Carotenoid Content and Bioaccessibility in Commercial Maize Hybrids
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. In Vitro Digestion Procedure
2.3. Extraction of Carotenoids from Whole Maize Grain
2.4. Extraction of Carotenoids from Micellar Fraction
2.5. Statistical Analysis
3. Results and Discussion
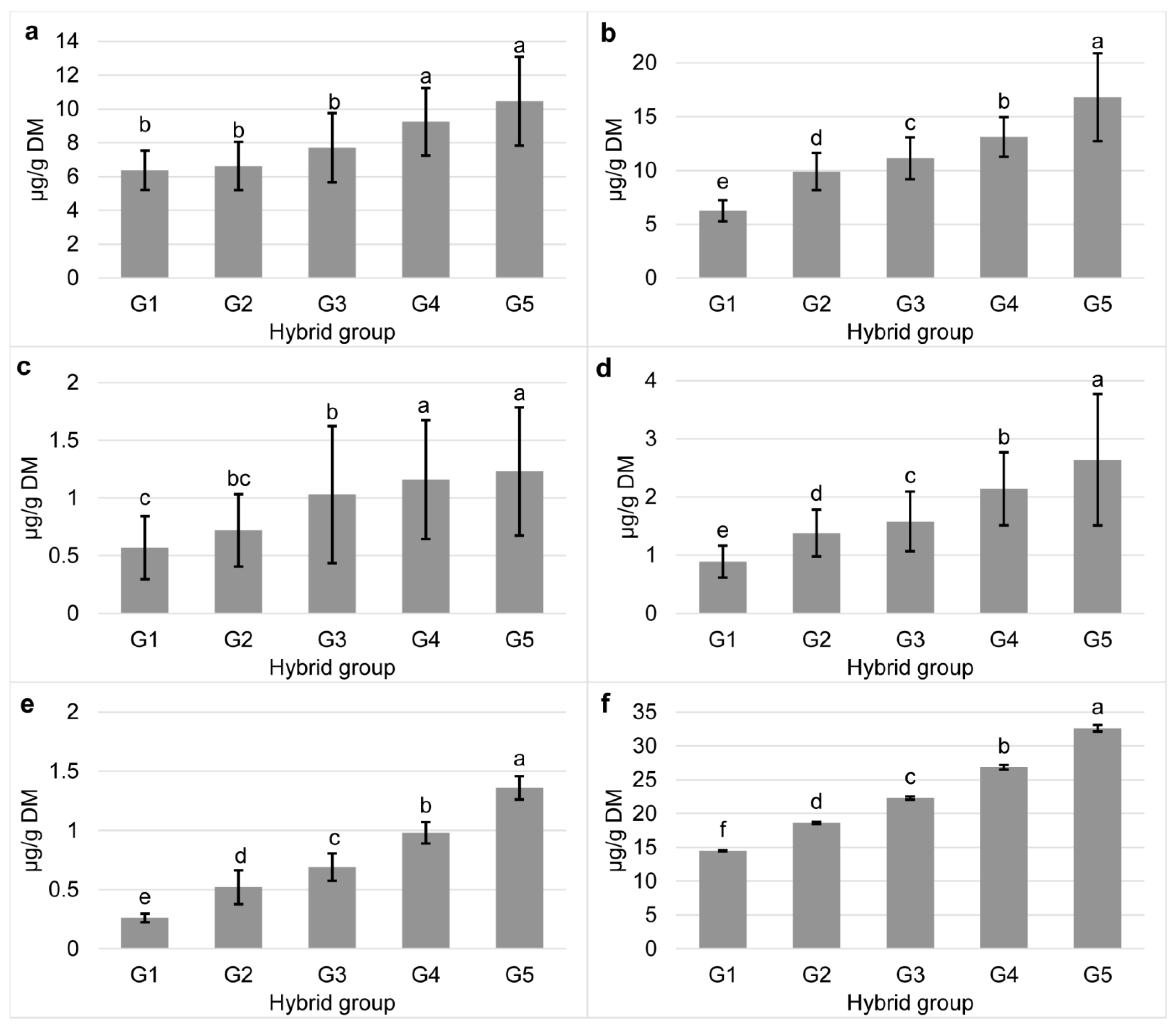
3.1. Carotenoid Content in Tested Commercial Maize Hybrids
3.2. Bioaccessibility of Carotenoids in Commercial Maize Hybrids
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mezzomo, N.; Ferreira, S.R.S. Carotenoids functionality, sources, and processing by supercritical technology: A review. J. Chem. 2016, 3164312. [Google Scholar] [CrossRef] [Green Version]
- Menkir, A.; Pixley, K.; Maziya-Dixon, B.; Gedil, M. Recent advances in breeding maize for enhanced provitamin A content. In Meeting the Challenges of Global Climate Change and Food Security through Innovative Maize Research, Proceedings of the Third National Maize Workshop of Ethiopia, Addis Ababa, Ethiopia, 18–20 April 2011; Worku, M., Twumasi Afriyie, S., Legesse, W., Berhanu Tadesse, E., Demisie, G., Bogale, G., Dagne Wegary, G., Prasanna, B.M., Eds.; International Maize and Wheat Improvement Center: Texcoco, Mexico, 2012; p. 290. [Google Scholar]
- Ashokkumar, K.; Govindaraj, M.; Karthikeyan, A.; Shobhana, V.G.; Warkentin, T.D. Genomics-integrated breeding for carotenoids and folates in staple cereal grains to reduce malnutrition. Front. Genet. 2020, 11, 414. [Google Scholar] [CrossRef] [PubMed]
- Meléndez-Martínez, A.J.; Mandić, A.I.; Bantis, F.; Böhm, V.; Borge, G.I.A.; Brnčić, M.; Bysted, A.; Cano, M.P.; Graça Dias, M.; Elgersma, A.; et al. A comprehensive review on carotenoids in foods and feeds: Status quo, applications, patents, and research needs. Crit. Rev. Food Sci. Nutr. 2021. [Google Scholar] [CrossRef] [PubMed]
- Muzhingi, T.; Yeum, K.J.; Russell, R.M.; Johnson, E.; Qin, J.; Tang, G. Determination of carotenoids in yellow maize, the effects of saponification and food preparations. Int. J. Vitam. Nutr. Res. 2008, 78, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Halilu, A.D.; Ado, S.G.; Aba, A.D.; Usman, S.I. Genetics of carotenoids for provitamin A biofortification in tropical-adapted maize. Crop. J. 2016, 4, 313–322. [Google Scholar] [CrossRef] [Green Version]
- Andersson, M.S.; Saltzman, A.; Virk, P.S.; Pfeiffer, W.H. Progress update: Crop development of biofortified staple food crops under HarvestPlus. Afr. J. Food Nutr. Dev. 2017, 17, 11905–11935. [Google Scholar] [CrossRef]
- Berardo, N.; Brenna, O.V.; Amato, A.; Valoti, P.; Pisacane, V.; Motto, M. Carotenoids concentration among maize genotypes measured by near infrared reflectance spectroscopy (NIRS). Innov. Food Sci. Emerg. Technol. 2004, 5, 393–398. [Google Scholar] [CrossRef]
- Schieber, A.; Weber, F. Carotenoids. In Handbook on Natural Pigments in Food and Beverages, Industrial Applications for Improving Food Color, 1st ed.; Carle, R., Schweiggert, R.M., Eds.; Woodhead Publishing Series in Food Science, Technology and Nutrition: Cambridge, UK, 2016; pp. 101–123. [Google Scholar] [CrossRef]
- Carbonell-Capella, J.M.; Buniowska, M.; Barba, F.J.; Esteve, M.J.; Frìgola, A. Analytical methods for determining bioavailability and bioaccessibility of bioactive compounds from fruits and vegetables: A review. Compr. Rev. Food Sci. Food Saf. 2014, 13, 155–171. [Google Scholar] [CrossRef]
- van Het Hof, K.H.; West, C.E.; Weststrate, J.A.; Hautvast, J.G. Dietary factors that affect the bioavailability of carotenoids. J. Nutr. 2000, 130, 503–506. [Google Scholar] [CrossRef]
- Dima, C.; Assadpour, E.; Dima, S.; Mahdi Jafari, S. Bioavailability of nutraceuticals: Role of the food matrix, processing conditions, the gastrointestinal tract, and nanodelivery systems. Compr. Rev. Food Sci. Food Saf. 2020, 19, 954–994. [Google Scholar] [CrossRef]
- Sugiura, M.; Nakamura, M.; Ikoma, Y.; Yano, M.; Ogawa, K.; Matsumoto, H.; Kato, M.; Ohshima, M.; Nagao, A. Serum carotenoid concentrations are inversely associated with serum aminotransferases in hyperglycemic subjects. Diabetes Res. Clin. Pract. 2006, 71, 82–91. [Google Scholar] [CrossRef]
- Zhai, S.; Xia, X.; He, Z. Carotenoids in staple cereals: Metabolism, regulation, and genetic manipulation. Front. Plant Sci. 2016, 7, 1197. [Google Scholar] [CrossRef] [Green Version]
- Kean, E.G.; Hamaker, B.R.; Ferruzzi, M.G. Carotenoid bioaccessibility from whole grain and degermed maize meal products. J. Agric. Food Chem. 2008, 56, 9918–9926. [Google Scholar] [CrossRef] [PubMed]
- Thakkar, S.K.; Failla, M.L. Bioaccessibility of pro-vitamin A carotenoids is minimally affected by non pro-vitamin A xanthophylls in maize (Zea mays sp.). J. Agric. Food Chem. 2008, 56, 11441–11446. [Google Scholar] [CrossRef] [PubMed]
- Hossain, A.; Jayadeep, P.A. Comparison of total carotenoids, lutein, zeaxanthin, and β-carotene content in maize employing solvent extraction and in vitro physiological methods. J. Food Biochem. 2018, 42, 1–9. [Google Scholar] [CrossRef]
- Brodkorb, A.; Egger, L.; Alminger, M.; Alvito, P.; Assunção, R.; Balance, S.; Bohn, T.; Bourlieu-Lacanal, C.; Boutrou, R.; Carrière, F.; et al. INFOGEST static in vitro simulation of gastrointestinal food digestion. Nat. Protoc. 2019, 14, 991–1014. [Google Scholar] [CrossRef] [PubMed]
- Estévez-Santiago, R.; Olmedilla-Alonso, B.; Fernández-Jalao, I. Bioaccessibility of provitamin A carotenoids from fruits: Application of a standardised static in vitro digestion method. Food Funct. 2016, 7, 1354–1366. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, D.B.; Mariutti, L.R.B.; Mercadante, A.Z. An in vitro digestion method adapted for carotenoids and carotenoid esters: Moving forward towards standardization. Food Funct. 2016, 7, 4992–5001. [Google Scholar] [CrossRef] [PubMed]
- Petry, F.C.; Mercadante, A.Z. Impact of in vitro digestion phases on the stability and bioaccessibility of carotenoids and their esters in mandarin pulps. Food Funct. 2017, 8, 3951–3963. [Google Scholar] [CrossRef]
- Tan, Y.; Zhang, Z.; Zhou, H.; Xiao, H.; McClements, D.J. Factors impacting lipid digestion and β-carotene bioaccessibility assessed by standardized gastrointestinal model (INFOGEST): Oil droplet concentration. Food Funct. 2020, 11, 7126–7137. [Google Scholar] [CrossRef]
- Egger, L.; Schlegel, P.; Baumann, C.; Stoffers, H.; Guggisberg, D.; Brügger, C.; Dürr, D.; Stoll, P.; Vergères, G.; Portmann, R. Physiological comparability of the harmonized INFOGEST in vitro digestion method to in vivo pig digestion. Food Res. Int. 2017, 102, 567–574. [Google Scholar] [CrossRef] [PubMed]
- Englyst, H.N.; Kingman, S.M.; Cummings, J.H. Classification and measurement of nutritionally important starch fractions. Eur. J. Clin. Nutr. 1992, 46, S33–S50. [Google Scholar] [PubMed]
- Weurding, R.E.; Veldman, A.; Veen, W.A.; van der Aar, P.J.; Verstegen, M.W. In vitro starch digestion correlates well with 414 rate and extent of starch digestion in broiler chickens. J. Nutr. 2001, 131, 2336–2342. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vinarov, Z.; Petkova, Y.; Tcholakova, S.; Denkov, N.; Stoyanov, S.; Pelan, E.; Lips, A. Effects of emulsifier charge and concentration on pancreatic lipolysis. 1. In the absence of bile salts. Langmuir 2012, 28, 8127–8139. [Google Scholar] [CrossRef]
- Mashurabad, P.C.; Palika, R.; Jyrwa, Y.W.; Bhaskarachary, K.; Pullakhandam, R. Dietary fat composition, food matrix and 420 relative polarity modulate the micellarization and intestinal uptake of carotenoids from vegetables and fruits. J. Food Sci. Technol. 2017, 54, 333–341. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kurilich, A.C.; Juvik, J.A. Quantification of carotenoid and tocopherol antioxidants in Zea mays. J. Agric. Food Chem. 1999, 47, 1948–1955. [Google Scholar] [CrossRef]
- Egesel, C.O.; Wong, J.C.; Lambert, R.J.; Rocheford, T.R. Combining ability of maize inbreds for carotenoids and tocopherols. Crop. Sci. 2003, 43, 818–823. [Google Scholar] [CrossRef]
- Ortiz-Covarrubias, Y.; Dhliwayo, T.; Palacios-Rojas, N.; Ndhlela, T.; Magorokosho, C.; Aguilar-Rincón, V.H.; Cruz-Morales, A.S.; Trachsel, S. Effects of drought and low nitrogen stress on provitamin A carotenoid content of biofortified maize hybrids. Crop. Sci. 2019, 59, 2521–2532. [Google Scholar] [CrossRef] [Green Version]
- Tiwari, A.; Prasanna, B.M.; Hossain, F.; Guruprasad, K.N. Analysis of genetic variability for kernel carotenoid concentration in selected maize inbred lines. Indian J. Genet. Plant Breed. 2012, 72, 1–6. [Google Scholar]
- Kumar, S.; Sangwan, S.; Yadav, R.; Langyan, S.; Singh, M. Maize carotenoid composition and biofortification for provitamin A activity. In Maize: Nutrition Dynamics and Novel Uses; Chaudhary, D.P., Kumar, S., Langyan, S., Eds.; Springer: New Delhi, India, 2014; pp. 83–91. [Google Scholar]
- Weber, E.J. Carotenoids and tocols of corn grain determined by HPLC. J. Am. Oil Chem. Soc. 1987, 64, 1129–1134. [Google Scholar] [CrossRef]
- Saenz, E.; Borrás, L.; Gerde, J.A. Carotenoid profiles in maize genotypes with contrasting kernel hardness. J. Cereal Sci. 2021, 99, 103206. [Google Scholar] [CrossRef]
- Kljak, K.; Grbeša, D. Carotenoid content and antioxidant activity of hexane extracts from selected Croatian corn hybrids. Food Chem. 2015, 167, 402–408. [Google Scholar] [CrossRef]
- Kandianis, C. Genetic dissection of carotenoid concentration and compositional traits in maize grain. Doctoral Dissertation, University of Illinois, Urbana, IL, USA, 2010. Available online: https://www.ideals.illinois.edu/handle/2142/14699 (accessed on 9 May 2021).
- Song, J.; Li, D.; He, M.; Chen, J.; Liu, C. Comparison of carotenoid composition in immature and mature grains of corn (Zea Mays L.) varieties. Int. J. Food Prop. 2016, 19, 351–358. [Google Scholar] [CrossRef]
- Menkir, A.; Gedil, M.; Tanumihardjo, S.; Adepoju, A.; Bossey, B. Carotenoid accumulation and agronomic performance of maize hybrids involving parental combinations from different marker-based groups. Food Chem. 2014, 148, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Menkir, A.; Liu, W.; White, W.S.; Maziya-Dixon, B.; Rocheford, T. Carotenoid diversity in tropical-adapted yellow maize inbred lines. Food Chem. 2008, 109, 521–529. [Google Scholar] [CrossRef]
- Menkir, A.; Maziya-Dixon, B.; Mengesha, W.; Rocheford, T.; Oladeji Alamu, E. Accruing genetic gain in pro-vitamin A enrichment from harnessing diverse maize germplasm. Euphytica 2017, 213, 105. [Google Scholar] [CrossRef]
- Wang, H.; Huang, Y.; Xiao, Q.; Huang, X.; Li, C.; Gao, X.; Wang, Q.; Xiang, X.; Zhu, Y.; Wang, J.; et al. Carotenoids modulate kernel texture in maize by influencing amyloplast envelope integrity. Nat. Commun. 2020, 11, 5346. [Google Scholar] [CrossRef]
- Rodriguez Concepcion, M.; Avalos, J.; Luisa Bonet, M.; Boronat, A.; Gomez-Gomez, L.; Hornero-Mendez, D.; Carmen Limon, M.; Meléndez-Martínez, A.J.; Olmedilla-Alonso, B.; Palou, A.; et al. A global perspective on carotenoids: Metabolism, biotechnology, and benefits for nutrition and health. Prog. Lipid Res. 2018, 70, 62–93. [Google Scholar] [CrossRef] [Green Version]
- Perera, C.O.; Mei Yen, G. Functional properties of carotenoids in human health. Int. J. Food Prop. 2007, 10, 201–230. [Google Scholar] [CrossRef]
- Tyssandier, V.; Lyan, B.; Borel, P. Main factors governing the transfer of carotenoids from emulsion lipid droplets to micelles. Biochim. Biophys. Acta 2001, 1533, 285–292. [Google Scholar] [CrossRef]
- Priyadarshani, A.M.B. A review on factors influencing bioaccessibility and bioefficacy of carotenoids. Crit. Rev. Food Sci. Nutr. 2017, 57, 1710–1717. [Google Scholar] [CrossRef] [PubMed]
- Dube, N.; Mashurabad, P.C.; Hossain, F.; Pullakhandam, R.; Thingnganing, L.; Kumar Bharatraj, D. β-Carotene bioaccessibility from biofortified maize (Zea mays) is related to its density and is negatively influenced by lutein and zeaxanthin. Food Funct. 2018, 9, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, D.B.; Chitchumroonchokchai, C.; Mariutti, L.R.B.; Mercadante, A.Z.; Failla, M. Comparison of two static in vitro digestion methods for screening the bioaccessibility of carotenoids in fruits, vegetables, and animal products. J. Agric. Food Chem. 2017, 65, 11220–11228. [Google Scholar] [CrossRef] [PubMed]


| Hybrid | Hybrid | Hybrid | Hybrid |
|---|---|---|---|
| Bc Institut Agram | KWS Kapitolis | Pioneer P0164 | PIO Os 522 |
| Bc Institut Alibi | KWS Kollegas | Pioneer P0200 | PIO Os 3850 |
| Bc Institut Bc 323 | KWS Kolumbaris | Pioneer P0216 | PIO Posavac 36 |
| Bc Institut Bc 344 | KWS Konfites | Pioneer P0217 | PIO Velimir |
| Bc Institut Bc 415 | KWS Kashmir | Pioneer P0412 | RWA ES Inventive |
| Bc Institut Bc 418 | KWS Orlando | Pioneer P0725 | RWA Ajowan |
| Bc Institut Bc 424 | KWS KxB 8386 | Pioneer P9241 | RWA Inclusiv |
| Bc Institut Bc 525 | KWS KxB 8453 | Pioneer P9300 | RWA Persic |
| Bc Institut Bc 572 | KWS Smaragd | Pioneer P9363 | RWA Gladiator |
| Bc Institut Instruktor | LG 30.3115 | Pioneer P9415 | RWA Glumanda |
| Bc Institut Kekec | LG 30.315 | Pioneer P9757 | RWA Ulyxxe |
| Bc Institut Majstor | LG 31.322 | Pioneer P9889 | RWA Hexagon |
| Bc Institut Pajdaš | LG 31.377 | Pioneer P9903 | RWA Tweetor |
| Bc Institut Tesla | LG 31.545 | Pioneer P9911 | RWA Urbanix |
| Bc Institut Thriler | LG 368/08 | Pioneer P9978 | Syngenta Sy Andromeda |
| DKC 4670 | LG Shannon | PIO 1 Tomasov | Syngenta Sy Atomic |
| DKC 4920 | MAS 34B | PIO Jablan | Syngenta Sy Bilbao |
| DKC 4943 | MAS 48L | PIO Kulak | Syngenta Sy Carioca |
| DKC 5031 | MAS 64P | PIO Os 3114 | Syngenta Sy Chorintos |
| DKC 5068 | NS seme 3022 | PIO Os 3150 | Syngenta Sy Kreon |
| DKC 5075 | NS seme 4015 | PIO Os 3450 | Syngenta Sy Lucius |
| DKC 5093 | NS seme 4051 | PIO Os 378 | Syngenta Sy Photon |
| DKC 5182 | NS seme 6102 | PIO Os 398 | Syngenta Sy Premeo |
| DKC 5685 | NS seme 6102 | PIO Os 4014 | Syngenta Sy Sandoro |
| DKC 5830 | NS seme Haris | PIO Os 4015 | Syngenta Sy Senko |
| KWS Balasco | P0023 | Os 403 | Syngenta Sy Zoan |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zurak, D.; Grbeša, D.; Duvnjak, M.; Kiš, G.; Međimurec, T.; Kljak, K. Carotenoid Content and Bioaccessibility in Commercial Maize Hybrids. Agriculture 2021, 11, 586. https://doi.org/10.3390/agriculture11070586
Zurak D, Grbeša D, Duvnjak M, Kiš G, Međimurec T, Kljak K. Carotenoid Content and Bioaccessibility in Commercial Maize Hybrids. Agriculture. 2021; 11(7):586. https://doi.org/10.3390/agriculture11070586
Chicago/Turabian StyleZurak, Dora, Darko Grbeša, Marija Duvnjak, Goran Kiš, Tatjana Međimurec, and Kristina Kljak. 2021. "Carotenoid Content and Bioaccessibility in Commercial Maize Hybrids" Agriculture 11, no. 7: 586. https://doi.org/10.3390/agriculture11070586
APA StyleZurak, D., Grbeša, D., Duvnjak, M., Kiš, G., Međimurec, T., & Kljak, K. (2021). Carotenoid Content and Bioaccessibility in Commercial Maize Hybrids. Agriculture, 11(7), 586. https://doi.org/10.3390/agriculture11070586










