Utilization of Shredded Steam-Exploded Pine Particles as a Dietary Ingredient to Modify Cecal Microbiota in Broilers
Abstract
:1. Introduction
2. Materials and Methods
2.1. Production of Shredded Steam-Exploded Pine Particles
2.2. Experimental Birds and Housing
2.3. Collection of Samples and Measurements
2.4. Extraction of DNA and Metagenome Analysis
2.5. Statistical Analysis
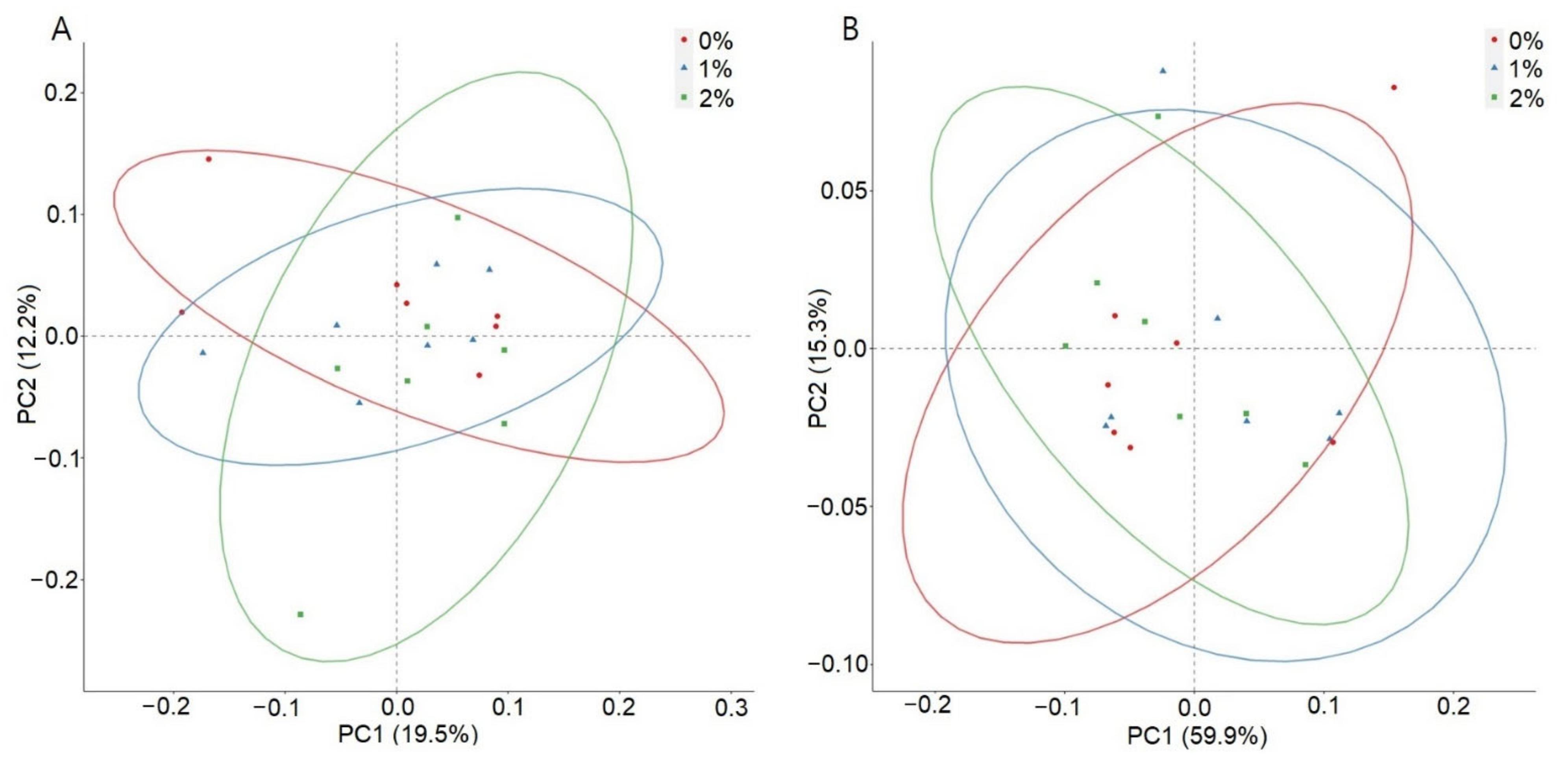
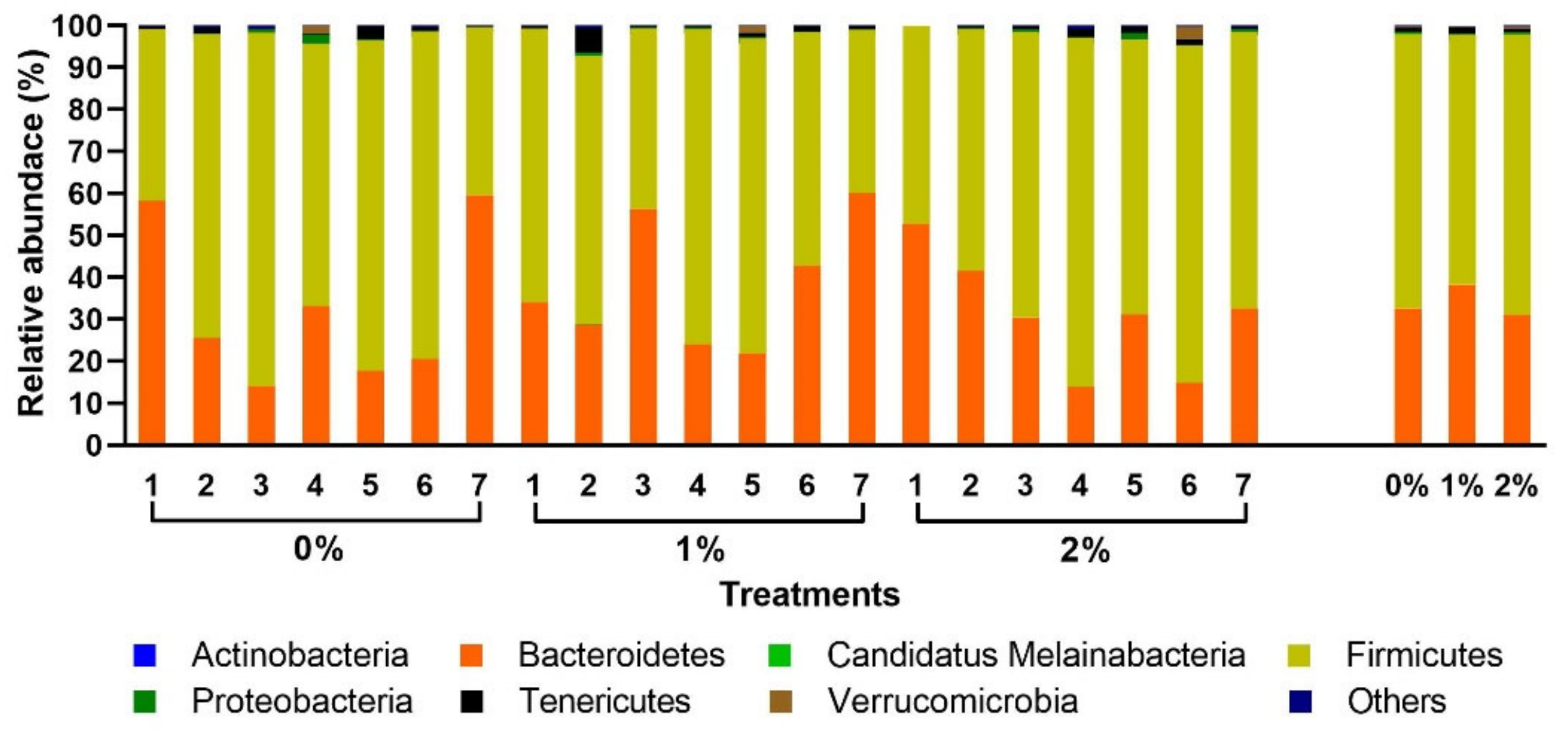
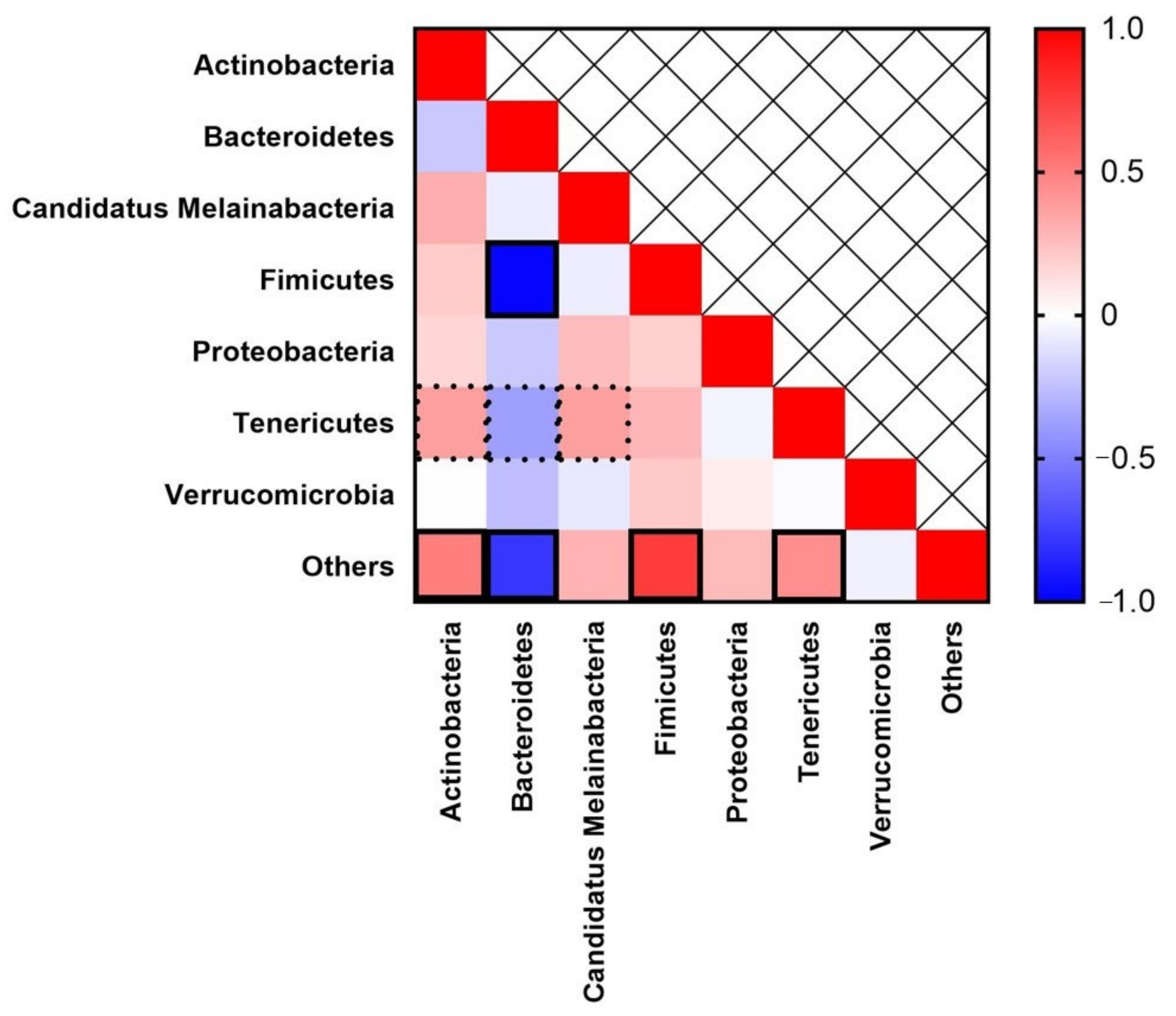
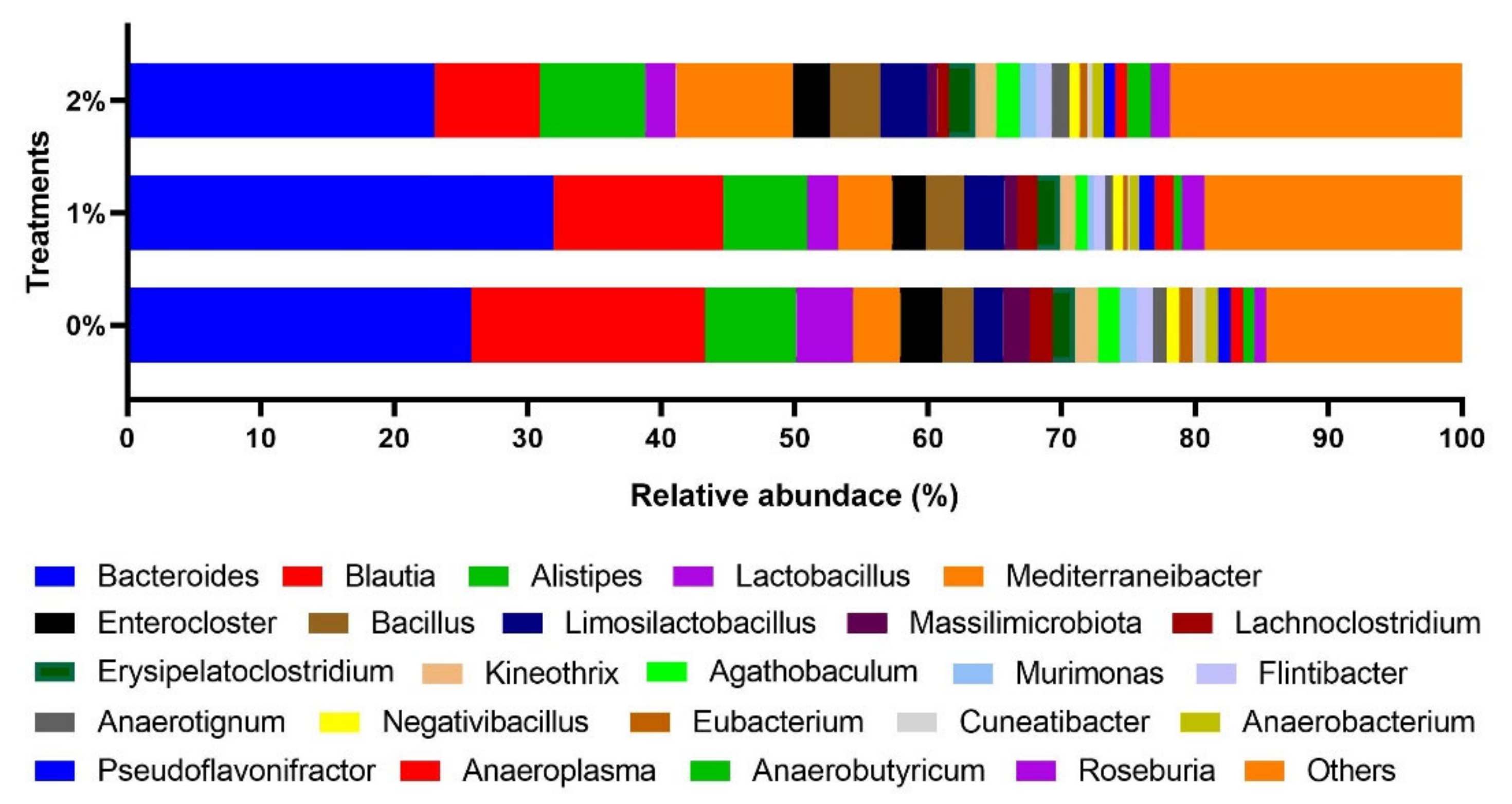
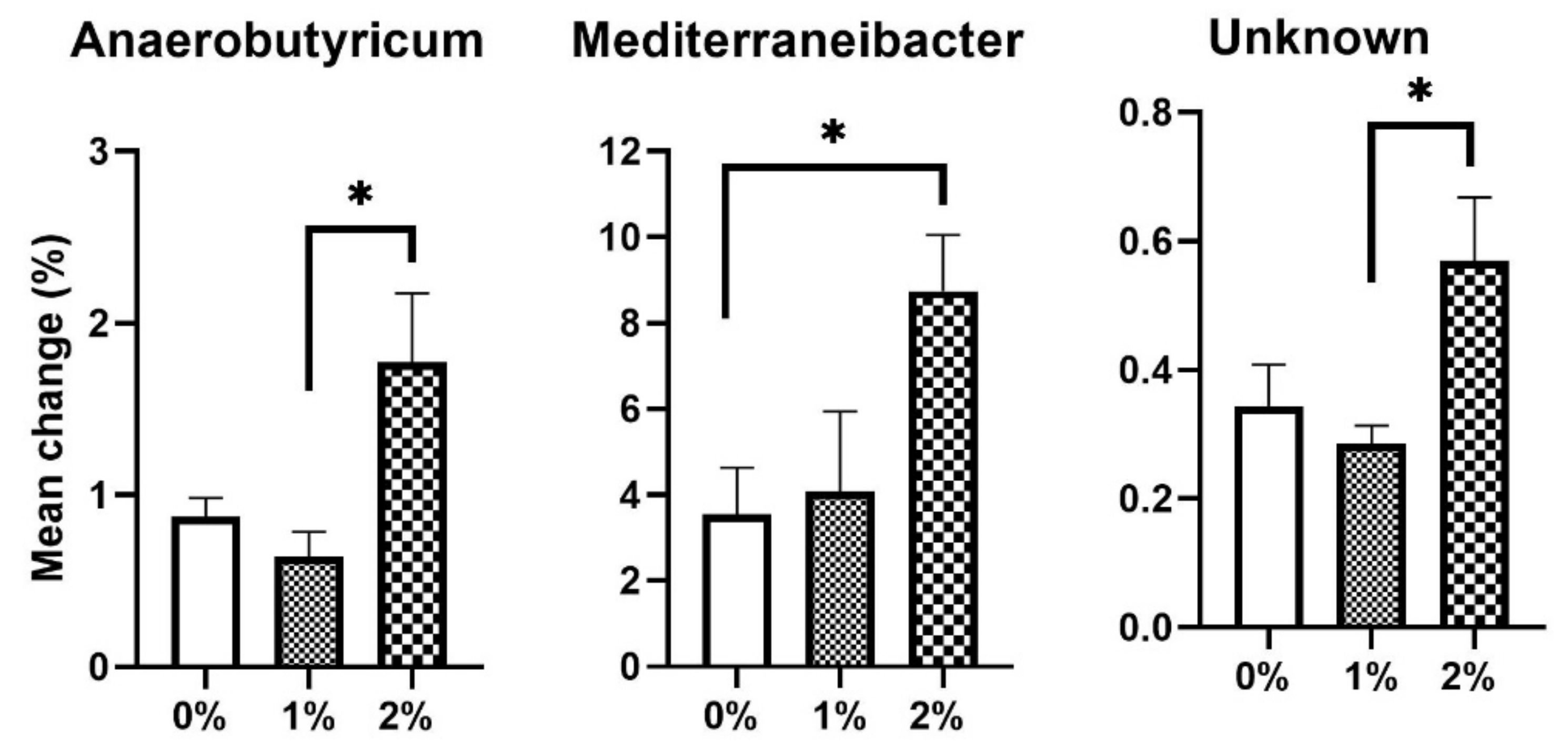
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tallentire, C.W.; Leinonen, I.; Kyriazakis, I. Breeding for efficiency in the broiler chicken: A review. Agron. Sustain. Dev. 2016, 36, 66. [Google Scholar] [CrossRef] [Green Version]
- Kamruzzaman, M.; Islam, S.; Rana, M.J. Financial and factor demand analysis of broiler production in Bangladesh. Heliyon 2021, 7, e07152. [Google Scholar] [CrossRef] [PubMed]
- Charis, G.; Danha, G.; Muzenda, E. A review of timber waste utilization: Challenges and opportunities in Zimbabwe. Procedia Manuf 2019, 35, 419–429. [Google Scholar] [CrossRef]
- Tejeda, O.J.; Kim, W.K. Role of Dietary Fiber in Poultry Nutrition. Animals 2021, 11, 461. [Google Scholar] [CrossRef] [PubMed]
- Röhe, I.; Zentek, J. Correction to: Lignocellulose as an insoluble fiber source in poultry nutrition: A review. J. Anim. Sci. Biotechnol. 2021, 12, 99. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.Y.; Heo, J.M.; Yang, J.-K. Effects of steam-exploded wood as an insoluble dietary fiber source on the performance characteristics of Broilers. BioResources 2019, 14, 1512–1524. [Google Scholar] [CrossRef]
- Li, J.; Henriksson, G.; Gellerstedt, G. Lignin depolymerization/repolymerization and its critical role for delignification of aspen wood by steam explosion. Bioresour. Technol. 2007, 98, 3061–3068. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.M.; Agblevor, F.A.; El-Zawawy, W.K. Isolation and characterization of cellulose and lignin from steam-exploded lignocellulosic biomass. BioResources 2010, 5, 397–418. [Google Scholar]
- Liu, Y.; Ren, X.; Wu, H.; Meng, Q.; Zhou, Z. Steam Explosion Treatment of Byproduct Feedstuffs for Potential Use as Ruminant Feed. Animals 2019, 9, 688. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patel, S.; Goyal, A. The current trends and future perspectives of prebiotics research: A review. 3 Biotech 2012, 2, 115–125. [Google Scholar] [CrossRef] [Green Version]
- Svihus, B.; Choct, M.; Classen, H.L. Function and nutritional roles of the avian caeca: A review. World’s Poult. Sci. J. 2013, 69, 249–264. [Google Scholar] [CrossRef]
- Saengkerdsub, S.; Ricke, S.C. Ecology and characteristics of methanogenic archaea in animals and humans. Crit. Rev. Microbiol. 2014, 40, 97–116. [Google Scholar] [CrossRef]
- Huang, Y.; Lv, H.; Song, Y.; Sun, C.; Zhang, Z.; Chen, S. Community composition of cecal microbiota in commercial yellow broilers with high and low feed efficiencies. Poult. Sci. 2021, 100, 100996. [Google Scholar] [CrossRef] [PubMed]
- Angelakis, E.; Raoult, D. The increase of Lactobacillus species in the gut flora of newborn broiler chicks and ducks is associated with weight gain. PLoS ONE 2010, 5, e10463. [Google Scholar] [CrossRef] [PubMed]
- Attia, Y.A.; Al-Hamid, A.E.A.; Ibrahim, M.S.; Al-Harthi, M.A.; Bovera, F.; Elnaggar, A.S. Productive performance, biochemical and hematological traits of broiler chickens supplemented with propolis, bee pollen, and mannan oligosaccharides continuously or intermittently. Livest. Sci. 2014, 164, 87–95. [Google Scholar] [CrossRef]
- Kang, S.; Kim, D.-H.; Lee, S.; Lee, T.; Lee, K.-W.; Chang, H.-H.; Moon, B.; Ayasan, T.; Choi, Y.-H. An Acute, Rather Than Progressive, Increase in Temperature-Humidity Index Has Severe Effects on Mortality in Laying Hens. Front. Vet. Sci. 2020, 7, 568093. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.H.; Kim, J.; Yoon, H.S.; Choi, Y.H. Effects of dietary corticosterone on yolk colors and eggshell quality in laying hens. Asian-Australas. J. Anim. Sci. 2015, 28, 840–846. [Google Scholar] [CrossRef] [Green Version]
- Chen, S.; Zhou, Y.; Chen, Y.; Gu, J. fastp: An ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 2018, 34, i884–i890. [Google Scholar] [CrossRef]
- Magoč, T.; Salzberg, S.L. FLASH: Fast length adjustment of short reads to improve genome assemblies. Bioinformatics 2011, 27, 2957–2963. [Google Scholar] [CrossRef]
- Li, W.; Fu, L.; Niu, B.; Wu, S.; Wooley, J. Ultrafast clustering algorithms for metagenomic sequence analysis. Brief. Bioinform. 2012, 13, 656–668. [Google Scholar] [CrossRef]
- Zhang, Z.; Schwartz, S.; Wagner, L.; Miller, W. A greedy algorithm for aligning DNA sequences. J. Comput. Biol. 2000, 7, 203–214. [Google Scholar] [CrossRef]
- Shang, Y.; Kumar, S.; Thippareddi, H.; Kim, W.K. Effect of Dietary Fructooligosaccharide (FOS) Supplementation on Ileal Microbiota in Broiler Chickens. Poult. Sci. 2018, 97, 3622–3634. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Tao, Y.; Lai, C.; Huang, C.; Zhou, Y.; Yong, Q. Effects of Mannanoligosaccharide Supplementation on the Growth Performance, Immunity, and Oxidative Status of Partridge Shank Chickens. Animals 2019, 9, 817. [Google Scholar] [CrossRef] [Green Version]
- Mookiah, S.; Sieo, C.C.; Ramasamy, K.; Abdullah, N.; Ho, Y.W. Effects of dietary prebiotics, probiotic and synbiotics on performance, caecal bacterial populations and caecal fermentation concentrations of broiler chickens. J. Sci. Food Agric. 2014, 94, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Farnell, Y.Z.; Peebles, E.D.; Kiess, A.S.; Wamsley, K.G.S.; Zhai, W. Effects of prebiotics, probiotics, and their combination on growth performance, small intestine morphology, and resident Lactobacillus of male broilers1. Poult. Sci. 2016, 95, 1332–1340. [Google Scholar] [CrossRef]
- Chen, T.C. Effect of adding chicory fructans in feed on broiler growth performance, serum cholesterol and intestinal length. Int. J. Poult. Sci. 2003, 2, 214–219. [Google Scholar] [CrossRef] [Green Version]
- Alshamy, Z.; Richardson, K.C.; Hünigen, H.; Hafez, H.M.; Plendl, J.; Al Masri, S. Comparison of the gastrointestinal tract of a dual-purpose to a broiler chicken line: A qualitative and quantitative macroscopic and microscopic study. PLoS ONE 2018, 13, e0204921. [Google Scholar] [CrossRef] [Green Version]
- Singh, J.; Suhag, M.; Dhaka, A. Augmented digestion of lignocellulose by steam explosion, acid and alkaline pretreatment methods: A review. Carbohydr. Polym. 2015, 117, 624–631. [Google Scholar] [CrossRef] [PubMed]
- Ballesteros, I.; Oliva, J.M.; Navarro, A.A.; González, A.; Carrasco, J.; Ballesteros, M. Effect of chip size on steam explosion pretreatment of softwood. Appl. Biochem. Biotechnol. 2000, 84–86, 97–110. [Google Scholar] [CrossRef]
- Kluth, H.; Rodehutscord, M. Effect of inclusion of cellulose in the diet on the inevitable endogenous amino acid losses in the ileum of broiler chicken. Poult. Sci. 2009, 88, 1199–1205. [Google Scholar] [CrossRef]
- Okrathok, S.; Khempaka, S. Modified-dietary fiber from cassava pulp reduces abdominal fat and meat cholesterol contents without affecting growth performance of broiler chickens. J. Appl. Poult. Res. 2020, 29, 229–239. [Google Scholar] [CrossRef]
- Zheng, M.; Mao, P.; Tian, X.; Guo, Q.; Meng, L. Effects of dietary supplementation of alfalfa meal on growth performance, carcass characteristics, meat and egg quality, and intestinal microbiota in Beijing-you chicken. Poult. Sci. 2019, 98, 2250–2259. [Google Scholar] [CrossRef] [PubMed]
- Zheng, M.; Mao, P.; Tian, X.; Meng, L. Growth performance, carcass characteristics, meat and egg quality, and intestinal microbiota in Beijing-you chicken on diets with inclusion of fresh chicory forage. Ital. J. Anim. Sci. 2019, 18, 1310–1320. [Google Scholar] [CrossRef] [Green Version]
- Vlaicu, P.A.; Untea, A.E.; Panaite, T.D.; Turcu, R.P. Effect of dietary orange and grapefruit peel on growth performance, health status, meat quality and intestinal microflora of broiler chickens. Ital. J. Anim. Sci. 2020, 19, 1394–1405. [Google Scholar] [CrossRef]
- Díaz Carrasco, J.M.; Redondo, E.A.; Pin Viso, N.D.; Redondo, L.M.; Farber, M.D.; Fernández Miyakawa, M.E. Tannins and Bacitracin Differentially Modulate Gut Microbiota of Broiler Chickens. BioMed Res. Int. 2018, 2018, 1879168. [Google Scholar] [CrossRef]
- Hou, L.; Sun, B.; Yang, Y. Effects of Added Dietary Fiber and Rearing System on the Gut Microbial Diversity and Gut Health of Chickens. Animals 2020, 10, 107. [Google Scholar] [CrossRef] [Green Version]
- Amit-Romach, E.; Sklan, D.; Uni, Z. Microflora ecology of the chicken intestine using 16S ribosomal DNA primers. Poult. Sci. 2004, 83, 1093–1098. [Google Scholar] [CrossRef]
- Mohd Shaufi, M.A.; Sieo, C.C.; Chong, C.W.; Gan, H.M.; Ho, Y.W. Deciphering chicken gut microbial dynamics based on high-throughput 16S rRNA metagenomics analyses. Gut Pathog. 2015, 7, 4. [Google Scholar] [CrossRef] [Green Version]
- Xiao, Y.; Xiang, Y.; Zhou, W.; Chen, J.; Li, K.; Yang, H. Microbial community mapping in intestinal tract of broiler chicken. Poult. Sci. 2017, 96, 1387–1393. [Google Scholar] [CrossRef]
- Dunkley, K.D.; Dunkley, C.S.; Njongmeta, N.L.; Callaway, T.R.; Hume, M.E.; Kubena, L.F.; Nisbet, D.J.; Ricke, S.C. Comparison of in vitro fermentation and molecular microbial profiles of high-fiber feed substrates incubated with chicken cecal inocula. Poult. Sci. 2007, 86, 801–810. [Google Scholar] [CrossRef]
- Chakraborti, C.K. New-found link between microbiota and obesity. World J. Gastrointest. Pathophysiol. 2015, 6, 110–119. [Google Scholar] [CrossRef] [PubMed]
- Shi, D.; Bai, L.; Qu, Q.; Zhou, S.; Yang, M.; Guo, S.; Li, Q.; Liu, C. Impact of gut microbiota structure in heat-stressed broilers. Poult. Sci. 2019, 98, 2405–2413. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Finegold, S.M.; Song, Y.; Lawson, P.A. Reclassification of Clostridium coccoides, Ruminococcus hansenii, Ruminococcus hydrogenotrophicus, Ruminococcus luti, Ruminococcus productus and Ruminococcus schinkii as Blautia coccoides gen. nov., comb. nov., Blautia hansenii comb. nov., Blautia hydrogenotrophica comb. nov., Blautia luti comb. nov., Blautia producta comb. nov., Blautia schinkii comb. nov. and description of Blautia wexlerae sp. nov., isolated from human faeces. Int. J. Syst. Evol. Microbiol. 2008, 58, 1896–1902. [Google Scholar]
- Biasato, I.; Ferrocino, I.; Dabbou, S.; Evangelista, R.; Gai, F.; Gasco, L.; Cocolin, L.; Capucchio, M.T.; Schiavone, A. Black soldier fly and gut health in broiler chickens: Insights into the relationship between cecal microbiota and intestinal mucin composition. J. Anim. Sci. Biotechnol. 2020, 11, 11. [Google Scholar] [CrossRef] [Green Version]
- Togo, A.H.; Diop, A.; Bittar, F.; Maraninchi, M.; Valero, R.; Armstrong, N.; Dubourg, G.; Labas, N.; Richez, M.; Delerce, J.; et al. Correction to: Description of Mediterraneibacter massiliensis, gen. nov., sp. nov., a new genus isolated from the gut microbiota of an obese patient and reclassification of Ruminococcus faecis, Ruminococcus lactaris, Ruminococcus torques, Ruminococcus gnavus and Clostridium glycyrrhizinilyticum as Mediterraneibacter faecis comb. nov., Mediterraneibacter lactaris comb. nov., Mediterraneibacter torques comb. nov., Mediterraneibacter gnavus comb. nov. and Mediterraneibacter glycyrrhizinilyticus comb. nov. Antonie Van Leeuwenhoek 2018, 111, 2129–2130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shetty, S.A.; Zuffa, S.; Bui, T.P.N.; Aalvink, S.; Smidt, H.; De Vos, W.M. Reclassification of Eubacterium hallii as Anaerobutyricum hallii gen. nov., comb. nov., and description of Anaerobutyricum soehngenii sp. nov., a butyrate and propionate-producing bacterium from infant faeces. Int. J. Syst. Evol. Microbiol. 2018, 68, 3741–3746. [Google Scholar] [CrossRef]
- Robert, C.; Bernalier-Donadille, A. The cellulolytic microflora of the human colon: Evidence of microcrystalline cellulose-degrading bacteria in methane-excreting subjects. FEMS Microbiol. Ecol. 2003, 46, 81–89. [Google Scholar] [CrossRef]
- Montgomery, L. Isolation of human colonic fibrolytic bacteria. Lett. Appl. Microbiol. 1988, 6, 55–57. [Google Scholar] [CrossRef]
- Flint, H.J.; Scott, K.P.; Duncan, S.H.; Louis, P.; Forano, E. Microbial degradation of complex carbohydrates in the gut. Gut Microbes 2012, 3, 289–306. [Google Scholar] [CrossRef] [Green Version]
| Items | Grower | Finisher | ||||
|---|---|---|---|---|---|---|
| 0% | 1% | 2% | 0% | 1% | 2% | |
| Ingredients (%) | ||||||
| Yellow corn, ground | 57.1 | 55.2 | 53.2 | 63.3 | 61.2 | 59.1 |
| Soybean meal | 33.3 | 33.5 | 33.8 | 28.7 | 29.0 | 29.3 |
| Corn gluten meal | 3.0 | 3.0 | 3.0 | 2.5 | 2.5 | 2.5 |
| Limestone | 1.6 | 1.6 | 1.5 | 1.5 | 1.5 | 1.5 |
| Tallow | 3.6 | 4.4 | 5.1 | 3.3 | 4.0 | 4.8 |
| Mono-dicalcium phosphate | 0.42 | 0.43 | 0.44 | 0.05 | 0.05 | 0.05 |
| Salt | 0.3 | 0.3 | 0.3 | 0.3 | 0.3 | 0.3 |
| Choline chloride (50%) | 0.07 | 0.07 | 0.07 | 0.03 | 0.03 | 0.03 |
| DL-methionine hydroxy analogue, 88% | 0.15 | 0.15 | 0.15 | 0.10 | 0.10 | 0.10 |
| L-Lysine | 0.17 | 0.16 | 0.15 | 0.04 | 0.05 | 0.05 |
| SPP | 0.0 | 1.0 | 2.0 | 0.0 | 1.0 | 2.0 |
| Vitamin mixture 1 | 0.2 | 0.2 | 0.2 | 0.2 | 0.2 | 0.2 |
| Mineral mixture 2 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 |
| Calculated values | ||||||
| TMEn, kcal/kg | 3103 | 3102 | 3102 | 3150 | 3150 | 3150 |
| Crude protein, % | 21.03 | 21.02 | 21.03 | 19.02 | 19.03 | 19.02 |
| Calcium, % | 0.91 | 0.92 | 0.91 | 0.82 | 0.81 | 0.82 |
| Available Phosphorus, % | 0.42 | 0.42 | 0.43 | 0.36 | 0.36 | 0.36 |
| Lysine, % | 1.15 | 1.15 | 1.15 | 1.01 | 1.01 | 1.01 |
| Methionine + Cystine, % | 0.84 | 0.83 | 0.83 | 0.71 | 0.72 | 0.71 |
| Analyzed values (%) | ||||||
| Moisture | 10.98 | 11.19 | 10.91 | 9.87 | 9.92 | 10.33 |
| Crude protein | 20.59 | 22.03 | 20.94 | 19.69 | 18.81 | 19.03 |
| Ether extract | 6.97 | 8.07 | 7.69 | 6.92 | 7.46 | 7.49 |
| Crude fiber | 2.70 | 3.12 | 4.11 | 2.77 | 3.43 | 4.13 |
| Ash | 4.32 | 4.34 | 4.31 | 4.80 | 3.87 | 3.72 |
| Parameters | 0% | 1% | 2% | pSEM | p-Values |
|---|---|---|---|---|---|
| Body weight (g) at day 8 | 202.9 | 202.8 | 202.9 | 0.05 | 0.747 |
| Body weight (g) at day 28 | 1458.4 | 1467.2 | 1502.7 | 19.29 | 0.624 |
| Day 8–14 | |||||
| ADG (g) | 38.9 | 38.1 | 39.9 | 0.70 | 0.601 |
| ADFI (g) | 45.3 | 43.8 | 46.0 | 0.76 | 0.505 |
| FCR (kg feed/kg body weight) | 1.15 | 1.17 | 1.16 | 0.01 | 0.811 |
| Day 15–21 | |||||
| ADG (g) | 64.4 | 64.4 | 66.5 | 1.24 | 0.736 |
| ADFI (g) | 92.2 | 89.9 | 93.4 | 1.61 | 0.682 |
| FCR (kg feed/kg body weight) | 1.41 | 1.42 | 1.41 | 0.01 | 0.967 |
| Day 22–28 | |||||
| ADG (g) | 76.1 | 77.7 | 79.3 | 1.19 | 0.545 |
| ADFI (g) | 128.9 | 127.4 | 130.7 | 2.00 | 0.810 |
| FCR (kg feed/kg body weight) | 1.67 | 1.66 | 1.65 | 0.01 | 0.690 |
| Parameters | 0% | 1% | 2% | pSEM | p-Values |
|---|---|---|---|---|---|
| Duodenum (%) | 0.46 | 0.44 | 0.45 | 0.017 | 0.871 |
| Jejunum (%) | 1.04 | 0.97 | 1.01 | 0.026 | 0.584 |
| Ileum (%) | 0.95 | 0.87 | 0.84 | 0.029 | 0.270 |
| Liver (%) | 2.62 | 2.68 | 2.78 | 0.085 | 0.769 |
| Spleen (%) | 0.11 | 0.10 | 0.10 | 0.004 | 0.629 |
| Parameters | 0% | 1% | 2% | pSEM | p-Values |
|---|---|---|---|---|---|
| Duodenum (cm) | 27.1 | 27.4 | 26.4 | 0.46 | 0.668 |
| Jejunum (cm) | 57.6 | 58.3 | 57.0 | 0.84 | 0.845 |
| Ileum (cm) | 55.9 | 56.3 | 53.3 | 0.95 | 0.389 |
| Cecum (cm) | 15.4 | 15.6 | 15.4 | 0.32 | 0.950 |
| Parameters | 0% | 1% | 2% | pSEM | p-Value |
|---|---|---|---|---|---|
| Glucose (mg/dL) | 260 | 2485 | 235 | 6.6 | 0.310 |
| Total Protein (g/dL) | 3.28 | 3.00 | 2.95 | 0.1 | 0.502 |
| Triglyceride (mg/dL) | 25.8 | 22.9 | 16.9 | 2.4 | 0.305 |
| Cholesterol (mg/dL) | 122 | 130 | 129 | 3.1 | 0.572 |
| Parameters | 0% | 1% | 2% | pSEM | p-Value |
|---|---|---|---|---|---|
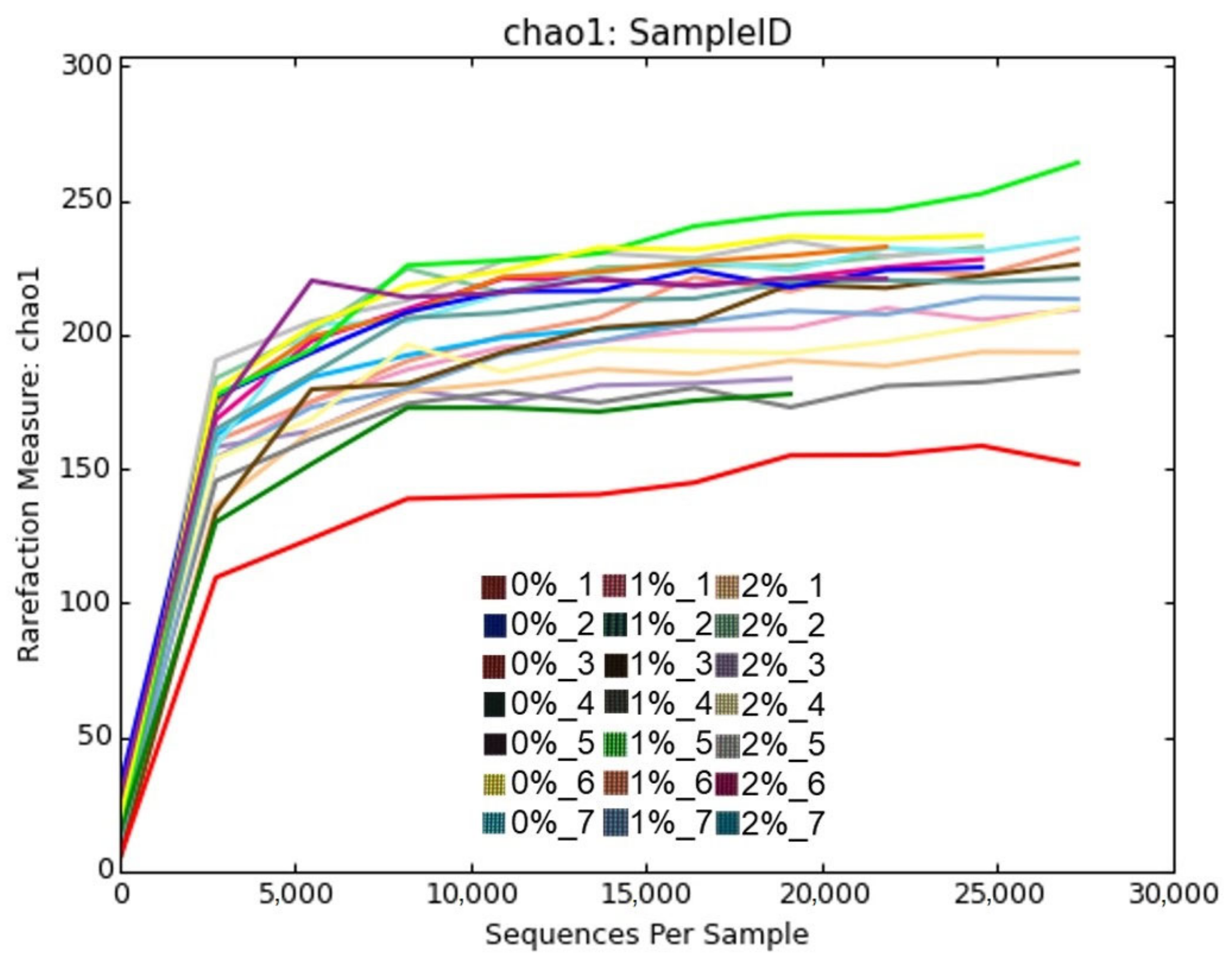
| OTUs | 199 | 203 | 202 | 4.6 | 0.988 |
| Chao1 | 218 | 222 | 220 | 5.6 | 0.953 |
| Shannon | 4.2 | 4.0 | 4.6 | 0.2 | 0.438 |
| Inverse Simpson | 0.82 | 0.823 | 0.869 | 0.02 | 0.610 |
| Goods coverage | 0.999 | 0.999 | 0.999 | 0 | 1.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goel, A.; Kim, B.-J.; Ncho, C.-M.; Jeong, C.-M.; Gupta, V.; Jung, J.-Y.; Ha, S.-Y.; Lee, D.-H.; Yang, J.-K.; Choi, Y.-H. Utilization of Shredded Steam-Exploded Pine Particles as a Dietary Ingredient to Modify Cecal Microbiota in Broilers. Agriculture 2021, 11, 1196. https://doi.org/10.3390/agriculture11121196
Goel A, Kim B-J, Ncho C-M, Jeong C-M, Gupta V, Jung J-Y, Ha S-Y, Lee D-H, Yang J-K, Choi Y-H. Utilization of Shredded Steam-Exploded Pine Particles as a Dietary Ingredient to Modify Cecal Microbiota in Broilers. Agriculture. 2021; 11(12):1196. https://doi.org/10.3390/agriculture11121196
Chicago/Turabian StyleGoel, Akshat, Beom-June Kim, Chris-Major Ncho, Chae-Mi Jeong, Vaishali Gupta, Ji-Young Jung, Si-Young Ha, Dong-Hwan Lee, Jae-Kyung Yang, and Yang-Ho Choi. 2021. "Utilization of Shredded Steam-Exploded Pine Particles as a Dietary Ingredient to Modify Cecal Microbiota in Broilers" Agriculture 11, no. 12: 1196. https://doi.org/10.3390/agriculture11121196
APA StyleGoel, A., Kim, B.-J., Ncho, C.-M., Jeong, C.-M., Gupta, V., Jung, J.-Y., Ha, S.-Y., Lee, D.-H., Yang, J.-K., & Choi, Y.-H. (2021). Utilization of Shredded Steam-Exploded Pine Particles as a Dietary Ingredient to Modify Cecal Microbiota in Broilers. Agriculture, 11(12), 1196. https://doi.org/10.3390/agriculture11121196







