Yield Component Responses of the Brachiaria brizantha Forage Grass to Soil Water Availability in the Brazilian Cerrado
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design and General Soil Properties
2.2. Cultivars Sowing and Plant Establishment
2.3. Response Variables
2.4. Statistical Analysis
3. Results and Discussion
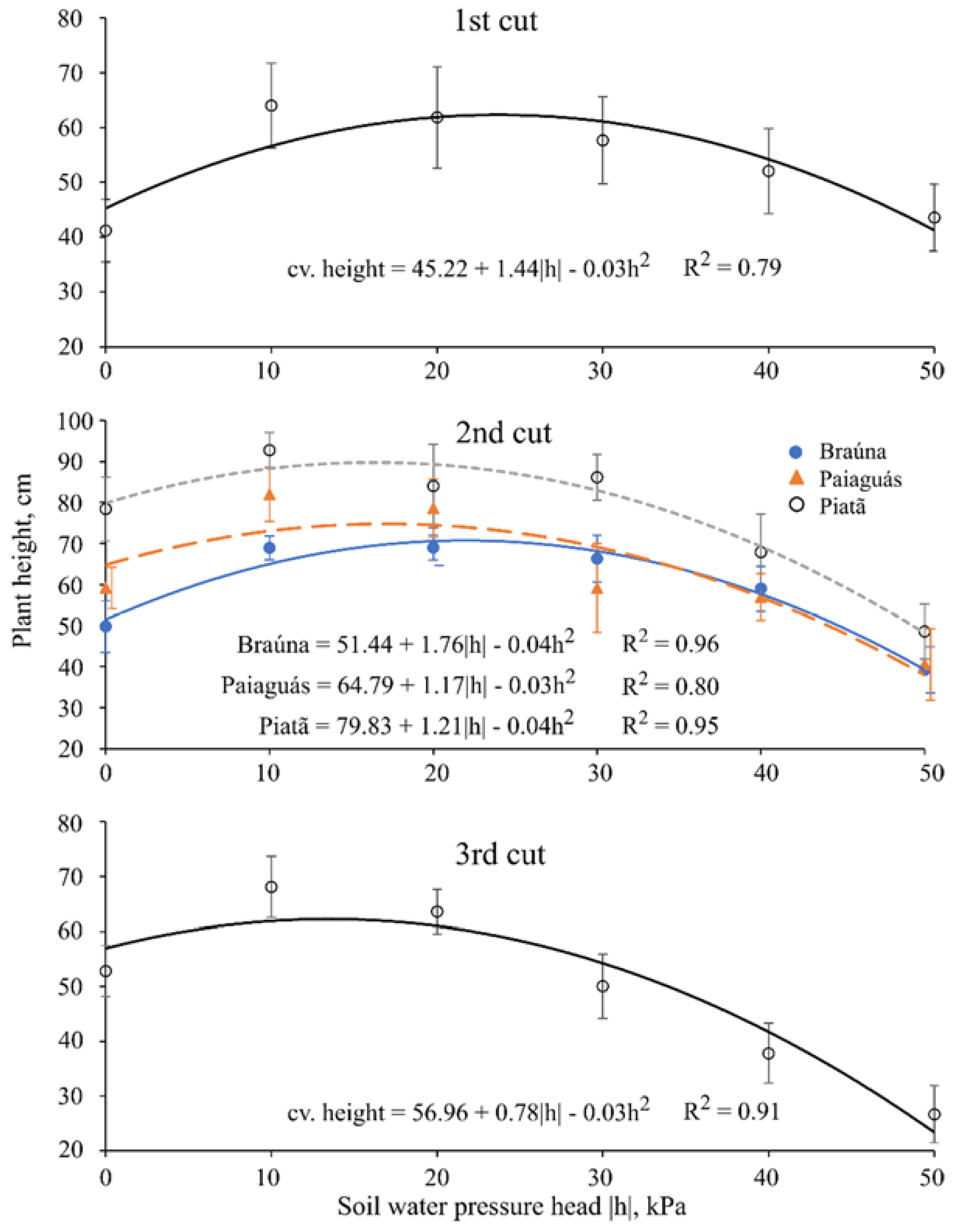
3.1. Plant Height
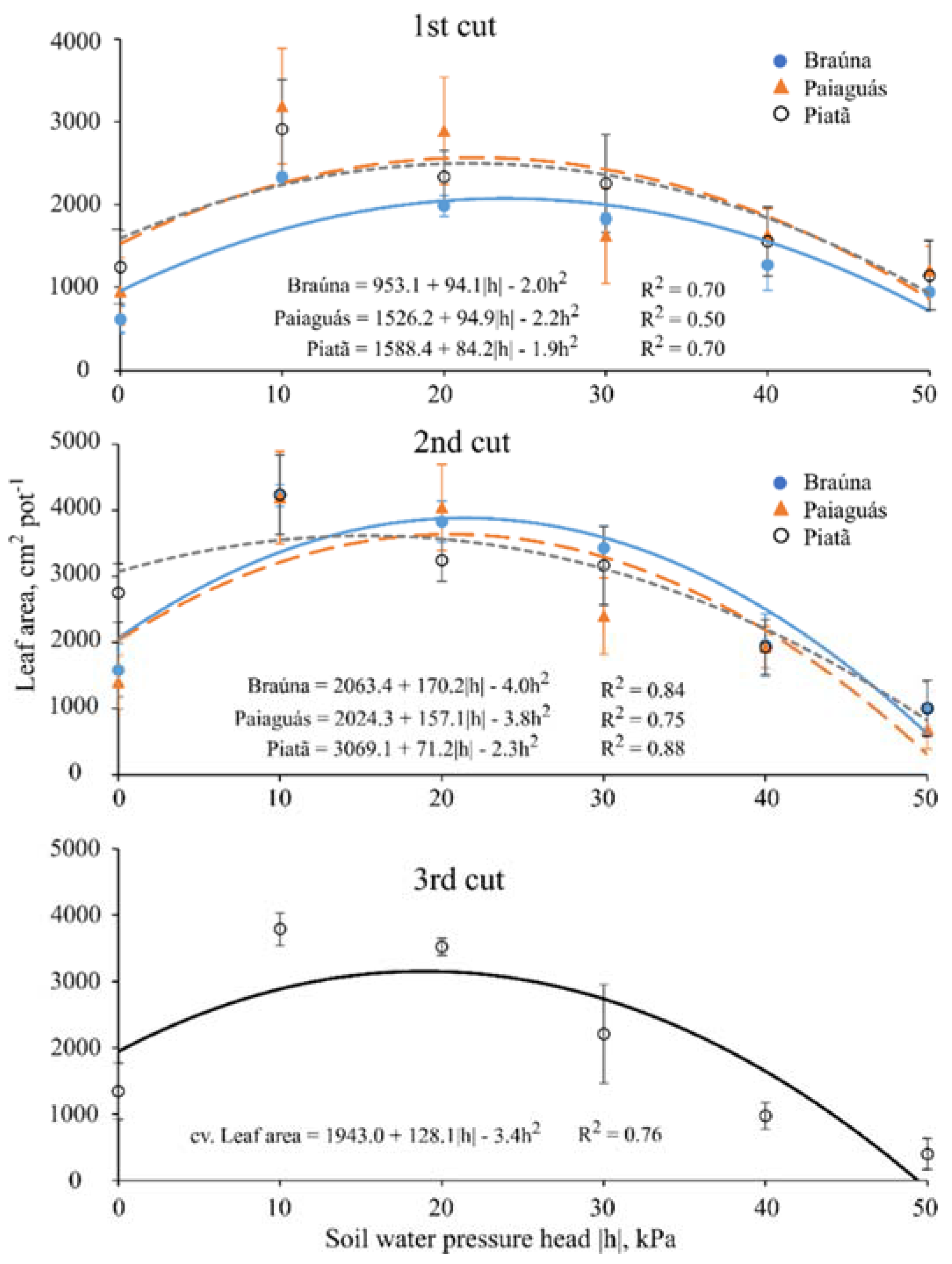
3.2. Leaf Area
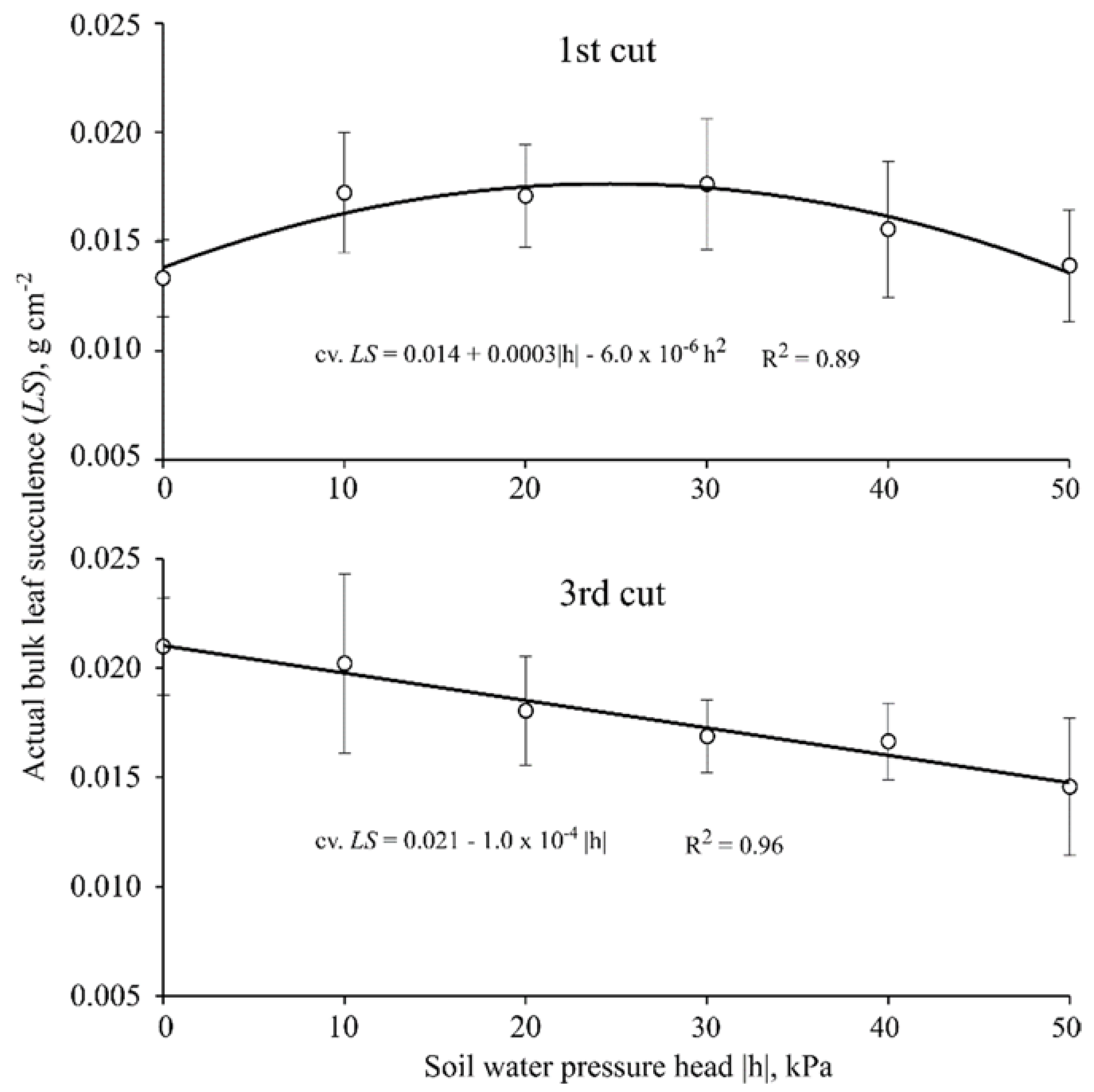
3.3. Bulk Leaf Succulence
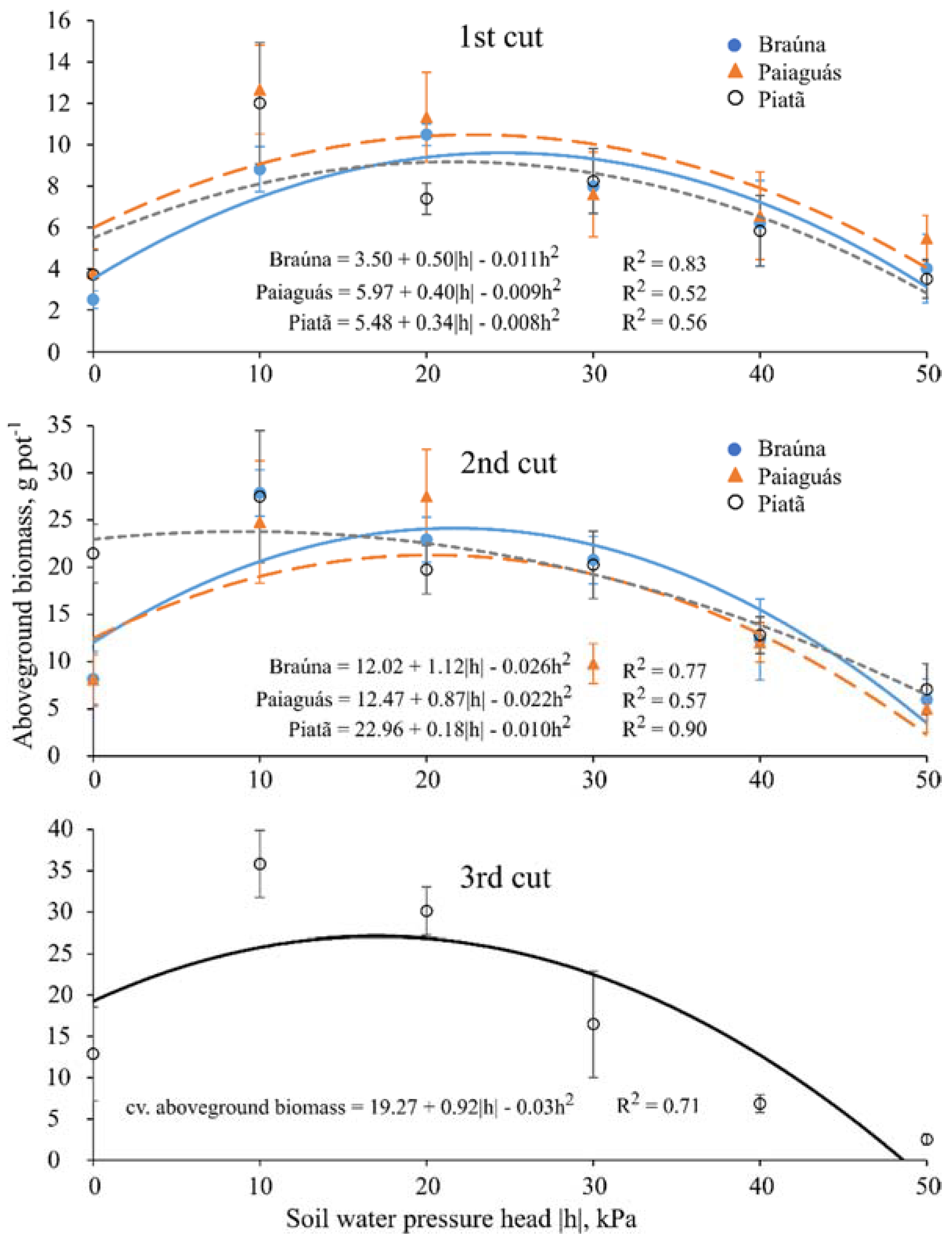
3.4. Aboveground Dry Biomass
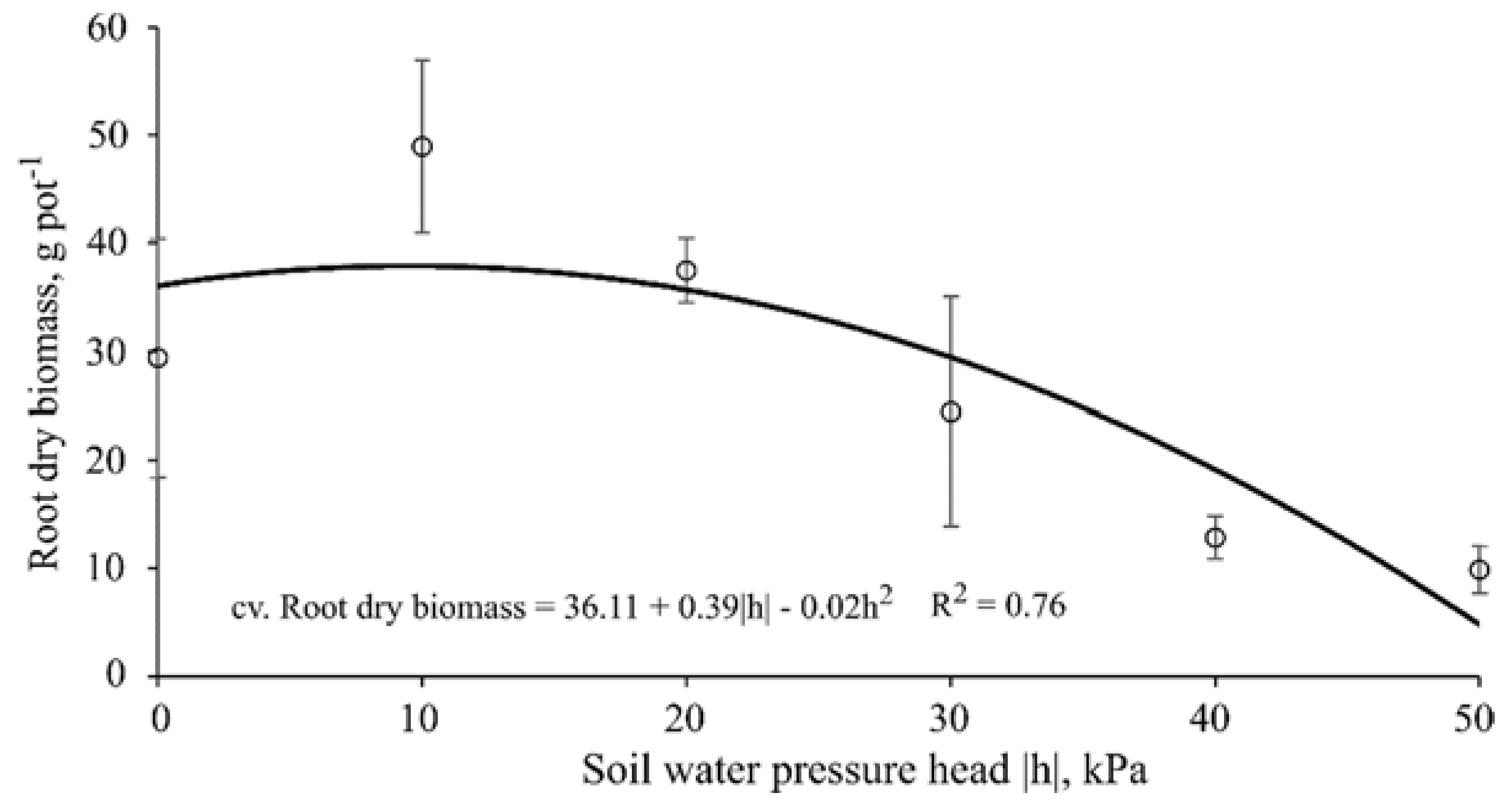
3.5. Root Dry Biomass
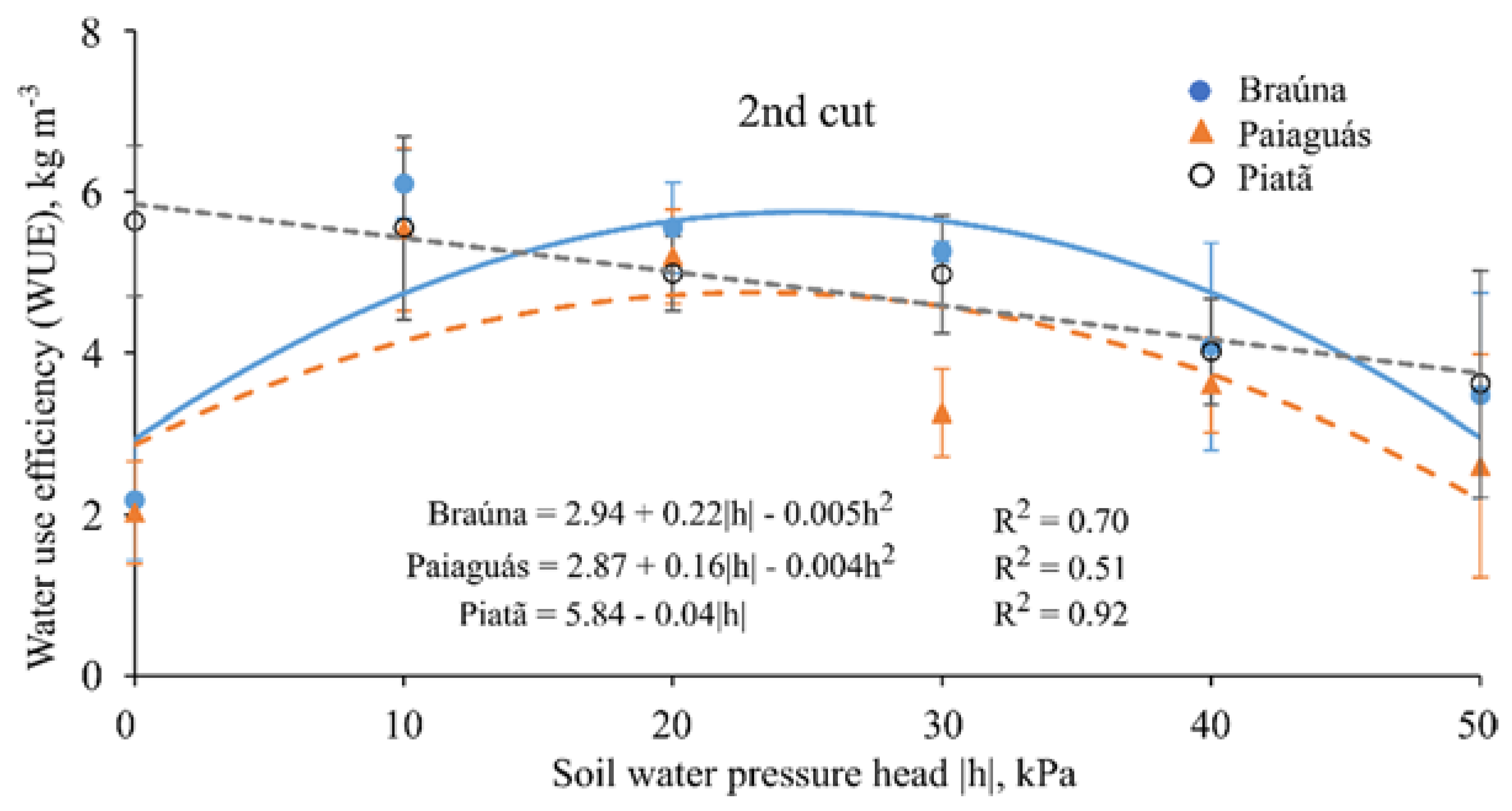
3.6. Water Use Efficiency
4. Conclusions
- The low drought-resistant trait observed for all evaluated Brachiaria brizantha cultivars, grown on Fluvisol, indicates the need for releasing of better-adapted cultivars to cope with scenarios of reduced soil water availability typical of drought-prone regions.
- Regarding the phenotypic traits response, the cultivar Piatã, followed closely by the cultivar BRS Paiaguás, showed superior results to most of the evaluated scenarios.
- The evaluated Brachiaria brizantha cultivars grown in a coarse-textured soil achieved higher performance at soil water pressure heads (h) within the range −15 kPa and −25 kPa; however, given the high dependence of soil water availability on soil hydraulic properties, other soil types should be tested in experiments under natural field conditions.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bowman, M.S.; Soares-Filho, B.S.; Merry, F.D.; Nepstad, D.C.; Rodrigues, H.; Almeida, O.T. Persistence of cattle ranching in the Brazilian Amazon: A spatial analysis of the rationale for beef production. Land Use Pol. 2012, 29, 558–568. [Google Scholar] [CrossRef]
- Jank, L.; Barrios, S.C.; Valle, C.B.; Simeão, R.M.; Alves, G.F. The value of improved pastures to Brazilian beef production. Crop. Pest. Sci. 2014, 65, 1132–1137. [Google Scholar] [CrossRef]
- Barreto, A.G.O.P.; Berndes, G.; Sparovek, G.; Wirsenius, S. Agricultural intensification in Brazil and its effects on land-use patterns: An analysis of the 1975–2006 period. Global Change Biol. 2013, 19, 1804–1815. [Google Scholar] [CrossRef]
- Baruch, Z. Responses to drought and flooding in tropical forage grasses I. Biomass allocation, leaf growth and mineral nutrients. Plant. Soil 1994, 164, 87–96. [Google Scholar] [CrossRef]
- Guenni, O.; Marín, D.; Baruch, Z. Response to drought of five Brachiaria species. I. Biomass production, leaf growth, root distribution, water use and forage quality. Plant. Soil 2002, 243, 229–241. [Google Scholar] [CrossRef]
- Meirelles, M.L.; Franco, A.C.; Farias, S.E.M.; Bracho, R. Evapotranspiration and plant—Atmospheric coupling in a Brachiaria brizantha pasture in the Brazilian savannah region. Grass For. Sci. 2011, 66, 206–213. [Google Scholar] [CrossRef]
- Foley, J.J.A.; Ramankutty, N.; Brauman, K.A.; Cassidy, E.S.; Gerber, J.S.; Johnston, M.; Mueller, N.D.; O’Connell, C.; Ray, D.K.; West, P.C.; et al. Solutions for a cultivated planet. Nature 2011, 478, 337–342. [Google Scholar] [CrossRef]
- MAPA/AGE. Projections of Agribusiness—Brazil 2009/10 to 2019/20 Ministry of Agriculture, Livestock and Food Supply—Strategic Management Advisory Board; MAPA/AGE: Brasília, Brazil, 2010; p. 48. [Google Scholar]
- ANUALPEC. ‘Anuário da Pecuária Brasileira’ (Yearbook of Brazilian Livestock); Instituto FNP, AGRA FNP Pesquisas Ltda: São Paulo, Brazil, 2015. [Google Scholar]
- Guenni, O.; Baruch, Z.; Marín, D. Response to drought of five Brachiaria species. II. Water relations and leaf gas exchange. Plant. Soil 2004, 258, 249–260. [Google Scholar] [CrossRef]
- Cezário, A.S.; Ribeiro, K.G.; Santos, S.A.; Valadares Filho, S.C.; Pereira, O.G. Silages of Brachiaria brizantha cv. Marandu harvested at two regrowth ages: Microbial inoculant responses in silage fermentation, ruminant digestion and beef cattle performance. Anim. Feed Sci. Technol. 2015, 208, 33–43. [Google Scholar] [CrossRef]
- Orrico Junior, M.A.P.; da Silveira, A.P.; Orrico, A.C.A.; Schwingel, A.W.; Carnavali, P.L.; Alves, D.C. Use of Organic Compost for the Fertilization of Piatã and Paiaguás Grasses: Effects of Dose on Morphogenetic, Structural, Nutritional, and Productive Characteristics. Compost Sci. Utiliz. 2018, 26, 201–208. [Google Scholar] [CrossRef]
- Santos, A.J.B.; Quesada, C.A.; da Silva, G.T.; Maia, J.F.; Miranda, H.S.; Miranda, A.C.; Lloyd, J. High rates of net ecosystem carbon assimilation by Brachiaria pasture in the Brazilian Cerrado. Glob. Chang. Biol. 2004, 10, 877–885. [Google Scholar] [CrossRef]
- Lascano, C.E. Managing the grazing resource for animal production in savannas of tropical America. Trop. Grassl. 1991, 25, 66–72. [Google Scholar]
- Pizarro, E.A.; do Valle, C.B.; Keller-Grein, G.; Schultze-Kraft, R.; Zimmer, A.H. Experiencia regional con Brachiaria: Región de América Tropical—Sabanas. In Brachiaria: Biología, Agronomía y Mejoramiento; Miles, J.W., Maass, B.L., do Valle, C.B., Eds.; CIAT: Cali, Colombia; EMBRAPA/CNPGC: Campo Grande, Brazil, 1998; pp. 247–269. [Google Scholar]
- Alvares, C.A.; Stape, J.L.; Sentelhas, P.C.; Gonçalves, J.L.M.; Sparovek, G. Köppen’s climate classification map for Brazil. Meteorol. Z. 2014, 22, 711–728. [Google Scholar] [CrossRef]
- IUSS Working Group WRB. World Reference Base for Soil Resources 2014, Update 2015. International Soil Classification System for Naming Soils and Creating Legends for Soil Maps; World Soil Resources Reports 106; FAO: Rome, Italy, 2015. [Google Scholar]
- Van Genuchten, M.T. A closed-form equation for predicting the hydraulic conductivity of unsaturated soils. Soil Sci. Soc. Am. J. 1980, 44, 892–897. [Google Scholar] [CrossRef]
- Van Genuchten, M.T.; Leij, F.J.; Yates, S.R. The RETC Code for Quantifying the Hydraulic Functions of Unsaturated Soils, Version 1.0; EPA Report 600/2-91/065; U.S. Salinity Laboratory, USDA, ARS: Riverside, CA, USA, 1991.
- Geerts, S.; Raes, D. Deficit irrigation as an on-farm strategy to maximize crop water productivity in dry areas. Agric. Water Manag. 2009, 96, 1275–1284. [Google Scholar] [CrossRef]
- Bastiaanssen, W.G.M.; Steduto, P. The water productivity score (WPS) at global and regional level: Methodology and first results from remote sensing measurements of wheat, rice and maize. Sci. Total Environ. 2017, 575, 595–611. [Google Scholar] [CrossRef]
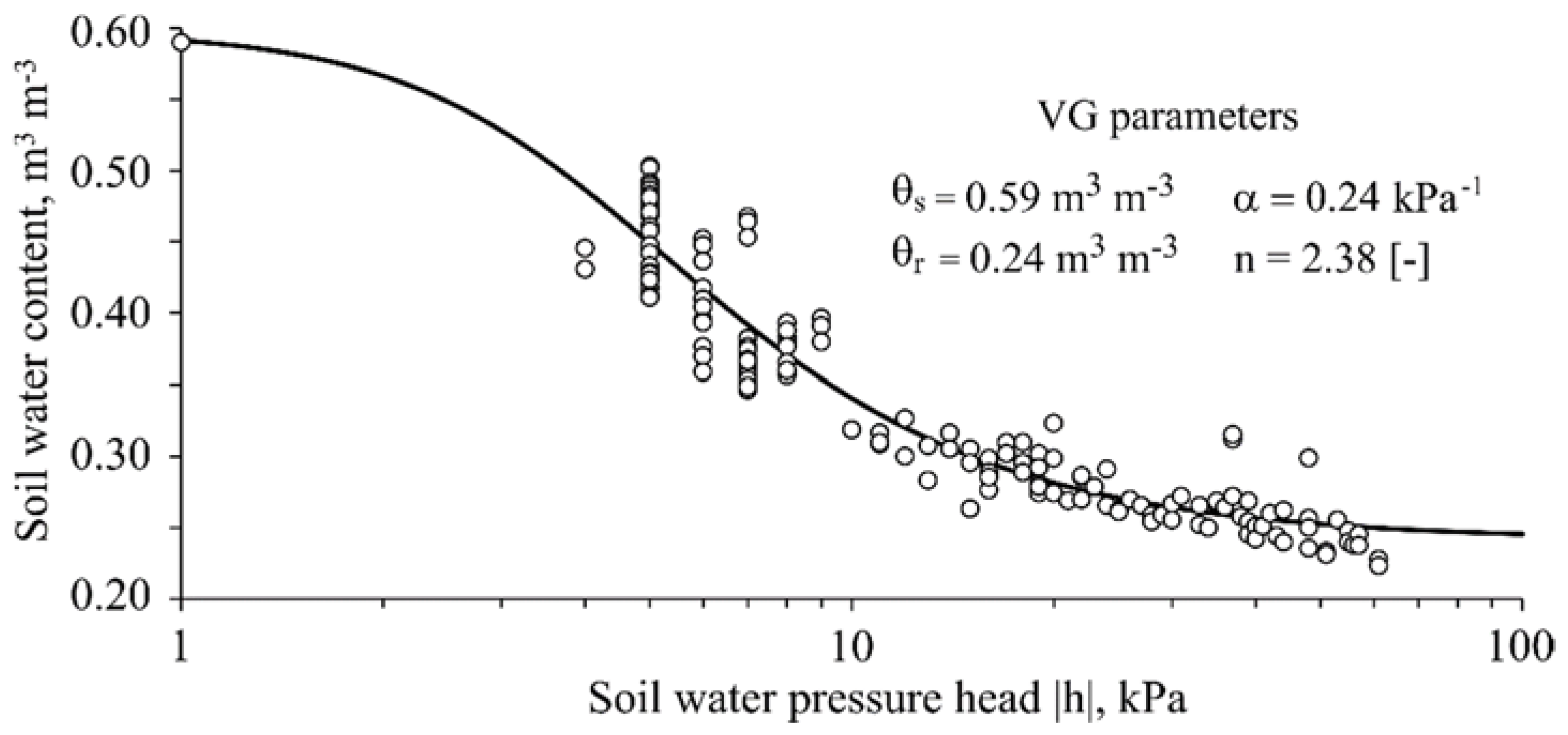
- De Jong van Lier, Q.; Pinheiro, E.A.R.; Inforsato, L. A One-Dimensional Physically Based Approach to Predict Soil Profile Aeration Requirements. Soil Sci. Soc. Am. J. 2018, 82, 593–600. [Google Scholar] [CrossRef]
- Bailey-Serres, J.; Voesenek, L.A.C.J. Flooding stress: Acclimations and genetic diversity. Annu. Rev. Plant. Biol. 2008, 59, 313–339. [Google Scholar] [CrossRef]
- Skirycz, A.; Inzé, D. More from less: Plant growth under limited water. Curr. Op. Biotechnol. 2010, 21, 197–203. [Google Scholar] [CrossRef]
- Geng, Y.; Wu, R.; Wee, C.W.; Xie, F.; Wei, X.; Chan, P.M.Y.; Tham, C.; Duan, L.; Dinneny, J.R. A spatio-temporal understanding of growth regulation during the salt stress response in Arabidopsis. Plant. Cell 2013, 25, 2132–2154. [Google Scholar] [CrossRef]
- Feng, W.; Lindner, H.; Robbins, N.E., II; Dinneny, J.R. Growing out of stress: The role of cell- and organ-scale growth control in plant water-stress responses. Plant. Cell 2016, 28, 1769–1782. [Google Scholar] [CrossRef]
- Metselaar, K.; Pinheiro, E.A.R.; de Jong van Lier, Q. Mathematical Description of Rooting Profiles of Agricultural Crops and its Effect on Transpiration Prediction by a Hydrological Model. Soil Syst. 2019, 3, 44. [Google Scholar] [CrossRef]
- De Jong van Lier, Q.; Metselaar, K.; Van Dam, J.C. Root Water Extraction and Limiting Soil Hydraulic Conditions Estimated by Numerical Simulation. Vadose Zone J. 2006, 5, 1264–1277. [Google Scholar] [CrossRef]
- Pinheiro, E.A.R.; de Jong van Lier, Q.; Metselaar, K. A Matric Flux Potential Approach to Assess Plant Water Availability in Two Climate Zones in Brazil. Vadose Zone J. 2018, 17, 160083. [Google Scholar] [CrossRef]
- Pinheiro, E.A.R.; de Jong van Lier, Q.; Inforsato, L.; Šimůnek, J. Measuring full-range soil hydraulic properties for the prediction of crop water availability using gamma-ray attenuation and inverse modeling. Agric. Water Manag. 2019, 216, 294–305. [Google Scholar] [CrossRef]
- Snyman, H.A. Evapotranspiration, water-use efficiency and quality of six dryland planted pasture species and natural vegetation, in a semi-arid rangeland. Afr. J. Range For. Sci. 1994, 11, 82–88. [Google Scholar] [CrossRef]
- White, T.A.; Snow, V.O. A modelling analysis to identify plant traits for enhanced water-use efficiency of pasture. Crop Past. Sci. 2012, 63, 63–76. [Google Scholar] [CrossRef]
- Moot, D.J.; Brown, H.E.; Pollock, K.; Mills, A. Yield and water use of temperate pastures in summer dry environments. Proc. N. Z. Grass. Assoc. 2008, 70, 51–57. [Google Scholar]
- Molden, D.; Oweis, T.; Steduto, P.; Bindraban, P.; Hanjra, M.A.; Kijne, J. Improving agricultural water productivity: Between optimism and caution. Agric. Water Manag. 2010, 97, 528–535. [Google Scholar] [CrossRef]
- Bodner, G.; Nakhforoosh, A.; Kaul, H.P. Management of crop water under drought: A review. Agron. Sustain. Dev. 2015, 35, 401–442. [Google Scholar] [CrossRef]


| pH CaCl2 | P | K | S | Ca | Mg | Al | H + Al | C.E.C. | O.M. | V | m |
|---|---|---|---|---|---|---|---|---|---|---|---|
| mg·dm−3 | cmolc·dm−3 | g·kg−1 | % | ||||||||
| 5.3 | 20.4 | 47 | 2 | 4.3 | 0.8 | 0.0 | 2.7 | 7.9 | 21.0 | 65.9 | 0.0 |
| Zn | Mn | Cu | Fe | B | Clay | Silt | Sand | ||||
| mg·dm−3 | g·kg−1 | ||||||||||
| 10.5 | 68.0 | 0.8 | 93 | 0.13 | 145 | 150 | 705 | ||||
| Plant Height 1 (cm) | ||||||
|---|---|---|---|---|---|---|
| Cultivar | First Cut (40 day after emergence (DAE)) | |||||
| Braúna | 46.1 c ± 8.1 | |||||
| Paiaguás | 53.0 b ± 10.7 | |||||
| Piatã | 61.0 a ± 10.1 | |||||
| Cultivar | Second cut (70 DAE) | |||||
| Soil water pressure head (kPa) | ||||||
| 0 | −10 | −20 | −30 | −40 | −50 | |
| Braúna | 49.8 b ± 6.3 | 68.9 b ± 2.9 | 69.0 b ± 3.0 | 66.3 b ± 5.7 | 59.0 a ± 5.5 | 39.2 a ± 5.6 |
| Paiaguás | 59.2 b ± 5.0 | 82.0 a ± 6.6 | 78.6 ab ± 7.0 | 59.2 b ± 10.8 | 56.9 a ± 5.7 | 40.6 a ± 8.7 |
| Piatã | 78.4 a ± 7.8 | 92.8 a ± 4.3 | 84.0 a ± 10.0 | 86.2 a ± 5.6 | 67.9 a ± 9.2 | 48.6 a ± 6.7 |
| Cultivar | Third cut (100 DAE) | |||||
| Braúna | 46.9 c ± 14.0 | |||||
| Paiaguás | 48.1 b ± 16.2 | |||||
| Piatã | 54.6 a ± 16.8 | |||||
| Leaf Area 1 (cm2·pot−1) | ||||||
|---|---|---|---|---|---|---|
| Cultivar | First cut (40 DAE) | |||||
| Soil water pressure head—h (kPa) | ||||||
| 0 | −10 | −20 | −30 | −40 | −50 | |
| Braúna | 609 b ± 157 | 2329 b ± 58 | 1982 b ± 125 | 1826 a ± 65 | 1267 a ± 308 | 939 a ± 29 |
| Paiaguás | 948 ab ± 160 | 3190 a ± 220 | 2892 a ± 204 | 1620 a ± 390 | 1632 a ± 295 | 1020 a ± 150 |
| Piatã | 1241 a ± 121 | 2910 a ± 362 | 2336 b ± 281 | 2254 b ± 269 | 1553 a ± 250 | 1138 a ± 222 |
| Cultivar | Second cut (70 DAE) | |||||
| Soil water pressure head—h (kPa) | ||||||
| 0 | −10 | −20 | −30 | −40 | −50 | |
| Braúna | 1581 b ± 404 | 4222 a ± 165 | 3826 a ± 313 | 3427 a ± 339 | 1960 a ± 474 | 999 a ± 398 |
| Paiaguás | 1403 b ± 405 | 4194 a ± 700 | 4042 a ± 650 | 1632 b ± 575 | 1930 a ± 316 | 690 a ± 299 |
| Piatã | 2750 a ± 442 | 4235 a ± 600 | 3241 a ± 318 | 3162 a ± 593 | 1930 a ± 417 | 1005 a ± 423 |
| Cultivar | Third cut (100 DAE) | |||||
| Braúna | 2150 a ± 1412 | |||||
| Paiaguás | 1752 a ± 1476 | |||||
| Piatã | 2211 a ± 1344 | |||||
| Bulk Leaf Succulence 1 (g·cm−2) | |
|---|---|
| Cultivar | First cut (40 DAE) |
| Braúna | 0.013 b ± 0.001 |
| Paiaguás | 0.017 a ± 0.003 |
| Piatã | 0.017 a ± 0.002 |
| Cultivar | Third cut (100 DAE) |
| Braúna | 0.015 b ± 0.002 |
| Paiaguás | 0.017 ab ± 0.001 |
| Piatã | 0.020 a ± 0.003 |
| Aboveground Dry Biomass 1 (g) | ||||||
|---|---|---|---|---|---|---|
| Cultivar | First cut (40 DAE) | |||||
| Soil water pressure head—h (kPa) | ||||||
| 0 | −10 | −20 | −30 | −40 | −50 | |
| Braúna | 2.5 a ± 0.4 | 8.8 b ± 1.1 | 1.5 b ± 0.5 | 8.0 a ± 1.3 | 6.2 a ± 2.1 | 4.0 b ± 1.7 |
| Paiaguás | 3.8 a ± 1.1 | 12.7 a ± 2.1 | 7.4 a ± 2.1 | 8.2 a ± 2.1 | 5.8 b ± 2.1 | 4.5 a ± 1.1 |
| Piatã | 3.7 a ± 1.2 | 12.0 a ± 0.8 | 7.4 a ± 0.8 | 8.2 a ± 1.6 | 5.8 b ± 1.7 | 3.5 b ± 0.9 |
| Cultivar | Second cut (70 DAE) | |||||
| Soil water pressure head—h (kPa) | ||||||
| 0 | −10 | −20 | −30 | −40 | −50 | |
| Braúna | 8.2 b ± 2.9 | 27.9 a ± 2.4 | 22.9 ab ± 2.4 | 20.8 a ± 2.5 | 12.3 a ± 4.3 | 6.0 a ± 2.2 |
| Paiaguás | 8.1 b ± 2.6 | 24.8 a ± 6.4 | 27.5 a ± 5.0 | 9.8 b ± 2.1 | 12.1 a ± 2.1 | 5.0 a ± 2.5 |
| Piatã | 21.5 a ± 3.1 | 27.5 a ± 7.0 | 19.8 b ± 2.6 | 20.3 a ± 3.5 | 12.8 a ± 1.9 | 7.7 a ± 2.7 |
| Cultivar | Third cut (100 DAE) | |||||
| Braúna | 18.4 ab ± 14.1 | |||||
| Paiaguás | 15.0 b ± 13.7 | |||||
| Piatã | 18.9 a ± 12.4 | |||||
| Water Use Efficiency 1 (kg·m−3) | ||||||
|---|---|---|---|---|---|---|
| Cultivars | Second cut (70 DAE) | |||||
| Soil water pressure head—h (kPa) | ||||||
| 0 | 10 | 20 | 30 | 40 | 50 | |
| Braúna | 2.19 b ± 0.7 | 6.10 a ± 0.4 | 5.55 a ± 0.6 | 5.25 a ± 0.1 | 4.08 a ± 1.2 | 3.47 a ± 1.3 |
| Paiaguás | 2.03 b ± 0.6 | 5.50 b ± 1.0 | 5.19 a ± 0.6 | 3.26 b ± 0.5 | 3.61 a ± 0.6 | 2.70 a ± 1.4 |
| Piatã | 5.63 a ± 0.9 | 5.55 a ± 1.1 | 5.00 a ± 0.5 | 4.98 a ± 0.7 | 4.02 a ± 0.7 | 3.62 a ± 1.4 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thaiana Rueda da Silva, C.; Bonfim-Silva, E.M.; de Araújo da Silva, T.J.; Alves Rodrigues Pinheiro, E.; Vieira José, J.; Pereira Freire Ferraz, A. Yield Component Responses of the Brachiaria brizantha Forage Grass to Soil Water Availability in the Brazilian Cerrado. Agriculture 2020, 10, 13. https://doi.org/10.3390/agriculture10010013
Thaiana Rueda da Silva C, Bonfim-Silva EM, de Araújo da Silva TJ, Alves Rodrigues Pinheiro E, Vieira José J, Pereira Freire Ferraz A. Yield Component Responses of the Brachiaria brizantha Forage Grass to Soil Water Availability in the Brazilian Cerrado. Agriculture. 2020; 10(1):13. https://doi.org/10.3390/agriculture10010013
Chicago/Turabian StyleThaiana Rueda da Silva, Camila, Edna Maria Bonfim-Silva, Tonny José de Araújo da Silva, Everton Alves Rodrigues Pinheiro, Jefferson Vieira José, and André Pereira Freire Ferraz. 2020. "Yield Component Responses of the Brachiaria brizantha Forage Grass to Soil Water Availability in the Brazilian Cerrado" Agriculture 10, no. 1: 13. https://doi.org/10.3390/agriculture10010013
APA StyleThaiana Rueda da Silva, C., Bonfim-Silva, E. M., de Araújo da Silva, T. J., Alves Rodrigues Pinheiro, E., Vieira José, J., & Pereira Freire Ferraz, A. (2020). Yield Component Responses of the Brachiaria brizantha Forage Grass to Soil Water Availability in the Brazilian Cerrado. Agriculture, 10(1), 13. https://doi.org/10.3390/agriculture10010013





