High Constitutive Cytokine Release by Primary Human Acute Myeloid Leukemia Cells Is Associated with a Specific Intercellular Communication Phenotype
Abstract
1. Introduction
2. Materials and Methods
2.1. AML Patients and Preparation of Primary AML Cells
2.2. Mutation Profiling, Flow Cytometric Analyses, and Analysis of Global Gene Expression Profiles
2.3. Analysis of Constitutive Mediator Release by Primary Human AML Cells
2.4. Proteomic Profiling: Selection of Patients, Sample Preparation, and Proteomic Analysis
2.5. Bioinformatical and Statistical Analyses and Presentation of the Data
3. Results
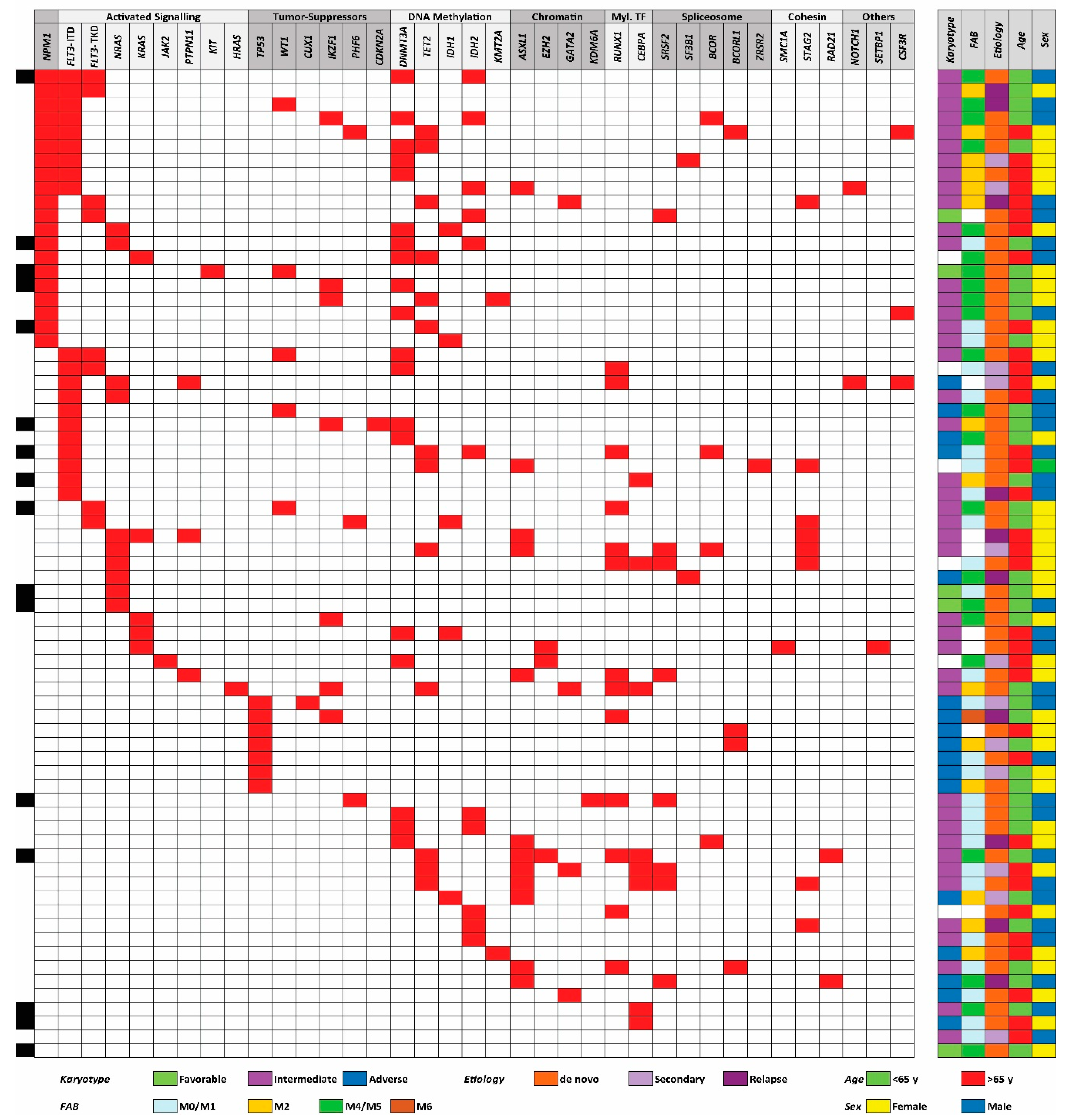
3.1. The Genetic Heterogeneity of AML Patients: TP53 Mutations are Associated with High-Risk Karyotypes and NPM1 Mutations are Associated with Mutations in DNA Methylation Genes
3.2. Expression of Molecular Differentiation Markers by Primary AML Cells: The Expression of the CD34 Stem Cell Markers Differs between Mutational Subsets
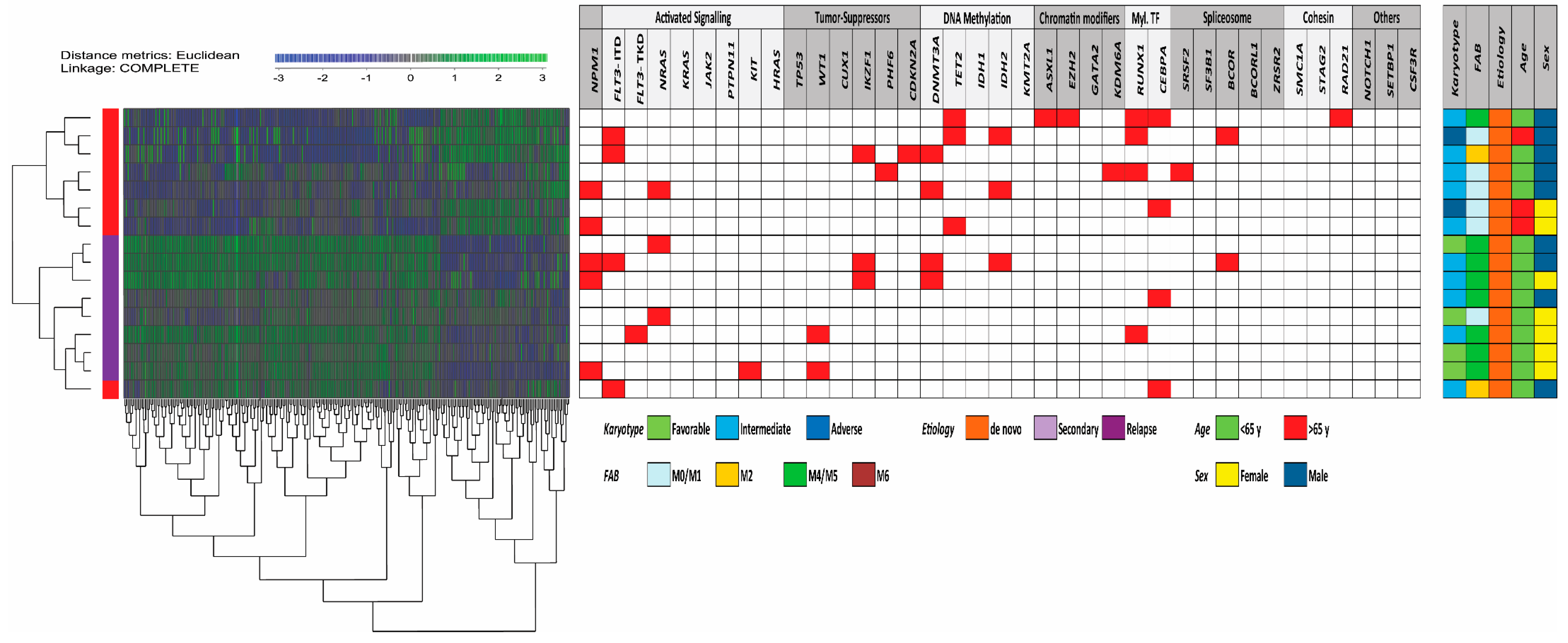
3.3. AML Patients Can Be Subclassified Based on Their Constitutive Release of Extracellular Mediators, but this Capacity Shows no Association with the Mutational Profile
3.4. Comparison of Global Gene Expression Profiles for Patients with Generally High and Low Constitutive Release of Extracellular Mediators
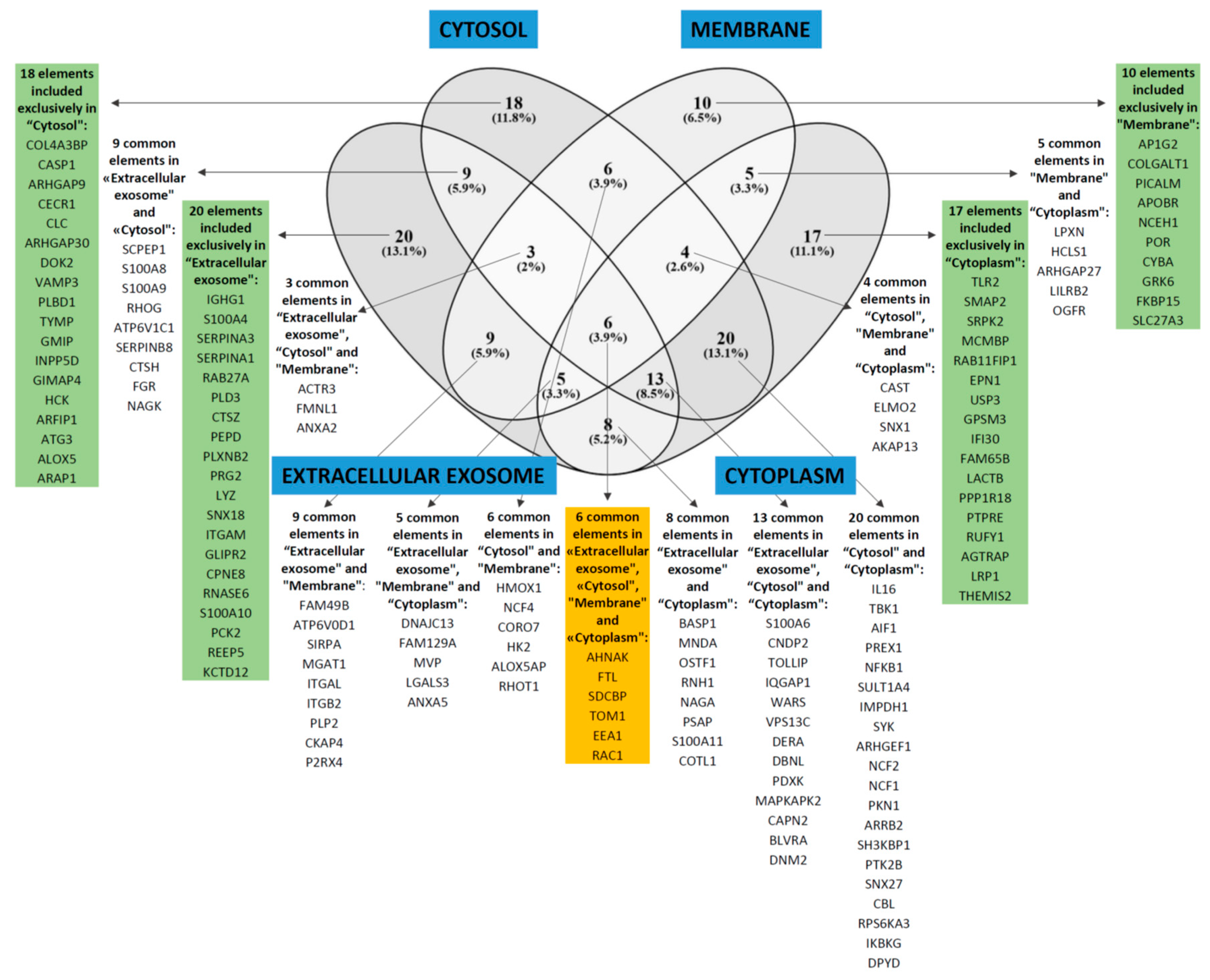
3.5. Comparison of Proteomic Profiles for AML Cell Populations Showing Generally High and Low Constitutive Release of Extracellular Mediators
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Döhner, H.; Weisdorf, D.J.; Bloomfield, C.D. Acute myeloid leukemia. N. Engl. J. Med. 2015, 373, 1136–1152. [Google Scholar] [PubMed]
- Döhner, H.; Estey, E.; Grimwade, D.; Amadori, S.; Appelbaum, F.R.; Buchner, T.; Dombret, H.; Ebert, B.L.; Fenaux, P.; Larson, R.A.; et al. Diagnosis and management of aml in adults: 2017 eln recommendations from an international expert panel. Blood 2017, 129, 424–447. [Google Scholar] [PubMed]
- Arber, D.A.; Orazi, A.; Hasserjian, R.; Thiele, J.; Borowitz, M.J.; Le Beau, M.M.; Bloomfield, C.D.; Cazzola, M.; Vardiman, J.W. The 2016 revision to the world health organization classification of myeloid neoplasms and acute leukemia. Blood 2016, 127, 2391–2405. [Google Scholar] [PubMed]
- Schlenk, R.F.; Dohner, K.; Krauter, J.; Frohling, S.; Corbacioglu, A.; Bullinger, L.; Habdank, M.; Spath, D.; Morgan, M.; Benner, A.; et al. Mutations and treatment outcome in cytogenetically normal acute myeloid leukemia. N. Engl. J. Med. 2008, 358, 1909–1918. [Google Scholar] [PubMed]
- Valk, P.J.; Verhaak, R.G.; Beijen, M.A.; Erpelinck, C.A.; Barjesteh van Waalwijk van Doorn-Khosrovani, S.; Boer, J.M.; Beverloo, H.B.; Moorhouse, M.J.; van der Spek, P.J.; Lowenberg, B.; et al. Prognostically useful gene-expression profiles in acute myeloid leukemia. N. Engl. J. Med. 2004, 350, 1617–1628. [Google Scholar] [PubMed]
- Papaemmanuil, E.; Gerstung, M.; Bullinger, L.; Gaidzik, V.I.; Paschka, P.; Roberts, N.D.; Potter, N.E.; Heuser, M.; Thol, F.; Bolli, N.; et al. Genomic classification and prognosis in acute myeloid leukemia. N. Engl. J. Med. 2016, 374, 2209–2221. [Google Scholar] [PubMed]
- Cancer Genome Atlas Research, N.; Ley, T.J.; Miller, C.; Ding, L.; Raphael, B.J.; Mungall, A.J.; Robertson, A.; Hoadley, K.; Triche, T.J., Jr.; Laird, P.W.; et al. Genomic and epigenomic landscapes of adult de novo acute myeloid leukemia. N. Engl. J. Med. 2013, 368, 2059–2074. [Google Scholar]
- Eppert, K.; Takenaka, K.; Lechman, E.R.; Waldron, L.; Nilsson, B.; van Galen, P.; Metzeler, K.H.; Poeppl, A.; Ling, V.; Beyene, J.; et al. Stem cell gene expression programs influence clinical outcome in human leukemia. Nat. Med. 2011, 17, 1086–1093. [Google Scholar]
- Brenner, A.K.; Reikvam, H.; Lavecchia, A.; Bruserud, O. Therapeutic targeting the cell division cycle 25 (cdc25) phosphatases in human acute myeloid leukemia--the possibility to target several kinases through inhibition of the various cdc25 isoforms. Molecules 2014, 19, 18414–18447. [Google Scholar]
- Hatfield, K.J.; Reikvam, H.; Bruserud, O. Identification of a subset of patients with acute myeloid leukemia characterized by long-term in vitro proliferation and altered cell cycle regulation of the leukemic cells. Expert. Opin. Therap. Targets 2014, 18, 1237–1251. [Google Scholar]
- Brenner, A.K.; Reikvam, H.; Bruserud, O. A subset of patients with acute myeloid leukemia has leukemia cells characterized by chemokine responsiveness and altered expression of transcriptional as well as angiogenic regulators. Front Immunol. 2016, 7, 205. [Google Scholar] [PubMed]
- Griessinger, E.; Anjos-Afonso, F.; Vargaftig, J.; Taussig, D.C.; Lassailly, F.; Prebet, T.; Imbert, V.; Nebout, M.; Vey, N.; Chabannon, C.; et al. Frequency and dynamics of leukemia-initiating cells during short-term ex vivo culture informs outcomes in acute myeloid leukemia patients. Cancer Res. 2016, 76, 2082–2086. [Google Scholar] [PubMed]
- Griessinger, E.; Anjos-Afonso, F.; Pizzitola, I.; Rouault-Pierre, K.; Vargaftig, J.; Taussig, D.; Gribben, J.; Lassailly, F.; Bonnet, D. A niche-like culture system allowing the maintenance of primary human acute myeloid leukemia-initiating cells: A new tool to decipher their chemoresistance and self-renewal mechanisms. Stem Cells Transl. Med. 2014, 3, 520–529. [Google Scholar] [PubMed]
- Hauge, M.; Bruserud, O.; Hatfield, K.J. Targeting of cell metabolism in human acute myeloid leukemia--more than targeting of isocitrate dehydrogenase mutations and pi3k/akt/mtor signaling? Eur. J. Haematol. 2016, 96, 211–221. [Google Scholar] [PubMed]
- Hernandez-Valladares, M.; Aasebo, E.; Mjaavatten, O.; Vaudel, M.; Bruserud, O.; Berven, F.; Selheim, F. Reliable fasp-based procedures for optimal quantitative proteomic and phosphoproteomic analysis on samples from acute myeloid leukemia patients. Biol. Proced. Online 2016, 18, 13. [Google Scholar] [PubMed]
- Aasebo, E.; Mjaavatten, O.; Vaudel, M.; Farag, Y.; Selheim, F.; Berven, F.; Bruserud, O.; Hernandez-Valladares, M. Freezing effects on the acute myeloid leukemia cell proteome and phosphoproteome revealed using optimal quantitative workflows. J. Proteomics 2016, 145, 214–225. [Google Scholar] [PubMed]
- Aasebo, E.; Vaudel, M.; Mjaavatten, O.; Gausdal, G.; Van der Burgh, A.; Gjertsen, B.T.; Doskeland, S.O.; Bruserud, O.; Berven, F.S.; Selheim, F. Performance of super-silac based quantitative proteomics for comparison of different acute myeloid leukemia (aml) cell lines. Proteomics 2014, 14, 1971–1976. [Google Scholar] [PubMed]
- Ossenkoppele, G.J.; Janssen, J.J.; van de Loosdrecht, A.A. Risk factors for relapse after allogeneic transplantation in acute myeloid leukemia. Haematologica 2016, 101, 20–25. [Google Scholar] [PubMed]
- Terwijn, M.; Zeijlemaker, W.; Kelder, A.; Rutten, A.P.; Snel, A.N.; Scholten, W.J.; Pabst, T.; Verhoef, G.; Lowenberg, B.; Zweegman, S.; et al. Leukemic stem cell frequency: A strong biomarker for clinical outcome in acute myeloid leukemia. PloS ONE 2014, 9, e107587. [Google Scholar]
- Wouters, R.; Cucchi, D.; Kaspers, G.J.; Schuurhuis, G.J.; Cloos, J. Relevance of leukemic stem cells in acute myeloid leukemia: Heterogeneity and influence on disease monitoring, prognosis and treatment design. Expert. Rev. Hematol. 2014, 7, 791–805. [Google Scholar]
- Majeti, R. Clonal evolution of pre-leukemic hematopoietic stem cells precedes human acute myeloid leukemia. Best Pract. Res. Clin. Haematol. 2014, 27, 229–234. [Google Scholar]
- Stapnes, C.; Gjertsen, B.T.; Reikvam, H.; Bruserud, O. Targeted therapy in acute myeloid leukaemia: Current status and future directions. Expert. Opin. Investig. Drugs 2009, 18, 433–455. [Google Scholar] [PubMed]
- Binder, S.; Luciano, M.; Horejs-Hoeck, J. The cytokine network in acute myeloid leukemia (aml): A focus on pro- and anti-inflammatory mediators. Cytokine Growth Factor Rev. 2018, 43, 8–15. [Google Scholar] [PubMed]
- Brenner, A.K.; Andersson Tvedt, T.H.; Bruserud, O. The complexity of targeting pi3k-akt-mtor signalling in human acute myeloid leukaemia: The importance of leukemic cell heterogeneity, neighbouring mesenchymal stem cells and immunocompetent cells. Molecules 2016, 21. [Google Scholar]
- Reikvam, H.; Hatfield, K.J.; Fredly, H.; Nepstad, I.; Mosevoll, K.A.; Bruserud, O. The angioregulatory cytokine network in human acute myeloid leukemia-from leukemogenesis via remission induction to stem cell transplantation. Eur. Cytokine Netw. 2012, 23, 140–153. [Google Scholar] [PubMed]
- Kupsa, T.; Horacek, J.M.; Jebavy, L. The role of cytokines in acute myeloid leukemia: A systematic review. Biomed. Pap. Med. Fac. Univ. Palacky Olomouc Czech Repub. 2012, 156, 291–301. [Google Scholar] [PubMed]
- Cho, B.S.; Kim, H.J.; Konopleva, M. Targeting the cxcl12/cxcr4 axis in acute myeloid leukemia: From bench to bedside. Korean J. Intern. Med. 2017, 32, 248–257. [Google Scholar] [PubMed]
- Bernasconi, P.; Farina, M.; Boni, M.; Dambruoso, I.; Calvello, C. Therapeutically targeting self-reinforcing leukemic niches in acute myeloid leukemia: A worthy endeavor? Am. J. Hematol. 2016, 91, 507–517. [Google Scholar] [PubMed]
- Brenner, A.K.; Tvedt, T.H.; Nepstad, I.; Rye, K.P.; Hagen, K.M.; Reikvam, H.; Bruserud, O. Patients with acute myeloid leukemia can be subclassified based on the constitutive cytokine release of the leukemic cells; the possible clinical relevance and the importance of cellular iron metabolism. Expert. Opin. Therap. Targets 2017, 21, 357–369. [Google Scholar]
- Bruserud, O.; Hovland, R.; Wergeland, L.; Huang, T.S.; Gjertsen, B.T. Flt3-mediated signaling in human acute myelogenous leukemia (aml) blasts: A functional characterization of flt3-ligand effects in aml cell populations with and without genetic flt3 abnormalities. Haematologica 2003, 88, 416–428. [Google Scholar]
- Reikvam, H.; Hovland, R.; Forthun, R.B.; Erdal, S.; Gjertsen, B.T.; Fredly, H.; Bruserud, O. Disease-stabilizing treatment based on all-trans retinoic acid and valproic acid in acute myeloid leukemia-identification of responders by gene expression profiling of pretreatment leukemic cells. BMC Cancer 2017, 17, 630. [Google Scholar]
- Staffas, A.; Kanduri, M.; Hovland, R.; Rosenquist, R.; Ommen, H.B.; Abrahamsson, J.; Forestier, E.; Jahnukainen, K.; Jonsson, O.G.; Zeller, B.; et al. Presence of flt3-itd and high baalc expression are independent prognostic markers in childhood acute myeloid leukemia. Blood 2011, 118, 5905–5913. [Google Scholar] [PubMed]
- Wangen, R.; Aasebo, E.; Trentani, A.; Doskeland, S.O.; Bruserud, O.; Selheim, F.; Hernandez-Valladares, M. Preservation method and phosphate buffered saline washing affect the acute myeloid leukemia proteome. Int. J. Mol. Sci. 2018, 19, 296. [Google Scholar]
- Aasebo, E.; Hernandez-Valladares, M.; Selheim, F.; Berven, F.S.; Brenner, A.K.; Bruserud, O. Proteomic profiling of primary human acute myeloid leukemia cells does not reflect their constitutive release of soluble mediators. Proteomes 2018, 7, 1. [Google Scholar]
- Cox, J.; Mann, M. Maxquant enables high peptide identification rates, individualized p.P.B.-range mass accuracies and proteome-wide protein quantification. Nat. Biotechnol. 2008, 26, 1367–1372. [Google Scholar] [PubMed]
- Cox, J.; Matic, I.; Hilger, M.; Nagaraj, N.; Selbach, M.; Olsen, J.V.; Mann, M. A practical guide to the maxquant computational platform for silac-based quantitative proteomics. Nat. Protoc. 2009, 4, 698–705. [Google Scholar] [PubMed]
- Stavrum, A.K.; Petersen, K.; Jonassen, I.; Dysvik, B. Analysis of gene-expression data using j-express. Curr. Protoc. Bioinformat. 2008, Chapter 7, Unit 7.3. [Google Scholar] [CrossRef]
- Mi, H.; Muruganujan, A.; Casagrande, J.T.; Thomas, P.D. Large-scale gene function analysis with the panther classification system. Nat. Protocols 2013, 8, 1551. [Google Scholar] [PubMed]
- Tyanova, S.; Temu, T.; Sinitcyn, P.; Carlson, A.; Hein, M.Y.; Geiger, T.; Mann, M.; Cox, J. The perseus computational platform for comprehensive analysis of (prote)omics data. Nat. Methods 2016, 13, 731–740. [Google Scholar]
- Arntzen, M.O.; Koehler, C.J.; Barsnes, H.; Berven, F.S.; Treumann, A.; Thiede, B. Isobariq: Software for isobaric quantitative proteomics using iptl, itraq, and tmt. J. Proteome Res. 2011, 10, 913–920. [Google Scholar]
- Huang, D.W.; Sherman, B.T.; Tan, Q.; Collins, J.R.; Alvord, W.G.; Roayaei, J.; Stephens, R.; Baseler, M.W.; Lane, H.C.; Lempicki, R.A. The david gene functional classification tool: A novel biological module-centric algorithm to functionally analyze large gene lists. Genome Biol. 2007, 8, R183. [Google Scholar] [CrossRef] [PubMed]
- Szklarczyk, D.; Morris, J.H.; Cook, H.; Kuhn, M.; Wyder, S.; Simonovic, M.; Santos, A.; Doncheva, N.T.; Roth, A.; Bork, P.; et al. The string database in 2017: Quality-controlled protein-protein association networks, made broadly accessible. Nucleic Acids Res. 2017, 45, D362–D368. [Google Scholar] [CrossRef] [PubMed]
- Shannon, P.; Markiel, A.; Ozier, O.; Baliga, N.S.; Wang, J.T.; Ramage, D.; Amin, N.; Schwikowski, B.; Ideker, T. Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Res. 2003, 13, 2498–2504. [Google Scholar] [CrossRef] [PubMed]
- Patel, J.L.; Schumacher, J.A.; Frizzell, K.; Sorrells, S.; Shen, W.; Clayton, A.; Jattani, R.; Kelley, T.W. Coexisting and cooperating mutations in npm1-mutated acute myeloid leukemia. Leukemia Res 2017, 56, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Tsykunova, G.; Reikvam, H.; Hovland, R.; Bruserud, O. The surface molecule signature of primary human acute myeloid leukemia (aml) cells is highly associated with npm1 mutation status. Leukemia 2012, 26, 557–559. [Google Scholar] [CrossRef] [PubMed]
- Honnemyr, M.; Bruserud, O.; Brenner, A.K. The constitutive protease release by primary human acute myeloid leukemia cells. J. Cancer Res. Clin. Oncol. 2017, 143, 1985–1998. [Google Scholar] [CrossRef]
- Brenner, A.K.; Nepstad, I.; Bruserud, O. Mesenchymal stem cells support survival and proliferation of primary human acute myeloid leukemia cells through heterogeneous molecular mechanisms. Front Immunol. 2017, 8, 106. [Google Scholar] [CrossRef]
- Johansen, S.; Brenner, A.K.; Bartaula-Brevik, S.; Reikvam, H.; Bruserud, O. The possible importance of beta3 integrins for leukemogenesis and chemoresistance in acute myeloid leukemia. Int. J. Mol. Sci. 2018, 19, 251. [Google Scholar] [CrossRef]
- Reikvam, H.; Olsnes, A.M.; Gjertsen, B.T.; Ersvar, E.; Bruserud, O. Nuclear factor-kappab signaling: A contributor in leukemogenesis and a target for pharmacological intervention in human acute myelogenous leukemia. Crit. Rev. Oncog. 2009, 15, 1–41. [Google Scholar] [CrossRef]
- Schmitt, A.; Li, L.; Giannopoulos, K.; Greiner, J.; Reinhardt, P.; Wiesneth, M.; Schmitt, M. Quantitative expression of toll-like receptor-2, -4, and -9 in dendritic cells generated from blasts of patients with acute myeloid leukemia. Transfusion 2008, 48, 861–870. [Google Scholar] [CrossRef]
- Bartaula-Brevik, S.; Lindstad Brattas, M.K.; Tvedt, T.H.A.; Reikvam, H.; Bruserud, O. Splenic tyrosine kinase (syk) inhibitors and their possible use in acute myeloid leukemia. Expert Opin. Investig. Drugs 2018, 27, 377–387. [Google Scholar] [CrossRef] [PubMed]
- Bruserud, O.; Ryningen, A.; Olsnes, A.M.; Stordrange, L.; Oyan, A.M.; Kalland, K.H.; Gjertsen, B.T. Subclassification of patients with acute myelogenous leukemia based on chemokine responsiveness and constitutive chemokine release by their leukemic cells. Haematologica 2007, 92, 332–341. [Google Scholar] [CrossRef] [PubMed]
- Godley, L.A. Profiles in leukemia. N. Engl. J. Med 2012, 366, 1152–1153. [Google Scholar] [CrossRef] [PubMed]
- Rucker, F.G.; Schlenk, R.F.; Bullinger, L.; Kayser, S.; Teleanu, V.; Kett, H.; Habdank, M.; Kugler, C.M.; Holzmann, K.; Gaidzik, V.I.; et al. Tp53 alterations in acute myeloid leukemia with complex karyotype correlate with specific copy number alterations, monosomal karyotype, and dismal outcome. Blood 2012, 119, 2114–2121. [Google Scholar] [CrossRef] [PubMed]
- Granfeldt Ostgard, L.S.; Medeiros, B.C.; Sengelov, H.; Norgaard, M.; Andersen, M.K.; Dufva, I.H.; Friis, L.S.; Kjeldsen, E.; Marcher, C.W.; Preiss, B.; et al. Epidemiology and clinical significance of secondary and therapy-related acute myeloid leukemia: A national population-based cohort study. J. Clin. Oncol. 2015, 33, 3641–3649. [Google Scholar] [CrossRef] [PubMed]
- Bruserud, O.; Gjertsen, B.T.; Foss, B.; Huang, T.S. New strategies in the treatment of acute myelogenous leukemia (aml): In vitro culture of aml cells--the present use in experimental studies and the possible importance for future therapeutic approaches. Stem Cells 2001, 19, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Bruserud, O.; Gjertsen, B.T.; von Volkman, H.L. In vitro culture of human acute myelogenous leukemia (aml) cells in serum-free media: Studies of native aml blasts and aml cell lines. J. Hematother. Stem Cell Res. 2000, 9, 923–932. [Google Scholar] [CrossRef]
- Patel, J.P.; Gonen, M.; Figueroa, M.E.; Fernandez, H.; Sun, Z.; Racevskis, J.; Van Vlierberghe, P.; Dolgalev, I.; Thomas, S.; Aminova, O.; et al. Prognostic relevance of integrated genetic profiling in acute myeloid leukemia. N. Engl. J. Med. 2012, 366, 1079–1089. [Google Scholar] [CrossRef]
- Brenner, A.K.; Aasebo, E.; Hernandez-Valladares, M.; Selheim, F.; Berven, F.; Gronningsaeter, I.S.; Bartaula-Brevik, S.; Bruserud, O. The capacity of long-term in vitro proliferation of acute myeloid leukemia cells supported only by exogenous cytokines is associated with a patient subset with adverse outcome. Cancers 2019, 11, 73. [Google Scholar] [CrossRef]
- Angelini, D.F.; Ottone, T.; Guerrera, G.; Lavorgna, S.; Cittadini, M.; Buccisano, F.; De Bardi, M.; Gargano, F.; Maurillo, L.; Divona, M.; et al. A leukemia-associated cd34/cd123/cd25/cd99+ immunophenotype identifies flt3-mutated clones in acute myeloid leukemia. Clin. Cancer Res. 2015, 21, 3977–3985. [Google Scholar] [CrossRef]
- Mannelli, F.; Ponziani, V.; Bencini, S.; Bonetti, M.I.; Benelli, M.; Cutini, I.; Gianfaldoni, G.; Scappini, B.; Pancani, F.; Piccini, M.; et al. Cebpa-double-mutated acute myeloid leukemia displays a unique phenotypic profile: A reliable screening method and insight into biological features. Haematologica 2017, 102, 529–540. [Google Scholar] [CrossRef] [PubMed]
- Falini, B.; Mecucci, C.; Tiacci, E.; Alcalay, M.; Rosati, R.; Pasqualucci, L.; La Starza, R.; Diverio, D.; Colombo, E.; Santucci, A.; et al. Cytoplasmic nucleophosmin in acute myelogenous leukemia with a normal karyotype. N. Engl. J. Med. 2005, 352, 254–266. [Google Scholar] [CrossRef] [PubMed]
- Reikvam, H.; Fredly, H.; Kittang, A.O.; Bruserud, O. The possible diagnostic and prognostic use of systemic chemokine profiles in clinical medicine;the experience in acute myeloid leukemia from disease development and diagnosis via conventional chemotherapy to allogeneic stem cell transplantation. Toxins 2013, 5, 336–362. [Google Scholar] [CrossRef] [PubMed]


| Age and gender | Etiology | ||
|---|---|---|---|
| Median (years) | 64 | Previous chemo-radiotherapy | 1 |
| Range (years) | 18–90 | CML | 1 |
| Females | 31 | Li–Fraumeni’s syndrome | 1 |
| Males | 40 | Polycythemia vera | 1 |
| MDS | 8 | ||
| Relapse | 10 | ||
| de novo | 49 | ||
| FAB1 classification | Cytogenetic abnormalities3 | ||
| M0/1 | 26 | Adverse | 17 |
| M2 | 14 | Favorable | 5 |
| M4/5 | 22 | Intermediate | 43 |
| M6 | 1 | Normal | 404 |
| Unknown | 8 | Unknown | 6 |
| CD34 expression | |||
| Negative (<20%) | 282 | ||
| Positive (>20%) | 43 | ||
| Classification | Mutation | Number with Mutation | Classification | Mutation | Number with Mutation |
|---|---|---|---|---|---|
| NPM1 | ↑NPM1 | 20 | Chromatin modification | ↓ASXL1 | 12 |
| Total group | 20–20 | EZH2 | 3 | ||
| Signaling | ↓FLT3-ITD | 20 | GATA2 | 4 | |
| FLT3-TKD | 8 | KDM6A | 1 | ||
| HRAS | 1 | Total group | 20–15 | ||
| JAK2 | 1 | Myeloid transcription factors | |||
| KIT | 1 | ↑CEBPA | 8 | ||
| KRAS | 5 | ↓RUNX1 | 13 | ||
| NRAS | 10 | Total group | 21–18 | ||
| PTPN11 | 3 | Spliceosome/transcription repressors | BCOR | 4 | |
| Total group | 49–42 | BCORL1 | 4 | ||
| Tumor suppressors | CDKN2A | 1 | SF3B1 | 2 | |
| CUX1 | 1 | SRSF2 | 8 | ||
| IKZF1 | 7 | ZRSB2 | 1 | ||
| PHF6 | 3 | Total group | 19–15 | ||
| TP53↓ | 7 | Cohesin | RAD21 | 2 | |
| WT1 | 5 | SMC1A | 1 | ||
| Total group | 24–21 | STAG2 | 8 | ||
| DNA methylation | DNMT3A | 19 | Total group | 11–11 | |
| IDH1 | 5 | Others | CSF3R | 3 | |
| IDH2 | 11 | NOTCH1 | 2 | ||
| KMT2A/MLL | 2 | SETBP1 | 1 | ||
| TET2 | 12 | Total group | 6–5 | ||
| Total group | 49–39 |
| Main Classification | Increased Protein Levels in Cells with High Constitutive Release | Increased Protein Levels in Cells with Low Constitutive Release |
|---|---|---|
| Nucleosome | MBD3 | |
| Chromatin, histone, transcription, RNA | TOLLIP, NFKB1 | HIF0, HISTIH2AJ, MTA1, SMARCE1, MEN1, MBD3, POLR1E, CLPX, POLR1A, POLR1B |
| DNA repair | CLPX, JUND, POG2 | |
| Oncogene | CBL, DBNL | |
| Cell cycle regulation | IL16 | |
| Intracellular signaling | SYK, HCLS1, AKAP1, TLR2, TOLLIP, AGTRAP, ANXA2, CECR1, INPP5D, LPKN, IKBKB, TBK1 | |
| Tyrosine kinase | SYK, HCLS1, FGR, PKN1 | |
| SRC tyrosine kinases | HCLS1, FGR, HCK, | |
| PI3K-Akt-mTOR | NCF4 | |
| RAC1 | RAC1, NCF4, RHOT1, ARHGEF1, PKN1, RHOG, ARHGAP30, PREX1, GMIP, DOK2, AKAP1 | |
| GTPase | DNM2, ARHGEF1, PKN1, RHOG, ARHGAP30, PREX1, GMIP, AKAP1, ARHGAP, RAB27A | |
| G-protein coupled receptors | ARRB2, ARHGEF1, PREX1, GRK6 | |
| Phagocytosis | CYBA, NCF2, NCF4, ELMO2 | |
| Protein degradation | CBL, SERPINA1 | |
| Intracellular trafficking | VAMP3, DNM2, PICALM, SNX18, ARAP1, ARAP1, TOLLIP, AP1G2, S100A10, S100A4, TOM1, SDCDP, DNAJC13, EPN1, APHGAP, RAB27A | |
| Microtubule, cytoskeleton, structure | DNM2, EPN1, SH3KBP1, PKN1, RHOG, AHNAK, SDCDP, S100A4, CKAP4, FAM49B | |
| Cell migration | PLXNB2, HCK, DNM2, RHOG, ELMO2, AHNAK | |
| Mitochondria, metabolism | FAM49B, FTL, IMPDH1, PDXK | CLPX |
| Lysosomes | CTSH, CTSS, CTSZ, LYZ, PSAP | |
| Cell metabolism, NADP | HCK, NCF4 | |
| Cytokinesis | FMNL1 | |
| Extracellular matrix, cell adhesion | EPN1, SH3KBP1 | |
| Extracellular mediators | IL16, TLR2, TOLLIP | |
| Cell surface molecules | ITGAL, ITGAM. ITGB2, SYK, LILRB2, PKN1, LPXN | |
| Integrins | ITGAL, ITGAM. ITGB2, SYK, FGR, LPXN | |
| Viability, apoptosis | SH3KBP1, PKN1, ARAP1, TLR2 | |
| AML | CBL, PICALM | |
| Differentiation | MNDA, NCF1, CECR1 |
| Low constitutive mediator release; list of significant GO-terms | Protein number | Fold enrichment | FDR | |
| Cell compartment | GO:0005654—nucleoplasm | 31 | 2.8 | 2.3 × 10–5 |
| GO:0000790—nuclear chromatin | 8 | 11 | 0.0099 | |
| GO:0005736—DNA-directed RNA polymerase I complex | 4 | 80 | 0.017 | |
| Molecular function | GO:0003713—transcription coactivator activity | 9 | 8.5 | 0.011 |
| GO:0001054—RNA polymerase I activity | 4 | 78 | 0.018 | |
| High constitutive mediator release; list of significant GO-terms | ||||
| Biological processes | GO:0006954—inflammatory response | 19 | 5.0 | 6.5 × 10–5 |
| GO:0045087—innate immune response | 20 | 4.7 | 8.3 × 10–5 | |
| GO:0048010—vascular endothelial growth factor receptor signaling pathway | 9 | 13 | 8.6 × 10–4 | |
| GO:0007229—integrin-mediated signaling pathway | 10 | 10 | 9.4 × 10–4 | |
| GO:0031623—receptor internalization | 7 | 16 | 0.0062 | |
| GO:0007165—signal transduction | 29 | 2.5 | 0.015 | |
| GO:0098609—cell–cell adhesion | 13 | 4.8 | 0.026 | |
| Cell compartment | GO:0070062—extracellular exosome | 73 | 2.7 | 1.4 × 10–13 |
| GO:0005829—cytosol | 79 | 2.5 | 5.7 × 10–13 | |
| GO:0016020—membrane | 48 | 2.3 | 7.8 × 10–5 | |
| GO:0043020—NADPH oxidase complex | 5 | 43 | 0.0048 | |
| GO:0005737—cytoplasm | 78 | 1.6 | 0.010 | |
| GO:0030670—phagocytic vesicle membrane | 7 | 12 | 0.026 | |
| GO:0005925—focal adhesion | 15 | 4.0 | 0.03 | |
| GO:0045121—membrane raft | 11 | 5.6 | 0.038 | |
| GO:0005884—actin filament | 7 | 11 | 0.046 | |
| Molecular function | GO:0005515—protein binding | 129 | 1.4 | 5.8 × 10–6 |
| GO:0017124—SH3 domain binding | 11 | 8.9 | 5.8 × 10–4 | |
| GO:0035325—Toll-like receptor binding | 4 | 96 | 0.0058 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reikvam, H.; Aasebø, E.; Brenner, A.K.; Bartaula-Brevik, S.; Grønningsæter, I.S.; Forthun, R.B.; Hovland, R.; Bruserud, Ø. High Constitutive Cytokine Release by Primary Human Acute Myeloid Leukemia Cells Is Associated with a Specific Intercellular Communication Phenotype. J. Clin. Med. 2019, 8, 970. https://doi.org/10.3390/jcm8070970
Reikvam H, Aasebø E, Brenner AK, Bartaula-Brevik S, Grønningsæter IS, Forthun RB, Hovland R, Bruserud Ø. High Constitutive Cytokine Release by Primary Human Acute Myeloid Leukemia Cells Is Associated with a Specific Intercellular Communication Phenotype. Journal of Clinical Medicine. 2019; 8(7):970. https://doi.org/10.3390/jcm8070970
Chicago/Turabian StyleReikvam, Håkon, Elise Aasebø, Annette K. Brenner, Sushma Bartaula-Brevik, Ida Sofie Grønningsæter, Rakel Brendsdal Forthun, Randi Hovland, and Øystein Bruserud. 2019. "High Constitutive Cytokine Release by Primary Human Acute Myeloid Leukemia Cells Is Associated with a Specific Intercellular Communication Phenotype" Journal of Clinical Medicine 8, no. 7: 970. https://doi.org/10.3390/jcm8070970
APA StyleReikvam, H., Aasebø, E., Brenner, A. K., Bartaula-Brevik, S., Grønningsæter, I. S., Forthun, R. B., Hovland, R., & Bruserud, Ø. (2019). High Constitutive Cytokine Release by Primary Human Acute Myeloid Leukemia Cells Is Associated with a Specific Intercellular Communication Phenotype. Journal of Clinical Medicine, 8(7), 970. https://doi.org/10.3390/jcm8070970







