trinidad and Tobago | 4232.5 |
| Nicaragua | 322,352.46 | Turkey | 618,820.19 |
| Niger | 76,839.92 | Turkmenistan | 80,366.54 |
| Nigeria | 4,464,072.04 | Uganda | 162,392.73 |
| North Macedonia | 18,914.5 | Ukraine | 90,938.69 |
| Pakistan | 8,394,983.38 | United Republic of Tanzania | 1,519,746.96 |
| Panama | 281,628.96 | United States of America | 9,157,048.46 |
| Papua New Guinea | 772.88 | Uruguay | 1,182,819.15 |
| Paraguay | 343,408.69 | Uganda | 162,392.73 |
| Peru | 2,392,461.08 | Ukraine | 90,938.69 |
| Philippines | 15,025,640.73 | United Republic of Tanzania | 1,519,746.96 |
| Poland | 0 | United States of America | 9,157,048.46 |
| Portugal | 158,642.77 | Uzbekistan | 289,830.31 |
| Puerto Rico | 36.38 | Venezuela (Bolivarian Republic of) | 852,740.23 |
| Republic of Korea | 6,244,369.88 | Vietnam | 36,613,011.5 |
| Réunion | 123.85 | Zambia | 25,104.19 |
| Romania | 29,653.5 | Zimbabwe | 869.04 |
| Russian Federation | 735,256.92 | ||
| Rwanda | 56,898.92 |
| Property | Amylose | Amylopectin |
|---|---|---|
| Molecular Structure | Linear (α-1–4) | Branched (α-1–4; α-1–6) |
| Dilute Solutions | Unstable | Stable |
| Gels | Stiff, irreversible | Soft, reversible |
| Films | Coherent | _ |
| Complex Formation | Favourable | Unfavourable |
| Iodine Colour | Blue | Red-Purple |
| Digestibility, β-Amylase | 100% | 60% |
| Degree of Polymerization | 1500–6000 | 3 × 105–3 × 106 |
| Property | Brown Rice | Wheat | Corn | Barley | Millet | Sorghum | Rye | Oat |
|---|---|---|---|---|---|---|---|---|
| Protein (N × 6.25) (%) | 7.3 | 10.6 | 9.8 | 11.0 | 11.5 | 8.3 | 8.7 | 9.3 |
| Fibre (%) | 0.8 | 1.0 | 2.0 | 3.7 | 1.5 | 4.1 | 2.2 | 5.6 |
| Net protein utilization (%) | 73.8 | 53.0 | 58.0 | 62.0 | 56.0 | 50.0 | 59.0 | 59.1 |
| Digestible energy (kJ (100 g)−1) | 1550 | 1360 | 1450 | 1320 | 1440 | 1290 | 1330 | 1160 |
| Nutrient | Brown Rice | Milled Rice |
|---|---|---|
| Digestible carbohydrates | 84.8–88.2 | 89.1–91.2 |
| Crude protein | 6.5–10.0 | 7.3–8.3 |
| Crude ash | 1.2–1.7 | 0.3–0.9 |
| Crude fat | 1.9–3.9 | 0.3–0.65 |
| Crude fibre | 1.6-2.8 | 0.3-0.5 |
| Carbohydrates | 85.2–88.9 | 91.07 |
| Water (% of fresh weight) | 11.37–16.4 | 12.31–15.5 |
| Starch | 77.2 | 90.2 |
| Free sugars | 0.8–1.5 | 0.3–0.5 |
| Neutral detergent fibre | 4.5 | 0.8–2.7 |
| Dietary fibre/insoluble | 0.5 | |
| Total dietary fibre | 3.9 | 0.5–2.8 |
| Crude fibre | 0.7–1.2 | 0.2–0.6 |
| Hemicelluloses | 0.1 | |
| Pentosans | 1.4–2.4 | 0.6–1.6 |
| Lignin | 0.1 | |
| Energy (kJ/g) | 17.2–18.7 | 17.0–18.1 |
| Protein fraction (% of total protein) | ||
| Albumin (soluble in water) | 2–5 | |
| Glutelin (soluble in aqueous alkaline solution) | 60–65 | |
| Prolamin (soluble in alcohol) | 20–25 | |
| Globulin (soluble in salt water) | 2–10 | |
| Amino acid composition (% of dry matter) | ||
| Aspartic acid | 0.59–0.96 | |
| Arginine | 0.52–0.88 | |
| Alanine | 0.37–0.59 | |
| Cystine | 0.15–0.28 | |
| Histidine | 0.16–0.27 | |
| Glycine | 0.32–0.48 | |
| Glutamic acid | 1.06–1.88 | |
| Isoleucine | 0.22–0.40 | |
| Leucine | 0.51–0.85 | |
| Lysine | 0.26–0.40 | |
| Methionine | 0.14–0.34 | |
| Phenylalanine | 0.32–0.55 | |
| Threonine | 0.23–0.38 | |
| Proline | 0.25–0.46 | |
| Serine | 0.30–0.53 | |
| Tyrosine | 0.21–0.51 | |
| Tryptophan | 0.05–0.13 | |
| Valine | 0.37–0.59 | |
| Fatty acid component (% of total fatty acids) | ||
| Myristic (14:0) | 0.5–1.1 | |
| Pentadecanoic (15:0) | 0.1–0.3 | |
| Palmitic (16:0) | 18.2–31.2 | |
| Palmitoleic (16:1) | 0.1–0.2 | |
| Heptadecanoic (17:0) | 0.1–0.6 | |
| Stearic (18:0) | 1.5–2.8 | |
| Oleic (18:1) | 30.9–40.2 | |
| Linoleic (18:2) | 26.1–39.0 | |
| Linolenic (18:3) | 0.9–1.6 | |
| Arachidic (20:0) | 0.4–0.7 | |
| Eicosenoic (20:1) | 0.4–0.6 | |
| Behenic (22:0) | 0.2–0.6 | |
| Docosenoic/erucic (22:1) | 0.1–0.2 | |
| Tetracosenoic (24:1) | 0.1–0.3 | |
| Lignoceric (24:0) | 0.4–0.9 | |
| Others | 4 | |
| Macro-minerals (mg/g dry matter) | ||
| Calcium | 0.1–0.6 | 0.1–0.3 |
| Magnesium | 0.2–1.7 | 0.2–0.6 |
| Potassium | 0.7–3.2 | 0.8–1.5 |
| Phosphorus | 2.0–5.0 | 0.9–1.7 |
| Sulphur | 0.3–2.2 | 0.9 |
| Silicon | 0.7–1.6 | 0.1–0.5 |
| Micro-minerals (μg/g dry matter) | ||
| Copper | 1–7 | 2–3 |
| Iron | 2–60 | 2–33 |
| Manganese | 2–42.24 | 7–20 |
| Sodium | 20–395 | 6–100 |
| Zinc | 7–33 | 7–27 |
| Vitamin (μg/g dry matter) | ||
| Retinol (A) | 0–0.13 | 0-trace |
| Thiamine (B1) | 3.4–8.1 | 0.2–1.3 |
| Ribofavin (B2) | 0.2–1.6 | 0.2–0.7 |
| Niacin (B3) | 41–134.7 | 15–28 |
| Pantothenic acid (B5) | 11–17 | 4.8 |
| Pyridoxine (B6) | 1.8–11 | 0.5–1.4 |
| Biotin (B7) | 0.05–0.12 | 0.01–0.07 |
| Choline, total | 1100 | 450–1020 |
| Folic acid (B9) | 0.1–0.6 | 0.03–0.16 |
| Cyanocobalamin (B12) | 0–0.005 | 0–0.0016 |
| Alpha-tocopherol (E) | 8.9–29 | Trace-3 |
| Beta-tocopherol | 0.5–1.4 | |
| Delta-tocopherol | 0.1–0.6 | |
| Gamma-tocopherol | 2.2–4.8 | |
| Challenges to the sustainability of the rice production system |
|
| How can we overcome the challenges? |
|
| Countries | Date | Policy Instrument | Summary |
|---|---|---|---|
| China (Mainland) | 5.2021 | Stock release | After auctioning 1.81 million tonnes of paddy on 14 May 2021, 155,007 tonnes of paddy harvested from state reserves were sold. |
| India | 5.2021 | Export promotion, trade facilitation | Rice shipments were inaugurated through the Odisha state’s Paradip International Cargo Terminal (PICT). |
| Mali | 4.2021 | Price controls | Set a ceiling of XOF 340 (USD 0.63) per kg on market prices of non-fragrant broken rice. A limit of XOF 290,000 (USD 537) for each tonne would implement |
| Turkey | 4.2021 | Government procurement, purchasing prices | Government purchase prices have been revised for the 2020 season, increasing them from TRY 3500 to 4750 for each tonne. |
| Bangladesh | 3.2021 | Import tariff | Imported non-parboiled (white, non-fragrant) rice duties and charges reduced from 62.5% to 25% |
| European Union | 3.2021 | Import tariff | Reduced tariffs on imported non-basmati husked rice from EUR 65 to EUR 30 per tonne outside of current trade agreements. |
| Malaysia | 12.2020 | Import rights | Renewed Padiberas Nasional Berhad’s (BERNAS) exclusive import rights for another ten years, beginning in January 2021. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mohidem, N.A.; Hashim, N.; Shamsudin, R.; Che Man, H. Rice for Food Security: Revisiting Its Production, Diversity, Rice Milling Process and Nutrient Content. Agriculture 2022, 12, 741. https://doi.org/10.3390/agriculture12060741
Mohidem NA, Hashim N, Shamsudin R, Che Man H. Rice for Food Security: Revisiting Its Production, Diversity, Rice Milling Process and Nutrient Content. Agriculture. 2022; 12(6):741. https://doi.org/10.3390/agriculture12060741
Chicago/Turabian StyleMohidem, Nur Atikah, Norhashila Hashim, Rosnah Shamsudin, and Hasfalina Che Man. 2022. "Rice for Food Security: Revisiting Its Production, Diversity, Rice Milling Process and Nutrient Content" Agriculture 12, no. 6: 741. https://doi.org/10.3390/agriculture12060741
APA StyleMohidem, N. A., Hashim, N., Shamsudin, R., & Che Man, H. (2022). Rice for Food Security: Revisiting Its Production, Diversity, Rice Milling Process and Nutrient Content. Agriculture, 12(6), 741. https://doi.org/10.3390/agriculture12060741







