Maternal Cardiovascular Risk Assessment 3-to-11 Years Postpartum in Relation to Previous Occurrence of Pregnancy-Related Complications
Abstract
1. Introduction
2. Results
2.1. The Clinical Characteristics of Normal and Complicated Pregnancies
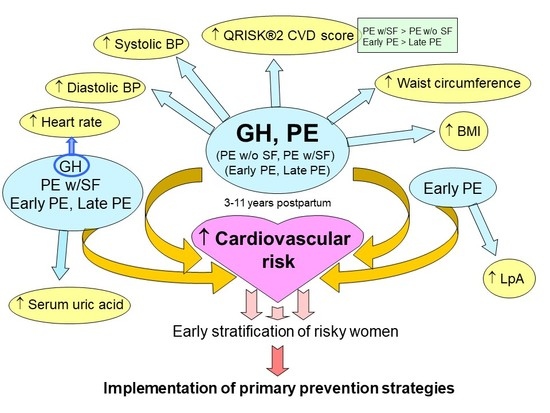
2.2. Impact of A History of Gestational Hypertension and Preeclampsia Irrespective of the Severity of the Disease on Maternal Cardiovascular Risk
2.3. Impact of a History of Preeclampsia without and with Severe Features on Maternal Cardiovascular Risk
2.4. Impact of A History of Early and Late Preeclampsia on Maternal Cardiovascular Risk
3. Discussion
4. Materials and Methods
4.1. Participants
4.2. Blood Pressure Measurements
4.3. BMI and Waist Circumference Measurements
4.4. Biological Sampling
4.5. Estimation of Individual and Relative Risks of Having a Heart Attack or Stroke Over the Next Ten Years
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| PE | Preeclampsia |
| FGR | Fetal growth restriction |
| GH | Gestational hypertension |
| SGA | Small for gestational age |
| DV | Ductus venosus |
| CPR | Cerebro-placental ratio |
| PI | Pulsatility index |
| HDL | High-density lipoprotein |
| LDL | Low-density lipoprotein |
| CRP | C-reactive protein |
| BMI | Body mass index |
| SBP | Systolic blood pressure |
| DBP | Diastolic blood pressure |
| FPR | False positive rate |
References
- Libby, G.; Murphy, D.J.; McEwan, N.F.; Greene, S.A.; Forsyth, J.S.; Chien, P.W.; Morris, A.D.; DARTS/MEMO Collaboration. Pre-eclampsia and the later development of type 2 diabetes in mothers and their children: An intergenerational study from the Walker cohort. Diabetologia 2007, 50, 523–530. [Google Scholar] [CrossRef] [PubMed]
- Garovic, V.D.; Hayman, S.R. Hypertension in pregnancy: An emerging risk factor for cardiovascular disease. Nat. Clin. Pract. Nephrol. 2007, 3, 613–622. [Google Scholar] [CrossRef]
- Männistö, T.; Mendla, P.; Vääräsmäki, M.; Järvelin, M.R.; Hartikainen, A.L.; Pouta, A.; Suvanto, E. Elevated blood pressure in pregnancy and subsequent chronic disease risk. Circulation 2013, 127, 681–690. [Google Scholar] [CrossRef] [PubMed]
- Bellamy, L.; Casas, J.P.; Hingorani, A.D.; Williams, D.J. Pre-eclampsia and risk of cardiovascular disease and cancer in later life: Systematic review and meta-analysis. BMJ 2007, 335, 974. [Google Scholar] [CrossRef] [PubMed]
- Craici, I.M.; Wagner, S.J.; Hayman, S.R.; Garovic, V.D. Pre-eclamptic pregnancies: An opportunity to identify women at risk for future cardiovascular disease. Womens Health 2008, 4, 133–135. [Google Scholar] [CrossRef] [PubMed]
- Haukkamaa, L.; Moilanen, L.; Kattainen, A.; Luoto, R.; Kahonen, M.; Leinonen, M.; Jula, A.; Kesäniemi, Y.A.; Kaaja, R. Pre-eclampsia is a risk factor of carotid artery atherosclerosis. Cerebrovasc. Dis. 2009, 27, 599–607. [Google Scholar] [CrossRef] [PubMed]
- Lykke, J.A.; Langhoff-Roos, J.; Sibai, B.M.; Funai, E.F.; Triche, E.W.; Paidas, M.J. Hypertensive pregnancy disorders and subsequent cardiovascular morbidity and type 2 diabetes mellitus in the mother. Hypertension 2009, 53, 944–951. [Google Scholar] [CrossRef]
- Berks, D.; Hoedjes, M.; Raat, H.; Duvekot, J.J.; Steegers, E.A.; Habbema, J.D. Risk of cardiovascular disease after pre-eclampsia and the effect of lifestyle interventions: A literature-based study. BJOG 2013, 120, 924–931. [Google Scholar] [CrossRef]
- McDonald, S.D.; Ray, J.; Teo, K.; Jung, H.; Salehian, O.; Yusuf, S.; Lonn, E. Measures of cardiovascular risk and subclinical atherosclerosis in a cohort of women with a remote history of preeclampsia. Atherosclerosis 2013, 229, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Veerbeek, J.H.; Hermes, W.; Breimer, A.Y.; van Rijn, B.B.; Koenen, S.V.; Mol, B.W.; Franx, A.; de Groot, C.J.; Koster, M.P. Cardiovascular disease risk factors after early-onset preeclampsia, late-onset preeclampsia, and pregnancy-induced hypertension. Hypertension 2015, 65, 600–606. [Google Scholar] [CrossRef]
- Irgens, H.U.; Reisaeter, L.; Irgens, L.M.; Lie, R.T. Long term mortality of mothers and fathers after pre-eclampsia: Population based cohort study. BMJ 2001, 323, 1213–1217. [Google Scholar] [CrossRef] [PubMed]
- Mongraw-Chaffin, M.L.; Cirillo, P.M.; John, B.A. Preeclampsia and cardiovascular disease death: Prospective evidence from the child health and development studies cohort. Hypertension 2010, 56, 166–171. [Google Scholar] [CrossRef] [PubMed]
- Ray, J.G.; Vermeulen, M.J.; Schull, M.J.; Redelmeier, D.A. Cardiovascular health after maternal placental syndromes (CHAMPS): Population-based retrospective cohort study. Lancet 2005, 366, 1797–1803. [Google Scholar] [CrossRef]
- American College of Obstetricians and Gynecologists; Task Force on Hypertension in Pregnancy. Hypertension in pregnancy. Report of the American College of Obstetricians and Gynecologists’ Task Force on Hypertension in Pregnancy. Obstet. Gynecol. 2013, 122, 1122–1131. [Google Scholar]
- Borna, S.; Neamatipoor, E.; Radman, N. Risk of coronary artery disease in women with history of pregnancies complicated by preeclampsia and LBW. J. Matern. Fetal Neonatal Med. 2012, 25, 1114–1116. [Google Scholar] [CrossRef]
- Thilaganathan, B. Placental syndromes: Getting to the heart of the matter. Ultrasound Obstet. Gynecol. 2017, 49, 7–9. [Google Scholar] [CrossRef]
- Thilaganathan, B. Association of Higher Maternal Blood Pressure with Lower Infant Birthweight: Placental Cause or Cardiovascular Effect? Hypertension 2016, 67, 499–500. [Google Scholar] [CrossRef]
- Brosens, I.; Pijnenborg, R.; Vercruysse, L.; Romero, R. The “Great Obstetrical Syndromes” are associated with disorders of deep placentation. Am. J. Obstet. Gynecol. 2011, 204, 193–201. [Google Scholar] [CrossRef]
- Mosca, L.; Benjamin, E.J.; Berra, K.; Bezanson, J.L.; Dolor, R.J.; Lloyd-Jones, D.M.; Newby, L.K.; Piña, I.L.; Roger, V.L.; Shaw, L.J.; et al. Effectiveness-based guidelines for the prevention of cardiovascular disease in women—2011 update: A guideline from the American Heart Association. Circulation 2011, 123, 1243–1262, Erratum in: Circulation 2011, 123, e624. Circulation 2011, 124, e427. [Google Scholar] [CrossRef]
- Karumanchi, S.A.; Granger, J.P. Preeclampsia and Pregnancy-Related Hypertensive Disorders. Hypertension 2016, 67, 238–242. [Google Scholar] [CrossRef]
- Pouta, A.; Hartikainen, A.L.; Sovio, U.; Gissler, M.; Laitinen, J.; McCarthy, M.I.; Ruokonen, A.; Elliott, P.; Järvelin, M.R. Manifestations of metabolic syndrome after hypertensive pregnancy. Hypertension 2004, 43, 825–831. [Google Scholar] [CrossRef]
- Hermes, W.; Tamsma, J.T.; Grootendorst, D.C.; Franx, A.; van der Post, J.; van Pampus, M.G.; Bloemenkamp, K.W.; Porath, M.; Mol, B.W.; de Groot, C.J. Cardiovascular risk estimation in women with a history of hypertensive pregnancy disorders at term: A longitudinal follow-up study. BMC Pregnancy Childbirth 2013, 13, 126. [Google Scholar] [CrossRef] [PubMed]
- Aykas, F.; Solak, Y.; Erden, A.; Bulut, K.; Dogan, S.; Sarli, B.; Acmaz, G.; Afsar, B.; Siriopol, D.; Covic, A.; et al. Persistence of cardiovascular risk factors in women with previous preeclampsia: A long-term follow-up study. J. Investig. Med. 2015, 63, 641–645. [Google Scholar] [CrossRef]
- Romagnuolo, I.; Sticchi, E.; Attanasio, M.; Tritoni, E.; Cioni, G.; Cella, A.P.; Abbate, R.; Fatini, C. Searching for a common mechanism for placenta-mediated pregnancy complications and cardiovascular disease: Role of lipoprotein(a). Fertil. Steril. 2016, 105, 1287–1293.e3. [Google Scholar] [CrossRef] [PubMed]
- Dunietz, G.L.; Strutz, K.L.; Holman, C.; Tian, Y.; Todem, D.; Bullen, B.L.; Dativ, J.M. Moderately elevated blood pressure during pregnancy and odds of hypertension later in life: The POUCHmoms longitudinal study. BJOG 2017, 124, 1606–1613. [Google Scholar] [CrossRef]
- Fatma, J.; Karoli, R.; Siddiqui, Z.; Gupta, H.P.; Chandra, A.; Pandey, M. Cardio-metabolic Risk Profile in Women with Previous History of Pre-Eclampsia. J. Assoc. Physicians India 2017, 65, 23–27. [Google Scholar]
- Bergen, N.E.; Schalekamp-Timmermans, S.; Roos-Hesselink, J.; Roeters van Lennep, J.E.; Jaddoe, V.V.W.; Steegers, E.A.P. Hypertensive disorders of pregnancy and subsequent maternal cardiovascular health. Eur. J. Epidemiol. 2018, 33, 763–771. [Google Scholar] [CrossRef]
- Benschop, L.; Bergen, N.E.; Schalekamp-Timmermans, S.; Jaddoe, V.W.V.; Mulder, M.T.; Steegers, E.A.P.; Roeters van Lennep, J.E. Maternal lipid profile 6 years after a gestational hypertensive disorder. J. Clin. Lipidol. 2018, 12, 428–436.e4. [Google Scholar] [CrossRef] [PubMed]
- Escouto, D.C.; Green, A.; Kurlak, L.; Walker, K.; Loughna, P.; Chappell, L.; Broughton Pipkin, F.; Bramham, K. Postpartum evaluation of cardiovascular disease risk for women with pregnancies complicated by hypertension. Pregnancy Hypertens. 2018, 13, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Drost, J.T.; Arpaci, G.; Ottervanger, J.P.; de Boer, M.J.; van Eyck, J.; van der Schouw, Y.T.; Maas, A.H. Cardiovascular risk factors in women 10 years post early preeclampsia: The Preeclampsia Risk EValuation in FEMales study (PREVFEM). Eur. J. Prev. Cardiol. 2012, 19, 1138–1144. [Google Scholar] [CrossRef] [PubMed]
- Nisell, H.; Erikssen, C.; Persson, B.; Carlström, K. Is carbohydrate metabolism altered among women who have undergone a preeclamptic pregnancy? Gynecol. Obstet. Investig. 1999, 48, 241–246. [Google Scholar] [CrossRef]
- Visser, S.; Hermes, W.; Blom, H.J.; Heijboer, A.C.; Franx, A.; Van Pampus, M.G.; Bloemenkamp, K.W.; Koopmans, C.; Mol, B.W.; De Groot, C.J. Homocysteinemia After Hypertensive Pregnancy Disorders at Term. J. Womens Health (Larchmt) 2015, 24, 524–529. [Google Scholar] [CrossRef] [PubMed]
- Orabona, R.; Sciatti, E.; Vizzardi, E.; Bonadei, I.; Prefumo, F.; Valcamonico, A.; Metra, M.; Frusca, T. Maternal endothelial function and vascular stiffness after HELLP syndrome: A case-control study. Ultrasound Obstet. Gynecol. 2017, 50, 596–602. [Google Scholar] [CrossRef]
- Markovitz, A.R.; Stuart, J.J.; Horn, J.; Williams, P.L.; Rimm, E.B.; Missmer, S.A.; Tanz, L.J.; Haug, E.B.; Fraser, A.; Timpka, S.; et al. Does pregnancy complication history improve cardiovascular disease risk prediction? Findings from the HUNT study in Norway. Eur. Heart J. 2018. [Google Scholar] [CrossRef]
- Timpka, S.; Fraser, A.; Schyman, T.; Start, J.J.; Åsvold, B.O.; Migrén, I.; Franks, P.W.; Rich-Edwards, J.W. The value of pregnancy complication history for 10-year cardiovascular disease risk prediction in middle-aged women. Eur. J. Epidemiol. 2018, 33, 1003–1010. [Google Scholar] [CrossRef]
- Cunningham, M.W., Jr.; LaMarca, B. Risk of cardiovascular disease, end-stage renal disease, and stroke in postpartum women and their fetuses after a hypertensive pregnancy. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2018, 315, R521–R528. [Google Scholar] [CrossRef] [PubMed]
- Groenhof, T.K.J.; Zoet, G.A.; Franx, A.; Gansevoort, R.T.; Bots, M.L.; Groen, H.; Lely, A.T.; PREVEND Group. Trajectory of Cardiovascular Risk Factors After Hypertensive Disorders of Pregnancy. Hypertension 2019, 73, 171–178. [Google Scholar] [CrossRef] [PubMed]
- Gladstone, R.A.; Pudwell, J.; Nerenberg, K.A.; Grover, S.A.; Smith, G.N. Cardiovascular Risk Assessment and Follow-Up of Women After Hypertensive Disorders of Pregnancy: A Prospective Cohort Study. J. Obstet. Gynaecol. Can. 2019, pii: S1701-2163(18)30836-3. [Google Scholar] [CrossRef] [PubMed]
- Alsnes, I.V.; Janszky, I.; Forman, M.R.; Vatten, L.J.; Økland, I. A population-based study of associations between preeclampsia and later cardiovascular risk factors. Am. J. Obstet. Gynecol. 2014, 211, 657.e1–657.e7. [Google Scholar] [CrossRef]
- Weissgerber, T.L.; Milic, N.M.; Turner, S.T.; Asad, R.A.; Mosley, T.H., Jr.; Kardiak, S.L.; Hanis, C.L.; Garovic, V.D. Uric Acid: A Missing Link Between Hypertensive Pregnancy Disorders and Future Cardiovascular Disease? Mayo Clin. Proc. 2015, 90, 1207–1216. [Google Scholar] [CrossRef]
- Borghi, C.; Verardi, F.M.; Pareo, I.; Bentivenga, C.; Cicero, A.F. Hyperuricemia and cardiovascular disease risk. Expert Rev. Cardiovasc. Ther. 2014, 12, 1219–1225. [Google Scholar] [CrossRef] [PubMed]
- Braga, F.; Pasqualetti, S.; Ferraro, S.; Panteghini, M. Hyperuricemia as risk factor for coronary heart disease incidence and mortality in the general population: A systematic review and meta-analysis. Clin. Chem. Lab. Med. 2016, 54, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Capuano, V.; Marchese, F.; Capuano, R.; Torre, S.; Iannone, A.G.; Capuano, E.; Lamaida, N.; Sonderegger, M.; Capuano, E. Hyperuricemia as an independent risk factor for major cardiovascular events: A 10-year cohort study from Southern Italy. J. Cardiovasc. Med. 2017, 18, 159–164. [Google Scholar] [CrossRef] [PubMed]
- Van Pampus, M.G.; Koopman, M.M.; Wolf, H.; Büller, H.R.; Prins, M.H.; van den Ende, A. Lipoprotein(a) concentrations in women with a history of severe preeclampsia—A case control study. Thromb. Haemost. 1999, 82, 10–13. [Google Scholar]
- Leerink, C.B.; de Vries, C.V.; van der Klis, F.R. Elevated levels of serum lipoprotein(a) and apolipoprotein(a) phenotype are not related to pre-eclampsia. Acta Obstet. Gynecol. Scand. 1997, 76, 625–628. [Google Scholar] [CrossRef] [PubMed]
- Manten, G.T.; Sikkema, M.J.; Voorbij, H.A.; Visser, G.H.; Bruinse, H.W.; Franx, A. Risk factors for cardiovascular disease in women with a history of pregnancy complicated by preeclampsia or intrauterine growth restriction. Hypertens. Pregnancy 2007, 26, 39–50. [Google Scholar] [CrossRef] [PubMed]
- Wilde, C. Hidden Causes of Heart Attack and Stroke: Inflammation, Cardiology’s New Frontier, 1st ed.; Abigon Press: Studio City, CA, USA, 2003; pp. 182–183. [Google Scholar]
- Schreiner, P.J.; Morrisett, J.D.; Sharrett, A.R.; Patsch, W.; Tyroler, H.A.; Wu, K.; Weiss, G. Lipoprotein[a] as a risk factor for preclinical atherosclerosis. Arterioscler. Thromb. 1993, 13, 826–833. [Google Scholar] [CrossRef]
- Sotiriou, S.N.; Orlova, V.V.; Al-Fakhri, N.; Ihanus, E.; Economopoulou, M.; Isermann, B.; Bdeir, K.; Nawroth, P.P.; Preissner, K.T.; Gahmberg, C.G.; et al. Lipoprotein(a) in atherosclerotic plaques recruits inflammatory cells through interaction with Mac-1 integrin. FASEB J. 2006, 20, 559–661. [Google Scholar] [CrossRef]
- Ichikawa, T.; Unoki, H.; Sun, H.; Shimoyamada, H.; Marcovina, S.; Shikama, H.; Watanabe, T.; Fan, J. Lipoprotein(a) promotes smooth muscle cell proliferation and dedifferentiation in atherosclerotic lesions of human apo(a) transgenic rabbits. Am. J. Pathol. 2002, 160, 227–236. [Google Scholar] [CrossRef]
- Tsimikas, S.; Witztum, J.L. The role of oxidized phospholipids in mediating lipoprotein(a) atherogenicity. Curr. Opin. Lipidol. 2008, 19, 369–377. [Google Scholar] [CrossRef]
- Gouni-Berthold, I.; Berthold, H.K. Lipoprotein(a): Current perspectives. Curr. Vasc. Pharmacol. 2011, 9, 682–692. [Google Scholar] [CrossRef] [PubMed]
- Germain, A.M.; Romanik, M.C.; Guerra, I.; Solari, S.; Reyes, M.S.; Johnson, R.J.; Price, K.; Karumanchi, S.A.; Valdés, G. Endothelial dysfunction: A link among preeclampsia, recurrent pregnancy loss, and future cardiovascular events? Hypertension 2007, 49, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Weissgerber, T.L.; Turner, S.T.; Bailey, K.R.; Mosley, T.H., Jr.; Kardia, S.L.; Wiste, H.J.; Miller, V.M.; Kullo, I.J.; Garovic, V.D. Hypertension in pregnancy is a risk factor for peripheral arterial disease decades after pregnancy. Atherosclerosis 2013, 229, 212–216. [Google Scholar] [CrossRef] [PubMed]
- Tanz, L.J.; Start, J.J.; Missmer, S.A.; SIMM, E.B.; Sumer, J.A.; Vadnais, M.A.; Rich-Edwards, J.W. Cardiovascular biomarkers in the years following pregnancies complicated by hypertensive disorders or delivered preterm. Pregnancy Hypertens. 2018, 13, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Sep, S.; Verbeek, J.; Koek, G.; Smits, L.; Spaanderman, M.; Peeters, L. Clinical differences between early-onset HELLP syndrome and early-onset preeclampsia during pregnancy and at least 6 months postpartum. Am. J. Obstet. Gynecol. 2010, 202, 271.e1–271.e5. [Google Scholar] [CrossRef]
- ACOG practice bulletin. Diagnosis and management of preeclampsia and eclampsia. Obstet. Gynecol. 2002, 99, 159–167. [Google Scholar]
- Vyas, S.; Nicolaides, K.H.; Bower, S.; Campbell, S. Middle cerebral artery flow velocity waveforms in fetal hypoxaemia. Br. J. Obstet. Gynaecol. 1990, 97, 797–803. [Google Scholar] [CrossRef]
- Cohn, H.E.; Sacks, E.J.; Heymann, M.A.; Rudolph, A.M. Cardiovascular responses to hypoxemia and acidemia in fetal lambs. Am. J. Obstet. Gynecol. 1974, 120, 817–824. [Google Scholar] [CrossRef]
- Practice Guidelines: New AHA Recommendations for Blood Pressure Measurement. Am. Fam. Physician 2005, 72, 1391–1398.
- Nordestgaard, B.G.; Chapman, M.J.; Ray, K.; Borén, J.; Andreotti, F.; Watts, G.F.; Ginsberg, H.; Amarenco, P.; Catapano, A.; Descamps, O.S.; et al. Lipoprotein(a) as a cardiovascular risk factor: Current status. Eur. Heart J. 2010, 31, 2844–2853. [Google Scholar] [CrossRef]
- Harvard Health Publishing. Harvard Medical School; Published: March, 2014. Available online: https://www.health.harvard.edu/newsletter_article/Exercise_C-reactive_protein_and_your_heart (accessed on 26 February 2019).
- Huitema, B. Analysis of Covariance and Alternatives, 1st ed.; Wiley: New York, NY, USA, 1980. [Google Scholar]
- Liu, D.; Zhou, X.H. ROC analysis in biomarker combination with covariate adjustment. Acad. Radiol. 2013, 20, 874–882. [Google Scholar] [CrossRef] [PubMed][Green Version]
| Normotensive Term Pregnancies (n = 90) | PE (n = 102) | FGR (n = 34) | GH (n = 50) | p-Value 1 | p-Value 2 | p-Value 3 | |
|---|---|---|---|---|---|---|---|
| Pre-existing cardiovascular risk factors before gestation | |||||||
| DM type I | 0 (0%) | 1 (0.98%) | 0 (0%) | 1 (2.0%) | - | - | - |
| DM type II | 0 (0%) | 0 (0%) | 1 (2.94%) | 0 (0%) | - | - | - |
| Rheumatoid arthritis | 0 (0%) | 0 (0%) | 1 (2.94%) | 2 (4.0%) | - | - | - |
| Angina or heart attack in a first degree relative before the age of 60 years | 2 (2.22%) | 0 (0%) | 0 (0%) | 1 (2.0%) | - | - | - |
| On blood pressure treatment | 0 (0%) | 7 (6.86%) | 1 (2.94%) | 0 (0%) | - | - | - |
| Hypercholesterolemia | 0 (0%) | 0 (0%) | 0 (0%) | 1 (2.0%) | - | - | - |
| Dispensarisation at Dpt. of Cardiology (valve problems and heart defects) | 0 (0%) | 1 (0.98%) Sinus tachycardia | 1 (2.94%) Leaky heart valve | 1 (2.0%) Mitral valve prolapse | - | - | - |
| Chronic venous insufficiency | 0 (0%) | 0 (0%) | 0 (0%) | 1 (2%) | - | - | - |
| Thrombosis | 0 (0%) | 2 (1.96%) | 0 (0%) | 0 (0%) | - | - | - |
| Presence of risk factors for chronic kidney disease | 0 (%) | 1 (0.98%) Haematuria | 0 (0%) | 3 (6.0%) Abnormal kidney structure (n = 2) Glomerulonephritis in childhood (n = 1) | - | - | - |
| Chronic kidney disease | 0 (%) | 1 (0.98%) Nephrotic syndrome | 0 (0%) | 0 (0%) | - | - | - |
| At follow-up | |||||||
| Age (years) | 38.33 ± 0.45 | 38.05 ± 0.42 | 37.0 ± 0.74 | 39.2 ± 0.61 | 1.000 | 0.765 | 1.000 |
| Time elapsed since delivery (years) | 5.73 ± 0.21 | 5.33 ± 0.20 | 5.05 ± 0.35 | 5.06 ± 0.28 | 1.000 | 0.610 | 0.374 |
| Glucose status | 0.654 | 0.346 | 0.096 | ||||
| Normal | 86 (95.56%) | 96 (94.12%) | 31 (91.18%) | 44 (88.00%) | |||
| DM/GDM | 4 (4.44%) | 6 (5.88%) | 3 (8.82%) | 6 (12.00%) | |||
| Smoking | 0.999 | 0.619 | 0.941 | ||||
| Non-Smoker | 55 (61.11%) | 62 (61.39%) | 24 (70.59%) | 32 (64.00%) | |||
| Ex-smoker | 21 (23.33%) | 24 (23.53%) | 6 (17.65%) | 11 (22.00%) | |||
| Smoker | 14 (15.56%) | 16 (15.69%) | 4 (11.76%) | 7 (14.00%) | |||
| Hormonal contraceptive use | 0.086 | 0.379 | 0.248 | ||||
| No | 37 (41.11%) | 30 (29.41%) | 10 (29.41%) | 18 (36.00%) | |||
| In the past | 31 (34.44%) | 51 (50.00%) | 16 (47.06%) | 24 (48.00%) | |||
| Yes | 22 (24.44%) | 21 (20.59) | 8 (23.53%) | 8 (16.00%) | |||
| Total number of pregnancies per patient | <0.001 | 0.007 | 0.169 | ||||
| 1 | 8 (8.89%) | 29 (28.43%) | 9 (26.47%) | 10 (20.00%) | |||
| 2 | 16 (51.11%) | 42 (41.18%) | 16 (47.06%) | 19 (38.00%) | |||
| 3+ | 36 (40.00%) | 31 (30.39%) | 9 (26.47%) | 21 (42.00%) | |||
| Total parity per patient | <0.001 | 0.046 | 0.025 | ||||
| 1 | 13 (14.44%) | 40 (39.22%) | 11 (32.36%) | 17 (34.00%) | |||
| 2 | 63 (70.00%) | 52 (50.98%) | 21 (61.76%) | 27 (54.00%) | |||
| 3+ | 14 (15.56%) | 10 (9.80%) | 2 (5.71%) | 6 (12%) | |||
| During gestation | |||||||
| Maternal age at delivery (years) | 32.64 ± 0.42 | 32.54 ± 0.40 | 31.91 ± 0.69 | 34.08 ± 0.57 | 1.000 | 1.000 | 0.274 |
| GA at delivery (weeks) | 39.91 ± 0.28 | 35.97 ± 0.27 | 35.65 ± 0.46 | 38.73 ± 0.38 | <0.001 | <0.001 | 0.987 |
| Fetal birth weight (g) | 3402.44 ± 70.01 | 2416.74 ± 65.76 | 1910.00 ± 113.91 | 3242.40 ± 93.93 | <0.001 | <0.001 | 1.000 |
| Mode of delivery | <0.001 | <0.001 | <0.001 | ||||
| Vaginal | 83 (92.22%) | 19 (18.63%) | 7 (20.59%) | 22 (44.00%) | |||
| CS | 7 (7.78%) | 83 (81.37%) | 27 (79.41%) | 28 (56.00%) | |||
| Fetal sex | 0.315 | 0.740 | 0.405 | ||||
| Boy | 48 (53.33%) | 47 (46.08%) | 17 (50.00%) | 23 (54.00%) | |||
| Girl | 42 (46.67%) | 55 (53.92%) | 17 (50.00%) | 27 (46.00%) | |||
| Blood pressure (mmHg) | |||||||
| Systolic | 120.57 ± 1.51 | 158.38 ± 1.42 | 128.08 ± 2.44 | 147.79 ± 2.03 | <0.001 | 0.092 | <0.001 |
| Diastolic | 75.77 ± 1.03 | 98.74 ± 0.97 | 79.26 ± 1.66 | 93.89 ± 1.38 | <0.001 | 0.451 | <0.001 |
| Infertility treatment | 0.027 | 0.004 | 0.096 | ||||
| Yes | 4 (4.44%) | 14 (13.73%) | 7 (20.59%) | 6 (12.00%) | |||
| No | 86 (95.65%) | 88 (86.27%) | 27 (79.41%) | 44 (88.00%) | |||
| NTP (n = 90) | FGR (n = 34) | GH (n = 50) | PE (n = 102) | Diagnostic Groups (Normal vs Diseased) | p Value (ANOVA, ANCOVA) | ||
|---|---|---|---|---|---|---|---|
| Serum uric acid (μmol/L) | Unadjusted data | 248.044 (6.212) | 278.529 (10.051) | 286.081 (8.372) | 276.792 (5.832) | NTP vs ↑FGR | p = 0.094 |
| NTP vs ↑ GH | p = 0.004 | ||||||
| NTP vs ↑ PE | p = 0.005 | ||||||
| BMI | Unadjusted data | 23.100 (0.517) | 23.928 (0.941) | 27.228 (0.694) | 25.946 (0.486) | NTP vs ↑ GH | p < 0.001 |
| NTP vs ↑ PE | p < 0.001 | ||||||
| Adjusted data | 23.138 (0.525) A | 24.529 (0.860) A | 26.987 (0.694) A | 25.945 (0.490) A | NTP vs ↑ GH | p = 0.002 | |
| NTP vs ↑ PE | p = 0.001 | ||||||
| Waist circumference (cm) | Unadjusted data | 76.605 (1.277) | 78.264 (2.078) | 86.770 (1.713) | 83.852 (1.199) | NTP vs ↑ GH | p < 0.001 |
| NTP vs ↑ PE | p < 0.001 | ||||||
| Adjusted data | 76.891 (1.298) A | 79.546 (2.128) A | 86.118 (1.716) A | 83.624 (1.212) A | NTP vs ↑ GH | p = 0.001 | |
| NTP vs ↑ PE | p = 0.001 | ||||||
| SBP (mmHg) | Unadjusted data | 112.911 (1.316) | 118.088 (2.142) | 129.580 (1.766) | 123.656 (1.236) | NTP vs ↑ GH | p < 0.001 |
| NTP vs ↑ PE | p < 0.001 | ||||||
| Adjusted data | 114.895 (1.286) A | 118.325 (2.058) A | 127.517 (1.683) A | 122.642 (1.178) A | NTP vs ↑ GH | p < 0.001 | |
| NTP vs ↑ PE | p = 0.029 | ||||||
| DBP (mmHg) | Unadjusted data | 72.100 (1.000) | 77.058 (1.627) | 82.940 (1.342) | 79.490 (0.939) | NTP vs ↑ GH | p < 0.001 |
| NTP vs ↑ PE | p < 0.001 | ||||||
| Adjusted data | 73.898 (0.977) A | 77.091 (1.564) A | 81.295 (1.279) A | 78.737 (0.895) A | NTP vs ↑ GH | p < 0.001 | |
| NTP vs ↑ PE | p = 0.002 | ||||||
| Heart rate (bpm) | Unadjusted data | 71.644 (1.079) | 73.969 (1.782) | 76.300 (1.447) | 72.627 (1.013) | NTP vs ↑GH | p = 0.062 |
| Adjusted data | 71.705 (1.141) A | 74.294 (1.861) A | 76.262 (1.497) A | 72.670 (1.048) A | NTP vs ↑GH | p = 0.119 | |
| Relative QRISK®2 risk score | Unadjusted data | 0.920 (0.134) | 1.379 (0.216) | 1.966 (0.178) | 1.617 (0.125) | NTP vs ↑ GH | p < 0.001 |
| NTP vs ↑ PE | p < 0.001 | ||||||
| Adjusted data | 0.865 (0.118) A | 1.420 (0.195) A | 1.984 (0.155) A | 1.578 (0.111) A | NTP vs ↑ GH | p < 0.001 | |
| NTP vs ↑ PE | p < 0.001 |
| Diagnostic Groups (Normal vs Diseased) | ROC Curve Parameters | Sensitivity at 10% FPR | Sensitivity and Specificity When Critical Values are Exceeded | ||
|---|---|---|---|---|---|
| Serum uric acid (μmol/L) | Unadjusted data | NTP vs FGR | AUC 0.615, p = 0.056 | 32.35% Criterion > 308.5 μmol/L | 26.46% sensitivity at 97.5% specificity Criterion > 340.55 μmol/L (hyperuricemia) |
| NTP vs GH | AUC 0.670, p < 0.001 | 36.53% Criterion > 308.5 μmol/L | 14.29% sensitivity at 97.5% specificity Criterion > 340.55 μmol/L (hyperuricemia) | ||
| NTP vs PE | AUC 0.644, p < 0.001 | 28.51% Criterion > 311.2 μmol/L | 13.09% sensitivity at 97.5% specificity Criterion > 340.775 μmol/L (hyperuricemia) | ||
| BMI | Unadjusted data | NTP vs GH | AUC 0.738, p < 0.001 | 42.00% Criterion > 27.78 (overweight) | 20.0% sensitivity at 97.5% specificity Criterion > 31.02 (obese class I, moderately obese) |
| NTP vs PE | AUC 0.670, p < 0.001 | 27.45% Criterion > 27.78 (overweight) | 18.63% sensitivity at 97.5% specificity Criterion > 31.02 (obese class I, moderately obese) | ||
| Adjusted data | NTP vs GH | AUC 0.899, p < 0.001 | 74.00% | - | |
| NTP vs PE | AUC 0.791, p < 0.001 | 53.06% | - | ||
| Waist circumference (cm) | Unadjusted data | NTP vs GH | AUC 0.743, p < 0.001 | 42.00% Criterion > 87 cm | 36.0% sensitivity at 91.11% specificity Criterion > 88 cm (obese, high cardiovascular risk) |
| NTP vs PE | AUC 0.688, p < 0.001 | 32.35% Criterion > 87 cm | 29.41% sensitivity at 91.11% specificity Criterion > 88 cm (obese, high cardiovascular risk) | ||
| Adjusted data | NTP vs GH | AUC 0.902, p < 0.001 | 70.00% | - | |
| NTP vs PE | AUC 0.796, p < 0.001 | 54.08% | - | ||
| SBP (mmHg) | Unadjusted data | NTP vs GH | AUC 0.843, p < 0.001 | 54.00% Criterion > 123.4 mmHg (prehypertension) | 18.00% sensitivity at 100.0% specificity Criterion > 141 mmHg (hypertension) |
| NTP vs PE | AUC 0.750, p < 0.001 | 46.86% Criterion > 123.4 mmHg (prehypertension) | 10.78% sensitivity at 100.0% specificity Criterion > 141 mm Hg (hypertension) | ||
| Adjusted data | NTP vs GH | AUC 0.822, p < 0.001 | 62.00% | - | |
| NTP vs PE | AUC 0.754, p < 0.001 | 51.10% | - | ||
| DBP (mmHg) | Unadjusted data | NTP vs GH | AUC 0.794, p < 0.001 | 51.00% Criterion > 80.5 mmHg (prehypertension) | 20.0% sensitivity at 100.0% specificity Criterion > 91 mmHg (hypertension) |
| NTP vs PE | AUC 0.714, p < 0.001 | 39.71% Criterion > 80.5 mmHg (prehypertension) | 12.75% sensitivity at 100.0% specificity Criterion > 91 mmHg (hypertension) | ||
| Adjusted data | NTP vs GH | AUC 0.875, p < 0.001 | 60.00% | - | |
| NTP vs PE | AUC 0.778, p < 0.001 | 47.96% | - | ||
| Heart rate (bpm) | Unadjusted data | NTP vs GH | AUC 0.619, p = 0.017 | 18.00% Criterion >84 bpm | 4.0% sensitivity at 100.0% specificity Criterion > 107 bpm (tachycardia) |
| Adjusted data | NTP vs GH | AUC 0.833, p < 0.001 | 54.00% | - | |
| Relative QRISK®2 risk score | Unadjusted data | NTP vs GH | AUC 0.789, p < 0.001 | 30.00% Criterion > 1.60 | 18.0% sensitivity at 100.0% specificity Criterion > 2.9 |
| NTP vs PE | AUC 0.711, p < 0.001 | 26.37% Criterion > 1.60 | 12.75% sensitivity at 100.0% specificity Criterion > 2.9 | ||
| Adjusted data | NTP vs GH | AUC 0.894, p < 0.001 | 74.00% | - | |
| NTP vs PE | AUC 0.788, p < 0.001 | 55.10% | - |
| NTP (n = 90) | PE w/o SF (n = 25) | PE w/SF (n = 77) | Diagnostic Groups (Normal vs Diseased) | p Value (ANOVA, ANCOVA) | ||
|---|---|---|---|---|---|---|
| Serum uric acid (μmol/L) | Unadjusted data | 248.044 (5.649) | 275.880 (10.659) | 277.092 (6.113) | NTP vs ↑ PE w/SF | p = 0.001 |
| BMI | Unadjusted data | 23.100 (0.480) | 26.037 (0.912) | 25.916 (0.519) | NTP vs ↑ PE w/o SF | p = 0.021 |
| NTP vs ↑ PE w/SF | p < 0.001 | |||||
| Adjusted data | 23.518 (0.479) A | 26.300 (0.883) A | 25.378 (0.518) A | NTP vs ↑ PE w/o SF | p = 0.021 | |
| NTP vs ↑ PE w/SF | p = 0.015 | |||||
| Waist circumference (cm) | Unadjusted data | 76.605 (1.191) | 83.480 (2.260) | 83.974 (1.287) | NTP vs ↑ PE w/o SF | p = 0.010 |
| NTP vs ↑ PE w/SF | p < 0.001 | |||||
| Adjusted data | 77.796 (1.206) A | 83.607 (2.225) A | 82.416 (1.304) A | NTP vs ↑ PE w/o SF | p = 0.029 | |
| NTP vs ↑ PE w/SF | p = 0.013 | |||||
| SBP (mmHg) | Unadjusted data | 112.911(1.203) | 122.160 (2.283) | 124.142 (1.301) | NTP vs ↑ PE w/o SF | p = 0.001 |
| NTP vs ↑ PE w/SF | p < 0.001 | |||||
| Adjusted data | 116.949 (0.743) A | 120.994 (1.379) A | 119.791 (0.810) A | NTP vs ↑ PE w/o SF | p = 0.055 | |
| NTP vs ↑ PE w/SF | p = 0.121 | |||||
| DBP (mmHg) | Unadjusted data | 72.100 (0.934) | 77.520 (1.773) | 80.129 (1.010) | NTP vs ↑ PE w/o SF | p = 0.014 |
| NTP vs ↑ PE w/SF | p < 0.001 | |||||
| Adjusted data | 72.896 (0.948) A | 77.841 (1.810) A | 79.376 (1.037) A | NTP vs ↑ PE w/o SF | p = 0.036 | |
| NTP vs ↑ PE w/SF | p < 0.001 | |||||
| Relative QRISK®2 risk score | Unadjusted data | 0.920 (0.116) | 1.228 (0.220) | 1.744 (0.125) | NTP vs ↑ PE w/o SF | p = 0.008 |
| NTP vs ↑ PE w/SF | p < 0.001 | |||||
| Adjusted data | 1.060 (0.098) A | 1.201 (0.182) A | 1.553 (0.106) A | NTP vs ↑ PE w/o SF | p = 0.003 | |
| NTP vs ↑ PE w/SF | p < 0.001 |
| Diagnostic Groups (Normal vs Diseased) | ROC Curve Parameters | Sensitivity at 10% FPR | Sensitivity and Specificity When Critical Values are Exceeded | ||
|---|---|---|---|---|---|
| Serum uric acid (μmol/L) | Unadjusted data | NTP vs PE w/SF | AUC 0.648, p < 0.001 | 28.82% Criterion > 311.2 μmol/L | 13.45% sensitivity at 97.5% specificity Criterion > 340.775 μmol/L (hyperuricemia) |
| BMI | Unadjusted data | NTP vs PE w/o SF | AUC 0.666, p = 0.007 | 20.00% Criterion > 27.78 (overweight) | 20.0% sensitivity at 97.5% specificity Criterion > 31.02 (obese class I, moderately obese) |
| NTP vs PE w/SF | AUC 0.672, p < 0.001 | 29.87% Criterion > 27.78 (overweight) | 18.18% sensitivity at 97.5% specificity Criterion > 31.02 (obese class I, moderately obese) | ||
| Adjusted data | NTP vs PE w/o SF | AUC 0.853, p < 0.001 | 62.50% | - | |
| NTP vs PE w/SF | AUC 0.781, p < 0.001 | 56.76% | - | ||
| Waist circumference (cm) | Unadjusted data | NTP vs PE w/o SF | AUC 0.702, p = 0.001 | 28.00% Criterion > 87 cm | 24.0% sensitivity at 91.11% specificity Criterion > 88 cm (obese, high cardiovascular risk) |
| NTP vs PE w/SF | AUC 0.683, p < 0.001 | 33.77% Criterion > 87 cm | 31.17% sensitivity at 91.11% specificity Criterion > 88 cm (obese, high cardiovascular risk) | ||
| Adjusted data | NTP vs PE w/o SF | AUC 0.847, p < 0.001 | 58.33% | - | |
| NTP vs PE w/SF | AUC 0.788, p < 0.001 | 55.41% | - | ||
| SBP (mmHg) | Unadjusted data | NTP vs PE w/o SF | AUC 0.740, p < 0.001 | 40.00% Criterion > 123.4 mmHg (prehypertension) | 4.00% sensitivity at 100.0% specificity Criterion > 141 mm Hg (hypertension) |
| NTP vs PE w/SF | AUC 0.753, p < 0.001 | 49.09% Criterion > 123.4 mmHg (prehypertension) | 12.99% sensitivity at 100.0% specificity Criterion > 141 mm Hg (hypertension) | ||
| Adjusted data | NTP vs PE w/o SF | AUC 0.805, p < 0.001 | 62.50% | - | |
| NTP vs PE w/SF | AUC 0.762, p < 0.001 | 56.76% | - | ||
| DBP (mmHg) | Unadjusted data | NTP vs PE w/o SF | AUC 0.669, p = 0.002 | 24.00% Criterion > 80.5 mmHg (prehypertension) | 12.00% sensitivity at 100.0% specificity Criterion > 91 mm Hg (hypertension) |
| NTP vs PE w/SF | AUC 0.729, p < 0.001 | 44.81% Criterion > 80.5 mmHg (prehypertension) | 12.99% sensitivity at 100.0% specificity Criterion > 91 mm Hg (hypertension) | ||
| Adjusted data | NTP vs PE w/o SF | AUC 0.729, p < 0.001 | 45.83% | - | |
| NTP vs PE w/SF | AUC 0.747, p < 0.001 | 50.00% | - | ||
| Relative QRISK®2 risk score | Unadjusted data | NTP vs PE w/o SF | AUC 0.723, p < 0.001 | 19.87% Criterion > 1.60 | 0.0% sensitivity at 100.0% specificity Criterion > 2.9 |
| NTP vs PE w/SF | AUC 0.707, p < 0.001 | 28.48% Criterion > 1.60 | 16.88% sensitivity at 100.0% specificity Criterion > 2.9 | ||
| Adjusted data | NTP vs PE w/o SF | AUC 0.843, p < 0.001 | 58.33% | - | |
| NTP vs PE w/SF | AUC 0.782, p < 0.001 | 54.05% | - |
| NTP (n = 90) | Early PE (n = 36) | Late PE (n = 66) | Diagnostic Groups (Normal vs Diseased) | p Value (ANOVA, ANCOVA) | ||
|---|---|---|---|---|---|---|
| Serum Lp(a) (nmol/L) | Unadjusted data | 36.449 (7.509) | 90.828 (11.974) | 45.483 (8.720) | NTP vs ↑ early PE | p = 0.037 |
| Adjusted data | 38.909 (8.094) A | 88.341 (13.631) A | 46.977 (8.921) A | NTP vs early PE | p = 0.192 | |
| Serum uric acid (μmol/L) | Unadjusted data | 248.044 (5.625) | 285.971 (8.971) | 271.924 (6.532) | NTP vs ↑ early PE | p = 0.001 |
| NTP vs ↑ late PE | p = 0.018 | |||||
| BMI | Unadjusted data | 23.100 (0.465) | 28.085 (0.735) | 24.779 (0.543) | NTP vs ↑ early PE | p < 0.001 |
| NTP vs ↑ late PE | p = 0.034 | |||||
| Adjusted data | 23.440 (0.475) A | 26.948 (0.784) A | 24.998 (0.535) A | NTP vs early PE | p = 0.003 | |
| NTP vs ↑ late PE | p = 0.031 | |||||
| Waist circumference (cm) | Unadjusted data | 76.605 (1.161) | 88.472 (1.836) | 81.333 (1.356) | NTP vs ↑ early PE | p < 0.001 |
| NTP vs ↑ late PE | p = 0.006 | |||||
| Adjusted data | 77.654 (1.202) A | 85.201 (1.983) A | 81.578 (1.353) A | NTP vs ↑ early PE | p = 0.011 | |
| NTP vs ↑ late PE | p = 0.018 | |||||
| SBP (mmHg) | Unadjusted data | 112.911(1.178) | 128.055 (1.864) | 121.257 (1.376) | NTP vs ↑ early PE | p < 0.001 |
| NTP vs ↑ late PE | p < 0.001 | |||||
| Adjusted data | 116.931 (0.746) A | 120.196 (1.243) A | 120.052 (0.845) A | NTP vs ↑early PE | p = 0.099 | |
| NTP vs ↑ late PE | p = 0.021 | |||||
| DBP (mmHg) | Unadjusted data | 72.100 (0.914) | 83.166 (1.446) | 77.484 (1.068) | NTP vs ↑ early PE | p < 0.001 |
| NTP vs ↑ late PE | p < 0.001 | |||||
| Adjusted data | 72.837 (0.937) A | 81.848 (1.554) A | 77.570 (1.091) A | NTP vs ↑ early PE | p < 0.001 | |
| NTP vs ↑ late PE | p = 0.004 | |||||
| Relative QRISK®2 risk score | Unadjusted data | 0.920 (0.108) | 2.436 (0.170) | 1.171 (0.125) | NTP vs ↑ early PE | p < 0.001 |
| NTP vs ↑ late PE | p = 0.009 | |||||
| Adjusted data | 1.036 (0.093) A | 2.099 (0.151) A | 1.162 (0.105) A | NTP vs ↑ early PE | p < 0.001 | |
| NTP vs ↑ late PE | p = 0.005 |
| Diagnostic Groups (Normal vs Diseased) | ROC Curve Parameters | Sensitivity at 10% FPR | Sensitivity and Specificity When Critical Values are Exceeded | ||
|---|---|---|---|---|---|
| Serum Lp(a) (nmol/L) | Unadjusted data | NTP vs early PE | AUC 0.632, p = 0.022 | 31.43% Criterion > 89.26 nmol/L (risk of CVD) | 34.29% sensitivity at 86.52% specificity Criterion > 73.20 nmol/L (risk of CVD) |
| Adjusted data | NTP vs early PE | AUC 0.905, p < 0.001 | 81.82% | - | |
| Serum uric acid (μmol/L) | Unadjusted data | NTP vs early PE | AUC 0.667, p = 0.004 | 42.86% Criterion > 308.5 μmol/L | 20.00% sensitivity at 97.5% specificity Criterion > 340.77 μmol/L (hyperuricemia) |
| NTP vs late PE | AUC 0.632, p = 0.003 | 20.91% Criterion > 311.2 μmol/L | 9.43% sensitivity at 97.5% specificity Criterion > 340.55 μmol/L (hyperuricemia) | ||
| BMI | Unadjusted data | NTP vs early PE | AUC 0.748, p < 0.001 | 38.89% Criterion > 27.78 (overweight) | 33.33% sensitivity at 97.5% specificity Criterion > 31.02 (obese class I, moderately obese) |
| NTP vs late PE | AUC 0.628, p = 0.004 | 21.21% Criterion >27.78 (overweight) | 10.61% sensitivity at 97.5% specificity Criterion > 31.02 (obese class I, moderately obese) | ||
| Adjusted data | NTP vs early PE | AUC 0.894, p < 0.001 | 67.65% | - | |
| NTP vs late PE | AUC 0.767, p < 0.001 | 48.44% | - | ||
| Waist circumference (cm) | Unadjusted data | NTP vs early PE | AUC 0.740, p < 0.001 | 50.00% Criterion > 87 cm | 44.44% sensitivity at 91.11% specificity Criterion > 88 cm (obese, high cardiovascular risk) |
| NTP vs late PE | AUC 0.659, p < 0.001 | 22.73%Criterion > 87 cm | 21.21% sensitivity at 91.11% specificity Criterion > 88 cm (obese, high cardiovascular risk) | ||
| Adjusted data | NTP vs early PE | AUC 0.890, p < 0.001 | 64.71% | - | |
| NTP vs late PE | AUC 0.775, p < 0.001 | 50.00% | - | ||
| SBP (mmHg) | Unadjusted data | NTP vs early PE | AUC 0.859, p < 0.001 | 62.78% Criterion > 123.4 mmHg (prehypertension) | 16.67% sensitivity at 100.0% specificity Criterion > 141 mmHg (hypertension) |
| NTP vs late PE | AUC 0.690, p < 0.001 | 38.18% Criterion > 123.4 mmHg (prehypertension) | 7.58% sensitivity at 100.0% specificity Criterion > 141 mmHg (hypertension) | ||
| Adjusted data | NTP vs early PE | AUC 0.884, p < 0.001 | 70.59% | - | |
| NTP vs late PE | AUC 0.724, p < 0.001 | 46.88% | - | ||
| DBP (mmHg) | Unadjusted data | NTP vs early PE | AUC 0.824, p < 0.001 | 54.17% Criterion > 80.5 mmHg (prehypertension) | 16.67% sensitivity at 100.0% specificity Criterion > 91 mmHg (hypertension) |
| NTP vs late PE | AUC 0.654, p < 0.001 | 31.82% Criterion > 80.5 mmHg (prehypertension) | 10.61% sensitivity at 100.0% specificity Criterion > 91 mmHg (hypertension) | ||
| Adjusted data | NTP vs early PE | AUC 0.874, p < 0.001 | 64.71% | - | |
| NTP vs late PE | AUC 0.704, p < 0.001 | 34.38% | - | ||
| Relative QRISK®2 risk score | Unadjusted data | NTP vs early PE | AUC 0.802, p < 0.001 | 41.67% Criterion > 1.60 | 33.33% sensitivity at 100.0% specificity Criterion > 2.9 |
| NTP vs late PE | AUC 0.661, p < 0.001 | 18.03% Criterion > 1.60 | 1.52% sensitivity at 100.0% specificity Criterion > 2.9 | ||
| Adjusted data | NTP vs early PE | AUC 0.886, p < 0.001 | 73.53% | - | |
| NTP vs late PE | AUC 0.749, p < 0.001 | 51.56% | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hromadnikova, I.; Kotlabova, K.; Dvorakova, L.; Krofta, L. Maternal Cardiovascular Risk Assessment 3-to-11 Years Postpartum in Relation to Previous Occurrence of Pregnancy-Related Complications. J. Clin. Med. 2019, 8, 544. https://doi.org/10.3390/jcm8040544
Hromadnikova I, Kotlabova K, Dvorakova L, Krofta L. Maternal Cardiovascular Risk Assessment 3-to-11 Years Postpartum in Relation to Previous Occurrence of Pregnancy-Related Complications. Journal of Clinical Medicine. 2019; 8(4):544. https://doi.org/10.3390/jcm8040544
Chicago/Turabian StyleHromadnikova, Ilona, Katerina Kotlabova, Lenka Dvorakova, and Ladislav Krofta. 2019. "Maternal Cardiovascular Risk Assessment 3-to-11 Years Postpartum in Relation to Previous Occurrence of Pregnancy-Related Complications" Journal of Clinical Medicine 8, no. 4: 544. https://doi.org/10.3390/jcm8040544
APA StyleHromadnikova, I., Kotlabova, K., Dvorakova, L., & Krofta, L. (2019). Maternal Cardiovascular Risk Assessment 3-to-11 Years Postpartum in Relation to Previous Occurrence of Pregnancy-Related Complications. Journal of Clinical Medicine, 8(4), 544. https://doi.org/10.3390/jcm8040544