Virus-Induced Asthma/Wheeze in Preschool Children: Longitudinal Assessment of Airflow Limitation Using Impulse Oscillometry
Abstract
1. Introduction
2. Methods
2.1. Study Population
2.2. Study Design
2.3. Allergic Sensitization
2.4. Lung Function Test Maneuvers
2.5. Statistical Analysis
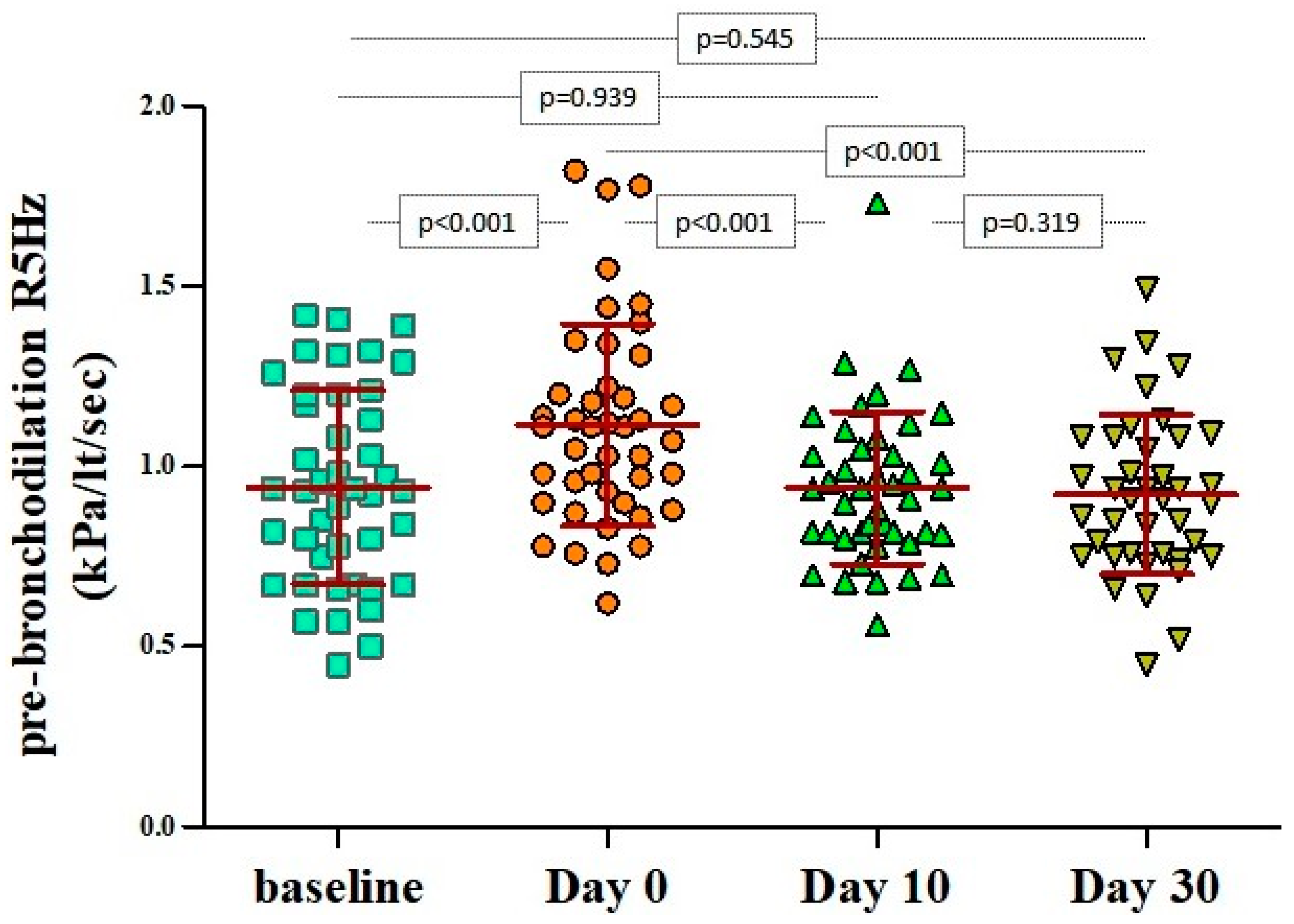
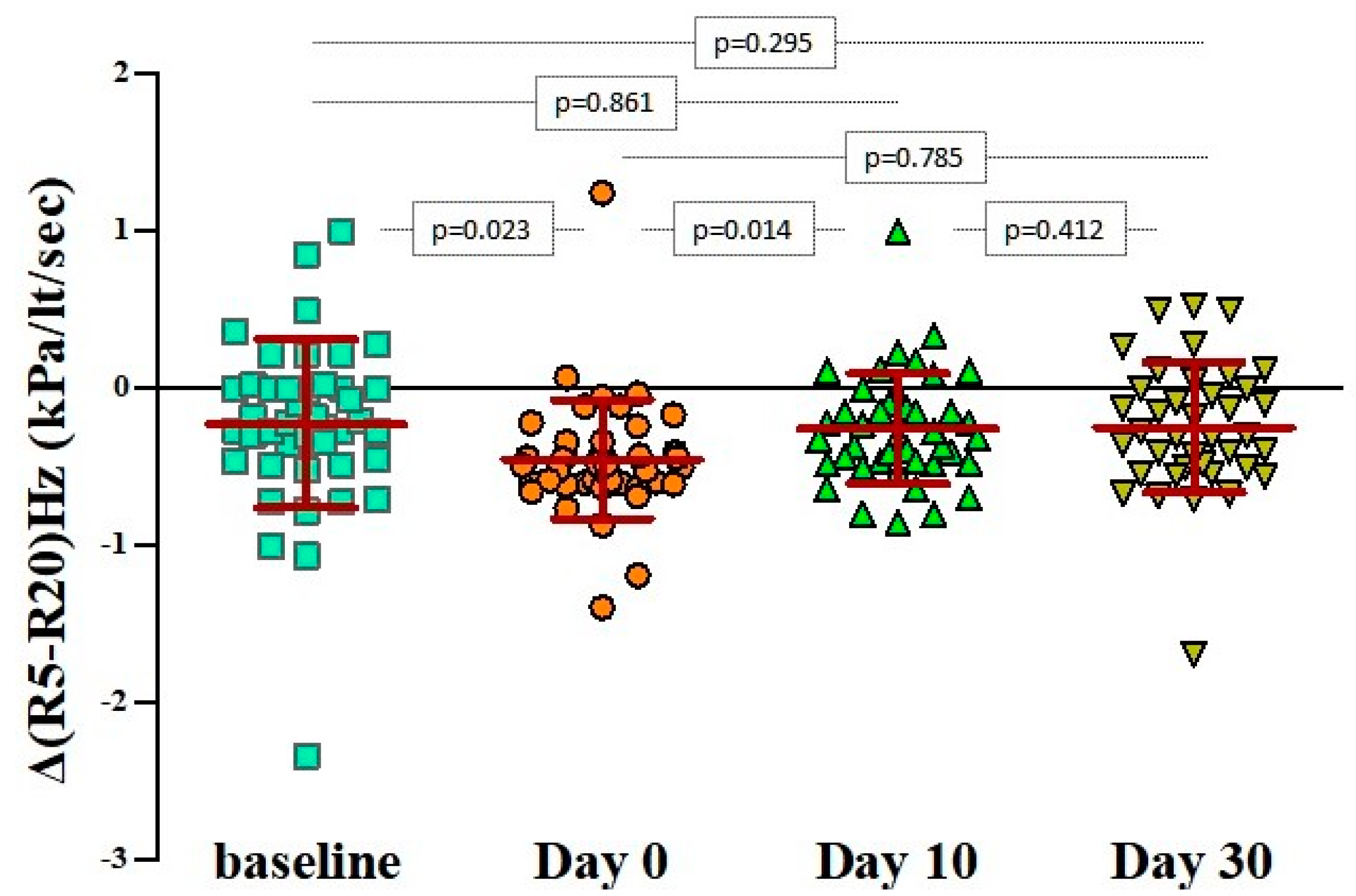
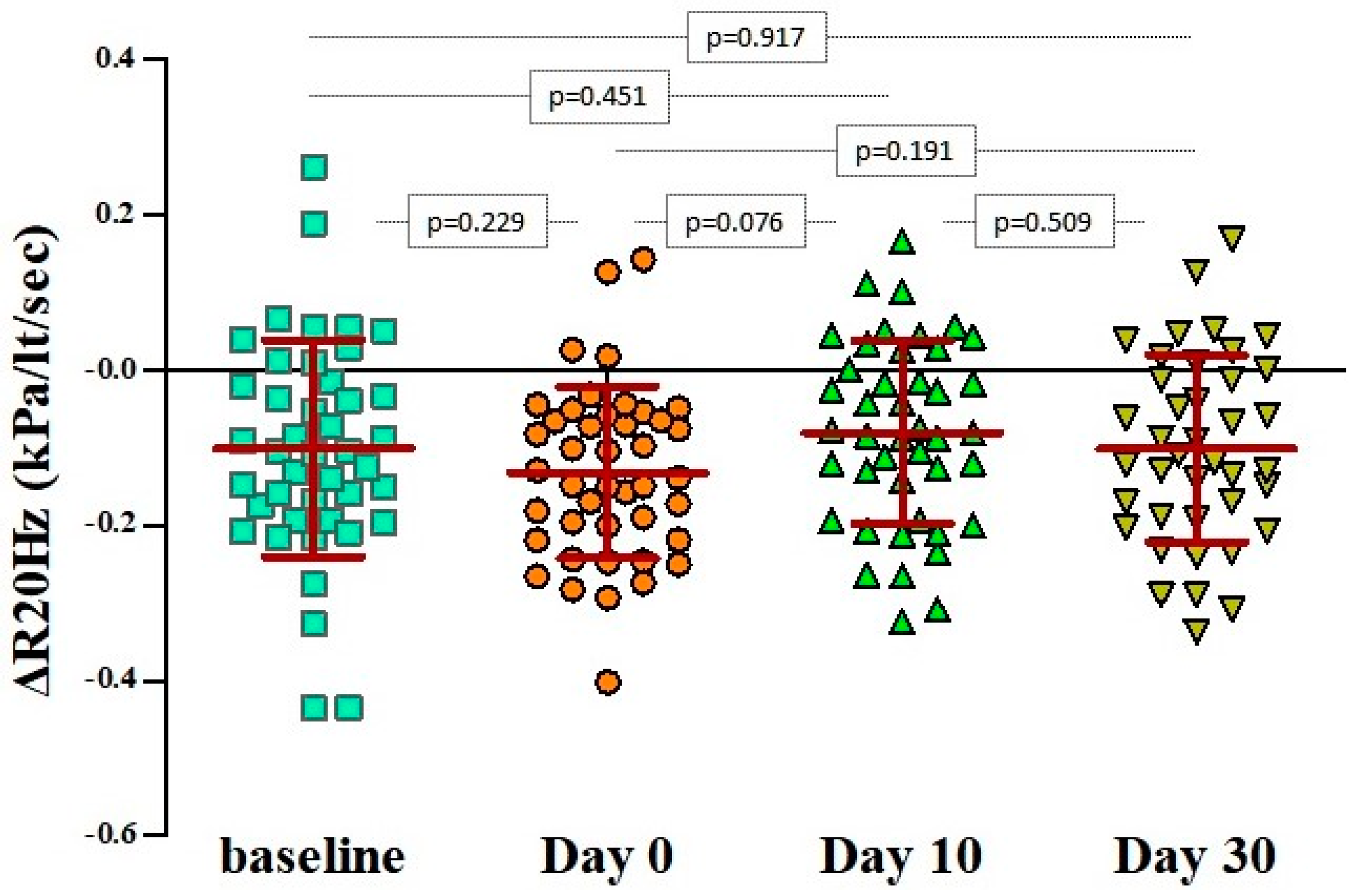
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Global Initiative for Asthma. Global Strategy for Asthma Management and Prevention. 2019. Available online: www.ginasthma.org (accessed on 1 June 2019).
- Bickel, S.; Popler, J.; Lesnick, B.; Eid, N. Impulse oscillometry: Interpretation and practical applications. Chest 2014, 146, 841–847. [Google Scholar] [CrossRef] [PubMed]
- Meraz, E.G.; Nazeran, H.; Ramos, C.D.; Nava, P.; Diong, B.; Goldman, M.D.; Goldman, C.A. Analysis of impulse oscillometric measures of lung function and respiratory system model parameters in small airway-impaired and healthy children over a 2-year period. Biomed. Eng. Online 2011, 10, 21. [Google Scholar] [CrossRef] [PubMed]
- Song, T.W.; Kim, K.W.; Kim, E.S.; Park, J.W.; Sohn, M.H.; Kim, K.E. Utility of impulse oscillometry in young children with asthma. Pediatric Allergy Immunol. Off. Publ. Eur. Soc. Pediatric Allergy Immunol. 2008, 19, 763–768. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Aledia, A.S.; Tatavoosian, A.V.; Vijayalakshmi, S.; Galant, S.P.; George, S.C. Relating small airways to asthma control by using impulse oscillometry in children. J. Allergy Clin. Immunol. 2012, 129, 671–678. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Aledia, A.S.; Galant, S.P.; George, S.C. Peripheral airway impairment measured by oscillometry predicts loss of asthma control in children. J. Allergy Clin. Immunol. 2013, 131, 718–723. [Google Scholar] [CrossRef] [PubMed]
- Komarow, H.D.; Skinner, J.; Young, M.; Gaskins, D.; Nelson, C.; Gergen, P.J.; Metcalfe, D.D. A study of the use of impulse oscillometry in the evaluation of children with asthma: analysis of lung parameters, order effect, and utility compared with spirometry. Pediatric Pulmonol. 2012, 47, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, K.; Fausto, L.L.; Camargos, P.A.M.; Kviecinski, M.R.; da Silva, J. Impulse oscillometry in the assessment of asthmatic children and adolescents: From a narrative to a systematic review. Paediatr. Respir. Rev. 2017, 23, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Frei, J.; Jutla, J.; Kramer, G.; Hatzakis, G.E.; Ducharme, F.M.; Davis, G.M. Impulse oscillometry: Reference values in children 100 to 150 cm in height and 3 to 10 years of age. Chest 2005, 128, 1266–1273. [Google Scholar] [CrossRef] [PubMed]
- Klug, B.; Bisgaard, H. Specific airway resistance, interrupter resistance, and respiratory impedance in healthy children aged 2–7 years. Pediatric Pulmonol. 1998, 25, 322–331. [Google Scholar] [CrossRef]
- Hellinckx, J.; De Boeck, K.; Bande-Knops, J.; van der Poel, M.; Demedts, M. Bronchodilator response in 3–6.5 years old healthy and stable asthmatic children. Eur. Respir. J. 1998, 12, 438–443. [Google Scholar] [CrossRef]
- Thamrin, C.; Gangell, C.L.; Udomittipong, K.; Kusel, M.M.; Patterson, H.; Fukushima, T.; Schultz, A.; Hall, G.L.; Stick, S.M.; Sly, P.D. Assessment of bronchodilator responsiveness in preschool children using forced oscillations. Thorax 2007, 62, 814–819. [Google Scholar] [CrossRef]
- Mansur, A.H.; Manney, S.; Ayres, J.G. Methacholine-induced asthma symptoms correlate with impulse oscillometry but not spirometry. Respir. Med. 2008, 102, 42–49. [Google Scholar] [CrossRef]
- Kalliola, S.; Malmberg, L.P.; Kajosaari, M.; Mattila, P.S.; Pelkonen, A.S.; Mäkelä, M.J. Assessing direct and indirect airway hyperresponsiveness in children using impulse oscillometry. Ann. Allergy Asthma Immunol. Off. Publ. Am. Coll. Allergy Asthma Immunol. 2014, 113, 166–172. [Google Scholar] [CrossRef] [PubMed]
- Brand, P.L.; Baraldi, E.; Bisgaard, H.; Boner, A.L.; Castro-Rodriguez, J.A.; Custovic, A.; de Blic, J.; de Jongste, J.C.; Eber, E.; Everard, M.L.; et al. Definition, assessment and treatment of wheezing disorders in preschool children: An evidence-based approach. Eur. Respir. J. 2008, 32, 1096–1110. [Google Scholar] [CrossRef]
- Bacharier, L.B.; Boner, A.; Carlsen, K.H.; Eigenmann, P.A.; Frischer, T.; Götz, M.; Helms, P.J.; Hunt, J.; Liu, A.; Papadopoulos, N.; et al. Diagnosis and treatment of asthma in childhood: A PRACTALL consensus report. Allergy 2008, 63, 5–34. [Google Scholar] [CrossRef] [PubMed]
- Beydon, N.; Davis, S.D.; Lombardi, E.; Allen, J.L.; Arets, H.G.; Aurora, P.; Bisgaard, H.; Davis, G.M.; Ducharme, F.M.; Eigen, H.; et al. An official American Thoracic Society/European Respiratory Society statement: Pulmonary function testing in preschool children. Am. J. Respir. Crit. Care Med. 2007, 175, 1304–1345. [Google Scholar] [CrossRef] [PubMed]
- Konstantinou, G.N.; Xepapadaki, P.; Manousakis, E.; Makrinioti, H.; Kouloufakou-Gratsia, K.; Saxoni-Papageorgiou, P.; Papadopoulos, N.G. Assessment of airflow limitation, airway inflammation, and symptoms during virus-induced wheezing episodes in 4- to 6-year-old children. J. Allergy Clin. Immunol. 2013, 131, 87–93.e1-5. [Google Scholar] [CrossRef]
- Oostveen, E.; MacLeod, D.; Lorino, H.; Farré, R.; Hantos, Z.; Desager, K.; Marchal, F. The forced oscillation technique in clinical practice: Methodology, recommendations and future developments. Eur. Respir. J. 2003, 22, 1026–1041. [Google Scholar] [CrossRef]
- Dencker, M.; Malmberg, L.P.; Valind, S.; Thorsson, O.; Karlsson, M.K.; Pelkonen, A.; Pohjanpalo, A.; Haahtela, T.; Turpeinen, M.; Wollmer, P. Reference values for respiratory system impedance by using impulse oscillometry in children aged 2–11 years. Clin. Physiol. Funct. Imaging 2006, 26, 247–250. [Google Scholar] [CrossRef]
- Nowowiejska, B.; Tomalak, W.; Radliński, J.; Siergiejko, G.; Latawiec, W.; Kaczmarski, M. Transient reference values for impulse oscillometry for children aged 3–18 years. Pediatric Pulmonol. 2008, 43, 1193–1197. [Google Scholar] [CrossRef]
- Lándsér, F.J.; Nagles, J.; Demedts, M.; Billiet, L.; van de Woestijne, K.P. A new method to determine frequency characteristics of the respiratory system. J. Appl. Physiol. 1976, 41, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Gochicoa-Rangel, L.; Torre-Bouscoulet, L.; Martínez-Briseño, D.; Rodríguez-Moreno, L.; Cantú-González, G.; Vargas, M.H. Values of impulse oscillometry in healthy Mexican children and adolescents. Respir. Care 2015, 60, 119–127. [Google Scholar] [CrossRef]
- Knihtilä, H.; Kotaniemi-Syrjänen, A.; Pelkonen, A.S.; Kalliola, S.; Mäkelä, M.J.; Malmberg, L.P. Small airway oscillometry indices: Repeatability and bronchodilator responsiveness in young children. Pediatric Pulmonol. 2017, 52, 1260–1267. [Google Scholar] [CrossRef] [PubMed]
- Clément, J.; Dumoulin, B.; Gubbelmans, R.; Hendriks, S.; van de Woestijne, K.P. Reference values of total respiratory resistance and reactance between 4 and 26 Hz in children and adolescents aged 4–20 years. Bull. Eur. De Physiopathol. Respir. 1987, 23, 441–448. [Google Scholar]
- De Assumpcao, M.S.; da Silva Goncalves, E.; Oliveira, M.S.; Ribeiro, J.D.; Dalbo Contrera Toro, A.A.; de Azevedo Barros-Filho, A.; de Monteiro, M.; Santos Schivinski, C.I. Impulse Oscillometry System and Anthropometric Variables of Preschoolers, Children and Adolescents Systematic Review. Curr. Pediatric Rev. 2017, 13, 126–135. [Google Scholar] [CrossRef] [PubMed]
- Xepapadaki, P.; Bachert, C.; Finotto, S.; Jartti, T.; Konstantinou, G.N.; Kiefer, A.; Kowalski, M.; Lewandowska-Polak, A.; Lukkarinen, H.; Roumpedaki, E.; et al. Contribution of repeated infections in asthma persistence from preschool to school age: Design and characteristics of the PreDicta cohort. Pediatric Allergy Immunol. Off. Publ. Eur. Soc. Pediatric Allergy Immunol. 2018, 29, 383–393. [Google Scholar] [CrossRef] [PubMed]
- Lands, L.C.; Allen, J.; Cloutier, M.; Leigh, M.; McColley, S.; Murphy, T.; Wilfond, B. Pediatric Assembly of American Thoracic Society Subcommittee. ATS Consensus Statement: Research opportunities and challenges in pediatric pulmonology. Am. J. Respir. Crit. Care Med. 2005, 172, 776–780. [Google Scholar] [CrossRef] [PubMed]

| Children with Wheezing Episode * (n = 43) | Children with no Episode † (n = 46) | |
|---|---|---|
| Age (years) | 5 ± 0.5 | 5 ± 0.7 |
| Male, n (%) | 23 (54%) | 20 (44%) |
| Height (m) | 1.15 ± 0.08 | 1.14 ± 0.08 |
| Weight (kg) | 22.9 ± 3.9 | 21.9 ± 4.3 |
| Atopic | 25 (58%) | 21 (46%) |
| Baseline pre-bronchodilation R5Hz (kPa/lt/sec) | 0.943 ± 0.269 | 0.980 ± 0.222 |
| Baseline post-bronchodilation R5Hz (kPa/lt/sec) | 0.817 ± 0.227 | 0.809 ± 0.186 |
| ΔR5Hz | −12% ± 13.5% | −15.8% ± 15.4% |
| Baseline pre-bronchodilation R20Hz (kPa/lt/sec) | 0.757 ± 0.191 | 0.764 ± 0.173 |
| Baseline post-bronchodilation R20Hz (kPa/lt/sec) | 0.669 ± 0.157 | 0.675 ± 0.153 |
| ΔR20Hz | −10.1% ± 13.9% | −11.5% ± 17.2% |
| Previously treated with Bronchodilators alone § | 32 (74.4%) | 32 (69.6%) |
| Atopics | Non-Atopics | |||||
|---|---|---|---|---|---|---|
| R5Hz Bronchodilation (kPa/lt/sec) | Mean ΔR5Hz%* | R5Hz Bronchodilation (kPa/lt/sec) | Mean ΔR5Hz%* | |||
| Time | Pre | Post | Pre | Post | ||
| Baseline | 0.930 ± 0.273 | 0.798 ± 0.230 | −12.8% ± 14.9% | 0.961 ± 0.271 | 0.844 ± 0.227 | −10.8% ± 11.6% |
| Day 0 | 1.106 ± 0.279 | 0.847 ± 0.218 † | −22.5 ± 12.8% ‡ | 1.125 ± 0.289 | 0.818 ± 0.171 † | −25.8% ± 11.1% ‡ |
| Day 10 | 0.959 ± 0.225 † | 0.831 ± 0.218 † | −12.9% ± 13.5% | 0.914 ± 0.192 | 0.803 ± 0.155 † | −11.2% ± 11.3% |
| Day 30 | 0.905 ± 0.248 † | 0.792 ± 0.159 † | 18.6% ± 10.7% | 0.947 ± 0.179 | 0.772 ± 0.191 † | −10.3% ± 12.6% |
| ΔR5Hz (%) | PPV | NPV | Sensitivity | Specificity | Accuracy |
|---|---|---|---|---|---|
| −46.4% | 100.00% | 80.38% | 4.65% | 100.00% | 80.57% |
| −35.1% | 30.77% | 80.30% | 9.30% | 94.64% | 77.25% |
| −31.3% | 32.00% | 81.18% | 18.60% | 89.88% | 75.36% |
| −20.5% | 42.25% | 90.71% | 69.77% | 75.60% | 74.41% |
| −12.4% | 28.57% | 90.22% | 79.07% | 49.40% | 55.45% |
| −8.3% | 26.03% | 92.31% | 88.37% | 35.71% | 46.45% |
| −7.1% | 25.16% | 94.23% | 93.02% | 29.17% | 42.18% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Konstantinou, G.N.; Papadopoulos, N.G.; Manousakis, E.; Xepapadaki, P. Virus-Induced Asthma/Wheeze in Preschool Children: Longitudinal Assessment of Airflow Limitation Using Impulse Oscillometry. J. Clin. Med. 2019, 8, 1475. https://doi.org/10.3390/jcm8091475
Konstantinou GN, Papadopoulos NG, Manousakis E, Xepapadaki P. Virus-Induced Asthma/Wheeze in Preschool Children: Longitudinal Assessment of Airflow Limitation Using Impulse Oscillometry. Journal of Clinical Medicine. 2019; 8(9):1475. https://doi.org/10.3390/jcm8091475
Chicago/Turabian StyleKonstantinou, George N, Nikolaos G Papadopoulos, Emmanouel Manousakis, and Paraskevi Xepapadaki. 2019. "Virus-Induced Asthma/Wheeze in Preschool Children: Longitudinal Assessment of Airflow Limitation Using Impulse Oscillometry" Journal of Clinical Medicine 8, no. 9: 1475. https://doi.org/10.3390/jcm8091475
APA StyleKonstantinou, G. N., Papadopoulos, N. G., Manousakis, E., & Xepapadaki, P. (2019). Virus-Induced Asthma/Wheeze in Preschool Children: Longitudinal Assessment of Airflow Limitation Using Impulse Oscillometry. Journal of Clinical Medicine, 8(9), 1475. https://doi.org/10.3390/jcm8091475





