Mitochondrial DNA Haplogroup JT is Related to Impaired Glycaemic Control and Renal Function in Type 2 Diabetic Patients
Abstract
1. Introduction
2. Experimental Section
2.1. Subjects
2.2. Anthropometric and Biochemical Parameters
2.3. Haplotyping
2.4. Statistical Analysis
3. Results
3.1. Clinical Characteristics of the Study Population
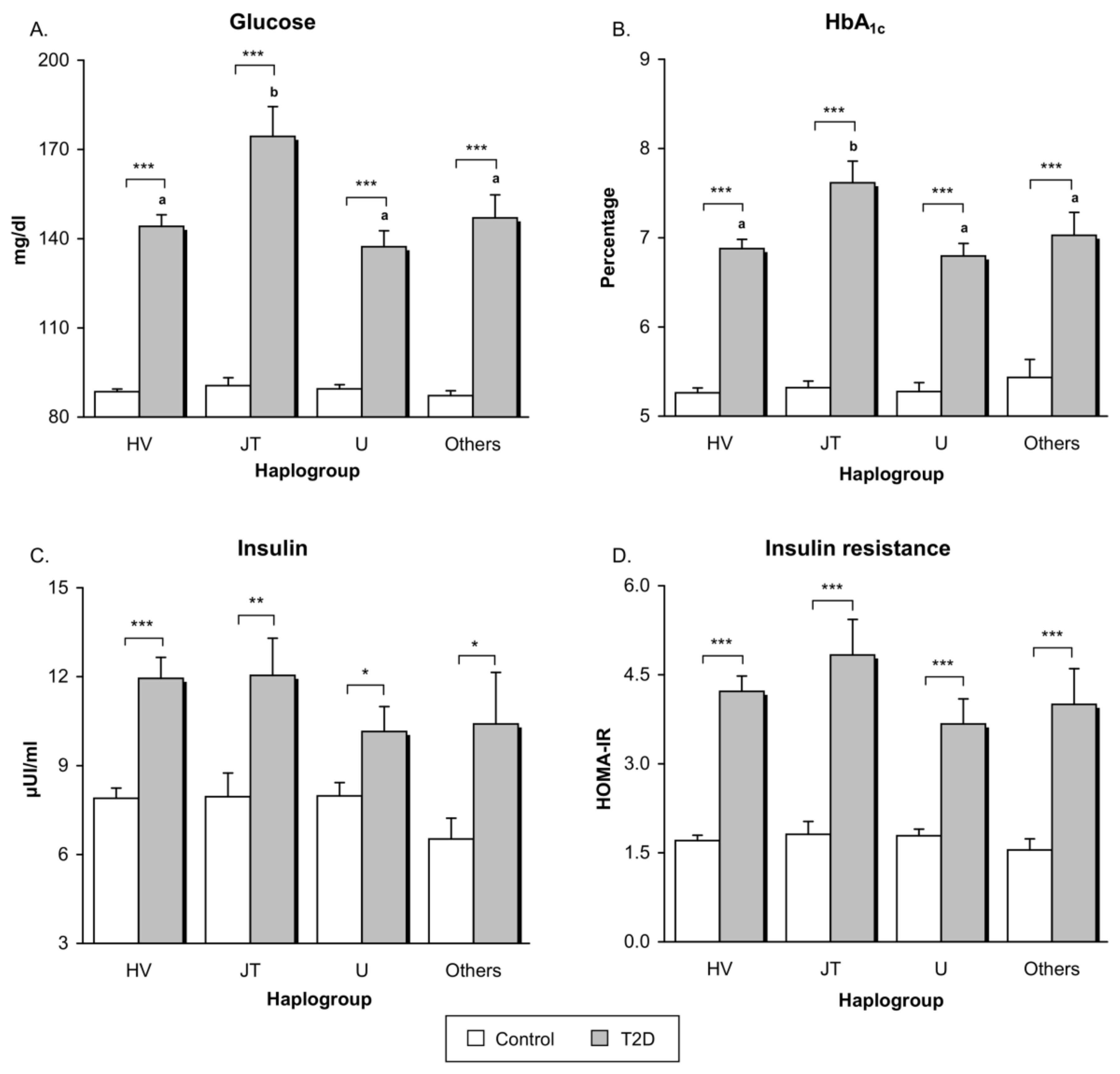
3.2. Glucose Metabolism
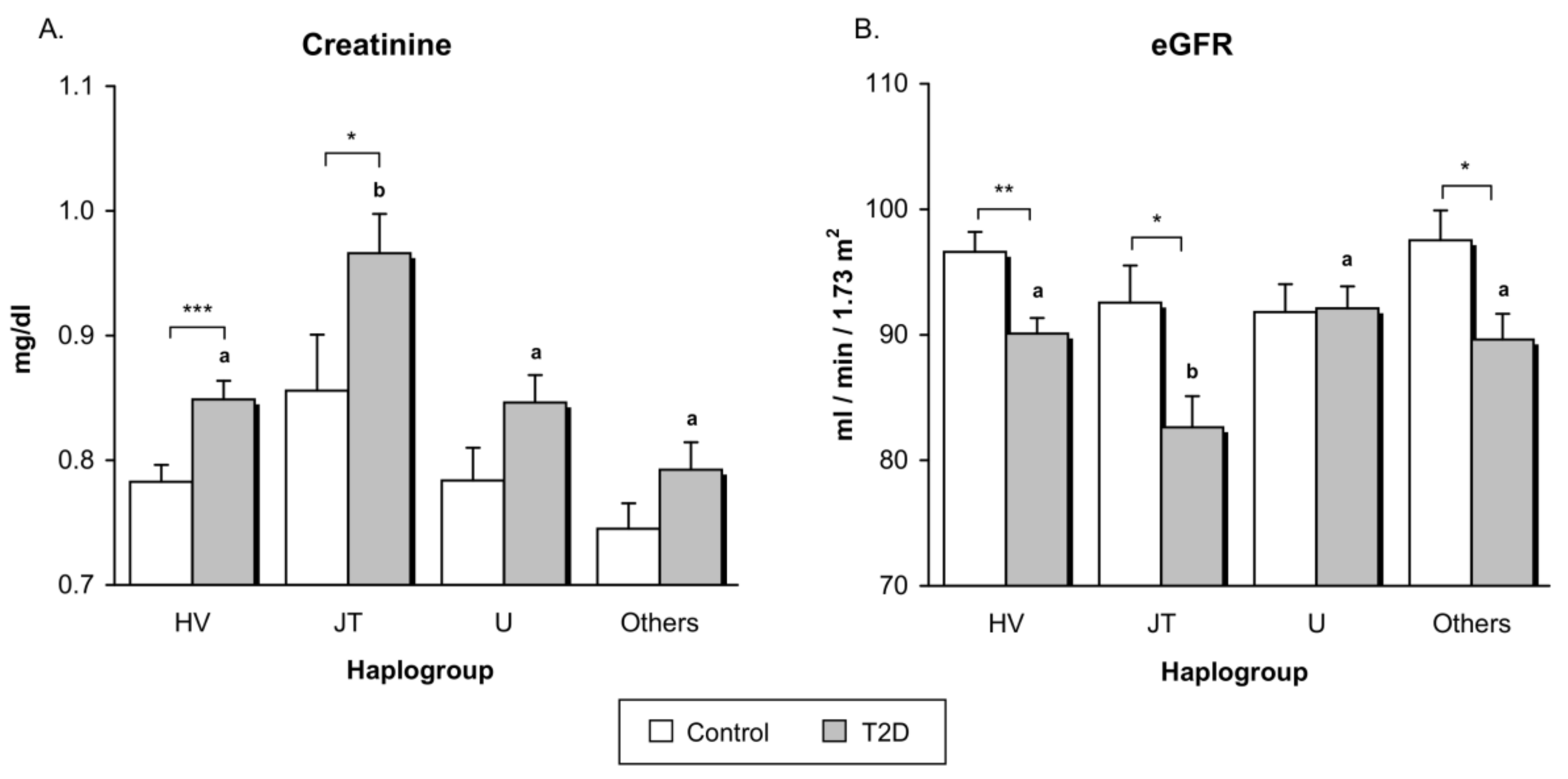
3.3. Renal Function
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix
| Healthy Controls | Type 2 Diabetic Patients | p-value | Age, Sex, and BMI Adjusted p-value | |
|---|---|---|---|---|
| Glucose (mg/dL) | 89.04 ± 8.93 | 147.39 ± 50.87 | <0.001 | <0.001 |
| HbA1c (%) | 5.28 ± 0.32 | 6.99 ± 1.32 | <0.001 | <0.001 |
| Insulin (μUI/mL) | 7.84 ± 2.98 | 11.50 ± 5.91 | <0.001 | 0.013 |
| HOMA-IR | 1.73 ± 0.76 | 4.21 ± 2.39 | <0.001 | <0.001 |
| Creatinine (mg/dL) | 0.79 ± 0.12 | 0.86 ± 0.17 | <0.001 | 0.12 |
| eGFR (mL/min/1.73 m2) | 94.92 ± 9.93 | 89.39 ± 13.19 | <0.001 | 0.04 |
References
- Ali, O. Genetics of type 2 diabetes. World J. Diabetes 2013, 4, 114–123. [Google Scholar] [CrossRef] [PubMed]
- Hara, K.; Shojima, N.; Hosoe, J.; Kadowaki, T. Genetic architecture of type 2 diabetes. Biochem. Biophys. Res. Commun. 2014, 452, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Supale, S.; Li, N.; Brun, T.; Maechler, P. Mitochondrial dysfunction in pancreatic beta cells. Trends Endocrinol. Metab. 2012, 23, 477–487. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Franquesa, A.; Patti, M.E. Insulin Resistance and Mitochondrial Dysfunction. Adv. Exp. Med. Biol. 2017, 982, 465–520. [Google Scholar] [CrossRef] [PubMed]
- Kwak, S.H.; Park, K.S. Role of mitochondrial DNA variation in the pathogenesis of diabetes mellitus. Front. Biosci. (Landmark Ed.) 2016, 21, 1151–1167. [Google Scholar] [PubMed]
- Van den Ouweland, J.M.; Lemkes, H.H.; Ruitenbeek, W.; Sandkuijl, L.A.; de Vijlder, M.F.; Struyvenberg, P.A.; van de Kamp, J.J.; Maassen, J.A. Mutation in mitochondrial tRNA(Leu)(UUR) gene in a large pedigree with maternally transmitted type II diabetes mellitus and deafness. Nat. Genet. 1992, 1, 368–371. [Google Scholar] [CrossRef] [PubMed]
- Reardon, W.; Ross, R.J.; Sweeney, M.G.; Luxon, L.M.; Pembrey, M.E.; Harding, A.E.; Trembath, R.C. Diabetes mellitus associated with a pathogenic point mutation in mitochondrial DNA. Lancet 1992, 340, 1376–1379. [Google Scholar] [CrossRef]
- Park, K.S.; Chan, J.C.; Chuang, L.M.; Suzuki, S.; Araki, E.; Nanjo, K.; Ji, L.; Ng, M.; Nishi, M.; Furuta, H.; et al. Study group of molecular diabetology in Asia A mitochondrial DNA variant at position 16189 is associated with type 2 diabetes mellitus in Asians. Diabetologia 2008, 51, 602–608. [Google Scholar] [CrossRef] [PubMed]
- Ye, Z.; Gillson, C.; Sims, M.; Khaw, K.T.; Plotka, M.; Poulton, J.; Langenberg, C.; Wareham, N.J. The association of the mitochondrial DNA OriB variant (16184–16193 polycytosine tract) with type 2 diabetes in Europid populations. Diabetologia 2013, 56, 1907–1913. [Google Scholar] [CrossRef] [PubMed]
- Crispim, D.; Canani, L.H.; Gross, J.L.; Tschiedel, B.; Souto, K.E.; Roisenberg, I. The European-specific mitochondrial cluster J/T could confer an increased risk of insulin-resistance and type 2 diabetes: An analysis of the m.4216T>C and m.4917A>G variants. Ann. Hum. Genet. 2006, 70, 488–495. [Google Scholar] [CrossRef] [PubMed]
- Niu, Q.; Zhang, W.; Wang, H.; Guan, X.; Lu, J.; Li, W. Effects of mitochondrial haplogroup N9a on type 2 diabetes mellitus and its associated complications. Exp. Ther. Med. 2015, 10, 1918–1924. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Li, R.; Zhang, Y.; Wang, P.; Wu, T.; Lin, J.; Yu, J.; Gu, M. Mitochondrial DNA Mutations Associated with Type 2 Diabetes Mellitus in Chinese Uyghur Population. Sci. Rep. 2017, 7, 16989. [Google Scholar] [CrossRef] [PubMed]
- Saxena, R.; de Bakker, P.I.; Singer, K.; Mootha, V.; Burtt, N.; Hirschhorn, J.N.; Gaudet, D.; Isomaa, B.; Daly, M.J.; Groop, L.; et al. Comprehensive association testing of common mitochondrial DNA variation in metabolic disease. Am. J. Hum. Genet. 2006, 79, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Chinnery, P.F.; Mowbray, C.; Patel, S.K.; Elson, J.L.; Sampson, M.; Hitman, G.A.; McCarthy, M.I.; Hattersley, A.T.; Walker, M. Mitochondrial DNA haplogroups and type 2 diabetes: A study of 897 cases and 1010 controls. J. Med. Genet. 2007, 44, e80. [Google Scholar] [CrossRef] [PubMed]
- Nathan, D.M.; Turgeon, H.; Regan, S. Relationship between glycated haemoglobin levels and mean glucose levels over time. Diabetologia 2007, 50, 2239–2244. [Google Scholar] [CrossRef] [PubMed]
- Klein, R. Hyperglycemia and microvascular and macrovascular disease in diabetes. Diabetes Care 1995, 18, 258–268. [Google Scholar] [CrossRef] [PubMed]
- Stratton, I.M.; Adler, A.I.; Neil, H.A.; Matthews, D.R.; Manley, S.E.; Cull, C.A.; Hadden, D.; Turner, R.C.; Holman, R.R. Association of glycaemia with macrovascular and microvascular complications of type 2 diabetes (UKPDS 35): Prospective observational study. BMJ 2000, 321, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Achilli, A.; Olivieri, A.; Pala, M.; Hooshiar Kashani, B.; Carossa, V.; Perego, U.A.; Gandini, F.; Santoro, A.; Battaglia, V.; Grugni, V.; et al. Mitochondrial DNA backgrounds might modulate diabetes complications rather than T2DM as a whole. PLoS ONE 2011, 6, e21029. [Google Scholar] [CrossRef] [PubMed]
- Estopinal, C.B.; Chocron, I.M.; Parks, M.B.; Wade, E.A.; Roberson, R.M.; Burgess, L.G.; Brantley, M.A., Jr.; Samuels, D.C. Mitochondrial haplogroups are associated with severity of diabetic retinopathy. Invest. Ophthalmol. Vis. Sci. 2014, 55, 5589–5595. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association 2. Classification and Diagnosis of Diabetes. Diabetes Care 2017, 40, S11–S24. [Google Scholar] [CrossRef] [PubMed]
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.L.; Castro, A.F., 3rd; Feldman, H.I.; Kusek, J.W.; Eggers, P.; Van Lente, F.; Greene, T.; et al. CKD-EPI (Chronic Kidney Disease Epidemiology Collaboration) A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 2009, 150, 604–612. [Google Scholar] [CrossRef] [PubMed]
- Van Oven, M.; Kayser, M. Updated comprehensive phylogenetic tree of global human mitochondrial DNA variation. Hum. Mutat. 2009, 30, E386–E394. [Google Scholar] [CrossRef] [PubMed]
- Dahmany, Y.; Marcuello, A.; Montiel-Sosa, F.; Martínez-Redondo, D.; Montoya, J.; Díez-Sánchez, C.; López-Pérez, M.; Ruiz-Pesini, E. Mitochondrial lineages distribution in the Spanish population: Anticipating association studies. An. R. Acad. Nac. Farm. 2006, 72, 37–47. [Google Scholar]
- Nogales-Gadea, G.; Pinos, T.; Ruiz, J.R.; Marzo, P.F.; Fiuza-Luces, C.; Lopez-Gallardo, E.; Ruiz-Pesini, E.; Martin, M.A.; Arenas, J.; Moran, M.; et al. Are mitochondrial haplogroups associated with elite athletic status? A study on a Spanish cohort. Mitochondrion 2011, 11, 905–908. [Google Scholar] [CrossRef] [PubMed]
- Olsson, A.H.; Yang, B.T.; Hall, E.; Taneera, J.; Salehi, A.; Nitert, M.D.; Ling, C. Decreased expression of genes involved in oxidative phosphorylation in human pancreatic islets from patients with type 2 diabetes. Eur. J. Endocrinol. 2011, 165, 589–595. [Google Scholar] [CrossRef] [PubMed]
- Sharoyko, V.V.; Abels, M.; Sun, J.; Nicholas, L.M.; Mollet, I.G.; Stamenkovic, J.A.; Gohring, I.; Malmgren, S.; Storm, P.; Fadista, J.; et al. Loss of TFB1M results in mitochondrial dysfunction that leads to impaired insulin secretion and diabetes. Hum. Mol. Genet. 2014, 23, 5733–5749. [Google Scholar] [CrossRef] [PubMed]
- Mishmar, D.; Ruiz-Pesini, E.; Golik, P.; Macaulay, V.; Clark, A.G.; Hosseini, S.; Brandon, M.; Easley, K.; Chen, E.; Brown, M.D.; et al. Natural selection shaped regional mtDNA variation in humans. Proc. Natl. Acad. Sci. USA 2003, 100, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Pesini, E.; Mishmar, D.; Brandon, M.; Procaccio, V.; Wallace, D.C. Effects of purifying and adaptive selection on regional variation in human mtDNA. Science 2004, 303, 223–226. [Google Scholar] [CrossRef] [PubMed]
- Feder, J.; Ovadia, O.; Blech, I.; Cohen, J.; Wainstein, J.; Harman-Boehm, I.; Glaser, B.; Mishmar, D. Parental diabetes status reveals association of mitochondrial DNA haplogroup J1 with type 2 diabetes. BMC Med. Genet. 2009, 10, 60. [Google Scholar] [CrossRef] [PubMed]
- Fuku, N.; Park, K.S.; Yamada, Y.; Nishigaki, Y.; Cho, Y.M.; Matsuo, H.; Segawa, T.; Watanabe, S.; Kato, K.; Yokoi, K.; et al. Mitochondrial haplogroup N9a confers resistance against type 2 diabetes in Asians. Am. J. Hum. Genet. 2007, 80, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, M.; Fuku, N.; Nishigaki, Y.; Matsuo, H.; Segawa, T.; Watanabe, S.; Kato, K.; Yokoi, K.; Ito, M.; Nozawa, Y.; et al. Women with mitochondrial haplogroup N9a are protected against metabolic syndrome. Diabetes 2007, 56, 518–521. [Google Scholar] [CrossRef] [PubMed]
- Mohlke, K.L.; Jackson, A.U.; Scott, L.J.; Peck, E.C.; Suh, Y.D.; Chines, P.S.; Watanabe, R.M.; Buchanan, T.A.; Conneely, K.N.; Erdos, M.R.; et al. Mitochondrial polymorphisms and susceptibility to type 2 diabetes-related traits in Finns. Hum. Genet. 2005, 118, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Feder, J.; Blech, I.; Ovadia, O.; Amar, S.; Wainstein, J.; Raz, I.; Dadon, S.; Arking, D.E.; Glaser, B.; Mishmar, D. Differences in mtDNA haplogroup distribution among 3 Jewish populations alter susceptibility to T2DM complications. BMC Genomics 2008, 9, 198. [Google Scholar] [CrossRef] [PubMed]
- Herrnstadt, C.; Elson, J.L.; Fahy, E.; Preston, G.; Turnbull, D.M.; Anderson, C.; Ghosh, S.S.; Olefsky, J.M.; Beal, M.F.; Davis, R.E.; et al. Reduced-median-network analysis of complete mitochondrial DNA coding-region sequences for the major African, Asian, and European haplogroups. Am. J. Hum. Genet. 2002, 70, 1152–1171. [Google Scholar] [CrossRef] [PubMed]
- Kabat, A.; Ponicke, K.; Salameh, A.; Mohr, F.W.; Dhein, S. Effect of a beta 2-adrenoceptor stimulation on hyperglycemia-induced endothelial dysfunction. J. Pharmacol. Exp. Ther. 2004, 308, 564–573. [Google Scholar] [CrossRef] [PubMed]
- Vinogradov, A.D.; Grivennikova, V.G. Oxidation of NADH and ROS production by respiratory complex I. Biochim. Biophys. Acta 2016, 1857, 863–871. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.J. Pathogenesis of chronic hyperglycemia: From reductive stress to oxidative stress. J. Diabetes Res. 2014, 2014, 137919. [Google Scholar] [CrossRef] [PubMed]
- He, Q.; Wang, M.; Petucci, C.; Gardell, S.J.; Han, X. Rotenone induces reductive stress and triacylglycerol deposition in C2C12 cells. Int. J. Biochem. Cell Biol. 2013, 45, 2749–2755. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.L.; Goldfine, I.D.; Maddux, B.A.; Grodsky, G.M. Are oxidative stress-activated signaling pathways mediators of insulin resistance and beta-cell dysfunction? Diabetes 2003, 52, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.; Iqbal, M.; Karam, J.; Salifu, M.; McFarlane, S.I. Oxidative stress, glucose metabolism, and the prevention of type 2 diabetes: Pathophysiological insights. Antioxid. Redox Signal. 2007, 9, 911–929. [Google Scholar] [CrossRef] [PubMed]
- Rocha, M.; Rovira-Llopis, S.; Banuls, C.; Bellod, L.; Falcon, R.; Castello, R.; Morillas, C.; Herance, J.R.; Hernandez-Mijares, A.; Victor, V.M. Mitochondrial dysfunction and oxidative stress in insulin resistance. Curr. Pharm. Des. 2013, 19, 5730–5741. [Google Scholar] [CrossRef] [PubMed]
- Hou, W.L.; Yin, J.; Alimujiang, M.; Yu, X.Y.; Ai, L.G.; Bao, Y.Q.; Liu, F.; Jia, W.P. Inhibition of mitochondrial complex I improves glucose metabolism independently of AMPK activation. J. Cell. Mol. Med. 2018, 22, 1316–1328. [Google Scholar] [CrossRef] [PubMed]
- Brunmair, B.; Staniek, K.; Gras, F.; Scharf, N.; Althaym, A.; Clara, R.; Roden, M.; Gnaiger, E.; Nohl, H.; Waldhausl, W.; et al. Thiazolidinediones, like metformin, inhibit respiratory complex I: A common mechanism contributing to their antidiabetic actions? Diabetes 2004, 53, 1052–1059. [Google Scholar] [CrossRef] [PubMed]
- Nishikawa, T.; Edelstein, D.; Du, X.L.; Yamagishi, S.; Matsumura, T.; Kaneda, Y.; Yorek, M.A.; Beebe, D.; Oates, P.J.; Hammes, H.P.; Giardino, I.; Brownlee, M. Normalizing mitochondrial superoxide production blocks three pathways of hyperglycaemic damage. Nature 2000, 404, 787–790. [Google Scholar] [CrossRef] [PubMed]
- Giacco, F.; Brownlee, M. Oxidative stress and diabetic complications. Circ. Res. 2010, 107, 1058–1070. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.K.; Winocour, P.; Farrington, K. Oxidative stress in early diabetic nephropathy: Fueling the fire. Nat. Rev. Endocrinol. 2011, 7, 176–184. [Google Scholar] [CrossRef] [PubMed]
| Healthy Controls | Type 2 Diabetic Patients | p-value * | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Haplogroup | HV | JT | U | Others | Total | p-value | HV | JT | U | Others | Total | p-value | |
| N (%) | 81 (52.9) | 26 (17.0) | 35 (22.9) | 11 (7.2) | 153 (100) | - | 165 (54.5) | 43 (14.2) | 65 (21.4) | 30 (9.9) | 303 (100) | - | 0.68 |
| Male % | 37.0 | 41.9 | 23.7 | 27.3 | 34.2 | 0.36 | 68.5 | 81.8 | 77.3 | 66.7 | 72.1 | 0.22 | <0.001 |
| Age (years) | 41.3 ± 16.4 | 45.3 ± 16.0 | 41.5 ± 15.7 | 40.0 ± 14.0 | 41.9 ±16.0 | 0.68 | 58.6 ± 9.8 | 59.5 ± 9.3 | 58.4 ± 9.9 | 59.3 ±7.8 | 58.8 ± 9.5 | 0.92 | <0.001 |
| BMI (kg/m2) | 24.4 ± 3.7 | 25.6 ± 4.5 | 25.2 ± 4.2 | 23.7 ± 2.3 | 24.8 ± 24.8 | 0.39 | 30.8 ± 4.8 | 30.2 ± 3.8 | 30.1 ± 4.4 | 31.2 ± 4.2 | 30.6 ± 4.5 | 0.68 | <0.001 |
| WHR | 0.83 ± 0.08 | 0.84 ± 0.13 | 0.85 ± 0.09 | 0.79 ± 0.08 | 0.83 ± 0.09 | 0.41 | 0.98 ± 0.08 | 0.97 ± 0.06 | 1.00 ± 0.08 | 0.98 ± 0.07 | 0.98 ± 0.08 | 0.58 | <0.001 |
| SBP (mmHg) | 122 ± 18 | 123 ± 21 | 122 ± 18 | 116 ± 16 | 122 ± 18 | 0.80 | 133 ± 18 | 132 ± 15 | 135 ± 18 | 137 ± 18 | 134 ± 17 | 0.79 | <0.001 |
| DBP (mmHg) | 73 ± 10 | 73 ± 10 | 77 ± 14 | 72 ± 11 | 74 ± 11 | 0.45 | 78 ± 10 | 79 ± 11 | 79 ± 9 | 77 ± 11 | 78 ± 10 | 0.88 | <0.001 |
| Duration of diabetes | - | - | - | - | - | - | 10.1 ± 8.4 | 9.7 ± 6.7 | 11.0 ± 8.5 | 10.6 ± 6.7 | 10.2 ± 8.0 | 0.87 | - |
| Hs-CRP (mg/L) | 0.82 (0.45–2.23) | 1.07 (0.40–3.16) | 1.04 (0.45–2.86) | 1.24 (0.31–2.60) | 1.01 (0.45–2.60) | 0.76 | 2.57 (1.19–5.76) | 2.34 (1.09–5.24) | 2.56 (0.95–5.61) | 2.53 (1.04–5.20) | 2.53 (1.08–5.50) | 0.86 | <0.001 |
| TC (mg/dL) | 189 ± 35 | 194 ± 29 | 190 ± 42 | 203 ± 32 | 191 ± 36 | 0.62 | 180 ± 41 | 181 ± 45 | 170 ± 36 | 182 ± 33 | 178 ± 40 | 0.35 | 0.001 |
| HDL-c (mg/dL) | 55.9 ± 16.5 | 54.5 ± 16.0 | 54.7 ± 11.6 | 59.9 ± 13.1 | 55.7 ± 15.1 | 0.76 | 42.8 ± 11.0 | 41.9 ± 11.4 | 41.4 ± 9.5 | 44.3 ± 9.5 | 42.5 ± 10.6 | 0.62 | <0.001 |
| LDL-c (mg/dL) | 115 ± 31 | 122 ± 28 | 118 ± 35 | 125 ± 24.9 | 118 ± 30.9 | 0.67 | 109 ± 36.0 | 108 ± 37.3 | 100 ± 33.1 | 109 ± 32.4 | 107 ± 35.3 | 0.34 | 0.002 |
| Triglycerides (mg/dL) | 69 (51–114) | 70 (47–104) | 75 (58–94) | 86 (60–117) | 73 (54–104) | 0.63 | 122 (88–176) | 123 (101–156) | 129 (94–200) | 134 (101–172) | 125 (93–174) | 0.94 | <0.001 |
| HV | JT | U | Others | Total | p-value | |
|---|---|---|---|---|---|---|
| Statins (%) | 56.5 | 50.0 | 53.2 | 56.0 | 54.7 | 0.91 |
| Fibrate (%) | 13.0 | 19.4 | 8.5 | 12.0 | 13.0 | 0.54 |
| Ezetimibe (%) | 3.2 | 5.0 | 6.7 | 0.0 | 4.0 | 0.73 |
| Metformin (%) | 63.8 | 83.3 | 77.6 | 60.0 | 69.5 | 0.06 |
| DPP-4 inhibitors (%) | 33.9 | 45.0 | 41.9 | 42.9 | 38.6 | 0.76 |
| Insulin (%) | 29.3 | 19.4 | 30.6 | 20.0 | 27.0 | 0.51 |
| Sulfonilureas (%) | 18.8 | 14.3 | 10.4 | 33.3 | 17.8 | 0.11 |
| Glitazones (%) | 7.8 | 19.4 | 4.1 | 12.0 | 9.3 | 0.09 |
| Glinides (%) | 21.6 | 16.7 | 8.2 | 32.0 | 19.0 | 0.07 |
| GLP-1 agonists (%) | 19.4 | 15.0 | 6.5 | 21.4 | 15.7 | 0.40 |
| Antihypertensive (%) | 45.2 | 43.2 | 49.0 | 48.0 | 46.0 | 0.95 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Diaz-Morales, N.; Lopez-Domenech, S.; Iannantuoni, F.; Lopez-Gallardo, E.; Sola, E.; Morillas, C.; Rocha, M.; Ruiz-Pesini, E.; Victor, V.M. Mitochondrial DNA Haplogroup JT is Related to Impaired Glycaemic Control and Renal Function in Type 2 Diabetic Patients. J. Clin. Med. 2018, 7, 220. https://doi.org/10.3390/jcm7080220
Diaz-Morales N, Lopez-Domenech S, Iannantuoni F, Lopez-Gallardo E, Sola E, Morillas C, Rocha M, Ruiz-Pesini E, Victor VM. Mitochondrial DNA Haplogroup JT is Related to Impaired Glycaemic Control and Renal Function in Type 2 Diabetic Patients. Journal of Clinical Medicine. 2018; 7(8):220. https://doi.org/10.3390/jcm7080220
Chicago/Turabian StyleDiaz-Morales, Noelia, Sandra Lopez-Domenech, Francesca Iannantuoni, Ester Lopez-Gallardo, Eva Sola, Carlos Morillas, Milagros Rocha, Eduardo Ruiz-Pesini, and Victor M. Victor. 2018. "Mitochondrial DNA Haplogroup JT is Related to Impaired Glycaemic Control and Renal Function in Type 2 Diabetic Patients" Journal of Clinical Medicine 7, no. 8: 220. https://doi.org/10.3390/jcm7080220
APA StyleDiaz-Morales, N., Lopez-Domenech, S., Iannantuoni, F., Lopez-Gallardo, E., Sola, E., Morillas, C., Rocha, M., Ruiz-Pesini, E., & Victor, V. M. (2018). Mitochondrial DNA Haplogroup JT is Related to Impaired Glycaemic Control and Renal Function in Type 2 Diabetic Patients. Journal of Clinical Medicine, 7(8), 220. https://doi.org/10.3390/jcm7080220








