The Potential of Novel Gene Editing-Based Approaches in Forages and Rumen Archaea for Reducing Livestock Methane Emissions
Abstract
1. Introduction
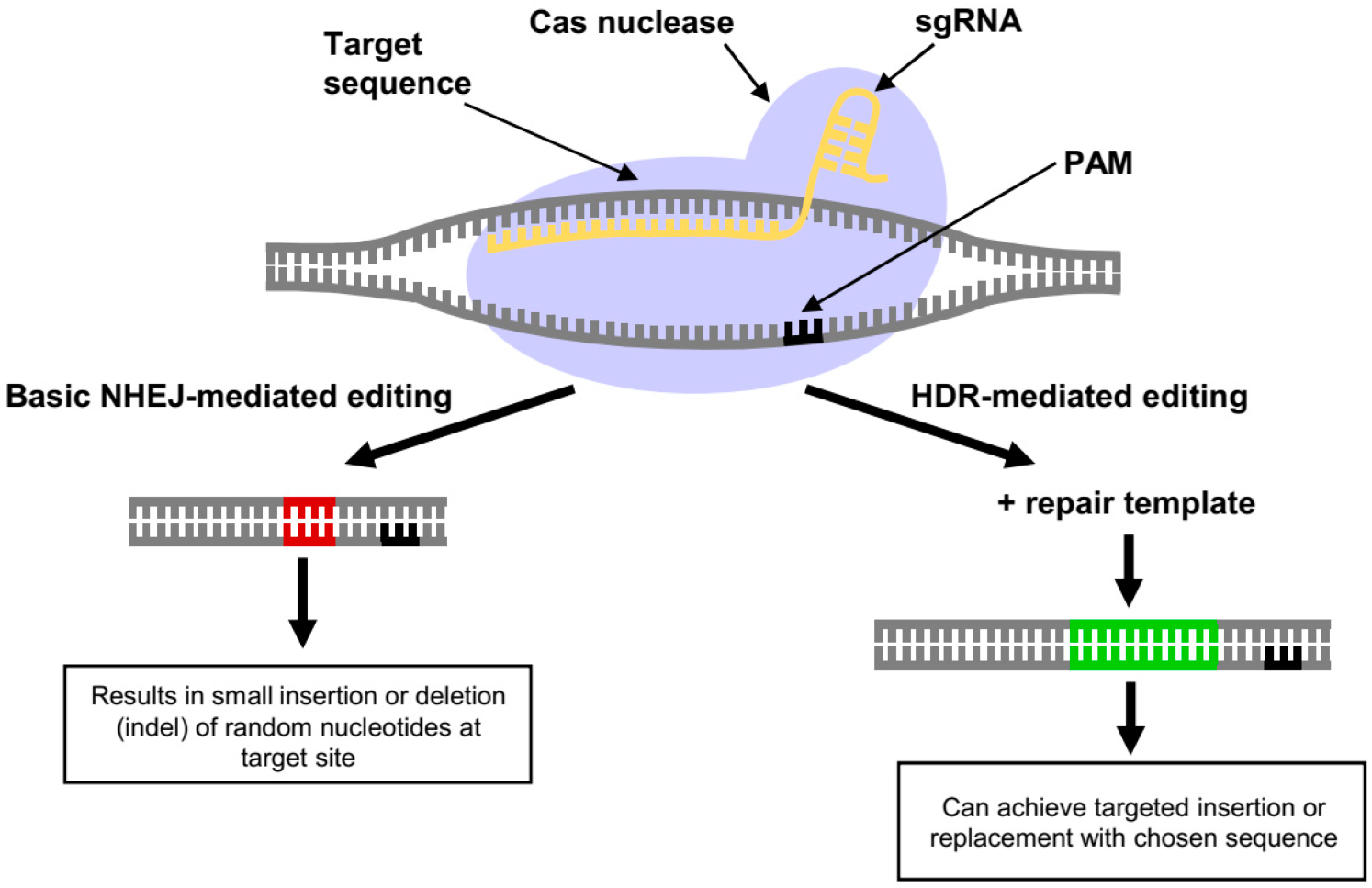
2. CRISPR/Cas Gene Editing
3. The Genetic Modulation of Forages for Reduced CH4 Emissions
3.1. CRISPR/Cas Gene Editing in Forages
3.2. The Potential of Forage Lipid Modulation as a Means of Reducing CH4 Emissions from Ruminants
3.3. The Potential of Other Forage Traits to Reduce CH4 Emissions from Ruminants
4. The Genetic Modulation of Rumen Methanogens for Reduced CH4 Emissions
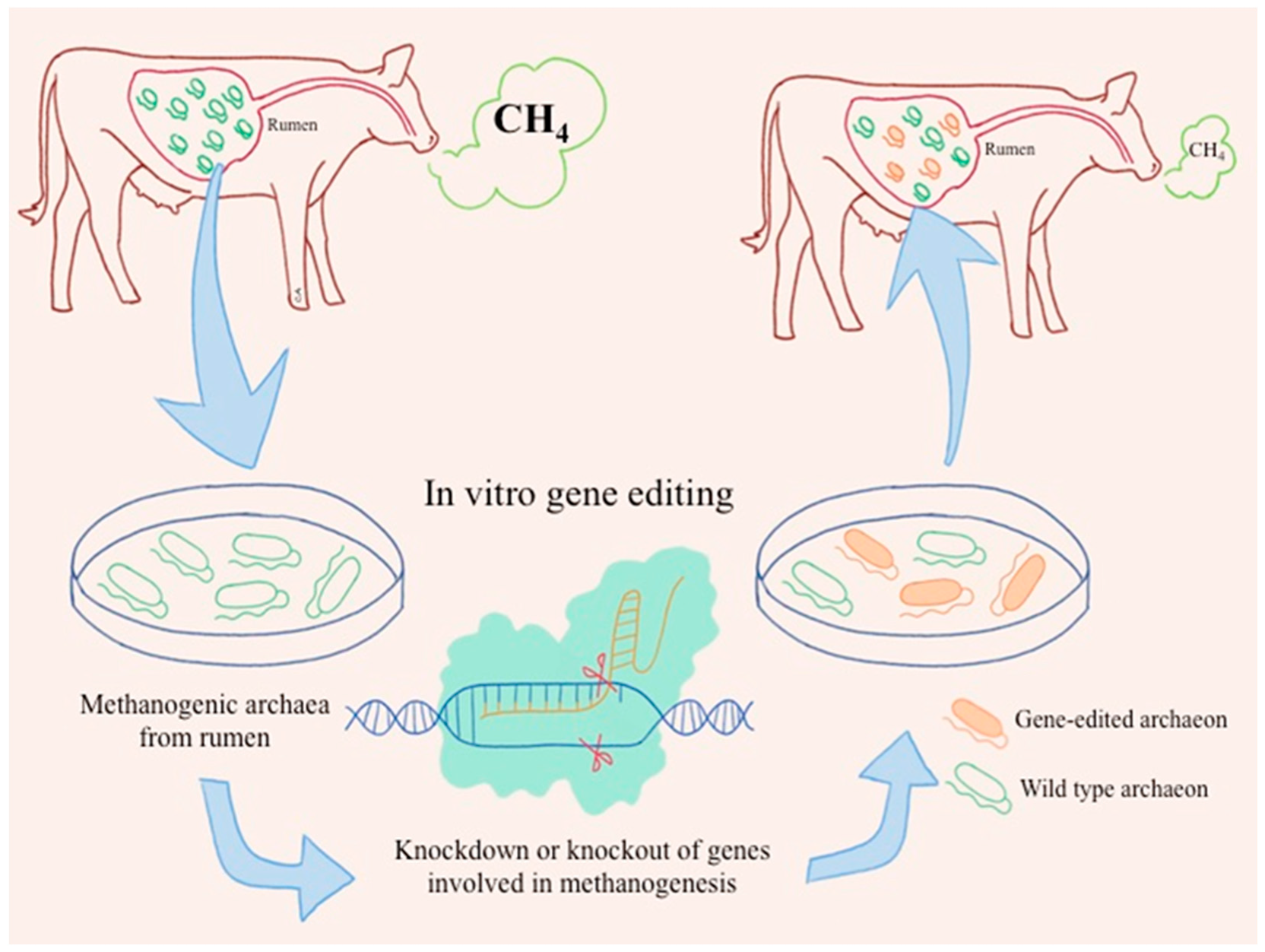
4.1. CRISPR/Cas Gene Editing in Archaea
4.2. Potential CRISPR/Cas-Based Approaches in Rumen Methanogens to Mitigate CH4 Emissions
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Minx, J.C.; Lamb, W.F.; Andrew, R.M.; Canadell, J.G.; Crippa, M.; Döbbeling, N.; Forster, P.M.; Guizzardi, D.; Olivier, J.; Peters, G.P.; et al. A comprehensive and synthetic dataset for global, regional, and national greenhouse gas emissions by sector 1970–2018 with an extension to 2019. Earth Syst. Sci. Data 2021, 13, 5213–5252. [Google Scholar] [CrossRef]
- Intergovernmental Panel on Climate Change (IPCC). Climate Change 2021: The Physical Science Basis. In Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change, Technical Summary; Masson-Delmotte, V.P., Zhai, P., Pirani, A., Connors, S.L., Péan, C., Chen, Y., Goldfarb, L., Gomi, M.I., Matthews, J.B.R., Berger, S., et al., Eds.; Cambridge University Press: Cambridge, UK, 2021; Available online: https://www.ipcc.ch/report/ar6/wg1/downloads/report/IPCC_AR6_WGI_TS.pdf (accessed on 28 July 2022).
- Tito, R.; Vasconcelos, H.L.; Feeley, K.J. Global climate change increases risk of crop yield losses and food insecurity in the tropical Andes. Glob. Chang. Biol. 2017, 24, e592–e602. [Google Scholar] [CrossRef] [PubMed]
- Arora, N.K. Impact of climate change on agriculture production and its sustainable solutions. Environ. Sustain. 2019, 2, 95–96. [Google Scholar] [CrossRef]
- Cammarano, D.; Ceccarelli, S.; Grando, S.; Romagosa, I.; Benbelkacem, A.; Akar, T.; Al-Yassin, A.; Pecchioni, N.; Francia, E.; Ronga, D. The impact of climate change on barley yield in the Mediterranean basin. Eur. J. Agron. 2019, 106, 1–11. [Google Scholar] [CrossRef]
- Malhi, Y.; Franklin, J.; Seddon, N.; Solan, M.; Turner, M.G.; Field, C.B.; Knowlton, N. Climate change and ecosystems: Threats, opportunities and solutions. Phil. Trans. R. Soc. B Biol. Sci. 2020, 375, 20190104. [Google Scholar] [CrossRef] [PubMed]
- Román-Palacios, C.; Wiens, J.J. Recent responses to climate change reveal the drivers of species extinction and survival. Proc. Natl. Acad. Sci. USA 2020, 117, 4211–4217. [Google Scholar] [CrossRef] [PubMed]
- Höhne, N.; Gidden, M.J.; den Elzen, M.; Hans, F.; Fyson, C.; Geiges, A.; Jeffery, M.L.; Gonzales-Zuñiga, S.; Mooldijk, S.; Hare, W.; et al. Wave of net zero emission targets opens window to meeting the Paris Agreement. Nat. Clim. Chang. 2021, 11, 820–822. [Google Scholar] [CrossRef]
- IPCC. Climate Change 2022: Mitigation of Climate Change. In Working Group III Contribution to the IPCC Sixth Assessment Report (AR6); IPCC: Geneva, Switzerland, 2022; Available online: https://report.ipcc.ch/ar6wg3/pdf/IPCC_AR6_WGIII_FinalDraft_FullReport.pdf (accessed on 28 July 2022).
- IPCC. Climate Change 2014: Mitigation of Climate Change. In Contribution of Working Group III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Edenhofer, O., Pichs-Madruga, R., Sokona, Y., Farahani, E., Kadner, S., Seyboth, K., Adler, A., Baum, I., Brunner, S., Eickemeier, P., et al., Eds.; Cambridge University Press: Cambridge, UK, 2014; Available online: https://www.ipcc.ch/site/assets/uploads/2018/02/ipcc_wg3_ar5_full.pdf (accessed on 28 July 2022).
- Jackson, R.B.; Saunois, M.; Bousquet, P.; Canadell, J.G.; Poulter, B.; Stavert, A.R.; Bergamaschi, P.; Niwa, Y.; Segers, A.; Tsuruta, A. Increasing anthropogenic methane emissions arise equally from agricultural and fossil fuel sources. Environ. Res. Lett. 2020, 15, 071002. [Google Scholar] [CrossRef]
- Gerber, P.J.; Steinfeld, H.; Henderson, B.; Motter, A.; Opio, C.; Dijkman, J.; Falcucci, A.; Tempio, G. Tackling Climate Change through Livestock—A Global Assessment of Emissions and Mitigation Opportunities; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2013; Available online: https://www.fao.org/3/i3437e/i3437e.pdf (accessed on 28 July 2022).
- Reisinger, A.; Clark, H.; Cowie, A.L.; Emmet-Booth, J.; Gonzalez Fischer, C.; Herrero, M.; Howden, M.; Leahy, S. How necessary and feasible are reductions of methane emissions from livestock to support stringent temperature goals? Phil. Trans. R. Soc. A 2021, 379, 20200452. [Google Scholar] [CrossRef] [PubMed]
- Searchinger, T.; Waite, R.; Hanson, C.; Ranganathan, J. Creating a Sustainable Food Future; World Resources Institute: Washington, DC, USA, 2019; Available online: https://files.wri.org/d8/s3fs-public/wrr-food-full-report.pdf (accessed on 30 August 2022).
- Leng, R.A. Interactions between microbial consortia in biofilms: A paradigm shift in rumen microbial ecology and enteric methane mitigation. Anim. Prod. Sci. 2014, 54, 519. [Google Scholar] [CrossRef]
- Beauchemin, K.A.; Ungerfield, E.M.; Eckard, R.J.; Wang, M. Fifty years of research on rumen methanogenesis: Lessons learned and future challenges for mitigation. Animal 2020, 14, s2–s16. [Google Scholar] [CrossRef] [PubMed]
- Duin, E.C.; Wagner, T.; Shima, S.; Prakash, D.; Cronin, B.; Yáñez-Ruiz, D.R.; Duval, S.; Rümbeli, R.; Stemmler, R.T.; Thauer, R.K.; et al. Mode of action uncovered for the specific reduction of methane emissions from ruminants by the small molecule 3-nitrooxypropanol. Proc. Natl. Acad. Sci. USA 2016, 113, 6172–6177. [Google Scholar] [CrossRef] [PubMed]
- Van Nevel, C.J.; Demeyer, D.I. Control of rumen methanogenesis. Environ. Monit. Assess. 1996, 42, 73–97. [Google Scholar] [CrossRef]
- Johnson, K.A.; Johnson, D.E. Methane emissions from cattle. J. Anim. Sci. 1995, 73, 2483–2492. [Google Scholar] [CrossRef] [PubMed]
- McGeough, E.J.; O’Kiely, P.; Hart, K.J.; Moloney, A.P.; Boland, T.M.; Kenny, D.A. Methane emissions, feed intake, performance, digestibility, and rumen fermentation of finishing beef cattle offered whole-crop wheat silages differing in grain content. J. Anim. Sci. 2010, 88, 2703–2716. [Google Scholar] [CrossRef] [PubMed]
- Van Wyngaard, J.D.V.; Meeske, R.; Erasmus, L.J. Effect of concentrate feeding level on methane emissions, production performance and rumen fermentation of Jersey cows grazing ryegrass pasture during spring. Anim. Feed. Sci. Technol. 2018, 241, 121–132. [Google Scholar] [CrossRef]
- DeRamus, H.A.; Clement, T.C.; Giampola, D.D.; Dickison, P.C. Methane emissions of beef cattle on forages. J. Environ. Qual. 2003, 32, 269–277. [Google Scholar] [CrossRef] [PubMed]
- Pickering, N.K.; Oddy, V.H.; Basarab, J.; Cammack, K.; Hayes, B.; Hegarty, R.S.; Lassen, J.; McEwan, J.C.; Miller, S.; Pinares-Patiño, C.S.; et al. Genetic possibilities to reduce enteric methane emissions from ruminants. Animal 2015, 9, 1431–1440. [Google Scholar] [CrossRef] [PubMed]
- McAllister, T.A.; Newbold, C.J. Redirecting rumen fermentation to reduce methanogenesis. Anim. Prod. Sci. 2008, 48, 7–13. [Google Scholar] [CrossRef]
- Lee, S.S.; Hsu, J.T.; Mantovani, H.C.; Russell, J.B. The effect of bovicin HC5, a bacteriocin from Streptococcus bovis HC5, on ruminal methane production in vitro. FEMS Microbiol. Lett. 2002, 217, 51–55. [Google Scholar] [CrossRef]
- Sar, C.; Mwenya, B.; Pen, B.; Morikawa, R.; Takaura, K.; Kobayashi, T.; Takahashi, J. Effect of nisin on ruminal methane production and nitrate/nitrite reduction in vitro. Aust. J. Agric. Res. 2005, 56, 803–810. [Google Scholar] [CrossRef]
- Baca-González, V.; Asensio-Calavia, P.; González-Acosta, S.; de la Lastra, J.M.P.; de la Nuez, A.M. Are vaccines the solution for methane emissions from ruminants? A systematic review. Vaccines 2020, 8, 460. [Google Scholar] [CrossRef] [PubMed]
- Beauchemin, K.A.; McGinn, S.M.; Petit, H.V. Methane abatement strategies for cattle: Lipid supplementation of diets. Can. J. Anim. Sci. 2007, 87, 431–440. [Google Scholar] [CrossRef]
- Animut, G.; Puchala, R.; Goetsch, A.L.; Patra, A.K.; Sahlu, T.; Varel, V.H.; Wells, J. Methane emission by goats consuming diets with different levels of condensed tannins from lespedeza. Anim. Feed Sci. Technol. 2008, 144, 212–227. [Google Scholar] [CrossRef]
- Beauchemin, K.A.; McAllister, T.A.; McGinn, S.M. Dietary mitigation of enteric methane from cattle. Anim. Nutr. 2020, 6, 231–246. [Google Scholar] [CrossRef]
- Tomkins, N.W.; Colegate, S.M.; Hunter, R.A. A bromochloromethane formulation reduces enteric methanogenesis in cattle fed grain-based diets. Anim. Prod. Sci. 2009, 49, 1053–1058. [Google Scholar] [CrossRef]
- Appuhamy, J.R.N.; Strathe, A.B.; Jayasundara, S.; Wagner-Riddle, C.; Dijkstra, J.; France, J.; Kebreab, E. Anti-methanogenic effects of monensin in dairy and beef cattle: A meta-analysis. J. Dairy Sci. 2013, 96, 5161–5173. [Google Scholar] [CrossRef]
- Hristov, A.N.; Lee, C.; Cassidy, T.T.; Heyler, K.; Tekippe, J.A.; Varga, G.A.; Corl, B.; Brandt, R.C. Effect of Origanum vulgare L. leaves on rumen fermentation, production, and milk fatty acid composition in lactating dairy cows. J. Dairy Sci. 2013, 96, 1189–1202. [Google Scholar] [CrossRef]
- Moate, P.J.; Williams, S.R.O.; Torok, V.A.; Hannah, M.C.; Ribaux, B.E.; Tavendale, M.H.; Eckard, R.J.; Jacobs, J.L.; Auldist, M.J.; Wales, W.J. Grape marc reduces methane emissions when fed to dairy cows. J. Dairy Sci. 2014, 97, 5073–5087. [Google Scholar] [CrossRef]
- Dijkstra, J.; Bannink, A.; France, J.; Kebreab, E.; van Gastelen, S. Antimethanogenic effects of 3-nitrooxypropanol depend on supplementation dose, dietary fiber content, and cattle type. J. Dairy Sci. 2018, 101, 9041–9047. [Google Scholar] [CrossRef]
- Martinez-Fernandez, G.; Duval, S.; Kindermann, M.; Schirra, H.J.; Denman, S.E.; McSweeney, C.S. 3-NOP vs. halogenated compound: Methane production, ruminal fermentation and microbial community response in forage fed cattle. Front. Microbiol. 2018, 9, 1582. [Google Scholar] [CrossRef] [PubMed]
- Hart, K.J.; Jones, H.G.; Waddams, K.E.; Worgan, H.J.; Zweifel, B.; Newbold, C.J. An essential oil blend decreases methane emissions and increases milk yield in dairy cows. Open J. Anim. Sci. 2019, 9, 259. [Google Scholar] [CrossRef]
- Roque, B.M.; Van Lingen, H.J.; Vrancken, H.; Kebreab, E. Effect of Mootral: A garlic- and citrus-extract-based feed additive: On enteric methane emissions in feedlot cattle. Transl. Anim. Sci. 2019, 3, 1383–1388. [Google Scholar] [CrossRef]
- Feng, X.Y.; Dijkstra, J.; Bannink, A.; van Gastelen, S.; France, J.; Kebreab, E. Anti-methanogenic effects of nitrate supplementation in cattle: A meta-analysis. J. Dairy Sci. 2020, 103, 11375–11385. [Google Scholar] [CrossRef] [PubMed]
- Roque, B.M.; Venegas, M.; Kinley, R.D.; de Nys, R.; Duarte, T.L.; Yang, X.; Kebreab, E. Red seaweed (Asparagopsis taxiformis) supplementation reduces enteric methane by over 80 percent in beef steers. PLoS ONE 2021, 16, e0247820. [Google Scholar] [CrossRef] [PubMed]
- Machado, L.; Magnusson, M.; Paul, N.A.; Kinley, R.; de Nys, R.; Tomkins, N. Identification of bioactives from the red seaweed Asparagopsis taxiformis that promote antimethanogenic activity in vitro. J. Appl. Phycol. 2016, 28, 3117–3126. [Google Scholar] [CrossRef]
- Roque, B.M.; Brooke, C.G.; Ladau, J.; Polley, T.; Marsh, L.J.; Najafi, N.; Pandey, P.; Singh, L.; Kinley, R.; Salwen, J.K.; et al. Effect of the macroalgae Asparagopsis taxiformis on methane production and rumen microbiome assemblage. Anim. Microbiome 2019, 1, 3. [Google Scholar] [CrossRef]
- Li, X.; Norman, H.C.; Kinley, R.D.; Laurence, M.; Wilmot, M.; Bender, H.; de Nys, R.; Tomkins, N. Asparagopsis taxiformis decreases enteric methane production from sheep. Anim. Prod. Sci. 2018, 58, 681–688. [Google Scholar] [CrossRef]
- Roque, B.M.; Salwen, J.K.; Kinley, R.; Kebreab, E. Inclusion of Asparagopsis armata in lactating dairy cows’ diet reduces enteric methane emission by over 50%. J. Clean Prod. 2019, 234, 132–138. [Google Scholar] [CrossRef]
- Kinley, R.D.; Martinez-Fernandez, G.; Matthews, M.K.; de Nys, R.; Magnusson, M.; Tomkins, N.W. Mitigating the carbon footprint and improving productivity of ruminant livestock agriculture using a red seaweed. J. Clean Prod. 2020, 59, 120836. [Google Scholar] [CrossRef]
- Stefenoni, H.A.; Räisänen, S.E.; Cueva, S.F.; Wasson, D.E.; Lage, C.F.A.; Melgar, A.; Fetter, M.E.; Smith, P.; Hennessy, M.; Vecchiarelli, B.; et al. Effects of the macroalga Asparagopsis taxiformis and oregano leaves on methane emission, rumen fermentation, and lactational performance of dairy cows. J. Dairy Sci. 2021, 104, 4157–4173. [Google Scholar] [CrossRef] [PubMed]
- Min, B.R.; Parker, D.; Brauer, D.; Waldrip, H.; Lockard, C.; Hales, K.; Akbay, A.; Augyte, S. The role of seaweed as a potential dietary supplementation for enteric methane mitigation in ruminants: Challenges and opportunities. Anim. Nut. 2021, 7, 1371–1387. [Google Scholar] [CrossRef] [PubMed]
- Haisan, J.; Sun, Y.; Guan, L.L.; Beauchemin, K.A.; Iwaasa, A.; Duval, S.; Barreda, D.R.; Oba, M. The effects of feeding 3-nitrooxypropanol on methane emissions and productivity of Holstein cows in mid lactation. J. Dairy Sci. 2014, 97, 3110–3119. [Google Scholar] [CrossRef] [PubMed]
- Hristov, A.N.; Oh, J.; Giallongo, F.; Frederick, T.W.; Harper, M.T.; Weeks, H.L.; Branco, A.F.; Moate, P.J.; Deighton, M.H.; Williams, S.R.O.; et al. An inhibitor persistently decreased enteric methane emission from dairy cows with no negative effect on milk production. Proc. Natl. Acad. Sci. USA 2015, 112, 10663–10668. [Google Scholar] [CrossRef] [PubMed]
- Lopes, J.C.; de Matos, L.F.; Harper, M.T.; Giallongo, F.; Oh, J.; Gruen, D.; Ono, S.; Kindermann, M.; Duval, S.; Hristov, A.N. Effect of 3-nitrooxypropanol on methane and hydrogen emissions, methane isotopic signature, and ruminal fermentation in dairy cows. J. Dairy Sci. 2016, 99, 5335–5344. [Google Scholar] [CrossRef]
- Vyas, D.; McGinn, S.M.; Duval, S.M.; Kindermann, M.; Beauchemin, K.A. Effects of sustained reduction of enteric methane emissions with dietary supplementation of 3-nitrooxypropanol on growth performance of growing and finishing beef cattle. J. Anim. Sci. 2016, 94, 2024–2034. [Google Scholar] [CrossRef]
- Vyas, D.; Alemu, A.W.; McGinn, S.M.; Duval, S.M.; Kindermann, M.; Beauchemin, K.A. The combined effects of supplementing monensin and 3-nitrooxypropanol on methane emissions, growth rate, and feed conversion efficiency in beef cattle fed high-forage and high-grain diets. J. Anim. Sci. 2018, 96, 2923–2938. [Google Scholar] [CrossRef]
- Melgar, A.; Welter, K.C.; Nedelkov, K.; Martins, C.M.M.R.; Harper, M.T.; Oh, J.; Räisänen, S.E.; Chen, X.; Cueva, S.F.; Duval, S.; et al. Dose-response effect of 3-nitrooxypropanol on enteric methane emissions in dairy cows. J. Dairy Sci. 2020, 103, 6145–6156. [Google Scholar] [CrossRef]
- Singer, S.D.; Hannoufa, A.; Acharya, S. Molecular improvement of alfalfa for enhanced productivity and adaptability in a changing environment. Plant Cell Environ. 2018, 41, 1955–1971. [Google Scholar] [CrossRef]
- Hünerberg, M.; Little, S.M.; Beauchemin, K.A.; McGinn, S.M.; O’Connor, D.; Okine, E.K.; Harstad, O.M.; Kröbel, R.; McAllister, T. Feeding high concentrations of corn dried distillers’ grains decreases methane, but increases nitrous oxide emissions from beef cattle production. Agric. Syst. 2014, 127, 19–27. [Google Scholar] [CrossRef]
- Lee, C.; Beauchemin, K.A. A review of feeding supplementary nitrate to ruminant animals: Nitrate toxicity, methane emissions, and production performance. Can. J. Anim. Sci. 2014, 94, 557–570. [Google Scholar] [CrossRef]
- Honan, M.; Feng, X.; Tricarico, J.M.; Kebreab, E. Feed additives as a strategic approach to reduce enteric methane production in cattle: Modes of action, effectiveness and safety. Anim. Prod. Sci. 2022, 62, 1303–1317. [Google Scholar] [CrossRef]
- Singer, S.D.; Laurie, J.; Bilichak, A.; Kumar, S.; Singh, J. Genetic variation and unintended risk in the context of old and new breeding techniques. Crit. Rev. Plant Sci. 2021, 40, 68–108. [Google Scholar] [CrossRef]
- Paques, F.; Duchateau, P. Meganucleases and DNA double-strand break-induced recombination: Perspectives for gene therapy. Curr. Gene Ther. 2007, 7, 49–66. [Google Scholar] [CrossRef] [PubMed]
- Papaioannou, I.; Simons, J.P.; Owen, J.S. Oligonucleotide-directed gene-editing technology: Mechanisms and future prospects. Exp. Opin. Biol. Ther. 2012, 12, 329–342. [Google Scholar] [CrossRef]
- Gaj, T.; Gersbach, C.A.; Barbas, C.F., III. ZFN, TALEN, and CRISPR/Cas-based methods for genome engineering. Trends Biotechnol. 2013, 31, 397–405. [Google Scholar] [CrossRef]
- Jinek, M.; Chylinski, K.; Fonfara, I.; Hauer, M.; Doudna, J.A.; Charpentier, E. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science 2012, 337, 816–821. [Google Scholar] [CrossRef]
- Meng, X.; Hu, X.; Liu, Q.; Song, X.; Gao, C.; Li, J.; Wang, K. Robust genome editing of CRISPR-Cas9 at NAG PAMs in rice. Sci. China Life Sci. 2018, 61, 122–125. [Google Scholar] [CrossRef]
- Puchta, H.; Fauser, F. Double-strand break repair and its application to genome engineering in plants. In Advances in New Technology for Targeted Modification of Plant Genomes; Zhang, F., Ed.; Springer: New York, NY, USA, 2015; pp. 1–20. [Google Scholar]
- White, M.F.; Allers, T. DNA repair in the archaea—An emerging picture. FEMS Microbiol. Rev. 2018, 42, 514–526. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhang, C.; Liu, W.; Gao, W.; Liu, C.; Song, G.; Li, W.-X.; Mao, L.; Chen, B.; Xu, Y.; et al. An alternative strategy for targeted gene replacement in plants using a dual-sgRNA/Cas9 design. Sci. Rep. 2016, 6, 23890. [Google Scholar] [CrossRef]
- Lu, Y.; Tian, Y.; Shen, R.; Yao, Q.; Wang, M.; Chen, M.; Dong, J.; Zhang, T.; Li, F.; Lei, M.; et al. Targeted, efficient sequence insertion and replacement in rice. Nat. Biotechnol. 2020, 38, 1402–1407. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Li, W. Development and application of CRISPR-Cas based tools. Front. Cell Dev. Biol. 2022, 10, 834646. [Google Scholar] [CrossRef] [PubMed]
- Zong, Y.; Wang, Y.; Li, C.; Zhang, R.; Chen, K.; Ran, Y.; Qiu, J.-L.; Wang, D.; Gao, C. Precise base editing in rice, wheat and maize with a Cas9-cytidine deaminase fusion. Nat. Biotechnol. 2017, 35, 438–440. [Google Scholar] [CrossRef]
- Huang, T.P.; Newby, G.A.; Liu, D.R. Precision genome editing using cytosine and adenine base editors in mammalian cells. Nat. Prot. 2021, 16, 1089–1128. [Google Scholar] [CrossRef]
- Anzalone, A.V.; Randolph, P.B.; Davis, J.R.; Sousa, A.A.; Koblan, L.W.; Levy, J.M.; Chen, P.J.; Wilson, C.; Newby, G.A.; Raguram, A.; et al. Search-and-replace genome editing without double-strand breaks or donor DNA. Nature 2019, 576, 149–157. [Google Scholar] [CrossRef]
- Lin, Q.; Zong, Y.; Wue, C.; Wang, S.; Jin, S.; Zhu, Z.; Wang, Y.; Anzalone, A.V.; Raguram, A.; Doman, J.L.; et al. Prime genome editing in rice and wheat. Nat. Biotechnol. 2020, 38, 582–585. [Google Scholar] [CrossRef] [PubMed]
- Hilton, I.B.; D’Ippolito, A.M.; Vockley, C.M.; Thakore, P.I.; Crawford, G.E.; Reddy, T.E.; Gersbach, C.A. Epigenome editing by a CRISPR-Cas9-based acetyltransferase activates genes from promoters and enhancers. Nat. Biotechnol. 2015, 33, 510–517. [Google Scholar] [CrossRef]
- Devesa-Guerra, I.; Morales-Ruiz, T.; Pérez-Roldán, J.; Parrilla-Doblas, J.T.; Dorado-León, M.; García-Ortiz, M.V.; Ariza, R.R.; Roldán-Arjona, T. DNA methylation editing by CRISPR-guided excision of 5-methylcytosine. J. Mol. Biol. 2020, 432, 2204–2216. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Wang, F.; Li, J.F. Targeted transcriptional activation in plants using a potent dead Cas9-derived synthetic gene activator. Curr. Prot. 2019, 127, e89. [Google Scholar] [CrossRef]
- Zheng, Y.; Su, T.; Qi, Q. Microbial CRISPRi and CRISPRa systems for metabolic engineering. Biotechnol. Bioprocess Eng. 2019, 24, 579–591. [Google Scholar] [CrossRef]
- Zhan, X.; Zhang, F.; Zhong, Z.; Chen, R.; Wang, Y.; Chang, L.; Bock, R.; Nie, B.; Zhang, J. Generation of virus-resistant potato plants by RNA genome targeting. Plant Biotechnol. J. 2019, 17, 1814–1822. [Google Scholar] [CrossRef]
- Lin, W.; Liu, Y.; Chemparathy, A.; Pande, T.; La Russa, M.; Qi, L.S. A comprehensive analysis and resource to use CRISPR-Cas13 for broad-spectrum targeting of RNA viruses. Cell Rep. Med. 2021, 2, 100245. [Google Scholar] [CrossRef] [PubMed]
- Sprink, T.; Wilhelm, R.; Hartung, F. Genome editing around the globe: An update on policies and perceptions. Plant Physiol. 2022, 190, 1579–1587. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Restall, J.; Crisp, P.; Godwin, I.; Liu, G. Current status and prospects of plant genome editing in Australia. Vitro Cell. Dev. Biol. Plant 2021, 57, 574–583. [Google Scholar] [CrossRef] [PubMed]
- Casler, M.D.; van Santen, E. Breeding objectives in forages. In Fodder Crops and Amenity Grasses. Handbook of Plant Breeding; Boller, B., Ed.; Springer: New York, NY, USA, 2010; Volume 5, pp. 115–136. [Google Scholar] [CrossRef]
- Chen, L.; Auh, C.-K.; Dowling, P.; Bell, J.; Chen, F.; Hopkins, A.; Dixon, R.A.; Wang, Z.-Y. Improved forage digestibility of tall fescue (Festuca arundinacea) by transgenic down-regulation of cinnamyl alcohol dehydrogenase. Plant Biotechnol. J. 2003, 1, 437–449. [Google Scholar] [CrossRef] [PubMed]
- Tang, L.; Cai, H.; Zhai, H.; Luo, X.; Wang, Z.; Cui, L.; Bai, X. Overexpression of Glycine soja WRKY20 enhances both drought and salt tolerance in transgenic alfalfa (Medicago sativa L.). Plant Cell Tiss. Org. Cult. 2014, 118, 77–86. [Google Scholar] [CrossRef]
- Arshad, M.; Feyissa, B.A.; Amyot, L.; Aung, B.; Hannoufa, A. MicroRNA156 improves drought stress tolerance in alfalfa (Medicago sativa) by silencing SPL13. Plant Sci. 2017, 258, 122–136. [Google Scholar] [CrossRef] [PubMed]
- Singer, S.D.; Weselake, R.J.; Acharya, S. Molecular enhancement of alfalfa: Improving quality traits for superior livestock performance and reduced environmental impact. Crop Sci. 2018, 58, 55–71. [Google Scholar] [CrossRef]
- Zhang, Y.; Ran, Y.; Nagy, I.; Lenk, I.; Qiu, J.-L.; Asp, T.; Jensen, C.S.; Gao, C. Targeted mutagenesis in ryegrass (Lolium spp.) using the CRISPR/Cas9 system. Plant Biotechnol. J. 2020, 18, 1854–1856. [Google Scholar] [CrossRef]
- Dinkins, R.D.; Hancock, J.; Coe, B.L.; May, J.B.; Goodman, J.P.; Bass, W.T.; Liu, J.; Fan, Y.; Zheng, Q.; Zhu, H. Isoflavone levels, nodulation and gene expression profiles of a CRISPR/Cas9 deletion mutant in the isoflavone synthase gene of red clover. Plant Cell Rep. 2021, 40, 517–528. [Google Scholar] [CrossRef]
- Singer, S.D.; Burton Hughes, K.; Subedi, U.; Dhariwal, G.K.; Kader, K.; Acharya, S.; Chen, G.; Hannoufa, A. The CRISPR/Cas9-mediated modulation of SQUAMOSA PROMOTER-BINDING PROTEIN-LIKE 8 in alfalfa leads to distinct phenotypic outcomes. Front. Plant Sci. 2022, 12, 774146. [Google Scholar] [CrossRef]
- Rodríguez-Leal, D.; Lemmon, Z.H.; Man, J.; Bartlett, M.E.; Lippman, Z.B. Engineering quantitative trait variation for crop improvement by genome editing. Cell 2017, 171, 470–480. [Google Scholar] [CrossRef]
- Zhang, H.; Si, X.; Ji, X.; Fan, R.; Liu, J.; Chen, K.; Wang, D.; Gao, C. Genome editing of upstream open reading frames enables translational control in plants. Nat. Biotechnol. 2018, 36, 894–898. [Google Scholar] [CrossRef]
- Beauchemin, K.A.; McGinn, S.M.; Benchaar, C.; Holtshausen, L. Crushed sunflower, flax, or canola seeds in lactating dairy cow diets: Effects on methane production, rumen fermentation, and milk production. J. Dairy Sci. 2009, 92, 2118–2127. [Google Scholar] [CrossRef]
- Grainger, C.; Beauchemin, K.A. Can enteric methane emissions from ruminants be lowered without lowering their production? Anim. Feed Sci. Technol. 2011, 166–167, 308–320. [Google Scholar] [CrossRef]
- Nogueira, R.G.S.; Perna, F.; Pereira, A.S.C.; Cassiano, E.C.O.; Carvalho, R.F.; Rodrigues, P.H.M. Methane mitigation and ruminal fermentation changes in cows fed cottonseed and vitamin E. Sci. Agrícola 2020, 77, e20180247. [Google Scholar] [CrossRef]
- Vargas, J.E.; Andrés, S.; López-Ferreras, L.; Snelling, T.J.; Yáñez-Ruíz, D.R.; García-Estrada, C.; López, S. Dietary supplemental plant oils reduce methanogenesis from anaerobic microbial fermentation in the rumen. Sci. Rep. 2020, 10, 1613. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, C.; Villalobos, R.; Peralta, A.M.T.; Morales, R.; Urrutia, N.L.; Ungerfield, E.M. Long-term and carryover effects of supplementation with whole oilseeds on methane emission, milk production and milk fatty acid profile of grazing dairy cows. Animals 2021, 11, 2978. [Google Scholar] [CrossRef]
- Beauchemin, K.A.; Kreuzer, M.; O’mara, F.; McAllister, T.A. Nutritional management for enteric methane abatement: A review. Aust. J. Exp. Agric. 2008, 48, 21–27. [Google Scholar] [CrossRef]
- Eugène, M.; Massé, D.; Chiquette, J.; Benchaar, C. Meta-analysis on the effects of lipid supplementation on methane production in lactating dairy cows. Can. J. Anim. Sci. 2008, 88, 331–337. [Google Scholar] [CrossRef]
- Bayat, A.R.; Tapio, I.; Vilkki, J.; Shingfield, K.J.; Leskinen, H. Plant oil supplements reduce methane emissions and improve milk fatty acid composition in dairy cows fed grass silage-based diets without affecting milk yield. J. Dairy Sci. 2018, 101, 1136–1151. [Google Scholar] [CrossRef] [PubMed]
- Boufaïed, H.; Chouinard, P.Y.; Tremblay, G.F.; Petit, H.V.; Michaud, R.; Bélanger, G. Fatty acids in forages. I. Factors affecting concentrations. Can. J. Anim. Sci. 2003, 83, 501–511. [Google Scholar] [CrossRef]
- Dewhurst, R.J.; Scollan, N.D.; Lee, M.R.F.; Ougham, H.J.; Humphreys, M.O. Forage breeding and management to increase the beneficial fatty acid content of ruminant products. Proc. Nutr. Soc. 2003, 62, 329–336. [Google Scholar] [CrossRef]
- Clapham, W.M.; Foster, J.G.; Neel, J.P.S.; Fedders, J.M. Fatty acid composition of traditional and novel forages. J. Agric. Food Chem. 2005, 53, 10068–10073. [Google Scholar] [CrossRef] [PubMed]
- Mel’uchová, B.; Blaško, J.; Kubinec, R.; Górová, R.; Dubravská, J.; Margetín, M.; Soják, L. Seasonal variations in fatty acid composition of pasture forage plants and CLA content in ewe milk fat. Small Rumin. Res. 2008, 78, 56–65. [Google Scholar] [CrossRef]
- Glasser, F.; Doreau, M.; Maxin, G.; Baumont, R. Fat and fatty acid content and composition of forages: A meta-analysis. Anim. Feed Sci. Technol. 2013, 185, 19–34. [Google Scholar] [CrossRef]
- Henderson, B.; Falcucci, A.; Mottet, A.; Early, L.; Werner, B.; Steinfeld, H.; Gerber, P. Marginal costs of abating greenhouse gases in the global ruminant livestock sector. Mitig. Adapt. Strateg. Glob. Chang. 2017, 22, 199–224. [Google Scholar] [CrossRef]
- Moate, P.J.; Williams, S.R.O.; Grainger, C.; Hannah, M.C.; Ponnampalam, E.N.; Eckard, R.J. Influence of cold-pressed canola, brewers grains and hominy meal as dietary supplements suitable for reducing enteric methane emissions from lactating dairy cows. Anim. Feed. Sci. Technol. 2011, 166–167, 254–264. [Google Scholar] [CrossRef]
- Li-Beisson, Y.; Shorrosh, B.; Beisson, F.; Andersson, M.X.; Arondel, V.; Bates, P.D.; Baud, S.; Bird, D.; DeBono, A.; Durrett, T.P.; et al. Acyl-lipid metabolism. Arab. Book 2013, 11, e0161. [Google Scholar] [CrossRef]
- Reszczyńska, E.; Hanaka, A. Lipids composition in plant membranes. Cell Biochem. Biophys. 2020, 78, 401–414. [Google Scholar] [CrossRef]
- Yang, Z.; Ohlrogge, J.B. Turnover of fatty acids during natural leaf senescence of Arabidopsis, Brachypodium, and switchgrass and in Arabidopsis β-oxidation mutants. Plant Physiol. 2009, 150, 1981–1989. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; Yu, L.; Xu, C. A central role for triacylglycerol in membrane lipid breakdown, fatty Acid β-oxidation, and plant survival under extended darkness. Plant Physiol. 2017, 174, 1517–1530. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Xu, Y.; Wang, J.; Singer, S.D.; Chen, G. The role of triacylglycerol in plant stress response. Plants 2020, 9, 472. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.; Oliver, D.J. Role of triacylglycerol in leaves. Plant Sci. 2008, 175, 233–237. [Google Scholar] [CrossRef]
- Ohlrogge, J.B.; Jaworski, J.G.; Post-Beittenmiller, D. De novo fatty acid biosynthesis. In Lipid Metabolism in Plants; Moore, T.S., Ed.; CRC Press: Boca Raton, FL, USA, 1993; pp. 4–32. [Google Scholar]
- He, M.; Qin, C.-W.; Wang, X.; Ding, N.-Z. Plant unsaturated fatty acids: Biosynthesis and regulation. Front. Plant Sci. 2020, 11, 390. [Google Scholar] [CrossRef]
- Jayawardhane, K.N.; Singer, S.D.; Weselake, R.J.; Chen, G. Plant sn-glycerol-3-phosphate acyltransferases: Biocatalysts involved in the biosynthesis of intracellular and extracellular lipids. Lipids 2018, 53, 469–480. [Google Scholar] [CrossRef]
- Wijekoon, C.P.; Singer, S.D.; Weselake, R.J.; Petrie, J.R.; Singh, S.; Jayawardhane, K.N.; Shah, S.; Chen, G.; Eastmond, P.J.; Acharya, S.N. Enhancement of total lipid production in vegetative tissues of alfalfa and sainfoin using chemical mutagenesis. Crop Sci. 2020, 60, 2990–3003. [Google Scholar] [CrossRef]
- Vanhercke, T.; Divi, U.K.; El Tahchy, A.; Liu, Q.; Mitchell, M.; Taylor, M.C. Step changes in leaf oil accumulation via iterative metabolic engineering. Metabol. Eng. 2017, 39, 237–246. [Google Scholar] [CrossRef]
- National Research Council (NRC). Nutrient Requirements of Dairy Cattle, 7th Revised ed.; National Academy Press: Washington, DC, USA, 2001. [Google Scholar]
- Liu, Y.-F.; Li, Z.-T.; Lu, X.; Song, Q.-X.; Lam, S.-M.; Zhang, W.-K.; Ma, B.; Lin, Q.; Man, W.-Q.; Du, W.-G.; et al. Soybean GmMYB73 promotes lipid accumulation in transgenic plants. BMC Plant Biol. 2014, 14, 73. [Google Scholar] [CrossRef]
- Beechey-Gradwell, Z.D.; Winichayakul, S.; Roberts, N.J. High lipid perennial ryegrass growth under variable nitrogen, water and carbon dioxide supply. J. N. Zeal. Grass. 2018, 80, 219–224. [Google Scholar] [CrossRef]
- Vanhercke, T.; Belide, S.; Taylor, M.C.; El Tahchy, A.; Okada, S.; Rolland, V.; Liu, Q.; Mitchell, M.; Shrestha, P.; Venables, I.; et al. Up-regulation of lipid biosynthesis increases the oil content in leaves of Sorghum bicolor. Plant Biotechnol. J. 2019, 17, 220–232. [Google Scholar] [CrossRef] [PubMed]
- Winichayakul, S.; Beechey-Gradwell, Z.; Muetzel, S.; Molano, G.; Crowther, T.; Lewis, S.; Xue, H.; Burke, J.; Bryan, G.; Roberts, N.J. In vitro gas production and rumen fermentation profile of fresh and ensiled genetically modified high-metabolizable energy ryegrass. J. Dairy Sci. 2020, 103, 2405–2418. [Google Scholar] [CrossRef] [PubMed]
- Subedi, U.; Jayawardhane, K.N.; Pan, X.; Ozga, J.; Chen, G.; Foroud, N.A.; Singer, S.D. The potential of genome editing for improving seed oil content and fatty acid composition in oilseed crops. Lipids 2020, 55, 495–512. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Liu, X.; Fan, C.; Li, T.; Qin, H.; Li, X.; Chen, K.; Zheng, Y.; Cheng, F.; Xu, Y. Enhancement of tobacco (Nicotiana tabacum L.) seed lipid content for biodiesel production by CRISPR-Cas9-mediated knockout of NtAn1. Front. Plant Sci. 2021, 11, 599474. [Google Scholar] [CrossRef]
- James, C.N.; Horn, P.J.; Case, C.R.; Gidda, S.K.; Zhang, D.; Mullen, R.T.; Dyer, J.M.; Anderson, R.G.W.; Chapman, K.D. Disruption of the Arabidopsis CGI-58 homologue produces Chanarin—Dorfman-like lipid droplet accumulation in plants. Proc. Natl. Acad. Sci. USA 2010, 107, 17833–17838. [Google Scholar] [CrossRef]
- Kelly, A.A.; van Erp, H.; Quettier, A.L.; Shaw, E.; Menard, G.; Kurup, S.; Eastmond, P.J. The SUGAR-DEPENDENT1 lipase limits triacylglycerol accumulation in vegetative tissues of Arabidopsis. Plant Physiol. 2013, 162, 1282–1289. [Google Scholar] [CrossRef]
- Wijekoon, C.; Singer, S.D.; Weselake, R.J.; Petrie, J.R.; Chen, G.; Singh, S.; Eastmond, P.J.; Acharya, S.N. Down-regulation of key genes involved in carbon metabolism in Medicago truncatula results in increased lipid accumulation in vegetative tissue. Crop Sci. 2020, 60, 1798–1808. [Google Scholar] [CrossRef]
- Zhai, Z.; Liu, H.; Xu, C.; Shanklin, J. Sugar potentiation of fatty acid and triacylglycerol accumulation. Plant Physiol. 2017, 175, 696–707. [Google Scholar] [CrossRef]
- Fan, J.; Zhai, Z.; Yan, C.; Xu, C. Arabidopsis TRIGALACTOSYLDIACYLGLYCEROL5 interacts with TGD1, TGD2, and TGD4 to facilitate lipid transfer from the endoplasmic reticulum to plastids. Plant Cell 2015, 27, 2941–2955. [Google Scholar] [CrossRef]
- Fan, J.; Yan, C.; Xu, C. Phospholipid: Diacylglycerol acyltransferase-mediated triacylglycerol biosynthesis is crucial for protection against fatty acid-induced cell death in growing tissues of Arabidopsis. Plant J. 2013, 76, 930–942. [Google Scholar] [CrossRef]
- Xu, C.; Fan, J.; Froehlich, J.E.; Awai, K.; Benning, C. Mutation of the TGD1 chloroplast envelope protein affects phosphatidate metabolism in Arabidopsis. Plant Cell 2005, 17, 3094–3110. [Google Scholar] [CrossRef] [PubMed]
- Jarvis, P.; Dörmann, P.; Peto, C.A.; Lutes, J.; Benning, C.; Chory, J. Galactolipid deficiency and abnormal chloroplast development in the Arabidopsis MGD synthase 1 mutant. Proc. Natl. Acad. Sci. USA 2000, 97, 8175–8179. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.Y.; Liu, C.; Li, M.C.; Zhao, M.M.; Gu, D.; Xu, Y.N. Effects of monogalactoglycerolipid deficiency and diacylglycerol acyltransferase overexpression on oil accumulation in transgenic tobacco. Plant Mol. Biol. Rep. 2013, 31, 1077–1088. [Google Scholar] [CrossRef]
- Yu, T.-S.; Lue, W.-L.; Wang, S.-M.; Chen, J. Mutation of Arabidopsis plastid phosphoglucose isomerase affects leaf starch synthesis and floral initiation. Plant Physiol. 2000, 123, 319–326. [Google Scholar] [CrossRef] [PubMed]
- Sanjaya; Durrett, T.P.; Weise, S.E.; Benning, C. Increasing the energy density of vegetative tissues by diverting carbon from starch to oil biosynthesis in transgenic Arabidopsis. Plant Biotechnol. J. 2011, 9, 874–883. [Google Scholar] [CrossRef]
- Lin, T.-P.; Caspar, T.; Somerville, C.; Preiss, J. Isolation and characterization of a starchless mutant of Arabidopsis thaliana (L.) Heynh lacking ADPglucose pyrophosphorylase activity. Plant Physiol. 1988, 86, 1131–1135. [Google Scholar] [CrossRef]
- Müller-Röber, B.; Sonnewald, U.; Willmitzer, L. Inhibition of the ADP-glucose pyrophosphorylase in transgenic potatoes leads to sugar-storing tubers and influences tuber formation and expression of tuber storage protein genes. EMBO J. 1992, 11, 1229–1238. [Google Scholar] [CrossRef]
- Rösti, S.; Rudi, H.; Rudi, K.; Opsahl-Sorteberg, H.-G.; Fahy, B.; Denyer, K. The gene encoding the cytosolic small subunit of ADP-glucose pyrophosphorylase in barley endosperm also encodes the major plastidial small subunit in the leaves. J. Exp. Bot. 2006, 57, 3619–3626. [Google Scholar] [CrossRef][Green Version]
- Lee, S.-K.; Hwang, S.-K.; Han, M.; Eom, J.-S.; Kang, H.-G.; Han, Y.; Choi, S.-B.; Cho, M.-H.; Bhoo, S.H.; An, G.; et al. Identification of the ADP-glucose pyrophosphorylase isoforms essential for starch synthesis in the leaf and seed endosperm of rice (Oryza sativa L.). Plant Mol. Biol. 2007, 65, 531–546. [Google Scholar] [CrossRef]
- Rösti, S.; Fahy, B.; Denyer, K. A mutant of rice lacking the leaf large subunit of ADP-glucose pyrophosphorylase has drastically reduced leaf starch content but grows normally. Funct. Plant Biol. 2007, 34, 480–489. [Google Scholar] [CrossRef]
- Cossegal, M.; Chambrier, P.; Mbelo, S.; Balzergue, S.; Martin-Magniette, M.-L.; Moing, A.; Deborde, C.; Guyon, V.; Perez, P.; Rogowsky, P. Transcriptional and metabolic adjustments in ADP-glucuse pyrophosphorylase-deficient bt2 maize kernels. Plant Physiol. 2008, 146, 1553–1570. [Google Scholar] [CrossRef] [PubMed]
- Slewinski, T.L.; Ma, Y.; Baker, F.; Huang, M.; Meeley, R.; Braun, D.M. Determining the role of Tie-dyed1 in starch metabolism: Epistasis analysis with a maize ADP-glucose pyrophosphorylase mutant lacking leaf starch. J. Hered. 2008, 99, 661–666. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Fan, J.; Yan, C.; Xu, C. Starch deficiency enhances lipid biosynthesis and turnover in leaves. Plant Physiol. 2018, 178, 118–129. [Google Scholar] [CrossRef] [PubMed]
- Khorrami, B.; Khiaosa-ard, R.; Zebeli, Q. Models to predict the risk of subacute ruminal acidosis in dairy cows based on dietary and cow factors: A meta-analysis. J. Dairy Sci. 2021, 104, 7761–7780. [Google Scholar] [CrossRef] [PubMed]
- Rivero, M.J.; Keim, J.P.; Balocchi, O.A.; Lee, M.R.F. In vitro fermentation patterns and methane output of perennial ryegrass differing in water-soluble carbohydrate and nitrogen concentrations. Animals 2020, 10, 1076. [Google Scholar] [CrossRef] [PubMed]
- Zale, J.; Jung, J.H.; Kim, J.Y.; Pathak, B.; Karan, R.; Liu, H.; Chen, X.; Wu, H.; Candreva, J.; Zhai, Z.; et al. Metabolic engineering of sugarcane to accumulate energy-dense triacylglycerols in vegetative biomass. Plant Biotechnol. J. 2016, 14, 661–669. [Google Scholar] [CrossRef] [PubMed]
- Eastmond, P.J. SUGAR-DEPENDENT1 encodes a patatin domain triacylglycerol lipase that initiates storage oil breakdown in germinating Arabidopsis seeds. Plant Cell 2006, 18, 665–675. [Google Scholar] [CrossRef]
- Park, S.; Gidda, S.K.; James, C.N.; Horn, P.J.; Khuu, N.; Seay, D.C.; Keereetaweep, J.; Chapman, K.D.; Mullen, R.T.; Dyer, J.M. The α/β hydrolase CGI-58 and peroxisomal transport protein PXA1 coregulate lipid homeostasis and signaling in Arabidopsis. Plant Cell 2013, 25, 1726–1739. [Google Scholar] [CrossRef]
- Kelly, A.A.; Quettier, A.-L.; Shaw, E.; Eastmond, P.J. Seed storage oil mobilization is important but not essential for germination or seedling establishment in Arabidopsis. Plant Physiol. 2011, 157, 866–875. [Google Scholar] [CrossRef]
- Xu, X.; Vanhercke, T.; Shrestha, P.; Luo, J.; Akbar, S.; Konik-Rose, C.; Venugoban, L.; Hussain, D.; Tian, L.; Singh, S.; et al. Upregulated lipid biosynthesis at the expense of starch production in potato (Solanum tuberosum) vegetative tissues via simultaneous downregulation of ADP-glucose pyrophosphorylase and sugar dependent1 expressions. Front. Plant Sci. 2019, 10, 1444. [Google Scholar] [CrossRef]
- Bhunia, R.K.; Medard, G.N.; Eastmond, P.J. A native promoter-gene fusion created by CRISPR/Cas9-mediated genomic deletion offers a transgene-free method to drive oil accumulation in leaves. FEBS Lett. 2022, 596, 1865–1870. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Harwood, J.L.; Lemieux, M.J.; Stone, S.J.; Weselake, R.J. Acyl-CoA: Diacylglycerol acyltransferase: Properties, physiological roles, metabolic engineering and intentional control. Prog. Lipid Res. 2022, 88, 101181. [Google Scholar] [CrossRef] [PubMed]
- Boadi, D.; Benchaar, C.; Chiquett, J.; Massé, D. Mitigation strategies to reduce enteric methane emissions from dairy cows: Update review. Can. J. Anim. Sci. 2004, 84, 319–335. [Google Scholar] [CrossRef]
- Haque, M.N. Dietary manipulation: A sustainable way to mitigate methane emissions from ruminants. J. Anim. Sci. Technol. 2018, 60, 15. [Google Scholar] [CrossRef]
- Verma, S.; Taube, F.; Malisch, C.S. Examining the variables leading to apparent incongruity between antimethanogenic potential of tannins and their observed effects in ruminants—A review. Sustainability 2021, 13, 2743. [Google Scholar] [CrossRef]
- Chung, Y.H.; McGeough, E.J.; Acharya, S.; McAllister, T.A.; McGinn, S.M.; Harstad, O.M.; Beauchemin, K.A. Enteric methane emission, diet digestibility, and nitrogen excretion from beef heifers fed sainfoin or alfalfa. J. Anim. Sci. 2013, 91, 4861–4874. [Google Scholar] [CrossRef] [PubMed]
- Huyen, N.T.; Desrues, O.; Alferink, S.J.J.; Zandstra, T.; Verstegen, M.W.A.; Hendriks, W.H.; Pellikaan, W.F. Inclusion of sainfoin (Onobrychis viciifolia) silage in dairy cow rations affects nutrient digestibility, nitrogen utilization, energy balance, and methane emissions. J. Dairy Sci. 2016, 99, 3566–3577. [Google Scholar] [CrossRef] [PubMed]
- Tavendale, M.H.; Meagher, L.P.; Pacheco, D.; Walker, N.; Attwood, G.T.; Sivakumaran, S. Methane production from in vitro rumen incubations with Lotus pedunculatus and Medicago sativa, and effects of extractable condensed tannin fractions on methanogenesis. Anim. Feed Sci. Technol. 2005, 123–124, 403–419. [Google Scholar] [CrossRef]
- Naumann, H.D.; Tedeschi, L.O.; Zeller, W.E.; Huntley, N.F. The role of condensed tannins in ruminant animal production: Advances, limitations and future directions. R. Bras. Zootec. 2017, 46, 929–949. [Google Scholar] [CrossRef]
- Hatew, B.; Carbonero, C.H.; Stringano, E.; Sales, L.F.; Smith, L.M.J.; Mueller-Harvey, I.; Hendriks, W.H.; Pellikaan, W.F. Diversity of condensed tannin structures affects rumen in vitro methane production in sainfoin (Onobrychis viciifolia) accessions. Grass Forage Sci. 2015, 70, 474–490. [Google Scholar] [CrossRef]
- Naumann, H.; Sepela, R.; Rezaire, A.; Masih, S.E.; Zeller, W.E.; Reinhardt, L.A.; Robe, J.T.; Sullivan, M.L.; Hagerman, A.E. Relationships between structures of condensed tannins from Texas legumes and methane production during in vitro rumen digestion. Molecules 2018, 23, 2123. [Google Scholar] [CrossRef] [PubMed]
- Aboagye, A.; Beauchemin, K.A. Potential of molecular weight and structure of tannins to reduce methane emissions from ruminants: A review. Animals 2019, 9, 856. [Google Scholar] [CrossRef] [PubMed]
- Jayanegara, A.; Marquardt, S.; Wina, E.; Kreuzer, M.; Leiber, F. In vitro indications for favourable non-additive effects on ruminal methane mitigation between high-phenolic and high-quality forages. Brit. J. Nutr. 2013, 109, 615–622. [Google Scholar] [CrossRef]
- Eugène, M.; Klumpp, K.; Sauvant, D. Methane mitigating options with forages fed to ruminants. Grass Forage Sci. 2021, 76, 196–204. [Google Scholar] [CrossRef]
- Van Gastelen, S.; Dijkstra, J.; Bannink, A. Are dietary strategies to mitigate enteric methane emission equally effective across dairy cattle, beef cattle, and sheep? J. Dairy Sci. 2019, 102, 6109–6130. [Google Scholar] [CrossRef]
- Nayak, D.D.; Metcalf, W.W. Cas9-mediated genome editing in the methanogenic archaeon Methanosarcina acetivorans. Proc. Natl. Acad. Sci. USA 2017, 114, 2976–2981. [Google Scholar] [CrossRef]
- Bartlett, E.J.; Brissett, N.C.; Doherty, A.J. Ribonucleolytic resection is required for repair of strand displaced nonhomologous end-joining intermediates. Proc. Natl. Acad. Sci. USA 2013, 110, E1984–E1991. [Google Scholar] [CrossRef]
- Li, Y.; Peng, N. Endogenous CRISPR-Cas system-based genome editing and antimicrobials: Review and prospects. Front. Microbiol. 2019, 10, 2471. [Google Scholar] [CrossRef]
- Li, Y.; Pan, S.; Zhang, Y.; Ren, M.; Feng, M.; Peng, N.; Chen, L.; Liang, Y.X.; She, Q. Harnessing type I and type III CRISPR-Cas systems for genome editing. Nucleic Acids Res. 2016, 44, e34. [Google Scholar] [CrossRef] [PubMed]
- Khelaifia, S.; Garibal, M.; Robert, C.; Raoult, D.; Drancourt, M. Draft genome sequence of a human-associated isolate of Methanobrevibacter arboriphilicus, the lowest-G + C-content Archaeon. Genome Announc. 2014, 2, e01181-13. [Google Scholar] [CrossRef] [PubMed]
- Khelaifia, S.; Garibal, M.; Robert, C.; Raoult, D.; Drancourt, M. Draft genome sequencing of Methanobrevibacter oralis strain JMR01, isolated from the human intestinal microbiota. Genome Announc. 2014, 2, e00073-14. [Google Scholar] [CrossRef] [PubMed]
- Weimer, P.J. Redundancy, resilience, and host specificity of the ruminal microbiota: Implications for engineering improved ruminal fermentations. Front. Microbiol. 2015, 6, 296. [Google Scholar] [CrossRef]
- Kamke, J.; Kittelmann, S.; Soni, P.; Li, Y.; Tavendale, M.; Ganesh, S.; Janssen, P.H.; Shi, W.; Froula, J.; Rubin, E.M.; et al. Rumen metagenome and metatranscriptome analyses of low methane yield sheep reveals a Sharpea-enriched microbiome characterised by lactic acid formation and utilisation. Microbiome 2016, 4, 56. [Google Scholar] [CrossRef]
- Auffret, M.D.; Stewart, R.D.; Dewhurst, R.J.; Duthie, C.-A.; Watson, M.; Roehe, R. Identification of microbial genetic capacities and potential mechanisms within the rumen microbiome explaining differences in beef cattle feed efficiency. Front. Microbiol. 2020, 11, 1229. [Google Scholar] [CrossRef] [PubMed]
- Newbold, D.J.; Ramos-Morales, E. Ruminal microbiome and microbial metabolome: Effects of diet and ruminant host. Animal 2020, 14, s78–s86. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.-Z.; Peng, K.-L.; Xue, M.-Y.; Liu, J.-X. Metagenomics analysis revealed the distinctive ruminal microbiome and resistive profiles in dairy buffaloes. Anim. Microbiome 2021, 3, 44. [Google Scholar] [CrossRef]
- Henderson, G.; Cox, F.; Ganesh, S.; Jonker, A.; Young, W.; Global Rumen Census Collaborators; Janssen, P.H. Rumen microbial community composition varies with diet and host, but a core microbiome is found across a wide geographical range. Sci. Rep. 2015, 5, 14567. [Google Scholar] [CrossRef]
- Islam, M.; Lee, S.S. Advanced estimation and mitigation strategies: A cumulative approach to enteric methane abatement from ruminants. J. Anim. Sci. Technol. 2019, 61, 122–137. [Google Scholar] [CrossRef]
- Janssen, P.H.; Kirs, M. Structure of the archaeal community of the rumen. Appl. Environ. Microbiol. 2008, 74, 3619–3625. [Google Scholar] [CrossRef] [PubMed]
- Patra, A.; Park, T.; Kim, M.; Yu, Z. Rumen methanogens and mitigation of methane emission by anti-methanogenic compounds and substances. J. Anim. Sci. Biotechnol. 2017, 8, 13. [Google Scholar] [CrossRef] [PubMed]
- Stams, A.J.; Plugge, C.M. Electron transfer in syntrophic communities of anaerobic bacteria and archaea. Nat. Rev. Microbiol. 2009, 7, 568–577. [Google Scholar] [CrossRef] [PubMed]
- Difford, G.F.; Plichta, D.R.; Løvendahl, P.; Lassen, J.; Noel, S.J.; Højberg, O.; Wright, A.G.; Zhu, Z.; Kristensen, L.; Nielsen, H.B.; et al. Host genetics and the rumen microbiome jointly associate with methane emissions in dairy cows. PLoS Genet. 2018, 14, e1007580. [Google Scholar] [CrossRef] [PubMed]
- Sasson, G.; Kruger Ben-Shabat, S.; Seroussi, E.; Doron-Faigenboim, A.; Shterzer, N.; Yaacoby, S.; Berg Miller, M.E.; White, B.A.; Halperin, E.; Mizrahi, I. Heritable bovine rumen bacteria are phylogenetically related and correlated with the cow’s capacity to harvest energy from its feed. mBio 2017, 8, e00703–e00717. [Google Scholar] [CrossRef]
- Abbas, W.; Howard, J.T.; Paz, H.A.; Hales, K.E.; Wells, J.E.; Kuehn, L.A.; Erickson, G.E.; Spangler, M.L.; Fernando, S.C. Influence of host genetics in shaping the rumen bacterial community in beef cattle. Sci. Rep. 2020, 10, 15101. [Google Scholar] [CrossRef]
- Leahy, S.C.; Kelly, W.J.; Altermann, E.; Ronimus, R.S.; Yeoman, C.J.; Pacheco, D.M.; Li, D.; Kong, Z.; McTavish, S.; Sang, C.; et al. The genome sequence of the rumen methanogen Methanobrevibacter ruminantium reveals new possibilities for controlling ruminant methane emissions. PLoS ONE 2010, 5, e8926. [Google Scholar] [CrossRef]
- Bharathi, M.; Senthil Kumar, N.; Chellapandi, P. Functional prediction and assignment of Methanobrevibacter ruminantium m1 operome using a combined bioinformatics approach. Front. Genet. 2020, 11, 593990. [Google Scholar] [CrossRef]
- Deppenmeier, U. The unique biochemistry of methanogenesis. Prog. Nucleic Acid Res. Mol. Biol. 2002, 71, 223–283. [Google Scholar] [CrossRef]
- Ferry, J.G. Fundamentals of methanogenic pathways that are key to the biomethanation of complex biomass. Curr. Opin. Biotechnol. 2011, 22, 351–357. [Google Scholar] [CrossRef]
- Shi, W.; Moon, C.D.; Leahy, S.C.; Kang, D.; Froula, J.; Kittelmann, S.; Fan, C.; Deutsch, S.; Gagic, D.; Seedorf, H.; et al. Methane yield phenotypes linked to differential gene expression in the sheep rumen microbiome. Genome Res. 2014, 24, 1517–1525. [Google Scholar] [CrossRef]
- Rother, M.; Boccazzi, P.; Bose, A.; Pritchett, M.A.; Metcalf, W.W. Methanol-dependent gene expression demonstrates that methyl-coenzyme M reductase is essential in Methanosarcina acetivorans C2A and allows isolation of mutants with defects in regulation of the methanol utilization pathway. J. Bacteriol. 2005, 187, 5552–5559. [Google Scholar] [CrossRef]
- Qi, L.S.; Larson, M.H.; Gilbert, L.A.; Doudna, J.A.; Weissman, J.S.; Arkin, A.P.; Lim, W.A. Repurposing CRISPR as an RNA-guided platform for sequence-specific control of gene expression. Cell 2013, 152, 1173–1183. [Google Scholar] [CrossRef] [PubMed]
- Mandegar, M.A.; Huebsch, N.; Frolov, E.B.; Shin, E.; Truong, A.; Olvera, M.P.; Chan, A.H.; Miyaoka, Y.; Holmes, K.; Spencer, C.I.; et al. CRISPR interference efficiently induces specific and reversible gene silencing in human iPSCs. Cell Stem Cell 2016, 8, 541–553. [Google Scholar] [CrossRef] [PubMed]
- Dhamad, A.E.; Lessner, D.J. A CRISPRi-dCas9 system for archaea and its use to examine gene function during nitrogen fixation by Methanosarcina acetivorans. Appl. Environ. Microbiol. 2020, 86, e01402–e01420. [Google Scholar] [CrossRef] [PubMed]
- Hale, C.R.; Cocozaki, A.; Li, H.; Terns, R.M.; Terns, M.P. Target RNA capture and cleavage by the Cmr type III-B CRISPR-Cas effector complex. Genes Dev. 2014, 28, 2432–2443. [Google Scholar] [CrossRef]
- Zink, I.A.; Wimmer, E.; Schleper, C. Heavily armed ancestors: CRISPR immunity and applications in archaea with a comparative analysis of CRISPR types in Sulfolobales. Biomolecules 2020, 10, 1523. [Google Scholar] [CrossRef] [PubMed]
- Zebec, Z.; Manica, A.; Zhang, J.; White, M.F.; Schleper, C. CRISPR-mediated targeted mRNA degradation in the archaeon Sulfolobus solfataricus. Nucleic Acids Res. 2014, 42, 5280–5288. [Google Scholar] [CrossRef]
- Zink, I.A.; Fouqueau, T.; Tarrason Risa, G.; Werner, F.; Baum, B.; Bläsi, U.; Schleper, C. Comparative CRISPR type III-based knockdown of essential genes in hyperthermophilic Sulfolobales and the evasion of lethal gene silencing. RNA Biol. 2021, 18, 421–434. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Subedi, U.; Kader, K.; Jayawardhane, K.N.; Poudel, H.; Chen, G.; Acharya, S.; Camargo, L.S.A.; Bittencourt, D.M.d.C.; Singer, S.D. The Potential of Novel Gene Editing-Based Approaches in Forages and Rumen Archaea for Reducing Livestock Methane Emissions. Agriculture 2022, 12, 1780. https://doi.org/10.3390/agriculture12111780
Subedi U, Kader K, Jayawardhane KN, Poudel H, Chen G, Acharya S, Camargo LSA, Bittencourt DMdC, Singer SD. The Potential of Novel Gene Editing-Based Approaches in Forages and Rumen Archaea for Reducing Livestock Methane Emissions. Agriculture. 2022; 12(11):1780. https://doi.org/10.3390/agriculture12111780
Chicago/Turabian StyleSubedi, Udaya, Kazi Kader, Kethmi N. Jayawardhane, Hari Poudel, Guanqun Chen, Surya Acharya, Luiz S. A. Camargo, Daniela Matias de C. Bittencourt, and Stacy D. Singer. 2022. "The Potential of Novel Gene Editing-Based Approaches in Forages and Rumen Archaea for Reducing Livestock Methane Emissions" Agriculture 12, no. 11: 1780. https://doi.org/10.3390/agriculture12111780
APA StyleSubedi, U., Kader, K., Jayawardhane, K. N., Poudel, H., Chen, G., Acharya, S., Camargo, L. S. A., Bittencourt, D. M. d. C., & Singer, S. D. (2022). The Potential of Novel Gene Editing-Based Approaches in Forages and Rumen Archaea for Reducing Livestock Methane Emissions. Agriculture, 12(11), 1780. https://doi.org/10.3390/agriculture12111780








