Post-Laser Twin Anemia Polycythemia Sequence: Diagnosis, Management, and Outcome in an International Cohort of 164 Cases
Abstract
1. Introduction
2. Experimental Section
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Center | Country | Post-Laser TAPS Cases |
|---|---|---|
| Leiden University Medical Center | The Netherlands | 66 |
| Children’s Hospital V. Buzzi Milan | Italy | 17 |
| Necker-Enfants Malades Hospital Paris | France | 16 |
| Leuven University Hospital | Belgium | 13 |
| Center Medico-Chirurgical Obstetrical Strasbourg | France | 10 |
| Mount Sinai Hospital Toronto | Canada | 10 |
| Saint George’s Hospital London | United Kingdom | 5 |
| Birmingham Women’s and Children’s NHS Foundation Trust | United Kingdom | 5 |
| Medical University of Graz | Austria | 3 |
| Mater Hospital Brisbane | Australia | 4 |
| University of Texas McGovern Medical School at Houston | United States of America | 4 |
| V.I. Kulakov National Medical Research Center of Obstetrics, Gynecology and Perinatology Moscow | Russia | 4 |
| Karolinska University Hospital Stockholm | Sweden | 3 |
| Hospital Universitari Vall d’Hebron Barcelona | Spain | 2 |
| Brugmann University Hospital | Belgium | 1 |
| University Medical Center Hamburg-Eppendorf | Germany | 1 |
Appendix B
| Death † (n = 60/304) | Alive † (n = 244/304) | Univariate Analysis OR (95% CI) | SE | p | Multivariate Analysis OR (95% CI) | SE | p | |
|---|---|---|---|---|---|---|---|---|
| GA at diagnosis TAPS | 23.3 ± 3.3 | 23.7 ± 3.7 | 1.0 (0.9–1.1) | 0.1 | 0.623 | |||
| GA at laser TTTS | 20.1 ± 2.4 | 20.8 ± 3.3 | 0.9 (0.8–1.0) | 0.1 | 0.233 | |||
| Days between TTTS laser and TAPS | 20.9 ± 18.4 | 21.8 ± 23.1 | 1.0 (1.0–1.0) | 0.0 | 0.829 | |||
| Quintero stage for TTTS ‡ | ||||||||
| 1 | 3/35 (9) | 32/35 (91) | - * | - * | ||||
| 2 | 27/140 (19) | 113/140 (81) | 2.5 (0.7–8.5) | 0.6 | 0.139 | 1.7 (0.4–6.9) | 0.7 | 0.439 |
| 3 | 27/111 (24) | 84/111(76) | 3.6 (1.1–12.0) | 0.6 | 0.039 | 3.5 (0.9–13.2) | 0.7 | 0.069 |
| 4 | 2/10 (20) | 8/10 (80) | 2.8 (0.2–32.4) | 1.3 | 0.418 | 3.4 (0.3–33.6) | 1.2 | 0.298 |
| Antenatal TAPS stage | ||||||||
| 1 | 6/47 (13) | 41/47 (87) | - * | - * | ||||
| 2 | 12/122 (10) | 110/122 (90) | 0.9 (0.3–3.2) | 0.6 | 0.864 | 0.8 (0.2–3.2) | 0.7 | 0.790 |
| 3 | 14/56 (25) | 50/56 (75) | 2.6 (0.7–9.1) | 0.6 | 0.136 | 2.6 (0.6–10.3) | 0.7 | 0.182 |
| 4 | 20/46 (44) | 26/46 (55) | 6.1 (1.8–20.4) | 0.6 | 0.003 | 6.1 (1.4–26.0) | 0.7 | 0.015 |
| TAPS recipient | 17/158 (11) | 146/158 (89) | - * | - * | ||||
| TAPS donor | 43/146 (30) | 103/146 (70) | 3.7 (2.2–6.3) | 0.3 | <0.001 | 4.2 (2.1–8.3) | 0.3 | <0.001 |
| Persistence of TTTS-TAPS donor-role, no § | 25/162 (15) | 137/162 (85) | - * | |||||
| Persistence of TTTS-TAPS donor-role, yes § | 34/137 (25) | 103/137 (77) | 1.8 (1.0–3.5) | 0.3 | 0.061 | |||
| Antenatal therapy | ||||||||
| Expectant management | 27/124 (22) | 97/124 (78) | - * | |||||
| Delivery | 4/18 (22) | 14/18 (78) | 1.4 (0.3–4.2) | 0.6 | 0.622 | |||
| IUT (± PET) | 16/81 (20) | 72/81 (82) | 0.9 (0.4–1.9) | 0.4 | 0.893 | |||
| Laser surgery | 9/44 (21) | 38/44 (79) | 1.0 (0.3–2.8) | 0.5 | 0.942 | |||
| Selective feticide (co-twin) | 0/11 (0) | 11/11 (100) | 0.3 (0.0–2.9) | 1.1 | 0.330 | |||
| GA at birth | 29.5 ± 5.0 | 31.6 ± 3.1 | 0.8 (0.7–1.0) | 0.1 | 0.006 | 0.8 (0.7–0.9) | 0.1 | 0.001 |
| SNM † (139/429) | No SNM † (290/429) | Univariate Analysis OR (95% CI) | SE | p | Multivariate Analysis OR (95% CI) | SE | p | |
|---|---|---|---|---|---|---|---|---|
| GA at diagnosis TAPS | 24.5 ± 3.1 | 23.4 ± 4.0 | 0.9 (0.8–1.0) | 0.1 | 0.090 | |||
| GA at laser TTTS | 21.1 ± 2.6 | 20.6 ± 3.6 | 0.9 (0.8–1.0) | 0.1 | 0.261 | |||
| Days between TTTS laser and TAPS | 22.3 ± 21.9 | 22.1 ± 23.9 | 1.0 (1.0–1.0) | 0.0 | 0.987 | |||
| Quintero stage for TTTS | ||||||||
| 1 | 8/34 (24) | 26/34 (76) | - * | - * | ||||
| 2 | 64/123 (52) | 59/123 (48) | 3.2 (1.1–9.7) | 0.6 | 0.038 | 2.8 (0.9–9.0) | 0.6 | 0.066 |
| 3 | 28/89 (32) | 61/89 (68) | 1.3 (0.4–4.2) | 0.6 | 0.617 | 1.3 (0.4–3.9) | 0.6 | 0.690 |
| 4 | 3/10 (30) | 3/10 (70) | 1.3 (0.2–9.3) | 1.0 | 0.782 | 1.1 (0.1–10.0) | 1.1 | 0.959 |
| Antenatal TAPS stage | ||||||||
| 1 | 18/45 (40) | 30/45 (60) | - * | |||||
| 2 | 41/112 (37) | 71/112 (63) | 1.0 (0.4–2.5) | 0.5 | 0.970 | |||
| 3 | 16/45 (36) | 29/45 (64) | 0.7 (0.2–2.2) | 0.6 | 0.570 | |||
| 4 | 17/32 (53) | 15/32 (47) | 1.6 (0.5–5.0) | 0.6 | 0.381 | |||
| TAPS recipient | 54/145(37) | 91/145 (63) | - * | |||||
| TAPS donor | 51/118 (43) | 67/118 (57) | 1.1 (0.8–1.5) | 0.2 | 0.568 | |||
| Persistence of TTTS-TAPS donor-role, no | 47/139 (34) | 92/139 (66) | - * | |||||
| Persistence of TTTS-TAPS donor-role, yes | 58/120 (48) | 62/120 (52) | 1.6 (0.9–3.0) | 0.3 | 0.116 | |||
| Antenatal therapy | ||||||||
| Expectant management | 34/100 (34) | 66/100 (66) | - * | |||||
| Delivery | 9/17 (53) | 8/17 (47) | 2.1 (0.6–8.1) | 0.7 | 0.270 | |||
| IUT (± PET) | 34/72 (47) | 38/72 (53) | 1.7 (0.8–3.7) | 0.4 | 0.158 | |||
| Laser surgery | 12/37 (32) | 25/37 (68) | 1.1 (0.4–2.9) | 0.5 | 0.851 | |||
| Selective feticide | 3/11 (27) | 8/11 (73) | 0.7 (0.2–3.2) | 0.7 | 0.720 | |||
| GA at birth | 29.4 ± 2.6 | 32.9 ± 2.8 | 1.5 (1.3–1.7) | 0.1 | <0.001 | 1.5 (1.3–1.7) | 0.1 | <0.001 |
| Severe growth restriction, no | 81/200 (41) | 119/200 (60) | - * | |||||
| Severe growth restriction, yes | 24/62 (39) | 38/62 (61) | 1.3 (0.8–2.2) | 0.3 | 0.351 | |||
| Postnatal TAPS, no | 23/58 (40) | 35/58 (60) | - * | |||||
| Postnatal TAPS, yes | 64/151 (42) | 87/151(58) | 1.1 (0.5–2.5) | 0.4 | 0.800 |
References
- Lopriore, E.; Middeldorp, J.M.; Oepkes, D.; Kanhai, H.H.; Walter, F.J.; Vandenbussche, F.P. Twin anemia-polycythemia sequence in two monochorionic twin pairs without oligo-polyhydramnios sequence. Placenta 2007, 28, 47–51. [Google Scholar] [CrossRef] [PubMed]
- Slaghekke, F.; Lopriore, E.; Lewi, L.; Middeldorp, J.M.; van Zwet, E.W.; Weingertner, A.S.; Klumper, F.J.; Dekoninck, P.; Devlieger, R.; Kilby, M.D.; et al. Fetoscopic laser coagulation of the vascular equator versus selective coagulation for twin-to-twin transfusion syndrome: An open-label randomised controlled trial. Lancet 2014, 383, 2144–2151. [Google Scholar] [CrossRef]
- Lewi, L.; Jani, J.; Blickstein, I.; Huber, A.; Gucciardo, L.; Van Mieghem, T.; Done, E.; Boes, A.S.; Hecher, K.; Gratacos, E.; et al. The outcome of monochorionic diamniotic twin gestations in the era of invasive fetal therapy: A prospective cohort study. Am. J. Obstet. Gynecol. 2008, 199, 514. [Google Scholar] [CrossRef] [PubMed]
- Lewi, L.; Jani, J.; Cannie, M.; Robyr, R.; Ville, Y.; Hecker, K.; Gratacos, E.; Vandecruys, H.; Vandecaveye, V.; Dymarkowski, S.; et al. Intertwin anastomoses in monochorionic placentas after fetoscopic laser coagulation for twin-to-twin transfusion syndrome: Is there more than meets the eye? Am. J. Obstet. Gynecol. 2006, 194, 790–795. [Google Scholar] [CrossRef]
- Knijnenburg, P.J.C.; Slaghekke, F.; Tollenaar, L.S.A.; van Klink, J.M.; Zhao, D.P.; Middeldorp, J.M.; Haak, M.C.; Klumper, F.J.; Oepkes, D.; Lopriore, E. Incidence of and Risk Factors for Residual Anastomoses in Twin-Twin Transfusion Syndrome Treated with Laser Surgery: A 15-Year Single-Center Experience. Fetal. Diagn. Ther. 2019, 45, 13–20. [Google Scholar] [CrossRef]
- Slaghekke, F.; Lewi, L.; Middeldorp, J.M.; Weingertner, A.S.; Klumper, F.J.; Dekoninck, P.; Devlieger, R.; Lanna, M.M.; Deprest, J.; Favre, R.; et al. Residual anastomoses in twin-twin transfusion syndrome after laser: The Solomon randomized trial. Am. J. Obstet. Gynecol. 2014, 211, 285 e1–285 e7. [Google Scholar] [CrossRef] [PubMed]
- Tollenaar, L.S.; Slaghekke, F.; Middeldorp, J.M.; Klumper, F.J.; Haak, M.C.; Oepkes, D.; Lopriore, E. Twin Anemia Polycythemia Sequence: Current Views on Pathogenesis, Diagnostic Criteria, Perinatal Management, and Outcome. Twin Res. Hum. Genet. 2016, 19, 222–233. [Google Scholar] [CrossRef] [PubMed]
- Tollenaar, L.S.A.; Slaghekke, F.; Lewi, L.; Colmant, C.; Lanna, M.M.; Weingertner, A.S.; Ryan, G.; Arevalo, S.; Tavares de Sousa, M. Spontaneous Twin Anemia Polycythemia Sequence: Management and Outcome in a Large International Cohort of 249 Cases. Submitted.
- Slaghekke, F.; Pasman, S.; Veujoz, M.; Middeldorp, J.M.; Lewi, L.; Devlieger, R.; Favra, R.; Lopriore, E.; Oepkes, D. Middle cerebral artery peak systolic velocity to predict fetal hemoglobin levels in twin anemia-polycythemia sequence. Ultrasound Obstet. Gynecol. 2015, 46, 432–436. [Google Scholar] [CrossRef]
- Lopriore, E.; Slaghekke, F.; Oepkes, D.; Middeldorp, J.M.; Vandenbussche, F.P.; Walther, F.J. Hematological characteristics in neonates with twin anemia-polycythemia sequence (TAPS). Prenat. Diagn. 2010, 30, 251–255. [Google Scholar]
- Lopriore, E.; Slaghekke, F.; Middeldorp, J.M.; Klumper, F.J.; Van Lith, J.M.; Walther, F.J.; Oepkes, D. Accurate and simple evaluation of vascular anastomoses in monochorionic placentas using colored dye. J. Vis. Exp. 2011, 55, e3208. [Google Scholar] [CrossRef] [PubMed]
- Trieu, N.T.; Weingertner, A.S.; Guerra, F.; Dautun, D.; Kohler, M.; Vayssiere, C.; Nisand, I.; Favre, R. Evaluation of the measurement of the middle cerebral artery peak systolic velocity before and after placental laser coagulation in twin-to-twin transfusion syndrome. Prenat. Diagn. 2012, 32, 127–130. [Google Scholar] [CrossRef] [PubMed]
- Slaghekke, F.; Kist, W.J.; Oepkes, D.; Pasman, S.A.; Middeldorp, J.M.; Klumper, F.J.; Walther, F.J.; Vandenbussche, F.P.; Lopriore, E. Twin anemia-polycythemia sequence: Diagnostic criteria, classification, perinatal management and outcome. Fetal Diagn. Ther. 2010, 27, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Bell, M.J.; Ternberg, J.L.; Feigin, R.D.; Keating, J.P.; Marshall, R.; Barton, L.; Brotherton, T.; Neonatal necrotizing enterocolitis. Therapeutic decisions based upon clinical staging. Ann. Surg. 1978, 187, 1–7. [Google Scholar] [CrossRef] [PubMed]
- An international classification of retinopathy of prematurity. The Committee for the Classification of Retinopathy of Prematurity. Arch. Ophthalmol. 1984, 74, 127–133. [Google Scholar]
- Volpe, J.J. Intraventricular hemorrhage and brain injury in the premature infant. Diagn. Progn. Prev. Clin. Perinatol. 1989, 16, 387–411. [Google Scholar] [CrossRef]
- Levene, M.I. Measurement of the growth of the lateral ventricles in preterm infants with real-time ultrasound. Arch. Dis. Child. 1981, 56, 900–904. [Google Scholar] [CrossRef]
- De Vries, L.S.; Eken, P.; Dubowitz, L.M. The spectrum of leukomalacia using cranial ultrasound. Behav. Brain Res. 1992, 49, 1–6. [Google Scholar] [CrossRef]
- Hoftiezer, L.; Hof, M.H.P.; Dijs-Elsinga, J.; Hogeveen, M.; Hukkelhoven, C.; van Lingen, R.A. From population reference to national standard: New and improved birthweight charts. Am. J. Obstet. Gynecol. 2019, 220, 383. [Google Scholar] [CrossRef]
- Lopriore, E.; Hecher, K.; Vandenbussche, F.P.; van den Wijngaard, J.P.; Klumper, F.J.; Oepkes, D. Fetoscopic laser treatment of twin-to-twin transfusion syndrome followed by severe twin anemia-polycythemia sequence with spontaneous resolution. Am. J. Obstet. Gynecol. 2008, 198, e4–e7. [Google Scholar] [CrossRef]
- Khalil, A.; Rodgers, M.; Baschat, A.; Bhide, A.; Gratacos, E.; Hecher, K.; Kilby, M.D.; Lewi, L.; Nicolaides, K.H.; Oepkes, D.; et al. ISUOG Practice Guidelines: Role of ultrasound in twin pregnancy. Ultrasound Obstet. Gynecol. 2016, 47, 247–263. [Google Scholar]
- Slaghekke, F.; van Klink, J.M.; Koopman, H.M.; Middeldorp, J.M.; Oepkes, D.; Lopriore, E. Neurodevelopmental outcome in twin anemia-polycythemia sequence after laser surgery for twin-twin transfusion syndrome. Ultrasound Obstet. Gynecol. 2014, 44, 316–321. [Google Scholar] [CrossRef]
- Senat, M.V.; Deprest, J.; Boulvain, M.; Paupe, A.; Winer, N.; Ville, Y. Endoscopic laser surgery versus serial amnioreduction for severe twin-to-twin transfusion syndrome. N. Engl. J Med. 2004, 351, 136–144. [Google Scholar] [CrossRef]
- De, Villiers, S.F.; Slaghekke, F.; Middeldorp, J.M.; Walther, F.J.; Oepkes, D.; Lopriore, E. Placental characteristics in monochorionic twins with spontaneous versus post-laser twin anemia-polycythemia sequence. Placenta 2013, 34, 456–459. [Google Scholar] [CrossRef] [PubMed]
- Lopriore, E.; Oepkes, D.; van den Wijngaard, J.P.; van Gemert, M.J.; Middeldorp, J.M.; Vandenbussche, F.P. Twin anemia-polycythemia sequence (TAPS) without a cause. Prenat. Diagn. 2008, 28, 559–560. [Google Scholar] [CrossRef] [PubMed]
- Tollenaar, L.S.A.; Slaghekke, F.; Lewi, L.; Ville, Y.; Lanna, M.; Weingertner, A.; Ryan, G.; Arevalo, S.; Khalil, A.; Brock, C.O.; et al. Treatment and outcome in 370 cases with spontaneous or post-laser twin anemia polycythemia sequence managed in 17 different fetal therapy centers. Ultrasound Obstet. Gynecol. 2020. [Google Scholar] [CrossRef]
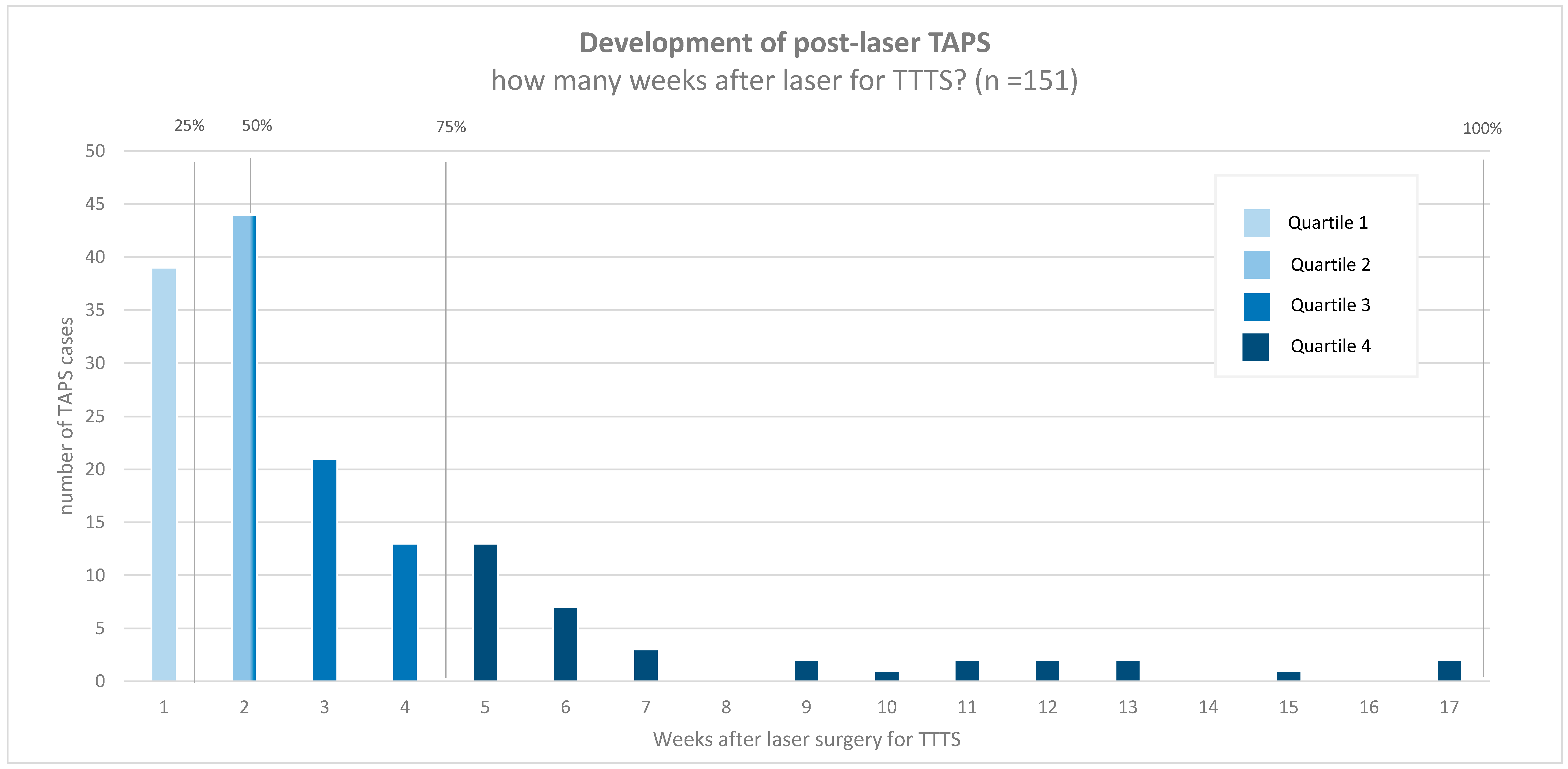
| Post-Laser TAPS (N = 164 Pregnancies, 328 Fetuses) | |
|---|---|
| Gravidity | 2 (1–3) |
| Parity | 0 (0–1) |
| Antenatal diagnosis of TAPS | 151/164 (92) |
| Location of placenta | |
| Anterior | 72/164 (44) |
| Posterior | 86/164 (52) |
| Other † | 6/164 (4) |
| TTTS stage ‡ | |
| Q1 | 20/159 (13) |
| Q2 | 75/159 (47) |
| Q3 | 59/159 (37) |
| Q4 | 5/159 (3) |
| Solomon technique for TTTS laser | 60/164 (37) |
| GA at laser for TTTS | 20.6 (18.0–23.0) |
| Time between laser and post-laser TAPS (days) | 14 (7–28) |
| Persistence of TTTS-TAPS donor-recipient role § | 73/161 (45) |
| Laser for TTTS complete (surgeon’s opinion) ¶ | 126/156 (81) |
| Post-Laser TAPS (N = 164 Pregnancies, 328 Fetuses) | |
|---|---|
| GA at diagnosis | 23.1 (20.4–25.9) |
| TAPS stage at diagnosis | |
| 1 | 43/151 (28) |
| 2 | 69/151 (48) |
| 3 | 27/151 (17) |
| 4 | 12/151 (8) |
| Highest TAPS stage | |
| 1 | 22/151 (15) |
| 2 | 62/151(40) |
| 3 | 26/151 (16) |
| 4 | 22/151 (14) |
| 5 | 19/151 (15) |
| Presence of additional ultrasound markers † | |
| Starry-sky liver (recipient) | 56/141 (40) |
| Difference in placental echogenicity | 35/151 (23) |
| Antenatal management | |
| Expectant management | 62/151 (43) |
| Delivery | 9/151 (6) |
| IUT (± PET) | 44/151 (29) |
| Laser surgery | 24/151 (15) |
| Selective feticide | 11/151 (7) |
| Termination of pregnancy | 1/151 (1) |
| Female ‡ | 162/308 (53) |
| Cesarean § | 206/326 (63) |
| Injected Post-Laser TAPS Placentas (N = 74) | |
|---|---|
| Total number of anastomoses | 1 (1–2) |
| Number of AV anastomoses | 1 (0–1) |
| Number of VA anastomoses | 0 (0–1) |
| Number of AA anastomoses | 0 (0–0) |
| Number of VV anastomoses | 0 (0–0) |
| Presence of anastomoses | |
| Presence of AV/VA anastomoses | 58/74 (80) |
| Presence of AA anastomoses | 6/74 (8) |
| Presence of VV anastomoses | 5/74 (7) |
| Type of anastomoses per placenta | |
| No anastomoses | 10/74 (14) |
| AV (one direction) | 40/74 (54) |
| AVs (both directions) | 14/74 (19) |
| AV/VA and AA | 3/74 (4) |
| AV/VA and VV | 2/74 (3) |
| Only AA | 2/74 (3) |
| Only VV | 2/74 (3) |
| AV/VA, AA and VV | 1/74 (1) |
| All anastomoses diameter < 1 mm | 62/64 (97) |
| Post-Laser TAPS (N = 164 Pregnancies; 328 Fetuses) | TAPS Donors (N = 164 Fetuses) | TAPS Recipients (N = 164 Fetuses) † | p-Value | |
|---|---|---|---|---|
| GA at birth | 31.7 (28.6–33.7; 19.0–41.3) | |||
| Fetal demise † | 56/327 (17) | 43/164 (26) | 13/163 (8) | <0.001 |
| Spontaneous | 33/327 (10) | 25/164 (15) | 8/163 (5) | <0.001 |
| Intended | 23/327 (7) | 18/164 (11) | 5/163 (3) | 0.007 |
| Neonatal mortality | 27/271 (10) | 18/121 (15) | 9/150 (6) | 0.008 |
| Perinatal mortality (overall) † | 83/327 (25) | 61/164 (37) | 22/163 (14) | <0.001 |
| Perinatal mortality (spontaneous) † | 60/327 (18) | 43/164 (26) | 17/163 (10) | <0.001 |
| Severe neonatal morbidity ‡ | 105/263 (40) | 51/118 (43) | 54/145 (37) | 0.568 |
| Respiratory distress syndrome | 88/263(34) | 42/118 (36) | 46/145 (32) | 0.945 |
| Patent ductus arteriosus | 20/263 (8) | 11/118 (9) | 9/145 (6) | 0.278 |
| Necrotizing enterocolitis | 6/263 (2) | 4/118 (3) | 2/145 (1) | 0.275 |
| Retinopathy of prematurity | 9/263 (3) | 6/118 (5) | 3/145 (2) | 0.165 |
| Amniotic band syndrome | 3/263 (1) | 2/118 (2) | 1/145 (1) | 0.451 |
| Severe cerebral injury | 28/263 (11) | 13/118 (11) | 15/145 (10) | 0.916 |
| Birth weight (g) § | 1390 ± 567 | 1346 ± 525 | 1533 ± 588 | <0.001 |
| Severe growth restriction (bw < p3) § | 62/270 (23) | 31/121 (26) | 31/149 (21) | 0.267 |
| Mild growth restriction (bw < p10) § | 122/270 (45) | 60/121 (50) | 61/149 (41) | 0.061 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tollenaar, L.S.A.; Lopriore, E.; Faiola, S.; Lanna, M.; Stirnemann, J.; Ville, Y.; Lewi, L.; Devlieger, R.; Weingertner, A.S.; Favre, R.; et al. Post-Laser Twin Anemia Polycythemia Sequence: Diagnosis, Management, and Outcome in an International Cohort of 164 Cases. J. Clin. Med. 2020, 9, 1759. https://doi.org/10.3390/jcm9061759
Tollenaar LSA, Lopriore E, Faiola S, Lanna M, Stirnemann J, Ville Y, Lewi L, Devlieger R, Weingertner AS, Favre R, et al. Post-Laser Twin Anemia Polycythemia Sequence: Diagnosis, Management, and Outcome in an International Cohort of 164 Cases. Journal of Clinical Medicine. 2020; 9(6):1759. https://doi.org/10.3390/jcm9061759
Chicago/Turabian StyleTollenaar, Lisanne S.A., Enrico Lopriore, Stefano Faiola, Mariano Lanna, Julien Stirnemann, Yves Ville, Liesbeth Lewi, Roland Devlieger, Anne Sophie Weingertner, Romain Favre, and et al. 2020. "Post-Laser Twin Anemia Polycythemia Sequence: Diagnosis, Management, and Outcome in an International Cohort of 164 Cases" Journal of Clinical Medicine 9, no. 6: 1759. https://doi.org/10.3390/jcm9061759
APA StyleTollenaar, L. S. A., Lopriore, E., Faiola, S., Lanna, M., Stirnemann, J., Ville, Y., Lewi, L., Devlieger, R., Weingertner, A. S., Favre, R., Hobson, S. R., Ryan, G., Rodo, C., Arévalo, S., Klaritsch, P., Greimel, P., Hecher, K., de Sousa, M. T., Khalil, A., ... Slaghekke, F. (2020). Post-Laser Twin Anemia Polycythemia Sequence: Diagnosis, Management, and Outcome in an International Cohort of 164 Cases. Journal of Clinical Medicine, 9(6), 1759. https://doi.org/10.3390/jcm9061759










