Changes in Circulating Lysyl Oxidase-Like-2 (LOXL2) Levels, HOMA, and Fibrosis after Sustained Virological Response by Direct Antiviral Therapy
Abstract
:1. Introduction
2. Patients and Methods
Methods
3. Results
3.1. Study Population
Baseline Characteristics and Impact of SVR at Two Years
3.2. Noninvasive Serological Scores
3.3. Influence of Metabolic Syndrome in Fibrosis Regression
3.4. Cirrhotic Patients
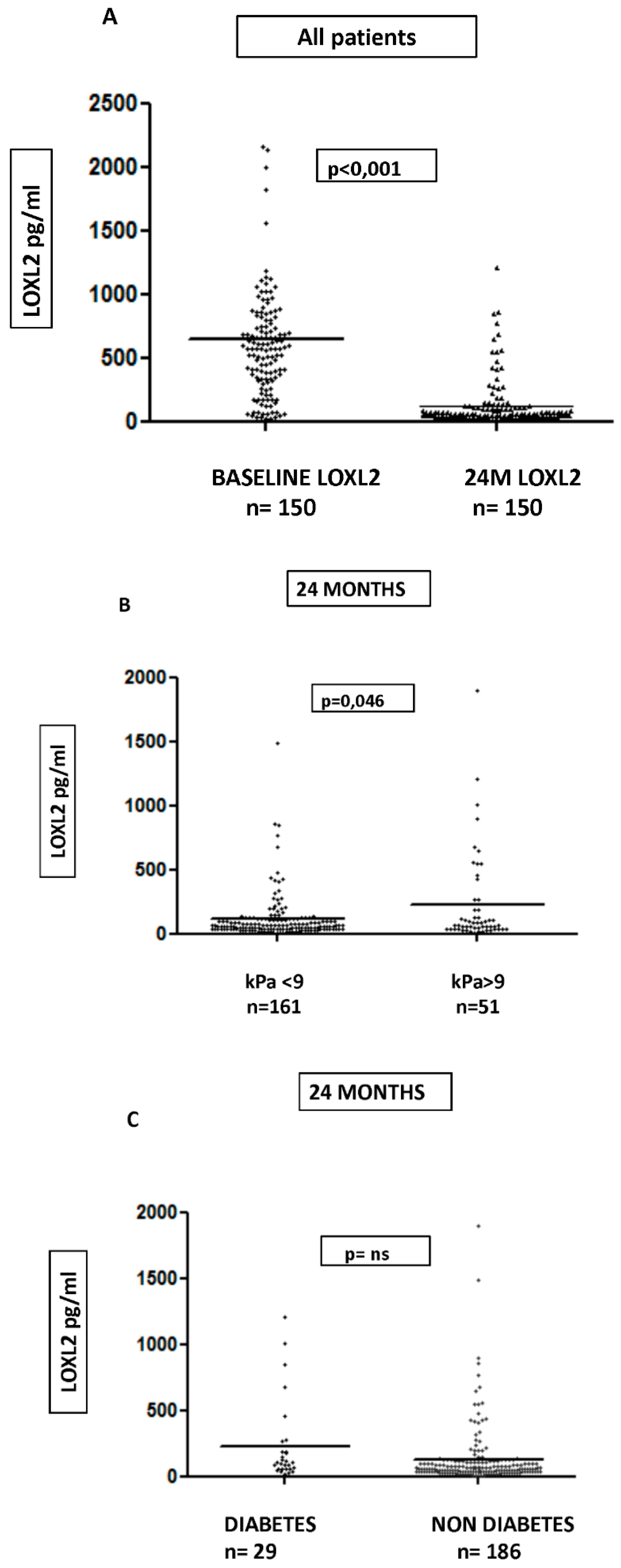
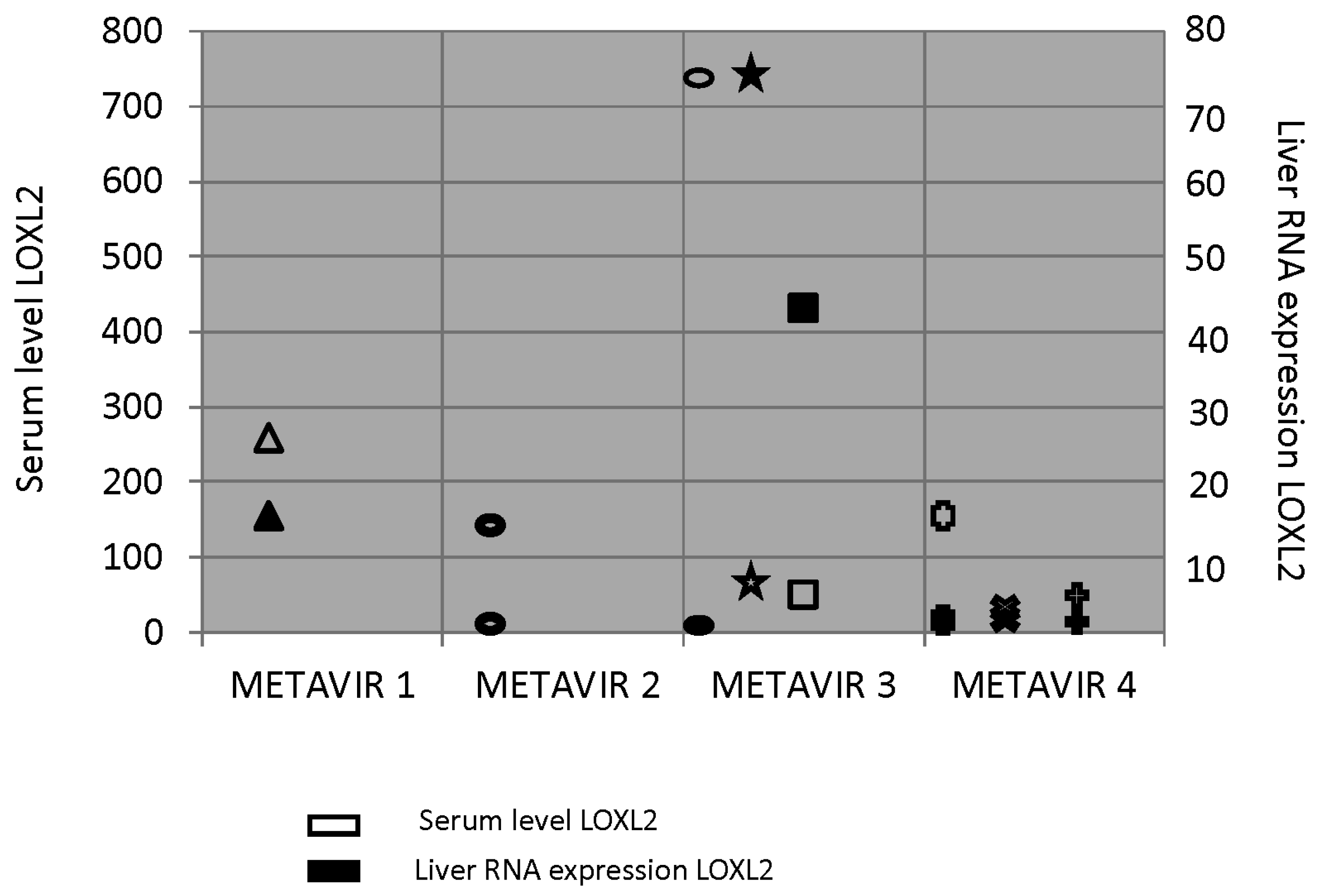
3.5. LOXL2 Serum and Tissue Expression
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Nonstandard Abbreviations Used
References
- European Association for the Study of the Liver. EASL Recommendations on Treatment of Hepatitis C 2018. J. Hepatol. 2018, 69, 461–511. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Garcia-Tsao, G. Regression of HCV cirrhosis: Time will tell. Hepatology. 2018, 67, 1651–1653. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van der Meer, A.J.; Hansen, B.E.; Janssen, H.L. Sustained virological response to treatment in patients with chronic hepatitis C—Reply. JAMA 2013, 309, 1457. [Google Scholar] [CrossRef] [PubMed]
- Petta, S.; Di Marco, V.; Bruno, S.; Enea, M.; Calvaruso, V.; Boccaccio, V.; Rossi, S.; Ceaxi, A.; Camma, C. Impact of virus eradication in patients with compensated hepatitis C virus-related cirrhosis: Competing risks and multistate model. Liver Int. 2016, 36, 1765–1773. [Google Scholar] [CrossRef] [PubMed]
- Mandorfer, M.; Kozbial, K.; Schwabl, P.; Freissmuth, C.; Schwarzer, R.; Stern, R.; Chromy, D.; Stattermayer, A.F.; Reiberger, T.; Beinhardt, S.; et al. Sustained virologic response to interferon-free therapies ameliorates HCV-induced portal hypertension. J. Hepatol. 2016, 65, 692–699. [Google Scholar] [CrossRef] [PubMed]
- Perelló, C.; Cabezas, J.; Llop, E.; Carrion, J.A.; Ruiz-Antoran, B.; Llerena, S.; Crespo, J.; Calleja, J.L.; Hernandez-Conde, M.; Crespo, J.; et al. LImpact of SVR in the development of all complications and fibrosis regression in a cohort of patients treated with interferon-base Triple Therapy and Direct Acting Antiviral. Hepatology 2017, 66, 125A. [Google Scholar]
- Ampuero, J.; Jimeno, C.; Quiles, R.; Rosales, J.M.; Llerena, S.; Palomo, N.l.; Cordero, P.; Serrano, F.J.; Urquijo, J.J.; Moreno-Planas, J.M.; et al. Impact of comorbidities on patient outcomes after interferon-free therapy-induced viral eradication in hepatitis C. J. Hepatol. 2018, 68, 940–948. [Google Scholar] [CrossRef]
- Hedenstierna, M.; Nangarhari, A.; El-Sabini, A.; Weiland, O.; Aleman, S. Cirrhosis, high age and high body mass index are risk factors for persisting advanced fibrosis after sustained virological response in chronic hepatitis C. J. Viral Hepat. 2018, 25, 802–810. [Google Scholar] [CrossRef]
- Rockey, D. Translating an Understanding of the Pathogenesis of Hepatic Fibrosis to Novel Therapies. Clin. Gastroenterol. Hepatol. 2013, 11, 224–231. [Google Scholar] [CrossRef]
- Pellicoro, A.; Ramachandran, P.; Iredale, J.P.; Fallowfield, J.A. Liver fibrosis and repair: Immune regulation of wound healing in a solid organ. Nat. Rev. Immunol. 2014, 14, 181–194. [Google Scholar] [CrossRef]
- Trautwein, C.; Friedman, S.; Schuppan, D.; Pinzani, M. Hepatic fibrosis: Concept to treatment. J. Hepatol. 2015, 62, S15–S24. [Google Scholar] [CrossRef] [Green Version]
- Tacke, F.; Zimmermann, H.W. Macrophage heterogeneity in liver injury and fibrosis. J. Hepatol. 2014, 60, 1090–1096. [Google Scholar] [CrossRef] [Green Version]
- Fernandez, M.; Semela, D.; Bruix, J.; Colle, I.; Pinzani, M.; Bosch, J. Angiogenesis in liver disease. J. Hepatol. 2009, 50, 604–620. [Google Scholar] [CrossRef]
- Schuppan, D.; Kim, Y. Evolving therapies for liver fibrosis. J. Clin. Invest. 2013, 123, 1887–1901. [Google Scholar] [CrossRef] [Green Version]
- Grau-Bove, X.; Ruiz-Trillo, I.; Pascual, F.R. Origin and evolution of lysyl oxidases. Sci. Rep. 2015, 29, 10568. [Google Scholar] [CrossRef]
- Puente, A.; Fortea, J.I.; Cabezas, J.; Arias Loste, M.T.; Iruzubieta, P.; Llerena, S.; Huelin, P.; Fabrega, E.; Crespo, J. Loxl-2. A new target in antifibrogenic therapy? Int. J. Mol. Sci. 2019, 20, 1634. [Google Scholar] [CrossRef]
- Wong, C.C.; Tse, A.P.; Huang, Y.P.; Zhu, Y.T.; Chiu, D.K.; Lai, R.K.; Au, S.L.; Kai, A.K.; Lee, J.M.; Wei, L.L.; et al. Lysyl oxidase-like 2 is critical to tumor microenvironment and metastatic niche formation in hepatocellular carcinoma. Hepatology 2014, 60, 1645–1658. [Google Scholar] [CrossRef] [PubMed]
- Dongiovanni, P.; Meroni, M.; Baselli, G.A.; Bassani, G.A.; Rametta, R.; Pietrelli, A.; Maggoini, M.; Facciotti, F.; Trunzo, V.; Badiali, S.; et al. Insulin resistance promotes Lysyl Oxidase Like 2 induction and fibrosis accumulation in nonalcoholic fatty liver disease. Clin. Sci. (Lond.) 2017, 131, 1301–1315. [Google Scholar] [CrossRef]
- Bosch, J.; Ratziu, V.; Rockey, D.; Ghalib, R.; Thuluvath, P.; Schiefke, I.; Flamm, S.; Abdelmalek, M.; Millers, R.; Aguilar, R.; et al. Ccorrelations between noninvasive markers of fibrosis and the hepatic venous pressure gradient (HVPG) in patients with compensated cirrhosis due to nonalcoholic steatohepatitis (NASH). Hepatology 2015, 62, 120. [Google Scholar]
- Afdha, P.; Fortea, J.I.; Cabezas, J.; Loste, M.T.A.; Iruzubieta, P.; Llerena, S.; Huelin, P.; Fabrega, E.; Crespo, J. Serum lysyl oxidase-like-2 (sLOXL2) is correlated with the hepatic venous pressure gradient (HVPG) in patients with cirrhosis due to hepatitis C. Hepatology 2015, 62, 121. [Google Scholar]
- Bourliere, M.; Loustaud-Ratti, V.; Metivier, S.; Leroy, V.; Abergel, A.; Myers, R.; Aguilar, R.; Hyland, R.; Subramanian, M.; McHutchison, J.; et al. Changes in liver stiffness by transient elastography (TE) and serum lysyl oxidase-like-2 (sLOXL2) in patients with cirrhosis treated with ledipasvir/sofosbuvir (LDV/ SOF)-based therapy. Hepatology 2015, 62, 123A. [Google Scholar]
- Barry-Hamilton, V.; Spangler, R.; Marshall, D.; McCauley, S.; Rodriguez, H.M.; Oyasu, M.; Mikekls, A.; Vaysberg, M.; Ghermazien, H.; Wai, C.; et al. Allosteric inhibition of lysyl oxidase-like-2 impedes the development of a pathologic microenvironment. Nat. Med. 2010, 16, 1009–1017. [Google Scholar] [CrossRef]
- Ikenaga, N.; Peng, Z.W.; Vaid, K.A.; Liu, S.B.; Yoshida, S.; Sverdlov, D.Y.; Mikels-Vigdal, A.; Smith, V.; Schuppan, D.; Popov, Y.V. Selective targeting of lysyl oxidase-like 2 (LOXL2) suppresses hepatic fibrosis progression and accelerates its reversal. Gut 2017, 66, 1697–1708. [Google Scholar] [CrossRef] [Green Version]
- Loomba, R.; Lawitz, E.; Mantry, P.S.; Jayakumar, S.; Caldwell, S.H.; Arnold, H.; Diehl, A.M.; Djedjos, C.S.; Han, L.; Myers, R.P.; et al. GS-US-384-1497 Investigators. The ASK1 inhibitor selonsertib in patients with nonalcoholic steatohepatitis: A randomized, phase 2 trial. Hepatology. 2018, 67, 549–559. [Google Scholar] [CrossRef]
- Pollheimer, M.J.; Racedo, S.; Mikels-Vigdal, A.; Marshall, D.; Bowlus, C.; Lackner, C.; Madl, T.; Karlsen, T.H.; Hov, J.R.; Lyman, S.K.; et al. Lysyl oxidase-like protein 2 (LOXL2) modulates barrier function in cholangiocytes in cholestasis. J. Hepatol. 2018, 69, 368–377. [Google Scholar] [CrossRef]
- Tatal, A.H.; Feron-Rigodon, M.; Subramanian, G.M.; Bornstein, J.D. Simtuzumab, an antifibrotic monoclonal antibody against lysyl oxidase like 2, appears safe and well tolerated in patients with liver disease of diverse etiology. J. Hepatol. 2013, 58, S409. [Google Scholar]
- D’Ambrosio, R.; Aghemo, A.; Fraquelli, M.; Rumi, M.G.; Donato, M.F.; Paradis, V.; Bedossa, P.; Colombo, M. The diagnostic accuracy of Fibroscan for cirrhosis is influenced by liver morphometry in HCV patients with a sustained virological response. J. Hepatol. 2013, 59, 251–256. [Google Scholar] [CrossRef]
- Lens, S.; Alvarado-Tapias, E.; Mariño, Z.; Londoño, M.C.; LLop, E.; Martinez, J.; Fortea, J.I.; Ibanea, L.; Ariza, X.; Baiges, A.; et al. Effects of All-Oral Anti-Viral Therapy on HVPG and Systemic Hemodynamics in Patients With Hepatitis C Virus-Associated Cirrhosis. Gastroenterology 2017, 153, 1273–1283. [Google Scholar] [CrossRef]
- Puente, Á.; Cabezas, J.; López Arias, M.J.; Fortea, J.I.; Arias, M.T.; Estébanez, Á.; Casafont, F.; Fabrega, E.; Crespo, J. Influence of sustained viral response on the regression of fibrosis and portal hypertension in cirrhotic HCV patients treated with antiviral triple therapy. Rev. Esp. Enferm. Dig. 2017, 109, 17–25. [Google Scholar] [CrossRef]
- Fernández Carrillo, C.; Lens, S.; Llop, E.; Pascasio, J.M.; Crespo, J.; Arenas, J.; Fernandez, I.; Baliellas, C.; Carrion, J.A.; de la Mata, M.; et al. Treatment of hepatitis C virus infection in patients with cirrhosis and predictive value of MELD: Analysis of data from the Hepa-C registry. Hepatology 2017, 65, 1810–1822. [Google Scholar] [CrossRef]
- Poynard, T.; McHutchison, J.; Manns, M.; Trepo, C.; Lindsay, K.; Goodman, Z.; Ling, M.H.; Albrecht, J. Impact of pegylated interferon alfa-2b and ribavirin on liver fibrosis in patients with chronic hepatitis C. Gastroenterology 2002, 122, 1303–1313. [Google Scholar] [CrossRef] [Green Version]
- Llerena, S.; Perello, C.; Hernandez, M.; Garcia, M.; Ramos, D.; Estebanez, A.; Cabezas, J.; Cuadrado, A.; Lopez Hoyos, M. Endothelial dysfunction, macrophage dysfunction and emerging cardiovascular risk factors in patients with hepatitis c virus infection. characterization and potential reversibility with direct acting antiviral agents. J. Hepatol. 2016, 64, S823. [Google Scholar] [CrossRef]


| Variable | n/Mean |
|---|---|
| Demographic Factors (n = 271) | |
| Age (years) | 59.29 ± 10.5 |
| Sex | 183 (66.8%) men / 91 (33.2%) woman |
| BMI (kg/m2) | Overall = 26.48 ± 4.21 − Normal (<25 kg/m2) n = 114 (41.6%) − Overweight (≥25 to <30 kg/m2) n = 109 (39.8%) − Obese (≥30 kg/m2) n = 51 (18.6%) |
| Metabolic Syndrome | n = 43 (17.3%) |
| Noninvasive Measures | |
| LSM (n = 271) | Mean: 12.79 kPa ± 10.7; F1 = 69 (25.2%) F2 = 59 (21.5%) F3 = 44 (16.1%) F4 = 102 (37.5%) |
| CAP (dB/m) (n = 25) | 223.04 ± 85.7 |
| NAFLD fibrosis score (n = 271) | −1.27 ± 1.34 |
| FIB4 (n = 271) | 3.75 ± 12.2 |
| HEPAMET score (n = 271) | 0.12 ± 3.94 |
| APRI (n = 271) | 1.59 ± 3.94 |
| FORNS (n = 271) | 6.82 ± 1.95 |
| Medical History (n = 271) | |
| Diabetes Mellitus | n = 36 (13.1%) − Diet n = 3 (1%) − OHAs n = 18 (6.5%) − Insulin n = 15 (5.4%) |
| Hypertension | n = 83 (30.2%) |
| Dislypemia | n = 45 (16.4%) – Diet 15 (5.4%) − Statins 10 (3.6%) |
| Psiquiatric Disorder | n = 55 (20%) – anxiety n = 45 (16.4%) – Psychotic disorder n = 10 (3.6% ) |
| Toxics | Drugs n = 136 (49.6%); ExPWID n = 98 ( 35.7%); Tobacco n = 208 (75.9%) |
| Coffee | n = 173 (63.1%) |
| Child Pugh Score (A/B/C) | n = 92 (33.57%)/n = 10 (3.6%)/n = 0 |
| Varices/Ascites/ Encephalopaty/HCC | n = 21 (7.7%)/n = 5 (1,8%) /n = 2 (0.7%)/ n = 3 (1.1) |
| Serum Biochemical Levels (n = 271) | |
| ALT, UI/mL | 84.20 ± 66.97 |
| AST, UI/mL | 66.19 ± 51.725 |
| GGT, UI/mL | 88.28 ± 125.92 |
| Bilirrubin, mg/dL | 1.13 ± 4.51 |
| Albumin, mg/dL | 4.30 ± 0.34 |
| Fasting Glucose Levels, mg/dL | 92.96 ± 28.1 |
| Insulin, µU/mL | 11.11 ± 6.31 |
| HOMA | 2.56 ± 2.256 |
| Platelets Count, 10*3/µL | 174.12 ± 64.82 |
| International normalized ratio (INR) | 2.19 ± 10.13 |
| Triglycerides, mg/dL | 99.39 ± 45.38 |
| Total Cholesterol, mg/dL | 166.51 ± 39.12 |
| High-Density Lipoprotein, mg/dL | 51.34 ± 15.6 |
| Low-Density Lipoprotein, mg/dL | 96.44 ± 31.16 |
| 24M Elastography | ||||
|---|---|---|---|---|
| Variable | Pearson Correlation | p Value | Multivariate Regression | p Value |
| Insulin | 0.356 | 0.0001* | −0.216 | 0.900 |
| Cholesterol | −0.073 | 0.230 | ||
| HDL-Cholesterol | −0.028 | 0.652 | ||
| LDL-Colesterol | −0.122 | 0.050 | 0.265 | 0.125 |
| HOMA | 0.385 | 0.0001* | 2.090 | 0.025* |
| Triglycerides | −0.009 | 0.886 | ||
| APRI | −0.165 | 0.006* | 0.614 | 0.540 |
| FIB4 | −0.093 | 0.128 | −0.111 | 0.903 |
| Variable | OR (IC 95%) | p Value | ||
| Metabolic Syndrome | 5.4 (1.9–15.4) | 0.001* | ||
| BMI > 25kg/m2 | (−0.656–3.423) | 0.183 | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Puente, A.; Fortea, J.I.; Posadas, M.; Garcia Blanco, A.; Rasines, L.; Cabezas, J.; Arias Loste, M.T.; Llerena, S.; Iruzubieta, P.; Fábrega, E.; et al. Changes in Circulating Lysyl Oxidase-Like-2 (LOXL2) Levels, HOMA, and Fibrosis after Sustained Virological Response by Direct Antiviral Therapy. J. Clin. Med. 2019, 8, 1242. https://doi.org/10.3390/jcm8081242
Puente A, Fortea JI, Posadas M, Garcia Blanco A, Rasines L, Cabezas J, Arias Loste MT, Llerena S, Iruzubieta P, Fábrega E, et al. Changes in Circulating Lysyl Oxidase-Like-2 (LOXL2) Levels, HOMA, and Fibrosis after Sustained Virological Response by Direct Antiviral Therapy. Journal of Clinical Medicine. 2019; 8(8):1242. https://doi.org/10.3390/jcm8081242
Chicago/Turabian StylePuente, Angela, Jose Ignacio Fortea, Miguel Posadas, Agustin Garcia Blanco, Laura Rasines, Joaquin Cabezas, Maria Teresa Arias Loste, Susana Llerena, Paula Iruzubieta, Emilio Fábrega, and et al. 2019. "Changes in Circulating Lysyl Oxidase-Like-2 (LOXL2) Levels, HOMA, and Fibrosis after Sustained Virological Response by Direct Antiviral Therapy" Journal of Clinical Medicine 8, no. 8: 1242. https://doi.org/10.3390/jcm8081242





