Long Non-Coding RNAs Modulate Sjögren’s Syndrome Associated Gene Expression and Are Involved in the Pathogenesis of the Disease
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Microarray Analysis
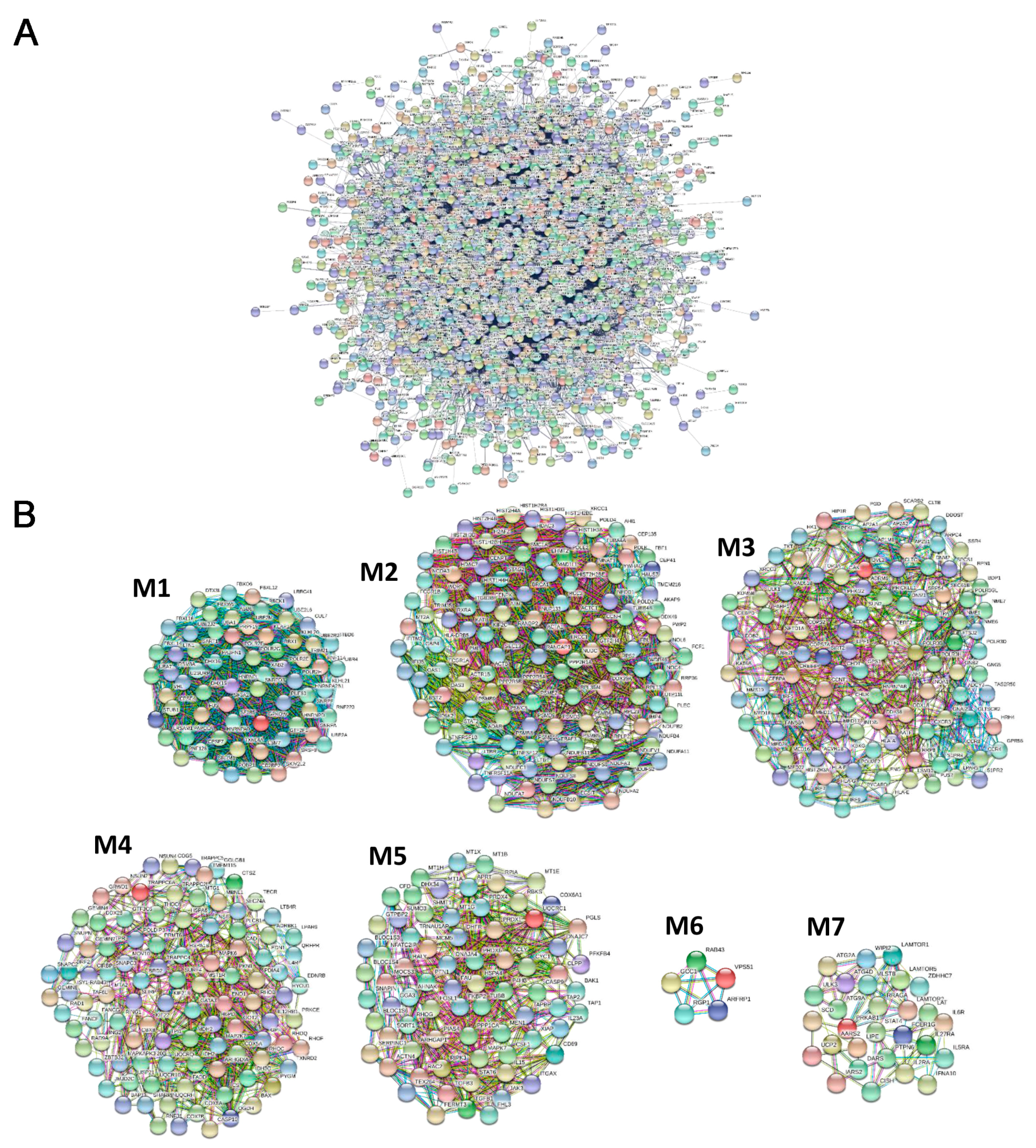
2.3. Protein–Protein Interaction (PPI) Network Construction and Network Clustering
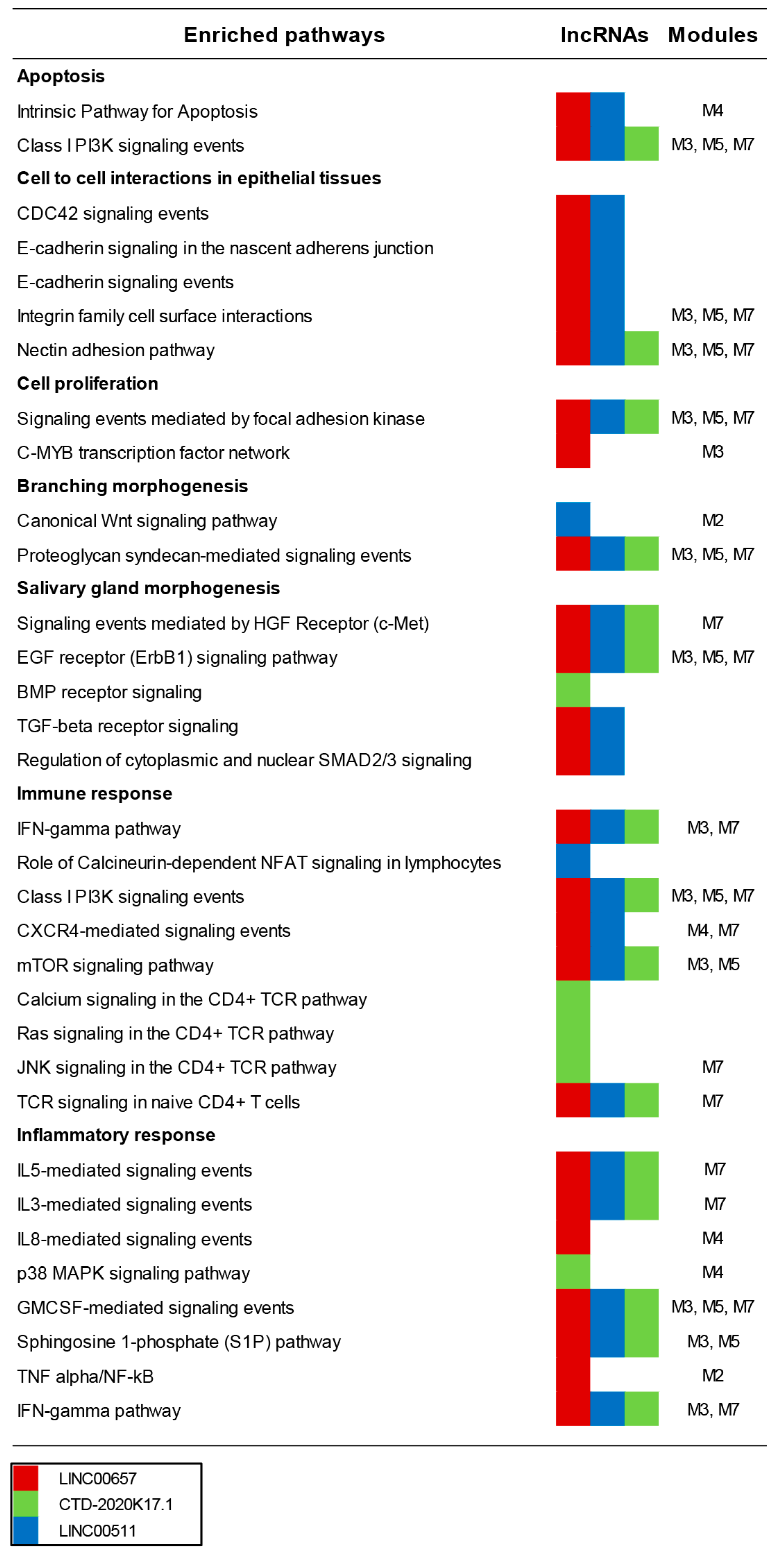
2.4. Gene Functional Classification and Enrichment Analysis
2.5. Real Time PCR of lncRNAs
2.6. Real Time PCR of Genes Modulated in pSjS Patients
2.7. Real Time PCR of microRNAs
3. Results
3.1. Patients Characteristics
3.2. High-Throughput Gene and Long Non-Coding RNA Expression Profiling in Peripheral Blood Mononuclear Cells of Patients with pSjS
3.3. PPI Network and Modular Analysis of Genes and lncRNAs Modulated in pSjS
3.4. Functional Analysis of Modulated Genes Targeted in the Whole Transcriptome
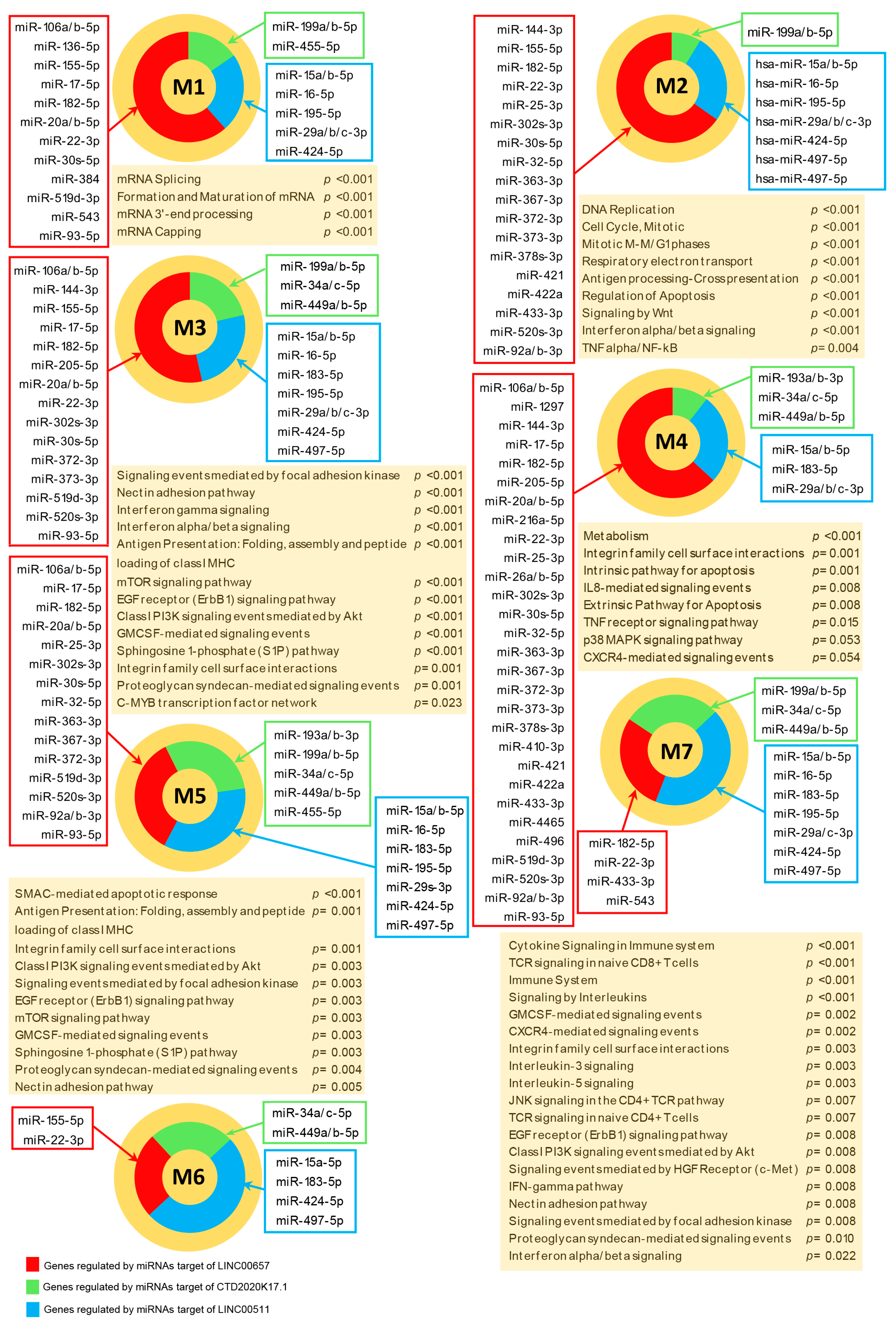
3.5. Functional Analysis of Highly Connected Genes that are Included in Modules
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Maldini, C.; Seror, R.; Fain, O.; Dhote, R.; Amoura, Z.; De Bandt, M.; Delassus, J.L.; Falgarone, G.; Guillevin, L.; Le Guern, V.; et al. Epidemiology of primary Sjögren’s syndrome in a French multiracial/multiethnic area. Arthritis Care Res. 2014, 66, 454–463. [Google Scholar] [CrossRef] [PubMed]
- Mariette, X.; Criswell, L.A. Primary Sjögren’s Syndrome. N. Engl. J. Med. 2018, 378, 931–939. [Google Scholar] [CrossRef] [PubMed]
- Tzioufas, A.G.; Kapsogeorgou, E.K.; Moutsopoulos, H.M. Pathogenesis of Sjögren’s syndrome: What we know and what we should learn. J. Autoimmun. 2012, 39, 4–8. [Google Scholar] [CrossRef] [PubMed]
- Bach, J.F. Infections and autoimmune diseases. J. Autoimmun. 2005, 25, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Shiboski, C.H.; Shiboski, S.C.; Seror, R.; Criswell, L.A.; Labetoulle, M.; Lietman, T.M.; Rasmussen, A.; Scofield, H.; Vitali, C.; Bowman, S.J.; et al. 2016 American College of Rheumatology/European League Against Rheumatism classification criteria for primary Sjögren’s syndrome: A consensus and data-driven methodology involving three international patient cohorts. Ann. Rheum. Dis. 2017, 76, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Li, J.H.; Liu, S.; Zhou, H.; Qu, L.H.; Yang, J.H. StarBase v2.0: Decoding miRNA-ceRNA, miRNA-ncRNA and protein-RNA interaction networks from large-scale CLIP-Seq data. Nucleic Acids Res. 2014, 42, D92–D97. [Google Scholar] [CrossRef] [PubMed]
- Pathan, M.; Keerthikumar, S.; Ang, C.S.; Gangoda, L.; Quek, C.Y.; Williamson, N.A.; Mouradov, D.; Sieber, O.M.; Simpson, R.J.; Salim, A.; et al. Funrich: An open access standalone functional enrichment and interaction network analysis tool. Proteomics 2015, 15, 2597–2601. [Google Scholar] [CrossRef]
- Jensen, L.J.; Kuhn, M.; Stark, M.; Chaffron, S.; Creevey, C.; Muller, J.; Doerks, T.; Julien, P.; Roth, A.; Simonovic, M.; et al. String 8—A global view on proteins and their functional interactions in 630 organisms. Nucleic Acids Res. 2009, 37, D412–D416. [Google Scholar] [CrossRef]
- Cline, M.S.; Smoot, M.; Cerami, E.; Kuchinsky, A.; Landys, N.; Workman, C.; Christmas, R.; Avila-Campilo, I.; Creech, M.; Gross, B.; et al. Integration of biological networks and gene expression data using cytoscape. Nat. Protoc. 2007, 2, 2366–2382. [Google Scholar] [CrossRef]
- Mi, H.; Muruganujan, A.; Casagrande, J.T.; Thomas, P.D. Large-scale gene function analysis with the Panther classification system. Nat. Protoc. 2013, 8, 1551–1566. [Google Scholar] [CrossRef]
- Fisher, B.A.; Jonsson, R.; Daniels, T.; Bombardieri, M.; Brown, R.M.; Morgan, P.; Bombardieri, S.; Ng, W.F.; Tzioufas, A.G.; Vitali, C.; et al. Standardisation of labial salivary gland histopathology in clinical trials in primary Sjogren’s syndrome. Ann. Rheum. Dis. 2017, 76, 1161–1168. [Google Scholar] [CrossRef] [PubMed]
- Seror, R.; Theander, E.; Brun, J.B.; Ramos-Casals, M.; Valim, V.; Dorner, T.; Bootsma, H.; Tzioufas, A.; Solans-Laquè, R.; Mandl, T.; et al. Validation of EULAR primary Sjogren’s syndrome disease activity (ESSDAI) and patient indexes (ESSPRI). Ann. Rheum. Dis. 2015, 74, 859–866. [Google Scholar] [CrossRef] [PubMed]
- Barabási, A.L.; Gulbahce, N.; Loscalzo, J. Network medicine: A network-based approach to human disease. Nat. Rev. Genet. 2011, 12, 56–68. [Google Scholar] [CrossRef] [PubMed]
- Coffre, M.; Koralov, S.B. miRNAs in B Cell Development and Lymphomagenesis. Trends Mol. Med. 2017, 23, 721–736. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Jima, D.D.; Jacobs, C.; Fischer, R.; Gottwein, E.; Huang, G.; Lugar, P.L.; Lagoo, A.S.; Rizzieri, D.A.; Friedman, D.; et al. Patterns of microRNA expression characterize stages of human B-cell differentiation. Blood 2009, 113, 4586–4594. [Google Scholar] [CrossRef]
- Ni, H.; Tong, R.; Zou, L.; Song, G.; Cho, W.C. MicroRNAs in diffuse large B-cell lymphoma. Oncol. Lett. 2016, 11, 1271–1280. [Google Scholar] [CrossRef]
- Wang, H.; Wang, A.; Hu, Z.; Xu, X.; Liu, Z.; Wang, Z. A Critical Role of miR-144 in Diffuse Large B-cell Lymphoma Proliferation and Invasion. Cancer Immunol. Res. 2016, 4, 337–344. [Google Scholar] [CrossRef]
- Zhu, F.Q.; Zeng, L.; Tang, N.; Tang, Y.P.; Zhou, B.P.; Li, F.F.; Wu, W.G.; Zeng, X.B.; Peng, S.S. MicroRNA-155 Downregulation Promotes Cell Cycle Arrest and Apoptosis in Diffuse Large B-Cell Lymphoma. Oncol. Res. 2016, 24, 415–427. [Google Scholar] [CrossRef]
- Hershkovitz-Rokah, O.; Geva, P.; Salmon-Divon, M.; Shpilberg, O.; Liberman-Aronov, S. Network analysis of microRNAs, genes and their regulation in diffuse and follicular B-cell lymphomas. Oncotarget 2018, 9, 7928–7941. [Google Scholar] [CrossRef]
- Arakawa, F.; Kimura, Y.; Yoshida, N.; Miyoshi, H.; Doi, A.; Yasuda, K.; Nakajima, K.; Kiyasu, J.; Niino, D.; Sugita, Y.; et al. Identification of miR-15b as a transformation-related factor in mantle cell lymphoma. Int. J. Oncol. 2016, 48, 485–492. [Google Scholar] [CrossRef]
- Borges, N.M.; do Vale Elias, M.; Fook-Alves, V.L.; Andrade, T.A.; de Conti, M.L.; Macedo, M.P.; Begnami, M.D.; Campos, A.H.; Etto, L.Y.; Bortoluzzo, A.B.; et al. Angiomirs expression profiling in diffuse large B-Cell lymphoma. Oncotarget 2016, 26, 4806–4816. [Google Scholar] [CrossRef] [PubMed]
- Di Lisio, L.; Sánchez-Beato, M.; Gómez-López, G.; Rodríguez, M.E.; Montes-Moreno, S.; Mollejo, M.; Menárguez, J.; Martínez, M.A.; Alves, F.J.; Pisano, D.G.; et al. MicroRNA signatures in B-cell lymphomas. Blood Cancer J. 2012, 2, e57. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Ushmorov, A.; Leithäuser, F.; Guan, H.; Steidl, C.; Färbinger, J.; Pelzer, C.; Vogel, M.J.; Maier, H.J.; Gascoyne, R.D.; et al. FOXO1 is a tumor suppressor in classical Hodgkin lymphoma. Blood 2012, 119, 3503–3511. [Google Scholar] [CrossRef] [PubMed]
- Caramuta, S.; Lee, L.; Ozata, D.M.; Akçakaya, P.; Georgii-Hemming, P.; Xie, H.; Amini, R.M.; Lawrie, C.H.; Enblad, G.; Larsson, C.; et al. Role of microRNAs and microRNA machinery in the pathogenesis of diffuse large B-cell lymphoma. Blood Cancer J. 2013, 3, e152. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Xue, W.; Wang, X.; Fu, X.; Sun, Z.; Li, Z.; Chang, Y.; Zhang, X.; Zhou, Z.; Chen, C.; et al. MiR-199a mediated the dissemination of human mantle cell lymphoma by interacting with the CCR7/CCL21 pair. Anti-Cancer Drugs 2018, 29, 861–870. [Google Scholar] [CrossRef] [PubMed]
- Mazan-Mamczarz, K.; Gartenhaus, R.B. Role of microRNA deregulation in the pathogenesis of diffuse large B-cell lymphoma (DLBCL). Leuk. Res. 2013, 37, 1420–1428. [Google Scholar] [CrossRef] [PubMed]
- Di Lisio, L.; Gómez-López, G.; Sánchez-Beato, M.; Gómez-Abad, C.; Rodríguez, M.E.; Villuendas, R.; Ferreira, B.I.; Carro, A.; Rico, D.; Mollejo, M.; et al. Mantle cell lymphoma: Transcriptional regulation by microRNAs. Leukemia 2010, 24, 1335–1342. [Google Scholar] [CrossRef]
- Marchesi, F.; Regazzo, G.; Palombi, F.; Terrenato, I.; Sacconi, A.; Spagnuolo, M.; Donzelli, S.; Marino, M.; Ercolani, C.; Di Benedetto, A.; et al. Serum miR-22 as potential non-invasive predictor of poor clinical outcome in newly diagnosed, uniformly treated patients with diffuse large B-cell lymphoma: An explorative pilot study. J. Exp. Clin. Cancer Res. 2018, 37, 95. [Google Scholar] [CrossRef]
- Gao, J.; Liu, Q.G. The role of miR-26 in tumors and normal tissues. Oncol. Lett. 2011, 2, 1019–1023. [Google Scholar] [CrossRef]
- Sandhu, S.K.; Croce, C.M.; Garzon, R. Micro-RNA Expression and Function in Lymphomas. Adv. Hematol. 2011, 2011, 347137. [Google Scholar] [CrossRef]
- Cordeiro, A.; Monzó, M.; Navarro, A. Non-Coding RNAs in Hodgkin Lymphoma. Int. J. Mol. Sci. 2017, 18, 1154. [Google Scholar] [CrossRef] [PubMed]
- Lawrie, C.H.; Chi, J.; Taylor, S.; Tramonti, D.; Ballabio, E.; Palazzo, S.; Saunders, N.J.; Pezzella, F.; Boultwood, J.; Wainscoat, J.S.; et al. Expression of microRNAs in diffuse large B cell lymphoma is associated with immunophenotype, survival and transformation from follicular lymphoma. J. Cell Mol. Med. 2009, 13, 1248–1260. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Lwin, T.; Zhao, J.J.; Tam, W.; Choi, Y.S.; Moscinski, L.C.; Dalton, W.S.; Sotomayor, E.M.; Wright, K.L.; Tao, J. Follicular dendritic cell-induced microRNA-mediated upregulation of PRDM1 and downregulation of BCL-6 in non-Hodgkin’s B-cell lymphomas. Leukemia 2011, 25, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Marques, S.C.; Ranjbar, B.; Laursen, M.B.; Falgreen, S.; Bilgrau, A.E.; Bødker, J.S.; Jørgensen, L.K.; Primo, M.N.; Schmitz, A.; Ettrup, M.S.; et al. High miR-34a expression improves response to doxorubicin in diffuse large B-cell lymphoma. Exp. Hematol. 2016, 44, 238–246. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Kluiver, J.; Koerts, J.; de Jong, D.; Rutgers, B.; Abdul Razak, F.R.; Terpstra, M.; Plaat, B.E.; Nolte, I.M.; Diepstra, A.; et al. miR-24-3p Is Overexpressed in Hodgkin Lymphoma and Protects Hodgkin and Reed-Sternberg Cells from Apoptosis. Am. J. Pathol. 2017, 187, 1343–1355. [Google Scholar] [CrossRef] [PubMed]
- Imig, J.; Motsch, N.; Zhu, J.Y.; Barth, S.; Okoniewski, M.; Reineke, T.; Tinguely, M.; Faggioni, A.; Trivedi, P.; Meister, G.; et al. microRNA profiling in Epstein-Barr virus-associated B-cell lymphoma. Nucleic Acids Res. 2011, 39, 1880–1893. [Google Scholar] [CrossRef] [PubMed]
- Condoluci, A.; Rossi, D.; Zucca, E.; Cavalli, F. Toward a Risk-Tailored Therapeutic Policy in Mantle Cell Lymphoma. Curr. Oncol. Rep. 2018, 20, 79. [Google Scholar] [CrossRef]
- Lim, E.L.; Marra, M.A. MicroRNA dysregulation in B-cell non-Hodgkin lymphoma. Blood Lymphat. Cancer Targets Ther. 2013, 3, 25–40. [Google Scholar]
- Khare, D.; Goldschmidt, N.; Bardugo, A.; Gur-Wahnon, D.; Ben-Dov, I.Z.; Avni, B. Plasma microRNA profiling: Exploring better biomarkers for lymphoma surveillance. PLoS ONE 2017, 12, e0187722. [Google Scholar] [CrossRef]
- Mazan-Mamczarz, K.; Zhao, X.F.; Dai, B.; Steinhardt, J.J.; Peroutka, R.J.; Berk, K.L.; Landon, A.L.; Sadowska, M.; Zhang, Y.; Lehrmann, E.; et al. Down-regulation of eIF4GII by miR-520c-3p represses diffuse large B cell lymphoma development. PLoS Genet. 2014, 10, e1004105. [Google Scholar] [CrossRef]
- Solé, C.; Arnaiz, E.; Lawrie, C.H. MicroRNAs as Biomarkers of B-cell Lymphoma. Biomark Insights 2018, 13, 1177271918806840. [Google Scholar] [CrossRef] [PubMed]
- Romero, M.; Gapihan, G.; Castro-Vega, L.J.; Acevedo, A.; Wang, L.; Li, Z.W.; El Bouchtaoui, M.; Di Benedetto, M.; Ratajczak, P.; Feugeas, J.P.; et al. Primary mediastinal large B-cell lymphoma: Transcriptional regulation by miR-92a through FOXP1 targeting. Oncotarget 2017, 8, 16243–16258. [Google Scholar] [CrossRef] [PubMed]
- Reale, M.; D’Angelo, C.; Costantini, E.; Laus, M.; Moretti, A.; Croce, A. MicroRNA in Sjögren’s Syndrome: Their Potential Roles in Pathogenesis and Diagnosis. J. Immunol. Res. 2018, 2018, 7510174. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.Q.; Zilahi, E.; Papp, G.; Sipka, S.; Zeher, M. Simultaneously increased expression of microRNA-155 and suppressor of cytokine signaling 1 (SOCS1) gene in the peripheral blood mononuclear cells of patients with primary Sjögren’s syndrome. Int. J. Rheum. Dis. 2017, 20, 609–613. [Google Scholar] [CrossRef] [PubMed]
- Perez, P.; Teos, L.; Tandon, M.; Kazmi, S.; Gallo, A.; Illei, G.; Alevizos, I. Downregulation of MicroRNA-183 in Sjogren’s Syndrome Minor Salivary Glands; Implications in the Control of ezrin Expression and Salivary Gland Function. In Proceedings of the 2014 ACR/ARHP Annual Meeting, Boston, MA, USA, 14–19 November 2014. [Google Scholar]
- Wang-Renault, S.F.; Boudaoud, S.; Nocturne, G.; Roche, E.; Sigrist, N.; Daviaud, C.; Bugge Tinggaard, A.; Renault, V.; Deleuze, J.F.; Mariette, X.; et al. Deregulation of microRNA expression in purified T and B lymphocytes from patients with primary Sjögren’s syndrome. Ann. Rheum. Dis. 2018, 77, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.C.; Pan, H.F.; Leng, R.X.; Wang, D.G.; Li, X.P.; Li, X.M.; Ye, D.Q. Emerging role of long noncoding RNAs in autoimmune diseases. Autoimmun. Rev. 2015, 14, 798–805. [Google Scholar] [CrossRef] [PubMed]
- Dolcino, M.; Tinazzi, E.; Puccetti, A.; Lunardi, C. In Systemic Sclerosis, a Unique Long Non Coding RNA Regulates Genes and Pathways Involved in the Three Main Features of the Disease (Vasculopathy, Fibrosis and Autoimmunity) and in Carcinogenesis. J. Clin. Med. 2019, 8, 320. [Google Scholar] [CrossRef]
- Konsta, O.D.; Thabet, Y.; Le Dantec, C.; Brooks, W.H.; Tzioufas, A.G.; Pers, J.O.; Renaudineau, Y. The contribution of epigenetics in Sjögren’s Syndrome. Front. Genet. 2014, 3, 5–71. [Google Scholar] [CrossRef]
- Muskardin, T.L.W.; Niewold, T.B. Type I interferon in rheumatic diseases. Nat. Rev. Rheumatol. 2018, 14, 214–228. [Google Scholar] [CrossRef]
- Nocturne, G.; Mariette, X. B cells in the pathogenesis of primary Sjögren syndrome. Nat. Rev. Rheumatol. 2018, 14, 133–145. [Google Scholar] [CrossRef]
- Han, S.B.; Moratz, C.; Huang, N.N.; Kelsall, B.; Cho, H.; Shi, C.S.; Schwartz, O.; Kehrl, J.H. Rgs1 and Gnai2 regulate the entrance of B lymphocytes into lymph nodes and B cell motility within lymph node follicles. Immunity 2005, 22, 343–354. [Google Scholar] [CrossRef] [PubMed]
- Coca, A.; Sanz, I. Updates on B-cell immunotherapies for systemic lupus erythematosus and Sjogren’s syndrome. Curr. Opin. Rheumatol. 2012, 24, 451–456. [Google Scholar] [CrossRef] [PubMed]
- Béguelin, W.; Teater, M.; Gearhart, M.D.; Calvo Fernández, M.T.; Goldstein, R.L.; Cárdenas, M.G.; Hatzi, K.; Rosen, M.; Shen, H.; Corcoran, C.M.; et al. EZH2 and BCL6 Cooperate to Assemble CBX8-BCOR Complex to Repress Bivalent Promoters, Mediate Germinal Center Formation and Lymphomagenesis. Cancer Cell 2016, 30, 197–213. [Google Scholar] [CrossRef] [PubMed]
- Goules, A.V.; Tzioufas, A.G. Lymphomagenesis in Sjögren’s syndrome: Predictive biomarkers towards precision medicine. Autoimmun. Rev. 2019, 18, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Bai, M.; Skyrlas, A.; Agnantis, N.J.; Kamina, S.; Tsanou, E.; Grepi, C.; Galani, V.; Kanavaros, P. Diffuse large B-cell lymphomas with germinal center B-cell-like differentiation immunophenotypic profile are associated with high apoptotic index, high expression of the proapoptotic proteins bax, bak and bid and low expression of the antiapoptotic protein bcl-xl. Mod. Pathol. 2004, 17, 847–856. [Google Scholar] [PubMed]
- Zhu, X.; Miao, X.; Wu, Y.; Li, C.; Guo, Y.; Liu, Y.; Chen, Y.; Lu, X.; Wang, Y.; He, S. ENO1 promotes tumor proliferation and cell adhesion mediated drug resistance (CAM-DR) in Non-Hodgkin’s Lymphomas. Exp. Cell Res. 2015, 335, 216–223. [Google Scholar] [CrossRef] [PubMed]
- Kishor, A.; Ge, Z.; Hogg, J.R. hnRNP L-dependent protection of normal mRNAs from NMD subverts quality control in B cell lymphoma. EMBO J. 2019, 38, e99128. [Google Scholar] [CrossRef] [PubMed]
- Moore, C.R.; Edwards, S.K.; Xie, P. Targeting TRAF3 Downstream Signaling Pathways in B cell Neoplasms. J. Cancer Sci. Ther. 2015, 7, 67–74. [Google Scholar]
- Togarrati, P.P.; Dinglasan, N.; Desai, S.; Ryan, W.R.; Muench, M.O. CD29 is highly expressed on epithelial, myoepithelial, and mesenchymal stromal cells of human salivary glands. Oral Dis. 2018, 24, 561–572. [Google Scholar] [CrossRef]
- Häärä, O.; Fujimori, S.; Schmidt-Ullrich, R.; Hartmann, C.; Thesleff, I.; Mikkola, M.L. Ectodysplasin and Wnt pathways are required for salivary gland branching morphogenesis. Development 2011, 138, 2681–2691. [Google Scholar] [CrossRef]
- Charras, A.; Konsta, O.D.; Le Dantec, C.; Bagacean, C.; Kapsogeorgou, E.K.; Tzioufas, A.G.; Pers, J.O.; Bordron, A.; Renaudineau, Y. Cell-specific epigenome-wide DNA methylation profile in long-term cultured minor salivary gland epithelial cells from patients with Sjögren’s syndrome. Ann. Rheum. Dis. 2017, 76, 625–628. [Google Scholar] [CrossRef] [PubMed]
- Hall, B.E.; Zheng, C.; Swaim, W.D.; Cho, A.; Nagineni, C.N.; Eckhaus, M.A.; Flanders, K.C.; Ambudkar, I.S.; Baum, B.J.; Kulkarni, A.B. Conditional overexpression of TGF-beta1 disrupts mouse salivary gland development and function. Lab. Investig. 2010, 90, 543–555. [Google Scholar] [CrossRef]
- Mattingly, A.; Finley, J.K.; Knox, S.M. Salivary gland development and disease. Wiley Interdiscip. Rev. Dev. Biol. 2015, 4, 573–590. [Google Scholar] [CrossRef] [PubMed]
- Dean, C.; Ito, M.; Makarenkova, H.P.; Faber, S.C.; Lang, R.A. Bmp7 regulates branching morphogenesis of the lacrimal gland by promoting mesenchymal proliferation and condensation. Development 2004, 131, 4155–4165. [Google Scholar] [CrossRef] [PubMed]
- Park, S.R.; Lee, E.K.; Kim, B.C.; Kim, P.H. p300 cooperates with Smad3/4 and Runx3 in TGFbeta1-induced IgA isotype expression. Eur. J. Immunol. 2003, 33, 3386–3392. [Google Scholar] [CrossRef] [PubMed]
- Harunaga, J.; Hsu, J.C.; Yamada, K.M. Dynamics of salivary gland morphogenesis. J. Dent. Res. 2011, 90, 1070–1077. [Google Scholar] [CrossRef] [PubMed]
- Rao, K.; Farley, W.J.; Pflugfelder, S.C. Association between high tear epidermal growth factor levels and corneal subepithelial fibrosis in dry eye conditions. Investig. Ophthalmol. Vis. Sci. 2010, 51, 844–849. [Google Scholar] [CrossRef]
- Loreto, C.; Caltabiano, R.; Musumeci, G.; Caltabiano, C.; Greco, M.G.; Leonardi, R. Hepatocyte growth factor receptor, c-Met, in human embryo salivary glands. An immunohistochemical study. Anat. Histol. Embryol. 2010, 39, 173–177. [Google Scholar] [CrossRef]
- Li, Q.; Weng, J.; Mohan, R.R.; Bennett, G.L.; Schwall, R.; Wang, Z.F.; Tabor, K.; Kim, J.; Hargrave, S.; Cuevas, K.H.; et al. Hepatocyte growth factor and hepatocyte growth factor receptor in the lacrimal gland, tears, and cornea. Investig. Ophthalmol. Vis. Sci. 1996, 37, 727–739. [Google Scholar]
- Barrera, M.J.; Bahamondes, V.; Sepúlveda, D.; Quest, A.F.; Castro, I.; Cortés, J.; Aguilera, S.; Urzúa, U.; Molina, C.; Pérez, P.; et al. Sjögren’s syndrome and the epithelial target: A comprehensive review. J. Autoimmun. 2013, 42, 7–18. [Google Scholar] [CrossRef]
- Velozo, J.; Aguilera, S.; Alliende, C.; Ewert, P.; Molina, C.; Pérez, P.; Leyton, L.; Quest, A.; Brito, M.; González, S.; et al. Severe alterations in expression and localisation of {alpha}6{beta}4 integrin in salivary gland acini from patients with Sjogren syndrome. Ann. Rheum. Dis. 2009, 68, 991–996. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, M.V.; Salomonsson, S.; Øijordsbakken, G.; Skarstein, K. Elevated serum levels of soluble E-cadherin in patients with primary Sjögren’s syndrome. Scand. J. Immunol. 2005, 62, 552–559. [Google Scholar] [CrossRef] [PubMed]
- Fukuhara, A.; Irie, K.; Yamada, A.; Katata, T.; Honda, T.; Shimizu, K.; Nakanishi, H.; Takai, Y. Role of nectin in organization of tight junctions in epithelial cells. Genes Cells 2002, 7, 1059–1072. [Google Scholar] [CrossRef] [PubMed]
- Cortés, J.; Hidalgo, J.; Aguilera, S.; Castro, I.; Brito, M.; Urra, H.; Pérez, P.; Barrera, M.J.; Carvajal, P.; Urzúa, U.; et al. Synaptotagmin-1 overexpression under inflammatory conditions affects secretion in salivary glands from Sjögren’s syndrome patients. J. Autoimmun. 2019, 97, 88–99. [Google Scholar] [CrossRef] [PubMed]
- Busamia, B.; Gonzalez-Moles, M.A.; Ruiz-Avila, I.; Brunotto, M.; Gil-Montoya, J.A.; Bravo, M.; Gobbi, C.; Finkelberg, A. Cell apoptosis and proliferation in salivary glands of Sjögren’s syndrome. J. Oral Pathol. Med. 2011, 40, 721–725. [Google Scholar] [CrossRef]
- Manganelli, P.; Fietta, P. Apoptosis and Sjögren syndrome. Semin. Arthritis Rheum. 2003, 33, 49–65. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, A.R.; Soares, R. Inflammation in Sjögren’s syndrome: Cause or consequence? Autoimmunity 2017, 50, 141–150. [Google Scholar] [CrossRef]
- Schütze, S.; Wiegmann, K.; Machleidt, T.; Krönke, M. TNF-induced activation of NF-kappa B. Immunobiology 1995, 193, 193–203. [Google Scholar] [CrossRef]
- Chen, W.; Lin, J.; Cao, H.; Xu, D.; Xu, B.; Xu, L.; Yue, L.; Sun, C.; Wu, G.; Qian, W. Local and Systemic IKKε and NF-κB Signaling Associated with Sjögren’s Syndrome Immunopathogenesis. J. Immunol. Res. 2015, 2015, 534648. [Google Scholar] [CrossRef]
- Sakai, N.; Wada, T.; Furuichi, K.; Iwata, Y.; Yoshimoto, K.; Kitagawa, K.; Kokubo, S.; Kobayashi, M.; Takeda, S.; Kida, H.; et al. p38 MAPK phosphorylation and NF-kappa B activation in human crescentic glomerulonephritis. Nephrol. Dial. Transplant. 2002, 17, 998–1004. [Google Scholar] [CrossRef]
- Sekiguchi, M.; Iwasaki, T.; Kitano, M.; Kuno, H.; Hashimoto, N.; Kawahito, Y.; Azuma, M.; Hla, T.; Sano, H. Role of sphingosine 1-phosphate in the pathogenesis of Sjögren’s syndrome. J. Immunol. 2008, 180, 1921–1928. [Google Scholar] [CrossRef] [PubMed]
- Pollard, K.M.; Cauvi, D.M.; Toomey, C.B.; Morris, K.V.; Kono, D.H. Interferon-γ and systemic autoimmunity. Discov. Med. 2013, 16, 123–131. [Google Scholar] [PubMed]
- Nezos, A.; Gravani, F.; Tassidou, A.; Kapsogeorgou, E.K.; Voulgarelis, M.; Koutsilieris, M.; Crow, M.K.; Mavragani, C.P. Type I and II interferon signatures in Sjogren’s syndrome pathogenesis: Contributions in distinct clinical phenotypes and Sjogren’s related lymphomagenesis. J. Autoimmun. 2015, 63, 47–58. [Google Scholar] [CrossRef] [PubMed]
- Baldini, C.; Rossi, C.; Ferro, F.; Santini, E.; Seccia, V.; Donati, V.; Solini, A. The P2X7 receptor-inflammasome complex has a role in modulating the inflammatory response in primary Sjögren’s syndrome. J. Intern. Med. 2013, 274, 480–489. [Google Scholar] [CrossRef] [PubMed]
- Both, T.; Dalm, V.A.; van Hagen, P.M.; van Daele, P.L. Reviewing primary Sjögren’s syndrome: Beyond the dryness—From pathophysiology to diagnosis and treatment. Int. J. Med. Sci. 2017, 14, 191–200. [Google Scholar] [CrossRef]
- Oh-hora, M.; Rao, A. The calcium/NFAT pathway: Role in development and function of regulatory T cells. Microbes Infect. 2009, 11, 612–619. [Google Scholar] [CrossRef] [PubMed]
- Limon, J.J.; Fruman, D.A. Akt and mTOR in B Cell Activation and Differentiation. Front Immunol. 2012, 3, 228. [Google Scholar] [CrossRef]
- Reth, M.; Nielsen, P. Signaling circuits in early B-cell development. Adv. Immunol. 2014, 122, 129–175. [Google Scholar] [CrossRef]
- Nie, Y.; Waite, J.; Brewer, F.; Sunshine, M.J.; Littman, D.R.; Zou, Y.R. The role of CXCR4 in maintaining peripheral B cell compartments and humoral immunity. J. Exp. Med. 2004, 200, 1145–1156. [Google Scholar] [CrossRef]
- Hansen, A.; Reiter, K.; Ziprian, T.; Jacobi, A.; Hoffmann, A.; Gosemann, M.; Scholze, J.; Lipsky, P.E.; Dörner, T. Dysregulation of chemokine receptor expression and function by B cells of patients with primary Sjögren’s syndrome. Arthritis Rheum. 2005, 52, 2109–2119. [Google Scholar] [CrossRef]

| Gene Symbol | Description | Fold Change | p-Value | mRNA Accession |
|---|---|---|---|---|
| Apoptosis | ||||
| CCAR2 | cell cycle and apoptosis regulator 2 | 1.83 | 0.002 | NM_021174 |
| CASP10 | caspase 10 | 2.26 | 0.004 | NM_001206524 |
| BCL2L13 | BCL2-like 13 (apoptosis facilitator) | 2.49 | <0.001 | NM_001270726 |
| BCL2L12 | BCL2-like 12 (proline rich) | 2.44 | 0.002 | NM_001040668 |
| PYCARD | PYD and CARD domain containing | 2.52 | 0.004 | NM_013258 |
| DAPK3 | death-associated protein kinase 3 | 2.09 | <0.001 | NM_001348 |
| PDCD6 | programmed cell death 6 | 2.93 | <0.001 | NM_001267556 |
| CASP9 | caspase 9 | 2.01 | <0.001 | NM_001229 |
| DEDD2 | death effector domain containing 2 | 3.82 | <0.001 | NM_001270614 |
| BMF | Bcl2 modifying factor | 1.88 | 0.003 | NM_001003940 |
| PDCD7 | programmed cell death 7 | 1.98 | 0.001 | NM_005707 |
| FADD | Fas (TNFRSF6)-associated via death domain | 2.06 | <0.001 | NM_003824 |
| ANP32B | acidic nuclear phosphoprotein 32 family member B | 2.34 | <0.001 | NM_006401 |
| BRI3 | brain protein I3 | 1.87 | 0.003 | NM_001159491 |
| CLPTM1L | CLPTM1-like | 2.8 | <0.001 | NM_030782 |
| ING2 | inhibitor of growth family member 2 | 1.97 | <0.001 | NM_001291959 |
| BFAR | bifunctional apoptosis regulator | 1.98 | 0.003 | NM_016561 |
| DAP | death-associated protein | 2.42 | 0.004 | NM_001291963 |
| SORT1 | sortilin 1 | 2.51 | 0.008 | NM_001205228 |
| RRAGA | Ras-related GTP binding A | 1.76 | 0.001 | NM_006570 |
| Cell adhesion | ||||
| LIN7C | lin-7 homolog C (C. elegans) | −1.76 | 0.001 | NM_018362 |
| ITGB3BP | integrin beta 3 binding protein (beta3-endonexin) | −2.87 | 0.001 | NM_001206739 |
| PRKCE | protein kinase C, epsilon | −1.62 | 0.005 | NM_005400 |
| FER | fer (fps/fes related) tyrosine kinase | −2.3 | 0.001 | NM_001308028 |
| ADAM8 | ADAM metallopeptidase domain 8 | 3.13 | <0.001 | NM_001109 |
| ADAM15 | ADAM metallopeptidase domain 15 | 3.59 | <0.001 | NM_001261464 |
| CLDN5 | claudin 5 | 1.69 | 0.007 | NM_001130861 |
| EMP2 | epithelial membrane protein 2 | 1.71 | 0.005 | NM_001424 |
| ICAM3 | intercellular adhesion molecule 3 | 2.86 | <0.001 | NM_002162 |
| HN1 | hematological and neurological expressed 1 | 2.71 | <0.001 | NM_001002032 |
| ITGA5 | integrin alpha 5 | 2.22 | <0.001 | NM_002205 |
| MISP | mitotic spindle positioning | 1.86 | 0.003 | NM_173481 |
| PTPRJ | protein tyrosine phosphatase, receptor type, J | 1.71 | 0.006 | NM_001098503 |
| RHOC | ras homolog family member C | 2.5 | 0.004 | NM_001042678 |
| ZEB1 | zinc finger E-box binding homeobox 1 | −4.18 | <0.001 | NM_001128128 |
| ZFYVE21 | zinc finger, FYVE domain containing 21 | 3.07 | 0.001 | NM_001198953 |
| Immune response | ||||
| LAMTOR2 | late endosomal/lysosomal adaptor, MAPK and MTOR activator 2 | 3.08 | 0.002 | NM_001145264 |
| IL6R | interleukin 6 receptor | 1.79 | 0.001 | NM_000565 |
| IGKV3D-7 | immunoglobulin kappa variable 3D-7 | 4.17 | 0.004 | OTTHUMT00000476805 |
| IGLV3-19 | immunoglobulin lambda variable 3-19 | 6.58 | 0.002 | OTTHUMT00000321830 |
| HLA-G | major histocompatibility complex, class I, G | 2.98 | <0.001 | NM_002127 |
| NCF1 | neutrophil cytosolic factor 1 | 2.17 | 0.003 | NM_000265 |
| CD68 | CD68 molecule | 2.18 | 0.002 | NM_001040059 |
| DEF6 | DEF6 guanine nucleotide exchange factor | 1.58 | 0.003 | NM_022047 |
| LILRA6 | leukocyte immunoglobulin-like receptor, subfamily A, member 6 | 2.37 | 0.004 | NM_024318 |
| TRBV24-1 | T cell receptor beta variable 24-1 | 3.57 | 0.001 | OTTHUMT00000352499 |
| IL5RA | interleukin 5 receptor, alpha | 2.14 | <0.001 | NM_000564 |
| CD6 | CD6 molecule | 3.71 | <0.001 | NM_001254750 |
| CD7 | CD7 molecule | 1.93 | <0.001 | NM_006137 |
| BTK | Bruton agammaglobulinemia tyrosine kinase | 1.79 | 0.006 | NM_000061 |
| BAX | BCL2-associated X protein | 3.14 | <0.001 | NM_001291428 |
| BAK1 | BCL2-antagonist/killer 1 | 2.46 | 0.005 | NM_001188 |
| GATA3 | GATA binding protein 3 | 1.74 | 0.008 | NM_001002295 |
| TAP1 | antigen peptide transporter 1 | 2.58 | <0.001 | NM_001292022 |
| TAP2 | antigen peptide transporter 2 | 2.23 | 0.002 | NM_018833 |
| IL17RA | interleukin 17 receptor A | 2.52 | 0.001 | NM_001289905 |
| IL23A | interleukin 23, alpha subunit p19 | −2.9 | 0.009 | NM_016584 |
| IL4R | interleukin 4 receptor | 2.37 | <0.001 | NM_000418 |
| CD33 | CD33 molecule | 3.39 | 0.002 | NM_001082618 |
| IL2RA | interleukin 2 receptor, alpha | 1.89 | 0.006 | NM_000417 |
| LAT | linker for activation of T-cells | 3.25 | <0.001 | NM_001014987 |
| C1RL | complement component 1, r subcomponent-like | 1.52 | 0.010 | NM_001297640 |
| FCGR1A | Fc fragment of IgG, high affinity Ia, receptor (CD64) | 3.2 | <0.001 | NM_000566 |
| CD81 | CD81 molecule | 2.04 | 0.008 | NM_001297649 |
| KLHL20 | kelch-like family member 20 | −1.94 | 0.004 | NM_014458 |
| KLHL6 | kelch-like family member 6 | 2.18 | 0.005 | NM_130446 |
| ORAI1 | ORAI calcium release-activated calcium modulator 1 | 2.97 | <0.001 | NM_032790 |
| CBX8 | chromobox homolog 8 | 1.62 | 0.008 | NM_020649 |
| ENO1 | enolase 1, (alpha) | 1.91 | 0.004 | NM_001201483 |
| GNAI2 | guanine nucleotide binding protein, alpha inhibiting activity polypeptide 2 | 1.67 | 0.007 | NM_001166425 |
| SPI1 | Spi-1 proto-oncogene | 1.73 | 0.007 | NM_001080547 |
| TRAF3 | TNF receptor-associated factor 3 | −1.63 | 0.009 | NM_001199427 |
| HNRNPL | heterogeneous nuclear ribonucleoprotein L | 2.16 | 0.001 | NM_001005335 |
| FOSL1 | FOS-like antigen 1 | −2.14 | 0.008 | NM_001300844 |
| Type I interferon signaling | ||||
| IRF5 | interferon regulatory factor 5 | 2.24 | 0.003 | NM_001098627 |
| IRF7 | interferon regulatory factor 7 | 3.14 | 0.002 | NM_001572 |
| IRF9 | interferon regulatory factor 9 | 2.68 | <0.001 | NM_006084 |
| MYD88 | myeloid differentiation primary response 88 | 1.86 | 0.005 | NM_001172566 |
| HLA-H | major histocompatibility complex, class I, H (pseudogene) | 2.18 | 0.004 | NR_001434 |
| OAS1 | 2-5-oligoadenylate synthetase 1 | 3.58 | 0.002 | NM_001032409 |
| IFI35 | interferon-induced protein 35 | 1.78 | 0.007 | NM_005533 |
| IFITM3 | interferon induced transmembrane protein 3 | 2.54 | 0.006 | NM_021034 |
| IFNA10 | interferon, alpha 10 | 1.99 | 0.002 | NM_002171 |
| ADAR | adenosine deaminase, RNA-specific | 3.15 | 0.005 | NM_001025107 |
| OAS3 | 2-5-oligoadenylate synthetase 3 | 4.27 | 0.008 | NM_006187 |
| STAT1 | signal transducer and activator of transcription 1 | 1.75 | 0.008 | NM_007315 |
| KLHL20 | kelch-like family member 20 | −1.94 | 0.004 | NM_014458 |
| Interferon-gamma signaling | ||||
| HLA-G | major histocompatibility complex, class I, G | 2.98 | <0.001 | NM_002127 |
| FCGR1B | Fc fragment of IgG, high affinity Ib, receptor (CD64) | 2.25 | 0.002 | NM_001004340 |
| PML | promyelocytic leukemia | 3.18 | 0.002 | NM_002675 |
| STAT1 | signal transducer and activator of transcription 1 | 1.75 | 0.008 | NM_007315 |
| TRIM21 | tripartite motif containing 21 | 2.13 | 0.002 | NM_003141 |
| FCGR1A | Fc fragment of IgG, high affinity Ia, receptor (CD64) | 3.2 | <0.001 | NM_000566 |
| Inflammatory response | ||||
| CCR4 | chemokine (C-C motif) receptor 4 | 3.73 | 0.005 | NM_005508 |
| CCR8 | chemokine (C-C motif) receptor 8 | 2.24 | 0.004 | NM_005201 |
| IL6R | interleukin 6 receptor | 1.79 | 0.001 | NM_000565 |
| MYD88 | myeloid differentiation primary response 88 | 1.86 | 0.005 | NM_001172566 |
| CSF1 | colony stimulating factor 1 (macrophage) | 2.23 | 0.005 | NM_000757 |
| MIF | macrophage migration inhibitory factor (glycosylation-inhibiting factor) | 2.48 | <0.001 | NM_002415 |
| TNFRSF1A | tumor necrosis factor receptor superfamily, member 1A | 4.39 | <0.001 | NM_001065 |
| TGFB1 | transforming growth factor beta 1 | 1.71 | 0.003 | NM_000660 |
| LTB4R | leukotriene B4 receptor | 2.46 | <0.001 | NM_001143919 |
| ALOX5 | arachidonate 5-lipoxygenase | 2.57 | 0.007 | NM_000698 |
| IL23A | interleukin 23, alpha subunit p19 | −2.9 | 0.009 | NM_016584 |
| CXCR3 | chemokine (C-X-C motif) receptor 3 | 2.25 | 0.002 | NM_001142797 |
| MIF | macrophage migration inhibitory factor (glycosylation-inhibiting factor) | 2.48 | <0.001 | NM_002415 |
| CEBPD | CCAAT/enhancer binding protein (C/EBP), delta | 1.94 | 0.007 | NM_005195 |
| MAP3K12 | mitogen-activated protein kinase kinase kinase 12 | 1.88 | 0.005 | NM_001193511 |
| TRIB2 | tribbles pseudokinase 2 | 2.19 | 0.005 | NM_021643 |
| ECM organization | ||||
| MMP9 | matrix metallopeptidase 9 | 3.31 | 0.002 | NM_004994 |
| EFEMP2 | EGF containing fibulin-like extracellular matrix protein 2 | 2.43 | 0.001 | NM_016938 |
| ADAM15 | ADAM metallopeptidase domain 15 | 3.59 | <0.001 | NM_001261464 |
| ADAM8 | ADAM metallopeptidase domain 8 | 3.13 | <0.001 | NM_001109 |
| BSG | basigin (Ok blood group) | 2.07 | 0.001 | NM_001728 |
| DAG1 | dystroglycan 1 (dystrophin-associated glycoprotein 1) | 1.57 | 0.003 | NM_001165928 |
| CTGF | connective tissue growth factor | 1.54 | 0.006 | NM_001901 |
| TGFB1 | transforming growth factor beta 1 | 1.71 | 0.003 | NM_000660 |
| LOXL3 | lysyl oxidase-like 3 | 2.02 | 0.002 | NM_001289164 |
| SPOCK2 | sparc/osteonectin, cwcv and kazal-like domains proteoglycan (testican) 2 | 2.2 | 0.006 | NM_001134434 |
| Morphogenesis of a branching epithelium | ||||
| CTSZ | cathepsin Z | 2.99 | 0.002 | NM_001336 |
| ILK | integrin linked kinase | 1.82 | 0.001 | NM_001014794 |
| DAG1 | dystroglycan 1 (dystrophin-associated glycoprotein 1) | 1.57 | 0.003 | NM_001165928 |
| EDN1 | endothelin 1 | −2.17 | 0.002 | NM_001168319 |
| TGFB1 | transforming growth factor beta 1 | 1.71 | 0.003 | NM_000660 |
| GREM1 | gremlin 1 | 1.53 | 0.009 | ENST00000633992 |
| ENG | endoglin | 2.42 | <0.001 | NM_000118 |
| Epithelial cells polarization | ||||
| ARF6 | ADP-ribosylation factor 6 | 1.53 | 0.008 | NM_001663 |
| FRMD4A | FERM domain containing 4A | 1.85 | 0.003 | NM_018027 |
| RHOQ | ras homolog family member Q | 1.79 | 0.005 | NM_012249 |
| Gene Symbol | Description | Fold Change | p-Value | Public Gene IDs |
| LINC00657 | long intergenic non-protein coding RNA 657 | 1.8 | 0.001 | NR_027451 |
| LINC00511 | long intergenic non-protein coding RNA 511 | −2.0 | 0.008 | NR_036488 |
| CTD-2020K17.1 | novel transcript, antisense to FMNL1 | 2.7 | 0.000 | ENST00000585471.1 |
| RP11-169K16.9 | uncharacterized LOC729614 | 1.7 | 0.008 | NR_024279 |
| RP11-214O1.2 | uncharacterized protein MGC12916 | −2.1 | 0.010 | NR_026880 |
| RP11-372K14.2 | novel transcript, antisense to SH3D19 | −2.1 | 0.004 | ENST00000603472.1 |
| Gene Symbol | miRNA Targets | Total Number of Targeted Modulated Genes | Targeted Modules | Total Number of Targeted Module-Associated Genes |
| LINC00657 | 67 | 313 | 7 | 75 |
| LINC00511 | 11 | 194 | 7 | 41 |
| CTD-2020K17.1 | 11 | 120 | 7 | 25 |
| RP11-169K16.9 | 13 | 90 | 5 | 15 |
| RP11-214O1.2 | 12 | 86 | 4 | 13 |
| RP11-372K14.2 | 12 | 75 | 4 | 12 |
| MiRNAs Previously Associated to Lymphoma | ||
| lncRNA | miRNA Target | References |
| LINC00657 | hsa-miR-106a-5p | Diffuse large B cell lymphoma [16] |
| LINC00657 | hsa-miR-106b-5p | Diffuse large B cell lymphoma [16] |
| LINC00657 | hsa-miR-144-3p | Diffuse large B cell lymphoma [17] |
| LINC00657 | hsa-miR-155-5p | Diffuse large B cell lymphoma [18] |
| LINC00511 | hsa-miR-15a-5p | Diffuse large B cell lymphoma [19] |
| LINC00511 | hsa-miR-15b-5p | Mantle cell lymphoma [20] |
| LINC00511 | hsa-miR-16-5p | Diffuse large B cell lymphoma [21] |
| LINC00657 | hsa-miR-17-5p | Burkitt’s lymphoma [22] |
| LINC00657 | hsa-miR-182-5p | Mantle cell lymphoma [22] |
| LINC00511 | hsa-miR-183-5p | Hodgkin’s lymphoma [23] |
| LINC00511 | hsa-miR-195-5p | Diffuse large B cell lymphoma [24] |
| CTD-2020K17.1 | hsa-miR-199a-5p | Mantle cell lymphoma [25] |
| CTD-2020K17.1 | hsa-miR-199b-5p | Diffuse large B cell lymphoma [26] |
| LINC00657 | hsa-miR-20a-5p | Diffuse large B cell lymphoma [16] |
| LINC00657 | hsa-miR-20b-5p | Mantle cell lymphoma [27] |
| LINC00657 | hsa-miR-22-3p | Diffuse large B cell lymphoma [28] |
| LINC00657 | hsa-miR-26a-5p | Burkitt’s lymphoma [29] |
| LINC00657 | hsa-miR-26b-5p | Burkitt’s lymphoma [29] |
| LINC00511 | hsa-miR-29a-3p | Mantle cell lymphoma [30] |
| LINC00511 | hsa-miR-29b-3p | Mantle cell lymphoma [30] |
| LINC00511 | hsa-miR-29c-3p | Mantle cell lymphoma [30] |
| LINC00657 | hsa-miR-302a-3p | Hodgkin Lymphoma [31] |
| LINC00657 | hsa-miR-302b-3p | Hodgkin’s Lymphoma [31]; diffuse large B cell lymphoma [32] |
| LINC00657 | hsa-miR-302c-3p | Hodgkin’s Lymphoma [31]; mantle cell lymphoma [27] |
| LINC00657 | hsa-miR-30a-5p | Non-Hodgkin’s B cell lymphoma [33] |
| LINC00657 | hsa-miR-30b-5p | Non-Hodgkin’s B cell lymphoma [33] |
| LINC00657 | hsa-miR-30c-5p | Non-Hodgkin’s B cell lymphoma [33] |
| LINC00657 | hsa-miR-30d-5p | Non-Hodgkin’s B cell lymphoma [33] |
| LINC00657 | hsa-miR-30e-5p | Non-Hodgkin’s B cell lymphoma [33] |
| CTD-2020K17.1 | hsa-miR-34a-5p | Diffuse large B cell lymphoma [34] |
| LINC00657 | hsa-miR-363-3p | Mantle cell lymphoma [22] |
| LINC00657 | hsa-miR-372-3p | Mantle cell lymphoma [27] |
| LINC00657 | hsa-miR-373-3p | Mantle cell lymphoma [27] |
| LINC00657 | hsa-miR-378a-3p | Hodgkin’s lymphoma [35] |
| LINC00657 | hsa-miR-378b | Epstein–Barr virus-associated B-cell lymphoma [36] |
| LINC00657 | hsa-miR-378c | Epstein–Barr virus-associated B-cell lymphoma [36] |
| LINC00657 | hsa-miR-378d | Mantle cell lymphoma [37]; Epstein–Barr virus-associated B-cell lymphoma [36] |
| LINC00657 | hsa-miR-378e | Epstein–Barr virus-associated B-cell lymphoma [36] |
| LINC00657 | hsa-miR-378f | Epstein–Barr virus-associated B-cell lymphoma [36] |
| LINC00657 | hsa-miR-378h | Epstein–Barr virus-associated B-cell lymphoma [36] |
| LINC00657 | hsa-miR-378i | Epstein–Barr virus-associated B-cell lymphoma [36] |
| LINC00657 | hsa-miR-421 | Diffuse large B cell lymphoma [32] |
| LINC00657 | hsa-miR-422a | Burkitt’s lymphoma [38] |
| LINC00511 | hsa-miR-424-5p | Diffuse large B cell lymphoma [39] |
| LINC00511 | hsa-miR-497-5p | Diffuse large B cell lymphoma [24] |
| LINC00657 | hsa-miR-519d-3p | Diffuse large B cell lymphoma [32] |
| LINC00657 | hsa-miR-520a-3p | Hodgkin’s Lymphoma [30] |
| LINC00657 | hsa-miR-520c-3p | Diffuse large B cell lymphoma [40] |
| LINC00657 | hsa-miR-520d-3p | Diffuse large B cell lymphoma [41] |
| LINC00657 | hsa-miR-92a-3p | Diffuse large B cell lymphoma [42]; Non-Hodgkin’s B cell lymphoma [22] |
| LINC00657 | hsa-miR-92b-3p | Mantle cell lymphoma [22] |
| MiRNAs Previously Associated to pSjS | ||
| lncRNA | miRNA target | References |
| LINC00657 | hsa-miR-106a-5p | [43] |
| LINC00657 | hsa-miR-155-5p | [44] |
| LINC00511 | hsa-miR-15a-5p | [43] |
| LINC00511 | hsa-miR-16-5p | [43] |
| LINC00657 | hsa-miR-17-5p | [43] |
| LINC00511 | hsa-miR-183-5p | [45] |
| LINC00511 | hsa-miR-195-5p | [46] |
| LINC00657 | hsa-miR-20a-5p | [43] |
| LINC00657 | hsa-miR-20b-5p | [43] |
| LINC00657 | hsa-miR-26a-5p | [43] |
| LINC00657 | hsa-miR-30b-5p | [46] |
| LINC00657 | hsa-miR-30c-5p | [46] |
| LINC00657 | hsa-miR-32-5p | [46] |
| CTD-2020K17.1 | hsa-miR-34a-5p | [43] |
| LINC00657 | hsa-miR-378a-3p | [43,46] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dolcino, M.; Tinazzi, E.; Vitali, C.; Papa, N.D.; Puccetti, A.; Lunardi, C. Long Non-Coding RNAs Modulate Sjögren’s Syndrome Associated Gene Expression and Are Involved in the Pathogenesis of the Disease. J. Clin. Med. 2019, 8, 1349. https://doi.org/10.3390/jcm8091349
Dolcino M, Tinazzi E, Vitali C, Papa ND, Puccetti A, Lunardi C. Long Non-Coding RNAs Modulate Sjögren’s Syndrome Associated Gene Expression and Are Involved in the Pathogenesis of the Disease. Journal of Clinical Medicine. 2019; 8(9):1349. https://doi.org/10.3390/jcm8091349
Chicago/Turabian StyleDolcino, Marzia, Elisa Tinazzi, Claudio Vitali, Nicoletta Del Papa, Antonio Puccetti, and Claudio Lunardi. 2019. "Long Non-Coding RNAs Modulate Sjögren’s Syndrome Associated Gene Expression and Are Involved in the Pathogenesis of the Disease" Journal of Clinical Medicine 8, no. 9: 1349. https://doi.org/10.3390/jcm8091349
APA StyleDolcino, M., Tinazzi, E., Vitali, C., Papa, N. D., Puccetti, A., & Lunardi, C. (2019). Long Non-Coding RNAs Modulate Sjögren’s Syndrome Associated Gene Expression and Are Involved in the Pathogenesis of the Disease. Journal of Clinical Medicine, 8(9), 1349. https://doi.org/10.3390/jcm8091349




