Salivary Cytokine Profile as a Possible Predictor of Autism Spectrum Disorder
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Groups
2.2. Dental Examination
2.3. Sample Collection
2.4. Measurement of Salivary IL-1β, IL-6, IL-8, Rantes, Eotaxin, MCP-1, and TNFα
2.5. Statistical Analysis
3. Results
3.1. Salivary IL-1β, IL-6, IL-8, Rantes, Eotaxin, MCP-1, and TNFα Levels
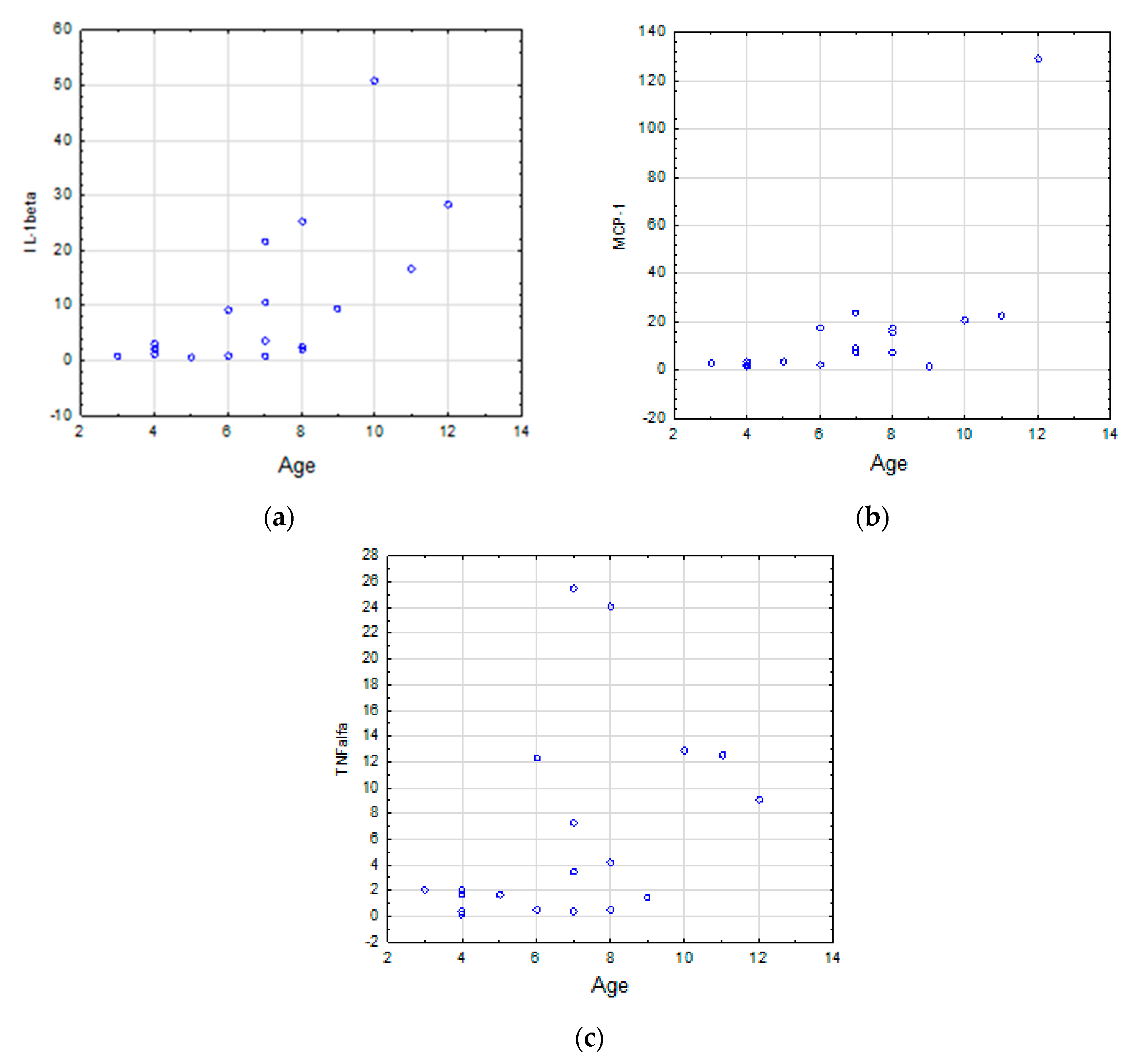
3.2. Correlations Between the Cytokine Levels and Age in TD and ASD groups
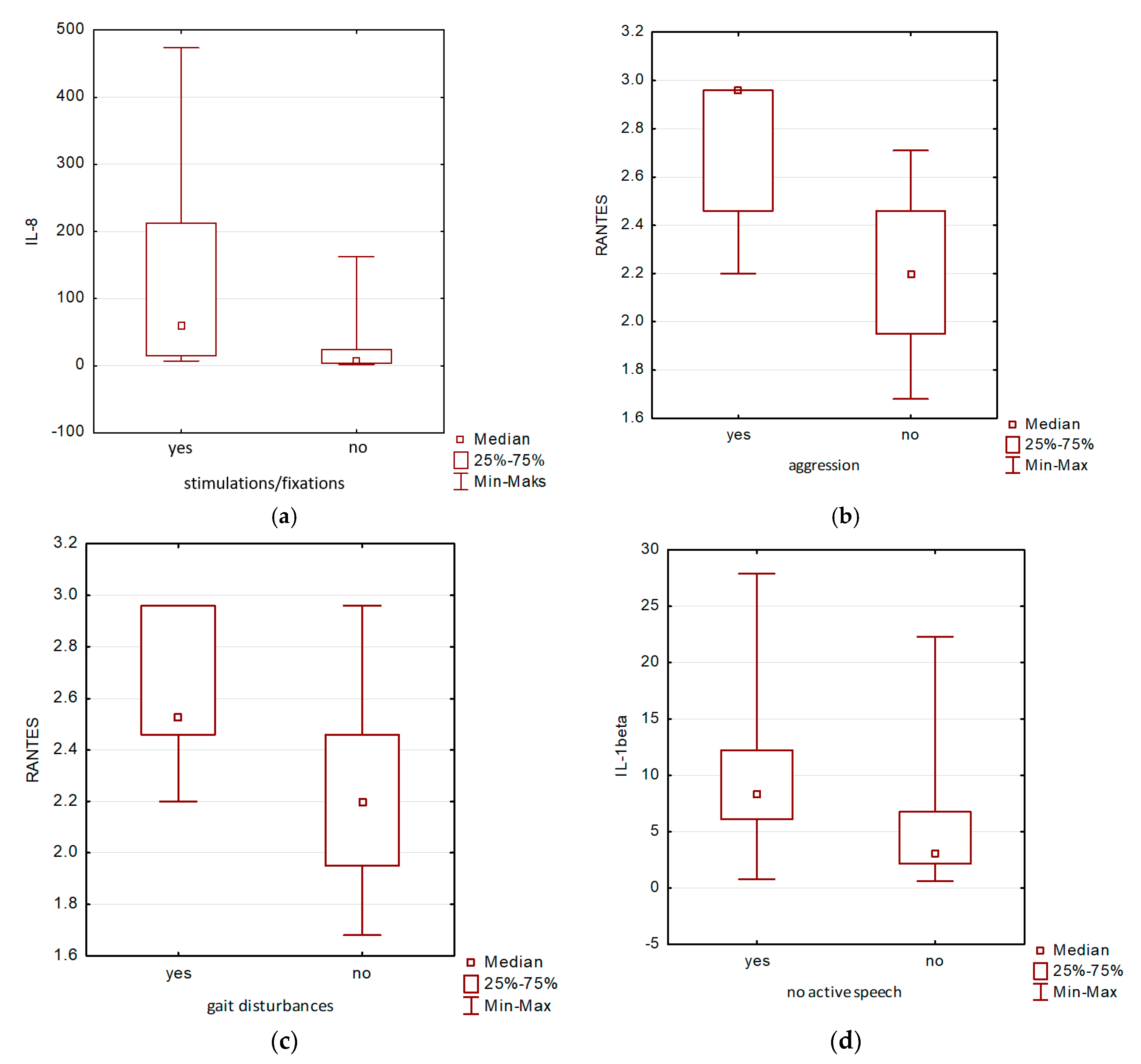
3.3. Correlations between Salivary Levels of Detected Cytokines and Neurodevelopmental Disorders
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Yoon, S.H.; Choi, J.; Lee, W.J.; Do, J.T. Genetic and Epigenetic Etiology Underlying Autism Spectrum Disorder. J. Clin. Med. 2020, 9, 966. [Google Scholar] [CrossRef]
- Gaugler, T.; Klei, L.; Sanders, S.J.; Bodea, C.A.; Goldberg, A.P.; Lee, A.B.; Mahajan, M.; Manaa, D.; Pawitan, Y.; Reichert, J.; et al. Most genetic risk for autism resides with common variation. Nat. Genet. 2014, 46, 881–885. [Google Scholar] [CrossRef] [PubMed]
- Lord, C.; Elsabbagh, M.; Baird, G.; Veenstra-Vanderweele, J. Autism spectrum disorder. Lancet 2018, 392, 508–520. [Google Scholar] [CrossRef]
- Graciarena, M. Cytokines and chemokines in novel roles: Exploring their potential as predictors of autism spectrum disorder. Biol. Psychiatry 2019, 86, e11–e12. [Google Scholar] [CrossRef] [PubMed]
- Hauer, L.S.; Croen, L.A.; Jones, K.L.; Yoshida, C.K.; Hansen, R.L.; Yolken, R.; Zerbo, O.; DeLorenze, G.; Kharrazi, M.; Ashwood, P.; et al. An exploratory examination of neonatal cytokines and chemokines as predictors of autism risk: The early markers for autism study. Biol. Psychiatry 2019, 04, 37. [Google Scholar] [CrossRef] [PubMed]
- Laila, A.A.; Halepoto, D.M. Role of proteomics in the discovery of autism biomarkers. J. Coll. Physicians Surg. Pak. 2013, 23, 137–143. [Google Scholar]
- Saghazadeh, A.; Ataeinia, B.; Keynejad, K.; Abdolalizadeh, A.; Hirbod-Mobarakeh, A.; Rezaei, N. A meta-analysis of pro-inflammatory cytokines in autism spectrum disorders: Effects of age, gender, and latitude. J. Psychiatry Res. 2015, 115, 90–102. [Google Scholar] [CrossRef] [PubMed]
- Masi, A.; Glozier, N.; Dale, R.; Guastella, A.J. The immune system, cytokines, and biomarkers in autism spectrum disorder. Neurosci. Bull. 2017, 33, 194–204. [Google Scholar] [CrossRef] [PubMed]
- Ratajczak, H.V.; Sothern, R.B. Measurement in saliva from neurotypical adults of biomarkers pertinent to autism spectrum disorders. Future Sci. OA 2015, 1, FSO70. [Google Scholar] [CrossRef]
- Walton, E.L. Saliva biomarkers in neurological disorders: A “spitting image” of brain health? Biomed. J. 2018, 41, 59–62. [Google Scholar] [CrossRef]
- Farah, R.; Haraty, H.; Salame, Z.; Fares, Y.; Ojcius, D.M.; Sadier, N.S. Salivary biomarkers for the diagnosis and monitoring of neurological diseases. Biomed. J. 2018, 41, 63–87. [Google Scholar] [CrossRef]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-5), 5th ed.; American Psychiatric Association Publishing: Washington, DC, USA, 2013. [Google Scholar]
- Chojnicka, I.; Pisula, E. Adaptation and Validation of the ADOS-2, Polish Version. Front. Psychol. 2017, 8, 1916. [Google Scholar] [CrossRef] [PubMed]
- Petersen, P.E.; Baez, R.J.; World Health Organization. Oral health Surveys: Basic Methods, 5th ed.; World Health Organization: Cham, Switzerland, 2013. [Google Scholar]
- González-Hernández, J.M.; Franco, L.; Colomer-Poveda, D.; Martinez-Subiela, S.; Cugat, R.; Cerón, J.J.; Márquez, G.; Martínez-Aranda, L.M.; Jimenez-Reyes, P.; Tvarijonaviciute, A. Influence of Sampling Conditions, Salivary Flow, and Total Protein Content in Uric Acid Measurements in Saliva. Antioxidants (Basel) 2019, 8, 389. [Google Scholar] [CrossRef] [PubMed]
- Little, F.F.; Delgado, D.M.; Wexler, F.J.; Oppenheim, F.G.; Mitchell, P.; Feldman, J.A.; Walt, D.R.; Peng, R.D.; Matsui, E.C. Salivary Inflammatory Mediator Profiling and Correlation to Clinical Disease Markers in Asthma. PLoS ONE 2014, 9, e84449. [Google Scholar] [CrossRef] [PubMed]
- Gottschalk, P.G.; Dunn, J.R., II; Brendan Scientific Corporation. Fitting Brendan’s Five-Parameter Logistic Curve; Bio-Plex® Suspension Array System; Bio-RadLaboratories, Inc.: Carlsbad, CA, USA, 2007. [Google Scholar]
- Davis, D.; Zhang, A.; Etienne, C.; Huang, I.; Malit, M. Principles of Curve Fitting for Multiplex Sandwich Immunoassays; Bio-Plex® Suspension Array System; Bio-RadLaboratories, Inc.: Carlsbad, CA, USA, 2007. [Google Scholar]
- Ashwood, P.; Krakowiak, P.; Hertz-Picciotto, I.; Hansen, R.; Pessah, I.N.; Van de Water, J. Altered T cell responses in children with autism. Brain Behav. Immun. 2011, 25, 840–849. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Fernandez, A.; de la Torre-Aguilar, M.; Gil-Campos, M.; Flores-Rojas, K.; Cruz-Rico, M.D.; Martin-Borreguero, P.; Perez-Navero, J.L. Children with autism spectrum disorder with regression exhibit a different profile in plasma cytokines and adhesion molecules compared to children without such regression. Front. Pediatr. 2018, 6, 264. [Google Scholar] [CrossRef]
- Manzardo, A.M.; Henkhaus, R.; Dhillon, S.; Butler, M.G. Plasma cytokine levels in children with autistic disorder and unrelated siblings. Int. J. Dev. Neurosci. 2012, 30, 121–127. [Google Scholar] [CrossRef]
- Gładysz, D.; Krzywdzińska, A.; Hozyasz, K.K. Immune Abnormalities in Autism Spectrum Disorder—Could They Hold Promise for Causative Treatment? Mol. Neurobiol. 2018, 55, 6387–6435. [Google Scholar] [CrossRef]
- Xu, N.; Li, X.; Zhong, Y. Inflammatory cytokines: Potential biomarkers of immunologic dysfunction in autism spectrum disorder. Med. Inflamm. 2015. [Google Scholar] [CrossRef]
- Ashwoodd, P.; Nguyen, D.V.; Hessl, D.; Hagerman, R.J.; Tassone, F. Plasma cytokine profiles in Fragile X subjects: Is there a role for cytokines in the pathogenesis? Brain Behav. Immun. 2010, 24, 898–902. [Google Scholar] [CrossRef]
- Abruzzo, P.M.; Matté, A.; Bolotta, A.; Federe, E.; Ghezzo, A.; Guarnieri, T.; Marini, M.; Posar, A.; Siciliano, A.; De Franceschi, L.; et al. Plasma peroxiredoxin changes and inflammatory cytokines suport the involvement of neuro-inflammation and oxidative stress in autism spectrum disorder. BMC 2019, 17, 332. [Google Scholar] [CrossRef]
- Abdallah, M.W.; Larsen, N.; Mortensen, E.L.; Atladóttir, H.Ó.; Nørgaard-Pedersen, B.; Bonefeld-Jørgensen, E.C.; Grove, J.; Hougaard, D.M. Neonatal levels of cytokines and risk of autism spectrum disorders: An exploratory register-based historic birth cohort study utilizing the Danish Newborn Screening Biobank. J. Neuroimmunol. 2012, 252, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Abdallah, M.W.; Larsen, N.; Grove, J.; Bonefeld-Jørgensen, E.C.; Nørgaard-Pedersen, B.; Hougaard, D.M.; Mortensen, E.L. Neonatal chemokine levels and risk of autism spectrum disorders: Findings from a Danish historic birth cohort follow-up study. Cytokine 2013, 61, 370–376. [Google Scholar] [CrossRef] [PubMed]
- Zafarullah, M.; Tassone, F. Molecular biomarkers in Fragile X syndrome. Brain Sci. 2019, 9, 96. [Google Scholar] [CrossRef]
- AL-Ayadhi, L.Y.; Mostafa, G.A. Elevated serum levels of interleukin-17A in children with autism. J. Neuroinflamm. 2012, 9, 158. [Google Scholar] [CrossRef]
- Olsen, I.; Hicks, S.D. Oral microbiota and autism spectrum disorder (ASD). J. Oral. Microbiol. 2010, 12, 1702806. [Google Scholar] [CrossRef]
- Qiao, Y.; Shi, H.; Wang, H.; Wang, M.; Chen, F. Oral Health Status of Chinese Children with Autism Spectrum Disorders. Front. Psychiatry 2020, 11, 398. [Google Scholar] [CrossRef]
- Qiao, Y.; Wu, M.; Feng, Y.; Zhou, Z.; Chen, L.; Chen, F. Alterations of oral microbiota distinguish children with autism spectrum disorders from healthy controls. Sci. Rep. 2018, 8, 1597. [Google Scholar] [CrossRef]
- Bartolomé-Villar, B.; Mourelle-Martínez, M.R.; Diéguez-Pérez, M.; de Nova-García, M.J. Incidence of oral health in paediatric patients with disabilities: Sensory disorders and autism spectrum disorder. Systematic review II. J. Clin. Exp. Dent. 2016, 8, e344–e351. [Google Scholar] [CrossRef]
- Ashwood, P.; Anthony, A.; Torrente, F.; Wakefield, A.W. Spontaneous Mucosal Lymphocyte Cytokine Profiles in Children with Autism and Gastrointestinal Symptoms: Mucosal Immune Activation and Reduced Counter Regulatory Interleukin-10. J. Clin. Immunol. 2004, 24, 664–673. [Google Scholar] [CrossRef]
- Rogers, G.B.; Keating, D.J.; Young, R.L.; Wong, M.L.; Licinio, J.; Wesselingh, S. From gut dysbiosis to altered brain function and mental illness: Mechanisms and pathways. Mol. Psychiatry 2016, 21, 738–748. [Google Scholar] [CrossRef] [PubMed]
- Jyonouchi, H.; Geng, L.; Davidow, A.L. Cytokine Profiles by Peripheral Blood Monocytes Are Associated With Changes in Behavioral Symptoms Following Immune Insults in a Subset of ASD Subjects: An Inflammatory Subtype? J. Neuroinflamm. 2014, 11, 1–13. [Google Scholar] [CrossRef]
- Roi, A.; Rusu, L.C.; Roi, C.I.; Luca, R.E.; Boia, S.; Munteanu, R.I. A New Approach for the Diagnosis of Systemic and Oral Diseases Based on Salivary Biomolecules. Dis. Markers 2019, 8761860. [Google Scholar] [CrossRef]
- Decker, M.L.; Gotta, V.; Wellmann, S.; Ritz, N. Cytokine profiling in healthy children shows association of age with cytokine concentrations. Sci. Rep. 2017, 7, 17842. [Google Scholar] [CrossRef] [PubMed]
- Decker, M.L.; Grobusch, M.P.; Ritz, N. Influence of Age and Other Factors on Cytokine Expression Profiles in Healthy Children-A Systematic Review. Front. Pediatr. 2017, 5, 255. [Google Scholar] [CrossRef] [PubMed]
- Kleiner, G.; Marcuzzi, A.; Zanin, V.; Monasta, L.; Zauli, G. Cytokine levels in the serum of healthy subjects. Mediat. Inflamm. 2013, 2013, 434010. [Google Scholar] [CrossRef] [PubMed]
- Masi, A.; Breen, E.J.; Alvares, G.A.; Glozier, N.; Hickie, I.B.; Hunt, A.; Hui, J.; Beilby, J.; Ravine, D.; Wray, J.; et al. Cytokine levels and associations with symptom severity in male and female children with autism spectrum disorder. Mol. Autism. 2017, 8, 63. [Google Scholar] [CrossRef] [PubMed]
- Bjørklund, G.; Saad, K.; Chirumbolo, S.; Kern, J.K.; Geier, D.; Geier, M.R.; Urbina, M.A. Immune dysfunction and neuroinflammation in autism spectrum disorder. Acta Neurobiol. Exp. 2016, 76, 257–268. [Google Scholar] [CrossRef]
- Estes, M.L.; McAllister, A.K. Immune mediators in the brain and peripheral tissues in autism spectrum disorder. Nat. Rev. Neurosci. 2015, 16, 469–486. [Google Scholar] [CrossRef]
- Ashwood, P.; Krakowiak, P.; Hertz-Picciotto, I.; Hansen, R.; Pessah, I.N.; Van de Water, J. Associations of impaired behaviors with elevated plasma chemokines in autism spectrum disorders. J. Neuroimmunol. 2011, 232, 196–199. [Google Scholar] [CrossRef]
- Ashwood, P.; Krakowiak, P.; Hertz-Picciotto, I.; Hansen, R.; Pessah, I.; Van de Water, J. Elevated plasma cytokines in autism spectrum disorders provide evidence of immune dysfunction and are associated with impaired behavioral outcome. Brain Behav. Immun. 2011, 25, 40–45. [Google Scholar]
- Han, Y.M.; Cheung, W.K.; Wong, C.K.; Sze, S.L.; Cheng, T.W.; Yeung, M.K.; Chan, A.S. Distinct cytokine and chemokine profiles in autism spectrum disorders. Front. Immunol. 2017, 8, 11. [Google Scholar] [CrossRef] [PubMed]
- Enstrom, A.M.; Onore, C.E.; Van de Water, J.A.; Ashwood, P. Differential monocyte responses to TLR ligands in children with autism spectrum disorders. Brain Behav. Immun. 2010, 24, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Emanuele, E.; Orsi, P.; Boso, M.; Broglia, D.; Brondino, N.; Barale, F.; di Nemi, S.U.; Politi, P. Low-grade endotoxemia in patients with severe autism. Neurosci. Lett. 2010, 471, 162–165. [Google Scholar] [CrossRef]
- Balestrieri, E.; Cipriani, C.; Matteucci, C.; Benvenuto, A.; Coniglio, A.; Agraw-Denboba, A.; Toschi, N.; Bucci, I.; Miele, M.T.; Grelli, S.; et al. Children with autism spectrum disorder and their mothers share abnormal expression of selected endogenous retroviruses families and cytokines. Front. Immun. 2019, 10, 2244. [Google Scholar]
- Delwel, S.; Binnekade, T.T.; Perez, R.S.G.M.; Hertogh, C.M.P.M.; Scherder, E.J.A.; Lobbezoo, F. Oral hygiene and oral health in older people with dementia: A comprehensive review with focus on oral soft tissues. Clin. Oral. Investig. 2018, 22, 93–108. [Google Scholar] [PubMed]
- Aragón, F.; Zea-Sevilla, M.A.; Montero, J.; Sancho, P.; Corral, R.; Tejedor, C.; Frades-Payo, B.; Paredes-Gallardo, V.; Albaladejo, A. Oral health in Alzheimer’s disease: A multicenter case-control study. Clin. Oral. Investig. 2018, 22, 3061–3070. [Google Scholar]
- Tvarijonaviciute, A.; Zamora, C.; Ceron, J.J.; Bravo-Cantero, A.F.; Pardo-Marin, L.; Valverde, S.; Lopez- Jornet, P. Salivary biomarkers in Alzheimer’s disease. Clin. Oral. Investig. 2020, 24, 3437–3444. [Google Scholar]
| Cytokine | Shapiro-Wilk Test p-Value | |
|---|---|---|
| ASD (n = 19) | TD (n = 19) | |
| IL-1β | 0.0149 | 0.0002 |
| IL-6 | 0.0001 | <0.0001 |
| IL-8 | 0.0009 | <0.0001 |
| Eotaxin | <0.0001 | 0.0838 |
| MCP-1 | 0.0677 | <0.0001 |
| RANTES | 0.3889 | 0.5419 |
| TNFα | 0.0008 | 0.0005 |
| Clinical, Demographic and Laboratory Data | Autism Spectrum Disorder (ASD) | Typically Developing (TDs) | p |
|---|---|---|---|
| Number of individuals, n | 19 | 19 | |
| Age (y) mean (±SD) | 6.78 (±2.80) | 6.84 (±2.52) | 0.7123 a |
| Gender, n (Female/Male) | 1/18 | 4/15 | 0.3397 b |
| Vision disturbances, n yes/no | 12/7 | 0/4 | 0.0023 c |
| Self-injury, n yes/no | 4/15 | 1/3 | 0.2253 c |
| Aggression, | 0.0323 c | ||
| 1n yes/rather yes | 0/2 | 2/0 | |
| 1 no/rather no | 13/2 | 2/0 | |
| Fixations simulations, | 0.0016 c | ||
| n yes/rather yes | 13/3 | 0/0 | |
| n no/rather no | 1/2 | 4/0 | |
| Gait disturbance, | <1.0000 c | ||
| n yes/rather yes | 2/3 | 1/0 | |
| n no/rather no | 12/2 | 3/0 | |
| Echolalia, | 0.1246 c | ||
| n yes/rather yes | 9/1 | 0/0 | |
| n no/rather no | 6/3 | 4/0 | |
| Sleeping disturbance, | 0.7431 c | ||
| n yes/rather yes | 2/1 | 0/0 | |
| n no/rather no | 11/5 | 4/0 | |
| Defecation disturbance, | 0.5138 c | ||
| n yes/rather yes | 1/1 | 0/0 | |
| n no/rather no | 11/6 | 4/0 | |
| Food selectivity, | 0.7976 c | ||
| n yes/rather yes | 6/4 | 2/0 | |
| n no/rather no | 6/3 | 2/0 | |
| Auditory hypersensitivity, | 0.1286 c | ||
| n yes/rather yes | 8/4 | 1/0 | |
| n no/rather no | 3/4 | 3/0 | |
| Olfactory hypersensitivity, | 0.7945 c | ||
| n yes/rather yes | 2/1 | 1/0 | |
| n no/rather no | 12/4 | 3/0 | |
| No active speech, | 0.2298 c | ||
| n yes/rather yes | 8/2 | 0/0 | |
| n no/rather no | 7/2 | 4/0 | |
| Tantrums of hysteria, crying, | 0.4814 c | ||
| n yes/rather yes | 6/4 | 2/0 | |
| n no/rather no | 4/5 | 2/0 | |
| Repetitive, stereotyped behaviors, | 0.6224 c | ||
| n yes/rather yes | 4/4 | 0/0 | |
| n no/rather no | 8/3 | 3/1 |
| Dental Data. | Autism Spectrum Disorder (ASD) | p | Typically Developing (TDs) |
|---|---|---|---|
| Previous contact with a dentist, n yes/no | 9/10 | 0.0911 a | 15/4 |
| Constant contact with a dentist, n yes/no | 2/17 | 0.0018 a | 12/7 |
| Number of teeth with caries, n yes/no, | 12/7 | 5/14 | |
| mean (±SD) | 2.73 (±3.19) | 0.0505 b | 1.42 (±3.04) |
| Oral hygiene, n good/bad | 7/12 | 0.5148 a | 10/9 |
| Presence of oral plaque, n yes/no | 6/13 | 1.0000 a | 7/12 |
| Presence of orthodontic defect, n yes/no | 2/17 | 0.6598 a | 4/15 |
| Presence of caries, n yes/no | 12/7 | 0.0488 a | 5/14 |
| Simplified Oral Hygiene Index (OHI-S) | |||
| mean (±SD) | 0.73 (±1.15) | 0.9312 b | 0.52 (±0.88) |
| median (a 25–75% confidence interval) | 0.00 (0.00–1.83) | 0.00 (0.00–0.83) | |
| Decayed Missing Filled Tooth (DMFT) index, | |||
| mean (±SD) | Dmft c 2.64 (±3.01) | 0.1975 b | Dmft c 2.00 (±3.59) |
| median (a 25%-75% confidence interval) | DMFTdmft d 2.89 (±3.22) | 0.1298 b | DMFTdmft d 2.00 (±3.59) |
| Dmft c 2.00 (0.00–4.00) | Dmft c 0.00 (0.00–3.00) | ||
| DMFTdmft d 2.00 (0.00–5.00) | DMFTdmft d 0.00 (0.00–3.00) |
| Cytokines | ASD (n = 19) Median (a 25–75% Confidence Interval) | TDs (n = 19) Median (a 25–75% Confidence Interval) | p |
|---|---|---|---|
| IL-1β (pg/mL) | 6.84 (3.84–12.2) | 2.98 (1.04–16.56) | 0.3972 b |
| IL-6 (pg/mL) | 0.67 (0.25–2.16) | 0.44 (0.25–2.11) | 0.5885 b |
| IL-8 (pg/mL) | 55.36 (15.02–162.75) | 47.59 (7.29–117.09) | 0.6300 b |
| Eotaxin (pg/mL) | 0.11 (0.08–0.15) | 0.08. (0.05–0.16) | 0.3248 b |
| RANTES (µg/mL) | 2.46 (2.20–2.59) | 2.46 (2.46–2.84) | 0.0314 a |
| MCP-1 (pg/mL) | 5.43 (3.77–10.39) | 7.19 (3.00–17.54) | 0.5592 b |
| TNFα (pg/mL) | 6.61 (2.06–21.48) | 2.06 (0.54–12.24) | 0.1018 b |
| Age & | n | rs | p | |
| IL-1β | 19 | −0.03 | 0.899 | |
| IL-6 | 19 | –0.14 | 0.563 | |
| IL-8 | 19 | –0.18 | 0.471 | |
| Eotaxin | 19 | –0.16 | 0.523 | |
| MCP-1 | 19 | –0.05 | 0.853 | |
| RANTES | 19 | 0.05 | 0.840 | |
| TNFα | 19 | 0.15 | 0.545 |
| Age & | n | rs | p | |
| IL-1β | 19 | 0.71 | <0.001 | |
| IL-6 | 19 | 0.19 | 0.438 | |
| IL-8 | 19 | 0.40 | 0.089 | |
| Eotaxin | 19 | 0.42 | 0.071 | |
| MCP-1 | 19 | 0.63 | 0.004 | |
| RANTES | 19 | 0.27 | 0.271 | |
| TNFα | 19 | 0.48 | 0.036 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Samborska-Mazur, J.; Kostiukow, A.; Miechowicz, I.; Sikorska, D.; Rutkowski, R.; Wyganowska-Świątkowska, M.; Błochowiak, K. Salivary Cytokine Profile as a Possible Predictor of Autism Spectrum Disorder. J. Clin. Med. 2020, 9, 3101. https://doi.org/10.3390/jcm9103101
Samborska-Mazur J, Kostiukow A, Miechowicz I, Sikorska D, Rutkowski R, Wyganowska-Świątkowska M, Błochowiak K. Salivary Cytokine Profile as a Possible Predictor of Autism Spectrum Disorder. Journal of Clinical Medicine. 2020; 9(10):3101. https://doi.org/10.3390/jcm9103101
Chicago/Turabian StyleSamborska-Mazur, Joanna, Anna Kostiukow, Izabela Miechowicz, Dorota Sikorska, Rafał Rutkowski, Marzena Wyganowska-Świątkowska, and Katarzyna Błochowiak. 2020. "Salivary Cytokine Profile as a Possible Predictor of Autism Spectrum Disorder" Journal of Clinical Medicine 9, no. 10: 3101. https://doi.org/10.3390/jcm9103101
APA StyleSamborska-Mazur, J., Kostiukow, A., Miechowicz, I., Sikorska, D., Rutkowski, R., Wyganowska-Świątkowska, M., & Błochowiak, K. (2020). Salivary Cytokine Profile as a Possible Predictor of Autism Spectrum Disorder. Journal of Clinical Medicine, 9(10), 3101. https://doi.org/10.3390/jcm9103101






