Use of Antimalarial Agents Is Associated with Favourable Physical Functioning in Patients with Systemic Lupus Erythematosus
Abstract
1. Introduction
2. Experimental Section
2.1. Study Design and Population
2.2. Evaluation of HRQoL
2.3. Evaluation of Disease Activity and Organ Damage
2.4. Patient Subgroups based on Organ-Specific Activity
2.5. Statistical Analysis
3. Results
3.1. Patient Characteristics
3.2. MOS SF-36
3.3. FACIT-Fatigue
3.4. EQ-5D
3.5. Associations with SF-36 Physical Functioning
3.6. Stratification into Subgroups Based on Organ-Specific Activity
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kaul, A.; Gordon, C.; Crow, M.K.; Touma, Z.; Urowitz, M.B.; van Vollenhoven, R.; Ruiz-Irastorza, G.; Hughes, G. Systemic lupus erythematosus. Nat. Rev. Dis. Prim. 2016, 2, 16039. [Google Scholar] [CrossRef]
- Waldheim, E.; Elkan, A.-C.; Pettersson, S.; Vollenhoven, R.V.; Bergman, S.; Frostegård, J.; Henriksson, E.W. Health-related quality of life, fatigue and mood in patients with SLE and high levels of pain compared to controls and patients with low levels of pain. Lupus 2013, 22, 1118–1127. [Google Scholar] [CrossRef] [PubMed]
- Jolly, M.; Toloza, S.; Goker, B.; Clarke, A.E.; Navarra, S.V.; Wallace, D.; Weisman, M.; Mok, C.C. Disease-specific quality of life in patients with lupus nephritis. Lupus 2018, 27, 257–264. [Google Scholar] [CrossRef]
- Bjork, M.; Dahlstrom, O.; Wettero, J.; Sjowall, C. Quality of life and acquired organ damage are intimately related to activity limitations in patients with systemic lupus erythematosus. BMC Musculoskelet. Disord. 2015, 16, 188. [Google Scholar] [CrossRef] [PubMed]
- Van Vollenhoven, R.; Voskuyl, A.; Bertsias, G.; Aranow, C.; Aringer, M.; Arnaud, L.; Askanase, A.; Balazova, P.; Bonfa, E.; Bootsma, H.; et al. A framework for remission in SLE: Consensus findings from a large international task force on definitions of remission in SLE (DORIS). Ann. Rheum. Dis. 2017, 76, 554–561. [Google Scholar] [CrossRef]
- Durcan, L.; O’Dwyer, T.; Petri, M. Management strategies and future directions for systemic lupus erythematosus in adults. Lancet 2019, 393, 2332–2343. [Google Scholar] [CrossRef]
- Fanouriakis, A.; Kostopoulou, M.; Alunno, A.; Aringer, M.; Bajema, I.; Boletis, J.N.; Cervera, R.; Doria, A.; Gordon, C.; Govoni, M.; et al. 2019 update of the EULAR recommendations for the management of systemic lupus erythematosus. Ann. Rheum. Dis. 2019, 78, 736–745. [Google Scholar] [CrossRef]
- Ruiz-Irastorza, G.; Egurbide, M.-V.; Pijoan, J.-I.; Garmendia, M.; Villar, I.; Martinez-Berriotxoa, A.; Erdozain, J.-G.; Aguirre, C. Effect of antimalarials on thrombosis and survival in patients with systemic lupus erythematosus. Lupus 2006, 15, 577–583. [Google Scholar] [CrossRef]
- Petri, M. Hydroxychloroquine use in the Baltimore Lupus Cohort: Effects on lipids, glucose and thrombosis. Lupus 1996, 5, 16–22. [Google Scholar] [CrossRef]
- Alarcón, G.S.; McGwin, G.; Bertoli, A.M.; Fessler, B.J.; Calvo-Alén, J.; Bastian, H.M.; Vilá, L.M.; Reveille, J.D. Effect of hydroxychloroquine on the survival of patients with systemic lupus erythematosus: Data from LUMINA, a multiethnic US cohort (LUMINA L). Ann. Rheum. Dis. 2007, 66, 1168–1172. [Google Scholar] [CrossRef]
- Annapureddy, N.; Devilliers, H.; Jolly, M. Patient-reported outcomes in lupus clinical trials with biologics. Lupus 2016, 25, 1111–1121. [Google Scholar] [CrossRef] [PubMed]
- Parodis, I.; Lopez Benavides, A.H.; Zickert, A.; Pettersson, S.; Moller, S.; Welin Henriksson, E.; Voss, A.; Gunnarsson, I. The Impact of Belimumab and Rituximab on Health-Related Quality of Life in Patients With Systemic Lupus Erythematosus. Arthritis Care Res. 2019, 71, 811–821. [Google Scholar] [CrossRef] [PubMed]
- Elera-Fitzcarrald, C.; Alva, M.; Gamboa-Cardenas, R.; Mora-Trujillo, C.S.; Zevallos, F.; García-Poma, A.; Medina, M.; Rodriguez-Bellido, Z.; Perich-Campos, R.A.; Pastor-Asurza, C.A.; et al. Factors associated with health-related quality of life in Peruvian patients with systemic lupus erythematosus. Lupus 2018, 27, 913–919. [Google Scholar] [CrossRef] [PubMed]
- Jolly, M.; Galicier, L.; Aumaître, O.; Francès, C.; Le Guern, V.; Lioté, F.; Smail, A.; Limal, N.; Perard, L.; Desmurs-Clavel, H.; et al. Quality of life in systemic lupus erythematosus: Description in a cohort of French patients and association with blood hydroxychloroquine levels. Lupus 2016, 25, 735–740. [Google Scholar] [CrossRef]
- Navarra, S.V.; Guzmán, R.M.; Gallacher, A.E.; Hall, S.; Levy, R.A.; Jimenez, R.E.; Li, E.K.M.; Thomas, M.; Kim, H.-Y.; León, M.G.; et al. Efficacy and safety of belimumab in patients with active systemic lupus erythematosus: A randomised, placebo-controlled, phase 3 trial. Lancet 2011, 377, 721–731. [Google Scholar] [CrossRef]
- Furie, R.; Petri, M.; Zamani, O.; Cervera, R.; Wallace, D.J.; Tegzova, D.; Sanchez-Guerrero, J.; Schwarting, A.; Merrill, J.T.; Chatham, W.W.; et al. A phase III, randomized, placebo-controlled study of belimumab, a monoclonal antibody that inhibits B lymphocyte stimulator, in patients with systemic lupus erythematosus. Arthritis Rheumat. 2011, 63, 3918–3930. [Google Scholar] [CrossRef]
- Hochberg, M.C. Updating the American college of rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheumat. 1997, 40, 1725. [Google Scholar] [CrossRef]
- Petri, M.; Kim, M.Y.; Kalunian, K.C.; Grossman, J.; Hahn, B.H.; Sammaritano, L.R.; Lockshin, M.; Merrill, J.T.; Belmont, H.M.; Askanase, A.D.; et al. Combined Oral Contraceptives in Women with Systemic Lupus Erythematosus. N. Engl. J. Med. 2005, 353, 2550–2558. [Google Scholar] [CrossRef]
- Ware, J.; Sherbourne, C. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med. Care 1992, 30, 473–483. [Google Scholar] [CrossRef]
- Webster, K.; Cella, D.; Yost, K. The Functional Assessment of Chronic Illness Therapy (FACIT) Measurement System: Properties, applications, and interpretation. Health Qual. Life Outcomes 2003, 1, 79. [Google Scholar] [CrossRef]
- EuroQol—A new facility for the measurement of health-related quality of life. Health Policy 1990, 16, 199–208. [CrossRef]
- Ware, J.E.J. SF-36 Health Survey Update. Spine 2000, 25, 3130–3139. [Google Scholar] [CrossRef] [PubMed]
- Shaw, J.W.; Johnson, J.A.; Coons, S.J. US Valuation of the EQ-5D Health States: Development and Testing of the D1 Valuation Model. Med. Care 2005, 43, 203–220. [Google Scholar] [CrossRef] [PubMed]
- EuroQol Research Foundation. EQ-5D-3L User Guide. 2018. Available online: https://euroqol.org/publications/user-guides (accessed on 9 June 2020).
- Strand, V.; Crawford, B. Improvement in health-related quality of life in patients with SLE following sustained reductions in anti-dsDNA antibodies. Expert Rev. Pharmacoecon. Outcomes Res. 2005, 5, 317–326. [Google Scholar] [CrossRef]
- Strand, V.; Levy, R.A.; Cervera, R.; Petri, M.A.; Birch, H.; Freimuth, W.W.; Zhong, Z.J.; Clarke, A.E. Improvements in health-related quality of life with belimumab, a B-lymphocyte stimulator-specific inhibitor, in patients with autoantibody-positive systemic lupus erythematosus from the randomised controlled BLISS trials. Ann. Rheum. Dis. 2014, 73, 838–844. [Google Scholar] [CrossRef] [PubMed]
- Coretti, S.; Ruggeri, M.; McNamee, P. The minimum clinically important difference for EQ-5D index: A critical review. Expert Rev. Pharmacoecon. Outcomes Res. 2014, 14, 221–233. [Google Scholar] [CrossRef]
- Bangert, E.; Wakani, L.; Merchant, M.; Strand, V.; Touma, Z. Impact of belimumab on patient-reported outcomes in systemic lupus erythematosus: Review of clinical studies. Patient Relat. Outcome Meas. 2019, 10, 1. [Google Scholar] [CrossRef]
- Hay, E.M.; Bacon, P.A.; Gordon, C.; Isenberg, D.A.; Maddison, P.; Snaith, M.L.; Symmons, D.P.M.; Viner, N.; Zoma, A. The BILAG index: A reliable and valid instrument for measuring clinical disease activity in systemic lupus erythematosus. QJM Int. J. Med. 1993, 86, 447–458. [Google Scholar] [CrossRef]
- Gladman, D.; Ginzler, E.; Goldsmith, C.; Fortin, P.; Liang, M.; Sanchez-Guerrero, J.; Urowitz, M.; Bacon, P.; Bombardieri, S.; Hanly, J.; et al. The development and initial validation of the systemic lupus international collaborating clinics/American college of rheumatology damage index for systemic lupus erythematosus. Arthritis Rheumat. 1996, 39, 363–369. [Google Scholar] [CrossRef]
- Azizoddin, D.R.; Gandhi, N.; Weinberg, S.; Sengupta, M.; Nicassio, P.M.; Jolly, M. Fatigue in systemic lupus: The role of disease activity and its correlates. Lupus 2019, 28, 163–173. [Google Scholar] [CrossRef]
- Tamayo, T.; Fischer-Betz, R.; Beer, S.; Winkler-Rohlfing, B.; Schneider, M. Factors influencing the health related quality of life in patients with systemic lupus erythematosus: Long-term results (2001–2005) of patients in the German Lupus Erythematosus Self-Help Organization (LULA Study). Lupus 2010, 19, 1606–1613. [Google Scholar] [CrossRef] [PubMed]
- Alarcon, G.S.; McGwin, G., Jr.; Uribe, A.; Friedman, A.W.; Roseman, J.M.; Fessler, B.J.; Bastian, H.M.; Baethge, B.A.; Vila, L.M.; Reveille, J.D. Systemic lupus erythematosus in a multiethnic lupus cohort (LUMINA). XVII. Predictors of self-reported health-related quality of life early in the disease course. Arthritis Care Res. 2004, 51, 465–474. [Google Scholar] [CrossRef] [PubMed]
- Rand, J.H.; Wu, X.X.; Quinn, A.S.; Chen, P.P.; Hathcock, J.J.; Taatjes, D.J. Hydroxychloroquine directly reduces the binding of antiphospholipid antibody-beta2-glycoprotein I complexes to phospholipid bilayers. Blood 2008, 112, 1687–1695. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; Silverman, E.; Bargman, J.M. The role of antimalarial agents in the treatment of SLE and lupus nephritis. Nat. Rev. Nephrol. 2011, 7, 718–729. [Google Scholar] [CrossRef]
- McElhone, K.; Abbott, J.; Shelmerdine, J.; Bruce, I.N.; Ahmad, Y.; Gordon, C.; Peers, K.; Isenberg, D.; Ferenkeh-Koroma, A.; Griffiths, B.; et al. Development and validation of a disease-specific health-related quality of life measure, the LupusQol, for adults with systemic lupus erythematosus. Arthritis Care Res. 2007, 57, 972–979. [Google Scholar] [CrossRef]
- Yazdany, J. Health-related quality of life measurement in adult systemic lupus erythematosus: Lupus Quality of Life (LupusQoL), Systemic Lupus Erythematosus-Specific Quality of Life Questionnaire (SLEQOL), and Systemic Lupus Erythematosus Quality of Life Questionnaire (L-QoL). Arthritis Care Res. 2011, 63 (Suppl. 11), S413–S419. [Google Scholar] [CrossRef]
- Kuhn, A.; Ruland, V.; Bonsmann, G. Cutaneous lupus erythematosus: Update of therapeutic options part I. J. Am. Acad. Dermatol. 2011, 65, e179–e193. [Google Scholar] [CrossRef]
- Iudici, M.; Pantano, I.; Fasano, S.; Pierro, L.; Charlier, B.; Pingeon, M.; Dal Piaz, F.; Filippelli, A.; Izzo, V. Health status and concomitant prescription of immunosuppressants are risk factors for hydroxychloroquine non-adherence in systemic lupus patients with prolonged inactive disease. Lupus 2018, 27, 265–272. [Google Scholar] [CrossRef]
- Chehab, G.; Sauer, G.M.; Richter, J.G.; Brinks, R.; Willers, R.; Fischer-Betz, R.; Winkler-Rohlfing, B.; Schneider, M. Medical adherence in patients with systemic lupus erythematosus in Germany: Predictors and reasons for non-adherence—A cross-sectional analysis of the LuLa-cohort. Lupus 2018, 27, 1652–1660. [Google Scholar] [CrossRef]
- Costedoat-Chalumeau, N.; Amoura, Z.; Hulot, J.S.; Aymard, G.; Leroux, G.; Marra, D.; Lechat, P.; Piette, J.C. Very low blood hydroxychloroquine concentration as an objective marker of poor adherence to treatment of systemic lupus erythematosus. Ann. Rheum. Dis. 2007, 66, 821–824. [Google Scholar] [CrossRef]
- Golder, V.; Ooi, J.J.Y.; Antony, A.S.; Ko, T.; Morton, S.; Kandane-Rathnayake, R.; Morand, E.F.; Hoi, A.Y. Discordance of patient and physician health status concerns in systemic lupus erythematosus. Lupus 2018, 27, 501–506. [Google Scholar] [CrossRef] [PubMed]
- Anker, S.D.; Agewall, S.; Borggrefe, M.; Calvert, M.; Jaime Caro, J.; Cowie, M.R.; Ford, I.; Paty, J.A.; Riley, J.P.; Swedberg, K.; et al. The importance of patient-reported outcomes: A call for their comprehensive integration in cardiovascular clinical trials. Eur. Heart J. 2014, 35, 2001–2009. [Google Scholar] [CrossRef] [PubMed]

| Patient Characteristics | Pooled BLISS | AMA Use | p Value | |
|---|---|---|---|---|
| + | − | |||
| Number of Patients | 1684 | 1098 | 586 | |
| Demographic Characteristics | ||||
| Age (years) | 37.8 (11.5) | 36.8 (11.4) | 39.6 (11.5) | <0.001 |
| Female sex | 1585 (94.1%) | 1033 (94.1%) | 552 (94.2%) | 0.922 |
| Ethnicity | ||||
| Asian | 353 (21.0%) | 243 (22.1%) | 110 (18.8%) | 0.107 |
| Black/African American | 146 (8.7%) | 98 (8.9%) | 48 (8.2%) | 0.610 |
| Indigenous American | 374 (22.2%) | 254 (23.1%) | 120 (20.5%) | 0.212 |
| White/Caucasian | 798 (47.4%) | 491 (44.7%) | 307 (52.4%) | 0.003 |
| Clinical Data | ||||
| SELENA-SLEDAI score | 9.7 (3.8) | 9.6 (3.6) | 10.0 (4.0) | 0.145 |
| SLE disease duration (years) | 6.4 (6.3) | 6.1 (6.2) | 7.0 (6.6) | 0.007 |
| SDI score | 0.78 (1.24) | 0.69 (1.15) | 0.95 (1.37) | <0.001 |
| SDI score = 0 | 977 (58.1%) | 673 (61.3%) | 304 (52.0%) | <0.001 |
| Glucocorticoid use | 1453 (86.3%) | 929 (84.6%) | 524 (89.4%) | 0.006 |
| Prednisone eq. dose (mg/day) | 10.8 (8.7) | 10.1 (8.5) | 12.1 (8.8) | <0.001 |
| AMA use | 1098 (64.8%) | 1098 (100%) | N/A | N/A |
| Hydroxychloroquine | 836 (49.6%) | 836 (76.1%) | N/A | N/A |
| Chloroquine | 265 (15.7%) | 265 (24.1%) | N/A | N/A |
| Other antimalarial agents * | 5 (0.3%) | 5 (0.5%) | N/A | N/A |
| Hydroxychloroquine eq. dose (mg/day) | 219.6 (183.5) | 336.2 (111.0) | N/A | N/A |
| Immunosuppressants | 816 (48.5%) | 476 (43.4%) | 340 (58.0%) | <0.001 |
| Azathioprine | 389 (23.1%) | 221 (20.1%) | 168 (28.7%) | <0.001 |
| Methotrexate | 231 (13.7%) | 144 (13.1%) | 87 (14.8%) | 0.325 |
| Mycophenolic acid | 189 (11.2%) | 104 (9.5%) | 85 (14.5%) | 0.002 |
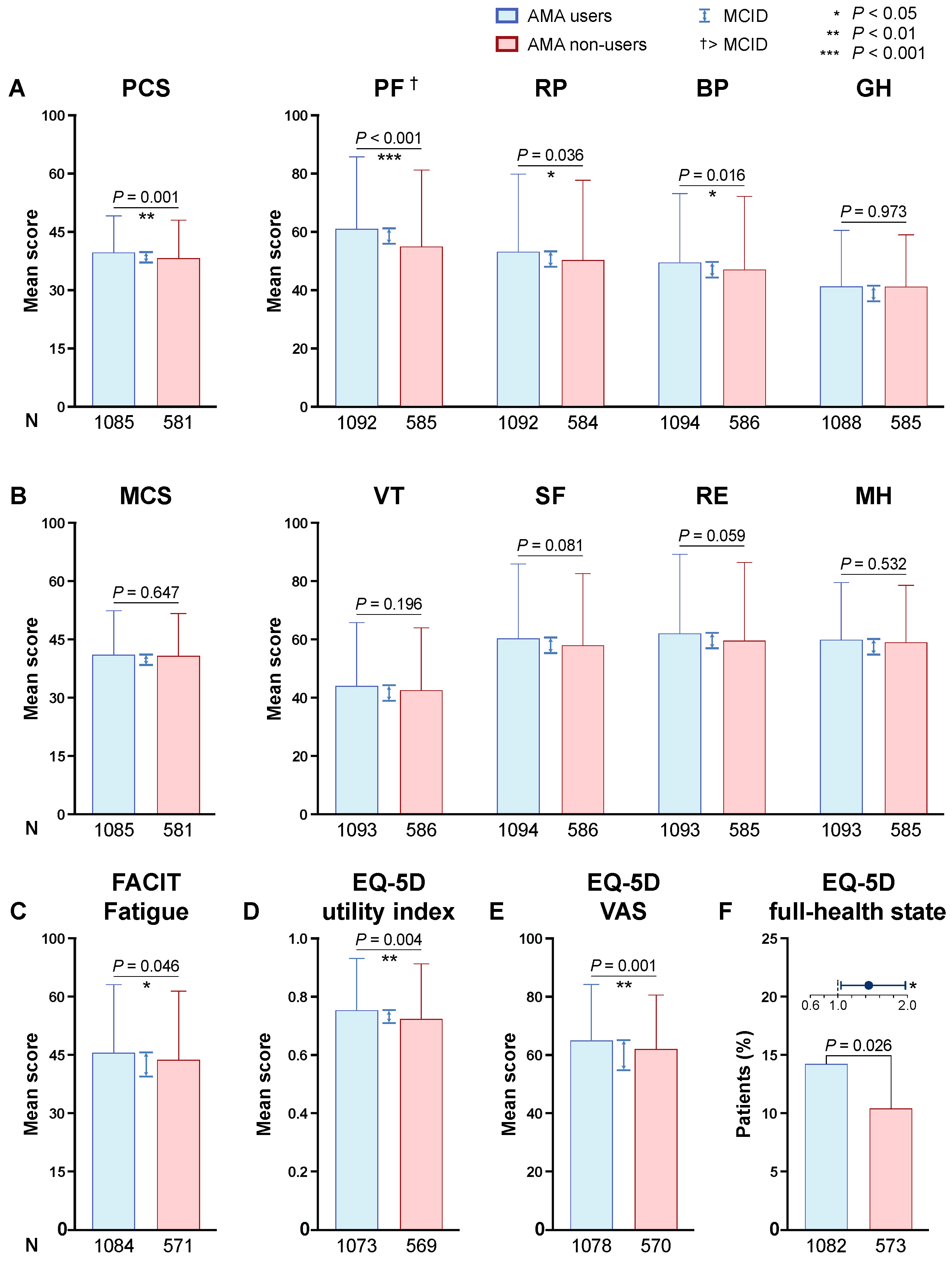
| HRQoL Items | AMA Users | AMA Non-Users | p Value | MCID | |||
|---|---|---|---|---|---|---|---|
| Number of Patients | 638 | 353 | |||||
| SF-36 | |||||||
| Physical component summary | 39.63 | (9.31) | 37.82 | (9.76) | 0.006 | No | |
| Mental component summary | 40.60 | (11.44) | 40.02 | (10.82) | 0.448 | No | |
| Physical functioning | 61.46 | (24.71) | 54.16 | (26.58) | <0.001 | Yes | |
| Role physical | 52.51 | (26.59) | 49.00 | (27.52) | 0.033 | No | |
| Bodily pain | 48.81 | (22.71) | 46.17 | (24.82) | 0.038 | No | |
| General health | 41.40 | (19.22) | 40.98 | (18.09) | 0.763 | No | |
| Vitality | 43.86 | (21.34) | 41.91 | (21.33) | 0.175 | No | |
| Social functioning | 60.63 | (25.14) | 57.01 | (25.01) | 0.043 | No | |
| Role emotional | 61.13 | (27.44) | 58.96 | (27.07) | 0.165 | No | |
| Mental health | 59.20 | (20.19) | 57.60 | (19.31) | 0.245 | No | |
| FACIT-Fatigue | |||||||
| Score | 30.32 | (11.74) | 28.87 | (12.09) | 0.077 | No | |
| EQ-5D | |||||||
| Utility index | 0.747 | (0.173) | 0.716 | (0.189) | 0.003 | No | |
| VAS | 65.10 | (19.27) | 60.53 | (18.99) | <0.001 | No | |
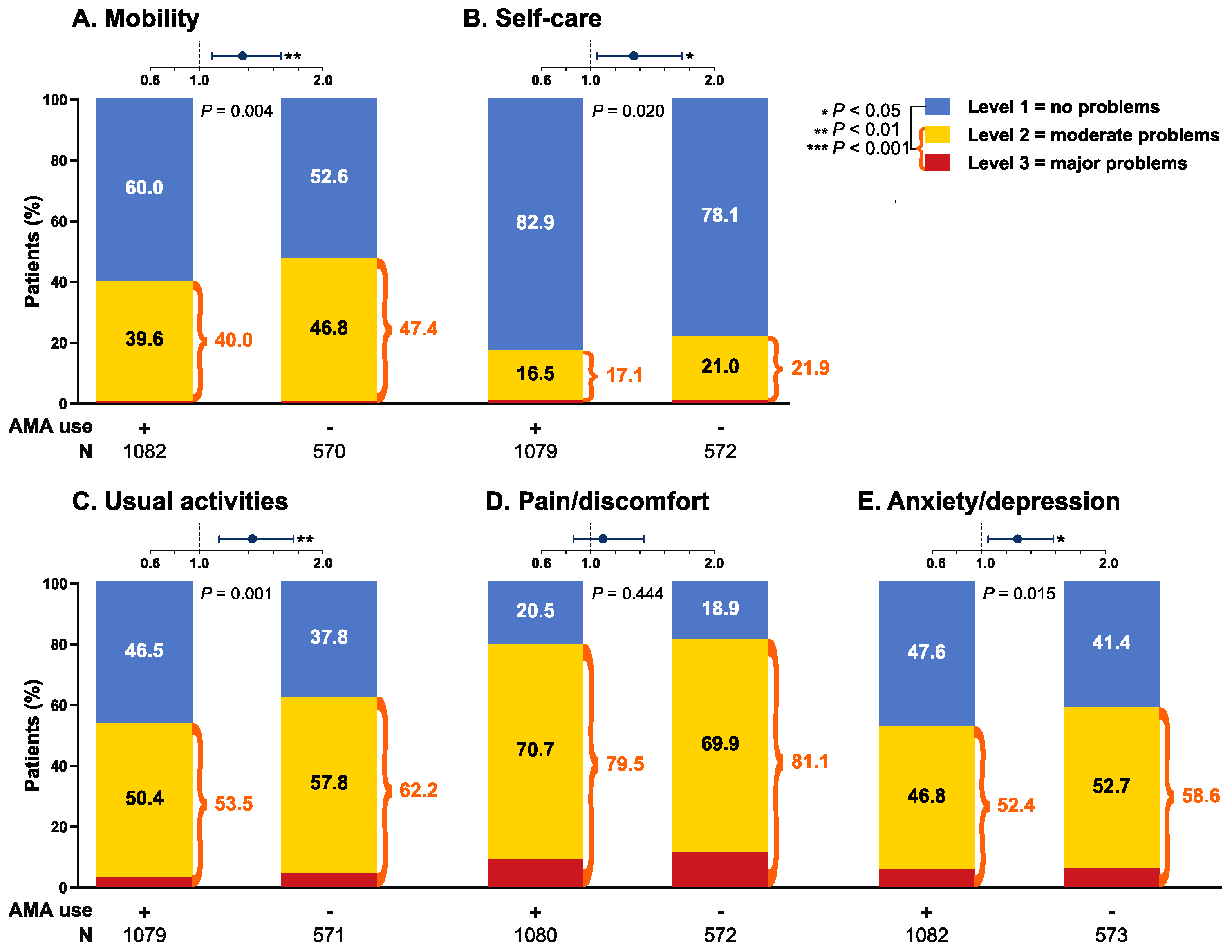
| EQ-5D Dimensions | |||||||
| Mobility | L 1 | 377 | (60.2%) | 169 | (49.4%) | 0.001 | N/A |
| L 2 | 249 | (39.8%) | 172 | (50.3%) | |||
| L 3 | 0 | (0%) | 1 | (0.3%) | |||
| Self-care | L 1 | 518 | (82.9%) | 261 | (75.9%) | 0.009 | N/A |
| L 2 | 103 | (16.5%) | 78 | (22.7%) | |||
| L 3 | 4 | (0.6%) | 5 | (1.5%) | |||
| Usual activities | L 1 | 281 | (44.9%) | 118 | (34.3%) | 0.001 | N/A |
| L 2 | 331 | (52.9%) | 213 | (61.9%) | |||
| L 3 | 14 | (2.2%) | 13 | (3.8%) | |||
| Pain or discomfort | L 1 | 116 | (18.6%) | 58 | (16.9%) | 0.510 | N/A |
| L 2 | 460 | (73.6%) | 248 | (72.1%) | |||
| L 3 | 49 | (7.8%) | 38 | (11.0%) | |||
| Anxiety or depression | L 1 | 295 | (47.1%) | 132 | (38.4%) | 0.009 | N/A |
| L 2 | 295 | (47.1%) | 196 | (57.0%) | |||
| L 3 | 36 | (5.8%) | 16 | (4.7%) | |||
| Full-health state | 77 | (12.3%) | 34 | (9.9%) | 0.262 | N/A | |
| HRQoL Items | AMA Users | AMA Non-Users | p Value | MCID | |||
|---|---|---|---|---|---|---|---|
| Number of Patients | 363 | 372 | |||||
| SF-36 | |||||||
| Physical component summary | 37.18 | (9.12) | 35.95 | (8.99) | 0.042 | No | |
| Mental component summary | 40.22 | (11.39) | 40.08 | (11.58) | 0.903 | No | |
| Physical functioning | 55.34 | (24.10) | 49.98 | (26.06) | 0.002 | Yes | |
| Role physical | 48.87 | (25.78) | 46.27 | (25.87) | 0.131 | No | |
| Bodily pain | 43.26 | (21.24) | 40.96 | (22.31) | 0.053 | No | |
| General health | 38.89 | (18.83) | 39.94 | (17.45) | 0.298 | No | |
| Vitality | 40.16 | (21.53) | 39.45 | (21.05) | 0.584 | No | |
| Social functioning | 56.91 | (25.85) | 55.01 | (24.47) | 0.361 | No | |
| Role emotional | 59.33 | (27.46) | 57.40 | (27.77) | 0.207 | No | |
| Mental health | 58.54 | (19.68) | 57.80 | (20.50) | 0.654 | No | |
| FACIT-Fatigue | |||||||
| Score | 28.37 | (11.93) | 27.22 | (11.96) | 0.167 | No | |
| EQ-5D | |||||||
| Utility index | 0.706 | (0.182) | 0.684 | (0.195) | 0.080 | No | |
| VAS | 61.76 | (19.91) | 59.21 | (18.28) | 0.042 | No | |
| EQ-5D Dimensions | |||||||
| Mobility | L 1 | 318 | (50.5%) | 157 | (43.9%) | 0.045 | N/A |
| L 2 | 311 | (49.4%) | 200 | (55.9%) | |||
| L 3 | 1 | (0.2%) | 1 | (0.3%) | |||
| Self-care | L 1 | 491 | (78.2%) | 258 | (71.9%) | 0.026 | N/A |
| L 2 | 132 | (21.0%) | 98 | (27.3%) | |||
| L 3 | 5 | (0.8%) | 3 | (0.8%) | |||
| Usual activities | L 1 | 230 | (36.7%) | 111 | (31.0%) | 0.072 | N/A |
| L 2 | 374 | (59.6%) | 224 | (62.6%) | |||
| L 3 | 23 | (3.7%) | 23 | (6.4%) | |||
| Pain or discomfort | L 1 | 66 | (10.5%) | 31 | (8.6%) | 0.345 | N/A |
| L 2 | 489 | (77.7%) | 276 | (76.9%) | |||
| L 3 | 74 | (11.8%) | 52 | (14.5%) | |||
| Anxiety or depression | L 1 | 261 | (41.4%) | 147 | (40.8%) | 0.855 | N/A |
| L 2 | 326 | (51.7%) | 188 | (52.2%) | |||
| L 3 | 43 | (6.8%) | 25 | (6.9%) | |||
| Full-health state | 43 | (6.8%) | 20 | (5.6%) | 0.431 | N/A | |
| HRQoL Items | AMA Users | AMA Non-Users | p Value | MCID | |||
|---|---|---|---|---|---|---|---|
| Number of Patients | 112 | 67 | |||||
| SF-36 | |||||||
| Physical component summary | 40.70 | (9.88) | 39.39 | (11.87) | 0.471 | No | |
| Mental component summary | 41.44 | (10.89) | 39.54 | (11.42) | 0.288 | No | |
| Physical functioning | 60.69 | (26.23) | 57.93 | (28.50) | 0.547 | No | |
| Role physical | 56.10 | (28.05) | 52.33 | (31.74) | 0.503 | No | |
| Bodily pain | 54.42 | (26.78) | 49.28 | (31.18) | 0.149 | Yes | |
| General health | 43.19 | (20.16) | 40.34 | (19.56) | 0.309 | No | |
| Vitality | 46.27 | (22.28) | 44.40 | (24.19) | 0.516 | No | |
| Social functioning | 62.39 | (25.14) | 56.90 | (26.68) | 0.138 | Yes | |
| Role emotional | 63.58 | (25.69) | 57.90 | (27.58) | 0.183 | Yes | |
| Mental health | 59.99 | (19.73) | 57.87 | (19.89) | 0.545 | No | |
| FACIT-Fatigue | |||||||
| Score | 32.19 | (11.64) | 30.03 | (13.11) | 0.357 | No | |
| EQ-5D | |||||||
| Utility index | 0.768 | (0.206) | 0.733 | (0.221) | 0.286 | No | |
| VAS | 65.61 | (21.59) | 59.86 | (19.48) | 0.041 | No | |
| EQ-5D Dimensions | |||||||
| Mobility | L 1 | 69 | (63.9%) | 35 | (53.0%) | 0.156 | N/A |
| L 2 | 39 | (36.1%) | 31 | (47.0%) | |||
| L 3 | 0 | (0%) | 0 | (0%) | |||
| Self-care | L 1 | 84 | (78.5%) | 54 | (81.8%) | 0.598 | N/A |
| L 2 | 22 | (20.6%) | 12 | (18.2%) | |||
| L 3 | 1 | (0.9%) | 0 | (0%) | |||
| Usual activities | L 1 | 52 | (48.6%) | 30 | (45.5%) | 0.688 | N/A |
| L 2 | 54 | (50.5%) | 32 | (48.5%) | |||
| L 3 | 1 | (0.9%) | 4 | (6.1%) | |||
| Pain or discomfort | L 1 | 33 | (30.8%) | 22 | (33.3%) | 0.732 | N/A |
| L 2 | 63 | (58.9%) | 35 | (53.0%) | |||
| L 3 | 11 | (10.3%) | 9 | (13.6%) | |||
| Anxiety or depression | L 1 | 59 | (54.6%) | 23 | (34.8%) | 0.011 | N/A |
| L 2 | 45 | (41.7%) | 37 | (56.1%) | |||
| L 3 | 4 | (3.7%) | 6 | (9.1%) | |||
| Full-health state | 27 | (25.0%) | 13 | (19.7%) | 0.420 | N/A | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gomez, A.; Soukka, S.; Johansson, P.; Åkerström, E.; Emamikia, S.; Enman, Y.; Chatzidionysiou, K.; Parodis, I. Use of Antimalarial Agents Is Associated with Favourable Physical Functioning in Patients with Systemic Lupus Erythematosus. J. Clin. Med. 2020, 9, 1813. https://doi.org/10.3390/jcm9061813
Gomez A, Soukka S, Johansson P, Åkerström E, Emamikia S, Enman Y, Chatzidionysiou K, Parodis I. Use of Antimalarial Agents Is Associated with Favourable Physical Functioning in Patients with Systemic Lupus Erythematosus. Journal of Clinical Medicine. 2020; 9(6):1813. https://doi.org/10.3390/jcm9061813
Chicago/Turabian StyleGomez, Alvaro, Sofia Soukka, Petter Johansson, Emil Åkerström, Sharzad Emamikia, Yvonne Enman, Katerina Chatzidionysiou, and Ioannis Parodis. 2020. "Use of Antimalarial Agents Is Associated with Favourable Physical Functioning in Patients with Systemic Lupus Erythematosus" Journal of Clinical Medicine 9, no. 6: 1813. https://doi.org/10.3390/jcm9061813
APA StyleGomez, A., Soukka, S., Johansson, P., Åkerström, E., Emamikia, S., Enman, Y., Chatzidionysiou, K., & Parodis, I. (2020). Use of Antimalarial Agents Is Associated with Favourable Physical Functioning in Patients with Systemic Lupus Erythematosus. Journal of Clinical Medicine, 9(6), 1813. https://doi.org/10.3390/jcm9061813






