MicroRNAs in Smoking-Related Carcinogenesis: Biomarkers, Functions, and Therapy
Abstract
1. Introduction
2. Correlation among Smoking, Cancer, and Expression of miRNAs
2.1. Lung
2.2. Urinary Tract
3. Relationship between Smoking and DNA Breaks
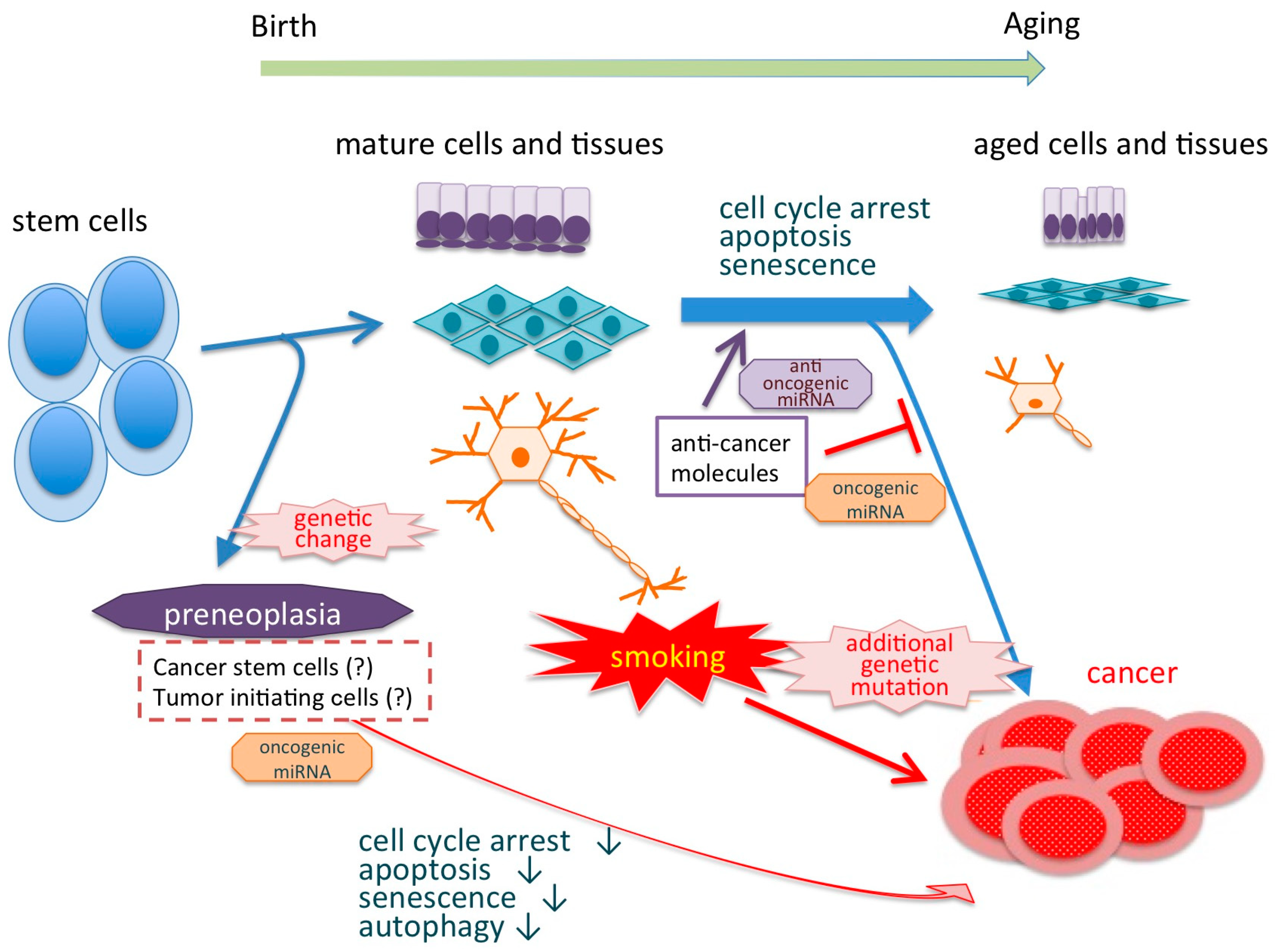
4. Smoking and Aging
5. MiRNA Detection
6. Conclusions
Author Contributions
Conflicts of Interest
References
- Gandini, S.; Botteri, E.; Iodice, S.; Boniol, M.; Lowenfels, A.B.; Maisonneuve, P.; Boyle, P. Tobacco smoking and cancer: A meta-analysis. Int. J. Cancer 2008, 122, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Momi, N.; Kaur, S.; Rachagani, S.; Ganti, A.K.; Batra, S.K. Smoking and microRNA dysregulation: A cancerous combination. Trends Mol. Med. 2014, 20, 36–47. [Google Scholar] [CrossRef] [PubMed]
- Franceschi, C.; Campisi, J. Chronic inflammation (inflammaging) and its potential contribution to age-associated diseases. J. Gerontol. A Biol. Sci. Med. Sci. 2014, 69 (Suppl. 1), S4–S9. [Google Scholar] [CrossRef] [PubMed]
- Hecht, S.S.; Hoffmann, D. Tobacco-specific nitrosamines, an important group of carcinogens in tobacco and tobacco smoke. Carcinogenesis 1988, 9, 875–884. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.L.; Hsueh, Y.M.; Chung, C.J.; Pu, Y.S.; Chang, L.W.; Hsieh, D.P.; Liou, S.H.; Lin, P. Correlation between the urine profile of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone metabolites and N7-methylguanine in urothelial carcinoma patients. Cancer Epidemiol. Biomark. Prev. 2008, 17, 3390–3395. [Google Scholar] [CrossRef] [PubMed]
- Hecht, S.S. Biochemistry, biology, and carcinogenicity of tobacco-specific N-nitrosamines. Chem. Res. Toxicol. 1998, 11, 559–603. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, D.; Brunnemann, K.D.; Prokopczyk, B.; Djordjevic, M.V. Tobacco-specific N-nitrosamines and Areca-derived N-nitrosamines: Chemistry, biochemistry, carcinogenicity, and relevance to humans. J. Toxicol. Environ. Health 1994, 41, 1–52. [Google Scholar] [CrossRef] [PubMed]
- Nagathihalli, N.S.; Massion, P.P.; Gonzalez, A.L.; Lu, P.; Datta, P.K. Smoking induces epithelial-to-mesenchymal transition in non-small cell lung cancer through HDAC-mediated downregulation of E-cadherin. Mol. Cancer Ther. 2012, 11, 2362–2372. [Google Scholar] [CrossRef] [PubMed]
- O’Driscoll, L. The emerging world of microRNAs. Anticancer Res. 2006, 26, 4271–4278. [Google Scholar] [PubMed]
- Ambros, V. The functions of animal microRNAs. Nature 2004, 431, 350–355. [Google Scholar] [CrossRef] [PubMed]
- Friedman, R.C.; Farh, K.K.; Burge, C.B.; Bartel, D.P. Most mammalian mRNAs are conserved targets of microRNAs. Genome Res. 2009, 19, 92–105. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, A.; Luettich, K. MicroRNAs as potential biomarkers of smoking-related diseases. Biomark. Med. 2012, 6, 671–684. [Google Scholar] [CrossRef] [PubMed]
- Landi, M.T.; Zhao, Y.; Rotunno, M.; Koshiol, J.; Liu, H.; Bergen, A.W.; Rubagotti, M.; Goldstein, A.M.; Linnoila, I.; Marincola, F.M.; et al. MicroRNA expression differentiates histology and predicts survival of lung cancer. Clin. Cancer Res. 2010, 16, 430–441. [Google Scholar] [CrossRef] [PubMed]
- Xi, S.; Xu, H.; Shan, J.; Tao, Y.; Hong, J.A.; Inchauste, S.; Zhang, M.; Kunst, T.F.; Mercedes, L.; Schrump, D.S. Cigarette smoke mediates epigenetic repression of miR-487b during pulmonary carcinogenesis. J. Clin. Investig. 2013, 123, 1241–1261. [Google Scholar] [CrossRef] [PubMed]
- Schembri, F.; Sridhar, S.; Perdomo, C.; Gustafson, A.M.; Zhang, X.; Ergun, A.; Lu, J.; Liu, G.; Zhang, X.; Bowers, J.; et al. MicroRNAs as modulators of smoking-induced gene expression changes in human airway epithelium. Proc. Natl. Acad. Sci. USA 2009, 106, 2319–2324. [Google Scholar] [CrossRef] [PubMed]
- Inamura, K.; Ishikawa, Y. MicroRNA in Lung Cancer: Novel Biomarkers and Potential Tools for Treatment. J. Clin. Med. 2016, 5, 36. [Google Scholar] [CrossRef] [PubMed]
- Brennan, P.; Bogillot, O.; Cordier, S.; Greiser, E.; Schill, W.; Vineis, P.; Lopez-Abente, G.; Tzonou, A.; Chang-Claude, J.; Bolm-Audorff, U.; et al. Cigarette smoking and bladder cancer in men: A pooled analysis of 11 case-control studies. Int. J. Cancer 2000, 86, 289–294. [Google Scholar] [CrossRef]
- Strope, S.A.; Montie, J.E. The causal role of cigarette smoking in bladder cancer initiation and progression, and the role of urologists in smoking cessation. J. Urol. 2008, 180, 31–37. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, J.K.; Silverman, D.T.; Hsing, A.W.; Ross, R.K.; Schoenberg, J.B.; Yu, M.C.; Stemhagen, A.; Lynch, C.F.; Blot, W.J.; Fraumeni, J.F., Jr. Cigarette smoking and cancers of the renal pelvis and ureter. Cancer Res. 1992, 52, 254–257. [Google Scholar] [PubMed]
- Mathers, J.C.; Strathdee, G.; Relton, C.L. Induction of epigenetic alterations by dietary and other environmental factors. Adv. Genet. 2010, 71, 3–39. [Google Scholar] [PubMed]
- Inoue, M.; Sawada, N.; Matsuda, T.; Iwasaki, M.; Sasazuki, S.; Shimazu, T.; Shibuya, K.; Tsugane, S. Attributable causes of cancer in Japan in 2005—Systematic assessment to estimate current burden of cancer attributable to known preventable risk factors in Japan. Ann. Oncol. 2012, 23, 1362–1369. [Google Scholar] [CrossRef] [PubMed]
- Park, S.L.; Bastani, D.; Goldstein, B.Y.; Chang, S.C.; Cozen, W.; Cai, L.; Cordon-Cardo, C.; Ding, B.; Greenland, S.; He, N.; et al. Associations between NBS1 polymorphisms, haplotypes and smoking-related cancers. Carcinogenesis 2010, 31, 1264–1271. [Google Scholar] [CrossRef] [PubMed]
- Lema, C.; Cunningham, M.J. MicroRNAs and their implications in toxicological research. Toxicol. Lett. 2010, 198, 100–105. [Google Scholar] [CrossRef] [PubMed]
- Stanitz, E.; Juhasz, K.; Gombos, K.; Gocze, K.; Toth, C.; Kiss, I. Alteration of miRNA expression correlates with lifestyle, social and environmental determinants in esophageal carcinoma. Anticancer Res. 2015, 35, 1091–1097. [Google Scholar] [PubMed]
- Cui, R.; Meng, W.; Sun, H.L.; Kim, T.; Ye, Z.; Fassan, M.; Jeon, Y.J.; Li, B.; Vicentini, C.; Peng, Y.; et al. MicroRNA-224 promotes tumor progression in non-small cell lung cancer. Proc. Natl. Acad. Sci. USA 2015, 112, E4288–E4297. [Google Scholar] [CrossRef] [PubMed]
- Sato, T.; Shiba-Ishii, A.; Kim, Y.; Dai, T.; Husni, R.E.; Hong, J.; Kano, J.; Sakashita, S.; Iijima, T.; Noguchi, M. miR-3941: A novel microRNA that controls IGBP1 expression and is associated with malignant progression of lung adenocarcinoma. Cancer Sci. 2017, 108, 536–542. [Google Scholar] [CrossRef] [PubMed]
- Budd, W.T.; Seashols-Williams, S.J.; Clark, G.C.; Weaver, D.; Calvert, V.; Petricoin, E.; Dragoescu, E.A.; O’Hanlon, K.; Zehner, Z.E. Dual action of miR-125b as a tumor suppressor and oncomiR-22 promotes prostate cancer tumorigenesis. PLoS ONE 2015, 10, e0142373. [Google Scholar] [CrossRef] [PubMed]
- Aunan, J.R.; Cho, W.C.; Soreide, K. The biology of aging and cancer: A brief overview of shared and divergent molecular hallmarks. Aging Dis. 2017, 8, 628–642. [Google Scholar] [CrossRef] [PubMed]
- Bernardes de Jesus, B.; Blasco, M.A. Telomerase at the intersection of cancer and aging. Trends Genet. 2013, 29, 513–520. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M. Smoking: Additional burden on aging and death. Genes Environ. 2016, 38. [Google Scholar] [CrossRef] [PubMed]
- Stern, M.C.; Lin, J.; Figueroa, J.D.; Kelsey, K.T.; Kiltie, A.E.; Yuan, J.M.; Matullo, G.; Fletcher, T.; Benhamou, S.; Taylor, J.A.; et al. Polymorphisms in DNA repair genes, smoking, and bladder cancer risk: Findings from the international consortium of bladder cancer. Cancer Res. 2009, 69, 6857–6864. [Google Scholar] [CrossRef] [PubMed]
- Vineis, P.; Talaska, G.; Malaveille, C.; Bartsch, H.; Martone, T.; Sithisarankul, P.; Strickland, P. DNA adducts in urothelial cells: Relationship with biomarkers of exposure to arylamines and polycyclic aromatic hydrocarbons from tobacco smoke. Int. J. Cancer 1996, 65, 314–316. [Google Scholar] [CrossRef]
- Turesky, R.J.; Freeman, J.P.; Holland, R.D.; Nestorick, D.M.; Miller, D.W.; Ratnasinghe, D.L.; Kadlubar, F.F. Identification of aminobiphenyl derivatives in commercial hair dyes. Chem. Res. Toxicol. 2003, 16, 1162–1173. [Google Scholar] [CrossRef] [PubMed]
- Pryor, W.A.; Hales, B.J.; Premovic, P.I.; Church, D.F. The radicals in cigarette tar: Their nature and suggested physiological implications. Science 1983, 220, 425–427. [Google Scholar] [CrossRef] [PubMed]
- Liz, J.; Esteller, M. lncRNAs and microRNAs with a role in cancer development. Biochim. Biophys. Acta 2016, 1859, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Ruan, K.; Fang, X.; Ouyang, G. MicroRNAs: Novel regulators in the hallmarks of human cancer. Cancer Lett. 2009, 285, 116–126. [Google Scholar] [CrossRef] [PubMed]
- Fujii, T.; Shimada, K.; Tatsumi, Y.; Tanaka, N.; Fujimoto, K.; Konishi, N. Syndecan-1 up-regulates microRNA-331-3p and mediates epithelial-to-mesenchymal transition in prostate cancer. Mol. Carcinog. 2016, 55, 1378–1386. [Google Scholar] [CrossRef] [PubMed]
- Nymark, P.; Guled, M.; Borze, I.; Faisal, A.; Lahti, L.; Salmenkivi, K.; Kettunen, E.; Anttila, S.; Knuutila, S. Integrative analysis of microRNA, mRNA and aCGH data reveals asbestos- and histology-related changes in lung cancer. Genes Chromosomes Cancer 2011, 50, 585–597. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Huang, Z.; Teng, F.; Xing, L.; Yu, J. Predictive biomarkers in PD-1/PD-L1 checkpoint blockade immunotherapy. Cancer Treat. Rev. 2015, 41, 868–876. [Google Scholar] [CrossRef] [PubMed]
- Brahmer, J.R.; Tykodi, S.S.; Chow, L.Q.; Hwu, W.J.; Topalian, S.L.; Hwu, P.; Drake, C.G.; Camacho, L.H.; Kauh, J.; Odunsi, K.; et al. Safety and activity of anti-PD-L1 antibody in patients with advanced cancer. N. Engl. J. Med. 2012, 366, 2455–2465. [Google Scholar] [CrossRef] [PubMed]
- Zarogoulidis, P.; Petanidis, S.; Domvri, K.; Kioseoglou, E.; Anestakis, D.; Freitag, L.; Zarogoulidis, K.; Hohenforst-Schmidt, W.; Eberhardt, W. Autophagy inhibition upregulates CD4(+) tumor infiltrating lymphocyte expression via miR-155 regulation and TRAIL activation. Mol. Oncol. 2016, 10, 1516–1531. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, P.C.; Mauer, A.M.; Vokes, E.E. Lung cancer. Lancet 2000, 355, 479–485. [Google Scholar] [CrossRef]
- Megiorni, F.; Pizzuti, A.; Frati, L. Clinical significance of microRNA expression profiles and polymorphisms in lung cancer development and management. Pathol. Res. Int. 2011, 2011, 780652. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Massion, P.P.; Carbone, D.P. The molecular basis of lung cancer: Molecular abnormalities and therapeutic implications. Respir. Res. 2003, 4, 12. [Google Scholar] [CrossRef] [PubMed]
- Angulo, M.; Lecuona, E.; Sznajder, J.I. Role of microRNAs in lung disease. Arch. Bronconeumol. 2012, 48, 325–330. [Google Scholar] [CrossRef] [PubMed]
- Brownson, R.C.; Alavanja, M.C.; Hock, E.T.; Loy, T.S. Passive smoking and lung cancer in nonsmoking women. Am. J. Public Health 1992, 82, 1525–1530. [Google Scholar] [CrossRef] [PubMed]
- Tessema, M.; Yingling, C.M.; Picchi, M.A.; Wu, G.; Ryba, T.; Lin, Y.; Bungum, A.O.; Edell, E.S.; Spira, A.; Belinsky, S.A. ANK1 Methylation regulates expression of MicroRNA-486-5p and discriminates lung tumors by histology and smoking status. Cancer Lett. 2017, 410, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Chung, G.T.; Sundaresan, V.; Hasleton, P.; Rudd, R.; Taylor, R.; Rabbitts, P.H. Sequential molecular genetic changes in lung cancer development. Oncogene 1995, 11, 2591–2598. [Google Scholar] [PubMed]
- Toyooka, S.; Matsuo, K.; Shigematsu, H.; Kosaka, T.; Tokumo, M.; Yatabe, Y.; Ichihara, S.; Inukai, M.; Suehisa, H.; Soh, J.; et al. The impact of sex and smoking status on the mutational spectrum of epidermal growth factor receptor gene in non-small cell lung cancer. Clin. Cancer Res. 2007, 13, 5763–5768. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Toyooka, S.; Tsuda, T.; Gazdar, A.F. The TP53 gene, tobacco exposure, and lung cancer. Hum. Mutat. 2003, 21, 229–239. [Google Scholar] [CrossRef] [PubMed]
- Vahakangas, K.H.; Bennett, W.P.; Castren, K.; Welsh, J.A.; Khan, M.A.; Blomeke, B.; Alavanja, M.C.; Harris, C.C. p53 and K-ras mutations in lung cancers from former and never-smoking women. Cancer Res. 2001, 61, 4350–4356. [Google Scholar] [PubMed]
- Zhai, X.; Xu, W. Long noncoding RNA ATB promotes proliferation, migration and invasion in bladder cancer by suppressing microRNA-126. Oncol. Res. 2018. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Wang, G.; Wang, H. MiR-126 functions as a tumor suppressor by targeting SRPK1 in human gastric cancer. Oncol. Res. 2018. [Google Scholar] [CrossRef] [PubMed]
- Jing, B.Q.; Ou, Y.; Zhao, L.; Xie, Q.; Zhang, Y.X. Experimental study on the prevention of liver cancer angiogenesis via miR-126. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 5096–5100. [Google Scholar] [PubMed]
- Grimolizzi, F.; Monaco, F.; Leoni, F.; Bracci, M.; Staffolani, S.; Bersaglieri, C.; Gaetani, S.; Valentino, M.; Amati, M.; Rubini, C.; et al. Exosomal miR-126 as a circulating biomarker in non-small-cell lung cancer regulating cancer progression. Sci. Rep. 2017, 7, 15277. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Zhou, Y.; Lu, J.; Xu, H.; Lei, L.; Chen, C.; Zhao, J.; Xu, L. The prognostic value of miR-126 expression in non-small-cell lung cancer: A meta-analysis. Cancer Cell Int. 2017, 17, 71. [Google Scholar] [CrossRef] [PubMed]
- Fiala, O.; Pitule, P.; Hosek, P.; Liska, V.; Sorejs, O.; Bruha, J.; Vycital, O.; Buchler, T.; Poprach, A.; Topolcan, O.; et al. The association of miR-126-3p, miR-126-5p and miR-664-3p expression profiles with outcomes of patients with metastatic colorectal cancer treated with bevacizumab. Tumour Biol. 2017, 39, 1010428317709283. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Peng, X.C.; Zheng, X.L.; Wang, J.; Qin, Y.W. MiR-126 restoration down-regulate VEGF and inhibit the growth of lung cancer cell lines in vitro and in vivo. Lung Cancer 2009, 66, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Izzotti, A.; Calin, G.A.; Arrigo, P.; Steele, V.E.; Croce, C.M.; De Flora, S. Downregulation of microRNA expression in the lungs of rats exposed to cigarette smoke. FASEB J. 2009, 23, 806–812. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.H.; Liu, C.S.; Liu, H.J.; Huang, C.P.; Huang, C.Y.; Hsu, H.T.; Liou, S.H.; Chung, C.J. Association between levels of urinary heavy metals and increased risk of urothelial carcinoma. Int. J. Urol. 2016, 23, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Stellman, S.D.; Djordjevic, M.V. Monitoring the tobacco use epidemic II: The agent: Current and emerging tobacco products. Prev. Med. 2009, 48 (Suppl. 1), S11–S15. [Google Scholar] [CrossRef] [PubMed]
- Burger, M.; Catto, J.W.; Dalbagni, G.; Grossman, H.B.; Herr, H.; Karakiewicz, P.; Kassouf, W.; Kiemeney, L.A.; La Vecchia, C.; Shariat, S.; et al. Epidemiology and risk factors of urothelial bladder cancer. Eur. Urol. 2013, 63, 234–241. [Google Scholar] [CrossRef] [PubMed]
- Schulz, W.A.; Goering, W. DNA methylation in urothelial carcinoma. Epigenomics 2016, 8, 1415–1428. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.C.; Huang, C.C.; Shen, C.H.; Lin, L.C.; Zhao, P.W.; Chen, S.Y.; Deng, Y.C.; Liu, Y.W. Gene expression and DNA methylation status of glutathione S-transferase Mu1 and Mu5 in urothelial carcinoma. PLoS ONE 2016, 11, e0159102. [Google Scholar] [CrossRef] [PubMed]
- Brait, M.; Munari, E.; LeBron, C.; Noordhuis, M.G.; Begum, S.; Michailidi, C.; Gonzalez-Roibon, N.; Maldonado, L.; Sen, T.; Guerrero-Preston, R.; et al. Genome-wide methylation profiling and the PI3K-AKT pathway analysis associated with smoking in urothelial cell carcinoma. Cell Cycle 2013, 12, 1058–1070. [Google Scholar] [CrossRef] [PubMed]
- Chung, C.J.; Lee, H.L.; Yang, H.Y.; Lin, P.; Pu, Y.S.; Shiue, H.S.; Su, C.T.; Hsueh, Y.M. Low ratio of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol-glucuronides (NNAL-Gluc)/free NNAL increases urothelial carcinoma risk. Sci. Total Environ. 2011, 409, 1638–1642. [Google Scholar] [CrossRef] [PubMed]
- Ganji, S.M.; Saidijam, M.; Amini, R.; Mousavi-Bahar, S.H.; Shabab, N.; Seyedabadi, S.; Mahdavinezhad, A. Evaluation of microRNA-99a and microRNA-205 expression levels in bladder cancer. Int. J. Mol. Cell. Med. 2017, 6, 87–95. [Google Scholar] [PubMed]
- Tsai, T.F.; Lin, J.F.; Chou, K.Y.; Lin, Y.C.; Chen, H.E.; Hwang, T.I. miR-99a-5p acts as tumor suppressor via targeting to mTOR and enhances RAD001-induced apoptosis in human urinary bladder urothelial carcinoma cells. OncoTargets Ther. 2018, 11, 239–252. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Li, H.; Liu, L.; Song, L.; Lv, Y.; Han, Y. Identification and functional analysis of risk-related microRNAs for the prognosis of patients with bladder urothelial carcinoma. Oncol. Lett. 2017, 14, 7297–7303. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Geva, G.A.; Gielchinsky, I.; Aviv, N.; Max, K.E.A.; Gofrit, O.N.; Gur-Wahnon, D.; Ben-Dov, I.Z. Urine cell-free microRNA as biomarkers for transitional cell carcinoma. BMC Res. Notes 2017, 10, 641. [Google Scholar] [CrossRef] [PubMed]
- Ke, H.L.; Li, W.M.; Lin, H.H.; Hsu, W.C.; Hsu, Y.L.; Chang, L.L.; Huang, C.N.; Li, C.C.; Chang, H.P.; Yeh, H.C.; et al. Hypoxia-regulated microRNA-210 overexpression is associated with tumor development and progression in upper tract urothelial carcinoma. Int. J. Med. Sci. 2017, 14, 578–584. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Liu, Y.; He, A.; Liu, Y.; Wu, J.; Liao, X.; Lv, Z.; Wang, F.; Mei, H. Hsa-miR-429 promotes bladder cancer cell proliferation via inhibiting CDKN2B. Oncotarget 2017, 8, 68721–68729. [Google Scholar] [CrossRef] [PubMed]
- Chung, Y.H.; Li, S.C.; Kao, Y.H.; Luo, H.L.; Cheng, Y.T.; Lin, P.R.; Tai, M.H.; Chiang, P.H. MiR-30a-5p inhibits epithelial-to-mesenchymal transition and upregulates expression of tight junction protein Claudin-5 in human upper tract urothelial carcinoma cells. Int. J. Mol. Sci. 2017, 18. [Google Scholar] [CrossRef] [PubMed]
- Lenherr, S.M.; Tsai, S.; Silva Neto, B.; Sullivan, T.B.; Cimmino, C.B.; Logvinenko, T.; Gee, J.; Huang, W.; Libertino, J.A.; Summerhayes, I.C.; et al. MicroRNA expression profile identifies high grade, non-muscle-invasive bladder tumors at elevated risk to progress to an invasive phenotype. Genes 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Matsuzaki, K.; Fujita, K.; Jingushi, K.; Kawashima, A.; Ujike, T.; Nagahara, A.; Ueda, Y.; Tanigawa, G.; Yoshioka, I.; Ueda, K.; et al. MiR-21-5p in urinary extracellular vesicles is a novel biomarker of urothelial carcinoma. Oncotarget 2017, 8, 24668–24678. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.R.; Yeh, H.C.; Wang, W.J.; Ke, H.L.; Lin, H.H.; Hsu, W.C.; Chao, S.Y.; Hour, T.C.; Wu, W.J.; Pu, Y.S.; et al. MiR-193b mediates CEBPD-induced cisplatin sensitization through targeting ETS1 and Cyclin D1 in human urothelial carcinoma cells. J. Cell. Biochem. 2017, 118, 1563–1573. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.B.; Wang, W.; Du, Y.H.; Li, H.; Xia, S.J.; Liu, H.T. MicroRNA-3713 regulates bladder cell invasion via MMP9. Sci. Rep. 2016, 6, 32374. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhao, X.; Shi, J.; Pan, Y.; Chen, Q.; Leng, P.; Wang, Y. miR-451 suppresses bladder cancer cell migration and invasion via directly targeting c-Myc. Oncol. Rep. 2016, 36, 2049–2058. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, Z.; Li, Z.; Gong, D.; Zhan, B.; Man, X.; Kong, C. MicroRNA-497 inhibits the proliferation, migration and invasion of human bladder transitional cell carcinoma cells by targeting E2F3. Oncol. Rep. 2016, 36, 1293–1300. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Zhu, Y.; Liang, Z.; Wang, X.; Meng, S.; Xu, X.; Xu, X.; Wu, J.; Ji, A.; Hu, Z.; et al. Up-regulation of p16 by miR-877-3p inhibits proliferation of bladder cancer. Oncotarget 2016, 7, 51773–51783. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; Kong, C.; Zhu, Y.; Zeng, Y.; Zhang, Z.; Liu, X.; Zhan, B.; Piao, C.; Jiang, Z. miR-130b, an onco-miRNA in bladder cancer, is directly regulated by NF-κB and sustains NF-κB activation by decreasing Cylindromatosis expression. Oncotarget 2016, 7, 48547–48561. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Wu, B.; Xu, Z.; Li, S.; Tan, S.; Liu, X.; Wang, K. Downregulation of miR-133b predict progression and poor prognosis in patients with urothelial carcinoma of bladder. Cancer Med. 2016, 5, 1856–1862. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, H.; Yoshiike, M.; Nozawa, S.; Usuba, W.; Katsuoka, Y.; Aida, K.; Kitajima, K.; Kudo, H.; Hoshikawa, M.; Yoshioka, Y.; et al. Expression level of urinary MicroRNA-146a-5p is increased in patients with bladder cancer and decreased in those after transurethral resection. Clin. Genitourin. Cancer 2016, 14, e493–e499. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.L.; Ho, J.Y.; Chou, S.C.; Yu, D.S. MiR-429 reverses epithelial-mesenchymal transition by restoring E-cadherin expression in bladder cancer. Oncotarget 2016, 7, 26593–26603. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Ma, X.; Du, J.; Yao, Z.; Shi, T.; Ai, Q.; Chen, X.; Zhang, Z.; Zhang, X.; Yao, X. MicroRNA-30a as a prognostic factor in urothelial carcinoma of bladder inhibits cellular malignancy by antagonising Notch1. BJU Int. 2016, 118, 578–589. [Google Scholar] [CrossRef] [PubMed]
- Fujii, T.; Shimada, K.; Tatsumi, Y.; Hatakeyama, K.; Obayashi, C.; Fujimoto, K.; Konishi, N. microRNA-145 promotes differentiation in human urothelial carcinoma through down-regulation of syndecan-1. BMC Cancer 2015, 15, 818. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Zhang, C.; Liu, W.; Zheng, W.; Zhang, Y.; Wang, S.; Huang, D.; Liu, X.; Bai, Z. MicroRNA-24 upregulation inhibits proliferation, metastasis and induces apoptosis in bladder cancer cells by targeting CARMA3. Int. J. Oncol. 2015, 47, 1351–1360. [Google Scholar] [CrossRef] [PubMed]
- Lombard, A.P.; Mooso, B.A.; Libertini, S.J.; Lim, R.M.; Nakagawa, R.M.; Vidallo, K.D.; Costanzo, N.C.; Ghosh, P.M.; Mudryj, M. miR-148a dependent apoptosis of bladder cancer cells is mediated in part by the epigenetic modifier DNMT1. Mol. Carcinog. 2016, 55, 757–767. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.; Bing, Z.; Yao, Y.; Master, S.R.; Gupta, P. Higher expression of miR-182 in cytology specimens of high-grade urothelial cell carcinoma: A potential diagnostic marker. Acta Cytol. 2015, 59, 109–112. [Google Scholar] [CrossRef] [PubMed]
- Xie, D.; Shang, C.; Zhang, H.; Guo, Y.; Tong, X. Up-regulation of miR-9 target CBX7 to regulate invasion ability of bladder transitional cell carcinoma. Med. Sci. Monit. 2015, 21, 225–230. [Google Scholar] [PubMed]
- Andrew, A.S.; Marsit, C.J.; Schned, A.R.; Seigne, J.D.; Kelsey, K.T.; Moore, J.H.; Perreard, L.; Karagas, M.R.; Sempere, L.F. Expression of tumor suppressive microRNA-34a is associated with a reduced risk of bladder cancer recurrence. Int. J. Cancer 2015, 137, 1158–1166. [Google Scholar] [CrossRef] [PubMed]
- Morais, D.R.; Reis, S.T.; Viana, N.; Piantino, C.B.; Massoco, C.; Moura, C.; Dip, N.; Silva, I.A.; Srougi, M.; Leite, K.R. The involvement of miR-100 in bladder urothelial carcinogenesis changing the expression levels of mRNA and proteins of genes related to cell proliferation, survival, apoptosis and chromosomal stability. Cancer Cell Int. 2014, 14, 119. [Google Scholar] [CrossRef] [PubMed]
- Drayton, R.M.; Peter, S.; Myers, K.; Miah, S.; Dudziec, E.; Bryant, H.E.; Catto, J.W. MicroRNA-99a and 100 mediated upregulation of FOXA1 in bladder cancer. Oncotarget 2014, 5, 6375–6386. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Yuan, J.; Feng, N.; Li, Y.; Lin, Z.; Jiang, Z.; Gui, Y. Hsa-miR-1 downregulates long non-coding RNA urothelial cancer associated 1 in bladder cancer. Tumour Biol. 2014, 35, 10075–10084. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.D.; Wu, X.H.; Fan, Y.R.; Tan, B.; Quan, Z.; Luo, C.L. Exosome-derived microRNA-29c induces apoptosis of BIU-87 cells by down regulating BCL-2 and MCL-1. Asian Pac. J. Cancer Prev. 2014, 15, 3471–3476. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Qi, F.; Cao, Y.; Chen, M.; Zu, X. Down-regulated microRNA-101 in bladder transitional cell carcinoma is associated with poor prognosis. Med. Sci. Monit. 2014, 20, 812–817. [Google Scholar] [PubMed]
- Jia, A.Y.; Castillo-Martin, M.; Bonal, D.M.; Sanchez-Carbayo, M.; Silva, J.M.; Cordon-Cardo, C. MicroRNA-126 inhibits invasion in bladder cancer via regulation of ADAM9. Br. J. Cancer 2014, 110, 2945–2954. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Zhou, M.; Wang, J.; Zhao, Y.; Li, S.; Zhou, B.; Su, Z.; Xu, C.; Xia, Y.; Qian, H.; et al. Role of microRNA-27a in down-regulation of angiogenic factor AGGF1 under hypoxia associated with high-grade bladder urothelial carcinoma. Biochim. Biophys. Acta 2014, 1842, 712–725. [Google Scholar] [CrossRef] [PubMed]
- Shang, C.; Zhang, H.; Guo, Y.; Hong, Y.; Liu, Y.; Xue, Y. MiR-320a down-regulation mediates bladder carcinoma invasion by targeting ITGB3. Mol. Biol. Rep. 2014, 41, 2521–2527. [Google Scholar] [CrossRef] [PubMed]
- Majid, S.; Dar, A.A.; Saini, S.; Deng, G.; Chang, I.; Greene, K.; Tanaka, Y.; Dahiya, R.; Yamamura, S. MicroRNA-23b functions as a tumor suppressor by regulating Zeb1 in bladder cancer. PLoS ONE 2013, 8, e67686. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Liu, H.; Zhang, H.; Shang, C.; Song, Y. miR-96 regulates FOXO1-mediated cell apoptosis in bladder cancer. Oncol. Lett. 2012, 4, 561–565. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Yao, Z.; Zhu, M.; Ma, X.; Shi, T.; Li, H.; Wang, B.; Ouyang, J.; Zhang, X. Inhibitory effects of microRNA-34a on cell migration and invasion of invasive urothelial bladder carcinoma by targeting Notch1. J. Huazhong Univ. Sci. Technol. Med. Sci. 2012, 32, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Song, T.; Zhang, X.; Wang, C.; Wu, Y.; Dong, J.; Gao, J.; Cai, W.; Hong, B. Expression of miR-143 reduces growth and migration of human bladder carcinoma cells by targeting cyclooxygenase-2. Asian Pac. J. Cancer Prev. 2011, 12, 929–933. [Google Scholar] [PubMed]
- Huang, L.; Luo, J.; Cai, Q.; Pan, Q.; Zeng, H.; Guo, Z.; Dong, W.; Huang, J.; Lin, T. MicroRNA-125b suppresses the development of bladder cancer by targeting E2F3. Int. J. Cancer 2011, 128, 1758–1769. [Google Scholar] [CrossRef] [PubMed]
- Friedman, J.M.; Liang, G.; Liu, C.C.; Wolff, E.M.; Tsai, Y.C.; Ye, W.; Zhou, X.; Jones, P.A. The putative tumor suppressor microRNA-101 modulates the cancer epigenome by repressing the polycomb group protein EZH2. Cancer Res. 2009, 69, 2623–2629. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.H.; Chiang, W.C.; Chou, T.Y.; Chang, S.Y.; Chen, P.M.; Teng, S.C.; Wu, K.J. Increased NBS1 expression is a marker of aggressive head and neck cancer and overexpression of NBS1 contributes to transformation. Clin. Cancer Res. 2006, 12, 507–515. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.C.; Su, Y.N.; Chou, P.C.; Chiang, W.C.; Chang, M.C.; Wang, L.S.; Teng, S.C.; Wu, K.J. Overexpression of NBS1 contributes to transformation through the activation of phosphatidylinositol 3-kinase/Akt. J. Biol. Chem. 2005, 280, 32505–32511. [Google Scholar] [CrossRef] [PubMed]
- Flanders, W.D.; Lally, C.A.; Zhu, B.P.; Henley, S.J.; Thun, M.J. Lung cancer mortality in relation to age, duration of smoking, and daily cigarette consumption: Results from Cancer Prevention Study II. Cancer Res. 2003, 63, 6556–6562. [Google Scholar] [PubMed]
- Kim, H.P.; Wang, X.; Chen, Z.H.; Lee, S.J.; Huang, M.H.; Wang, Y.; Ryter, S.W.; Choi, A.M. Autophagic proteins regulate cigarette smoke-induced apoptosis: Protective role of heme oxygenase-1. Autophagy 2008, 4, 887–895. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Ridzon, D.A.; Broomer, A.J.; Zhou, Z.; Lee, D.H.; Nguyen, J.T.; Barbisin, M.; Xu, N.L.; Mahuvakar, V.R.; Andersen, M.R.; et al. Real-time quantification of microRNAs by stem-loop RT-PCR. Nucleic Acids Res. 2005, 33, e179. [Google Scholar] [CrossRef] [PubMed]
- Castoldi, M.; Schmidt, S.; Benes, V.; Noerholm, M.; Kulozik, A.E.; Hentze, M.W.; Muckenthaler, M.U. A sensitive array for microRNA expression profiling (miChip) based on locked nucleic acids (LNA). RNA 2006, 12, 913–920. [Google Scholar] [CrossRef] [PubMed]
- Schulte, J.H.; Marschall, T.; Martin, M.; Rosenstiel, P.; Mestdagh, P.; Schlierf, S.; Thor, T.; Vandesompele, J.; Eggert, A.; Schreiber, S.; et al. Deep sequencing reveals differential expression of microRNAs in favorable versus unfavorable neuroblastoma. Nucleic Acids Res. 2010, 38, 5919–5928. [Google Scholar] [CrossRef] [PubMed]
- Mensah, M.; Borzi, C.; Verri, C.; Suatoni, P.; Conte, D.; Pastorino, U.; Orazio, F.; Sozzi, G.; Boeri, M. MicroRNA based liquid biopsy: The experience of the plasma miRNA signature classifier (MSC) for lung cancer screening. J. Vis. Exp. 2017, 128. [Google Scholar] [CrossRef] [PubMed]
- Li, L.L.; Qu, L.L.; Fu, H.J.; Zheng, X.F.; Tang, C.H.; Li, X.Y.; Chen, J.; Wang, W.X.; Yang, S.X.; Wang, L.; et al. Circulating microRNAs as novel biomarkers of ALK-positive non-small cell lung cancer and predictors of response to crizotinib therapy. Oncotarget 2017, 8, 45399–45414. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Ba, Y.; Ma, L.; Cai, X.; Yin, Y.; Wang, K.; Guo, J.; Zhang, Y.; Chen, J.; Guo, X.; et al. Characterization of microRNAs in serum: A novel class of biomarkers for diagnosis of cancer and other diseases. Cell Res. 2008, 18, 997–1006. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, P.S.; Parkin, R.K.; Kroh, E.M.; Fritz, B.R.; Wyman, S.K.; Pogosova-Agadjanyan, E.L.; Peterson, A.; Noteboom, J.; O’Briant, K.C.; Allen, A.; et al. Circulating microRNAs as stable blood-based markers for cancer detection. Proc. Natl. Acad. Sci. USA 2008, 105, 10513–10518. [Google Scholar] [CrossRef] [PubMed]
- Fujii, T.; Asano, A.; Shimada, K.; Tatsumi, Y.; Obayashi, C.; Konishi, N. Evaluation of RNA and DNA extraction from liquid-based cytology specimens. Diagn. Cytopathol. 2016, 44, 833–840. [Google Scholar] [CrossRef] [PubMed]
| miRNA | Target Molecules | Function | Reference |
|---|---|---|---|
| miR-99a-5p | mTOR | Decreased phosphorylation of mTOR and AKT | [68] |
| miR-29a | DNMT3A, DNMT3B, MAT2A, SMS | Involvement in the cysteine and methionine metabolism | [69] |
| miR-210 | HIF-1α | Promoting upper tract urothelial carcinoma carcinogenesis | [70,71] |
| miR-429 | CDKN2B | Promoted cell growth and decreased apoptosis | [72] |
| miR-30a-5p | claudin-5 | Suppressed cell proliferation, migration and EMT | [73] |
| miR-32-5p, -224-5p, -412-3p, -203a-3p, -205-5p | Cancer specific survival, tumor progression, EMT | [74] | |
| miR-21-5p | Novel biomarker of urothelial carcinoma in urine | [75] | |
| miR-193b | ETS1, Cyclin D1 | Inhibited cell migration activity, arrested cell at G1 phase; sensitized CDDP treatment | [76] |
| miR-3713 | MMP9 | Control of cell invasiveness | [77] |
| miR-451 | c-Myc | Suppressed cell migration and invasion | [78] |
| miR-497 | E2F3 | Inhibited cell proliferation, migration and invasion | [79] |
| miR-877-3p | p16 | Increased the expression of p16, inhibited cell proliferation and tumorigenicity | [80] |
| miR-130b | NF-κB | Persistent activation of NF-κB; promote the malignant progression of urothelial carcinoma | [81] |
| miR-133b | Novel biomarker of urothelial carcinoma in the tissue | [82] | |
| miR-146a-5p | Novel biomarker of urothelial carcinoma in urine | [83] | |
| miR-429 | E-cadherin | Decreased cell migration and invasion through reducing ZEB1 and β-catenin | [84] |
| miR-30a | Notch1 | Decreased cell proliferation and migration, activated cell cycle arrest | [85] |
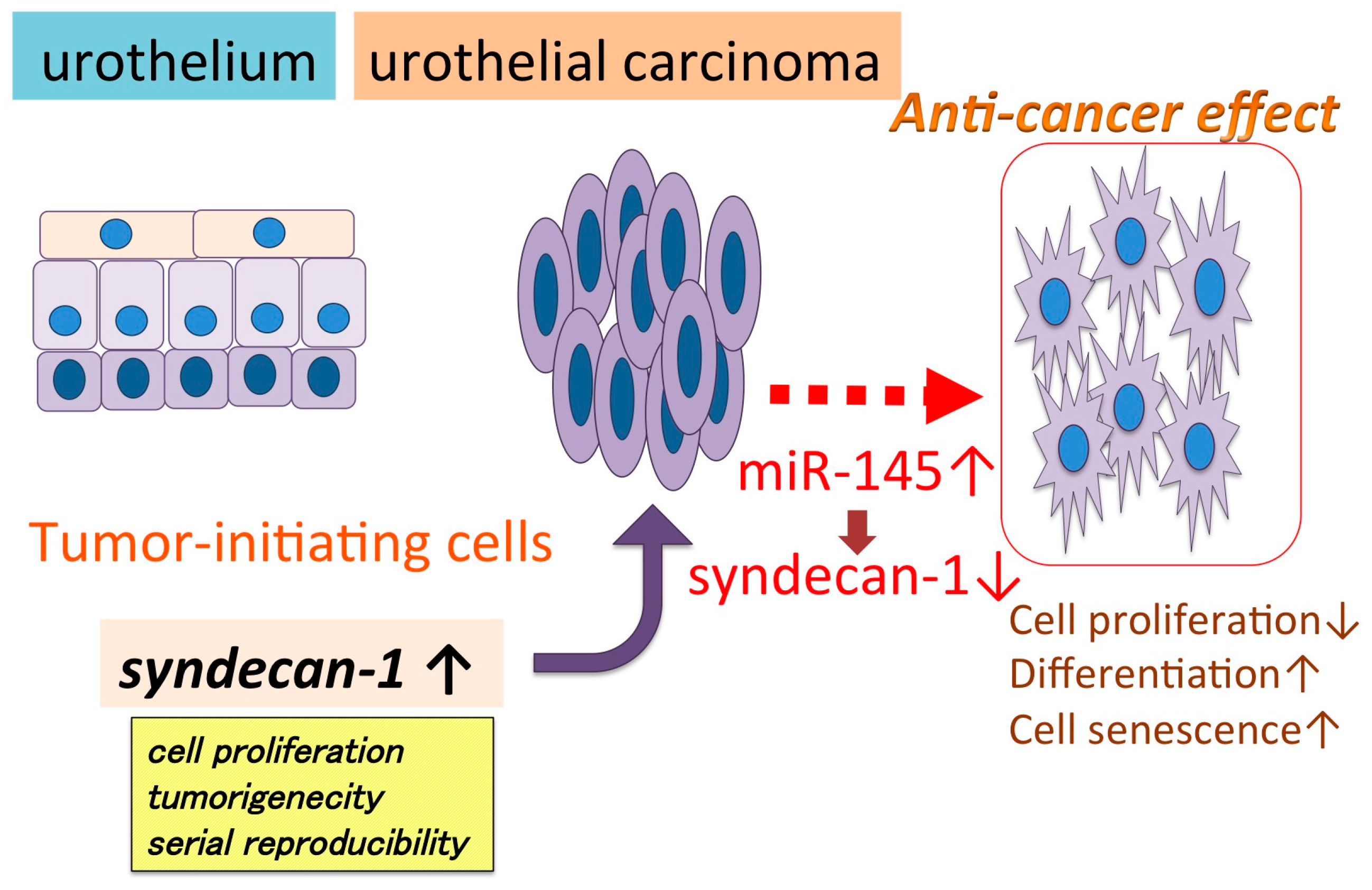
| miR-145 | syndecan-1 | Suppressed cell proliferation, induced cell senescence, differentiation | [86] |
| miR-24 | CARMA3 | Inhibited cell proliferation, invasion and EMT | [87] |
| miR-148a | DNMT1 | Reduced cell viability through apoptosis | [88] |
| miR-182 | Novel biomarker of urothelial carcinoma in urine | [89] | |
| miR-9 | CBX7 | Decreased cell invasion ability | [90] |
| miR-34a | S100P | Decreased cell invasion ability | [91] |
| miR-100 | BAZ2A, mTOR, SMARCA5 | Increased cell proliferation, anti-apoptosis | [92] |
| miR-99a, -100 | FGFR3, FOXA1 | Associated with regional hypomethylation | [93] |
| miR-1 | UCA1 | Decreased cell proliferation and motility, induced apoptosis | [94] |
| miR-29c | BCL-2, MCL-1 | Induced apoptosis | [95] |
| miR-101 | Novel biomarker of urothelial carcinoma in the tissue | [96] | |
| miR-126 | ADAM9 | Decreased cell invasion | [97] |
| miR27a | AGGF1 | Regulation of hypoxia-induced apoptosis | [98] |
| miR-320a | ITGB3. | Decreased cell invasion ability | [99] |
| miR-23b | Zeb1 | Inhibited cell proliferation, induced G0/G1 cell cycle arreset | [100] |
| miR-96 | FOXO1 | Tumorigenesis, control cell apoptosis | [101] |
| miR-34a | Notch1 | Decreased cell invasion and migration | [102] |
| miR-143 | cyclooxygenase-2 | Decreased cell proliferation and motility | [103] |
| miR-125b | E2F3 | Regulate G1/S transition through the E2F3-cyclin A2 signaling pathway | [104] |
| miR-101 | EZH2 | Inhibited cell proliferation and colony formation | [105] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fujii, T.; Shimada, K.; Nakai, T.; Ohbayashi, C. MicroRNAs in Smoking-Related Carcinogenesis: Biomarkers, Functions, and Therapy. J. Clin. Med. 2018, 7, 98. https://doi.org/10.3390/jcm7050098
Fujii T, Shimada K, Nakai T, Ohbayashi C. MicroRNAs in Smoking-Related Carcinogenesis: Biomarkers, Functions, and Therapy. Journal of Clinical Medicine. 2018; 7(5):98. https://doi.org/10.3390/jcm7050098
Chicago/Turabian StyleFujii, Tomomi, Keiji Shimada, Tokiko Nakai, and Chiho Ohbayashi. 2018. "MicroRNAs in Smoking-Related Carcinogenesis: Biomarkers, Functions, and Therapy" Journal of Clinical Medicine 7, no. 5: 98. https://doi.org/10.3390/jcm7050098
APA StyleFujii, T., Shimada, K., Nakai, T., & Ohbayashi, C. (2018). MicroRNAs in Smoking-Related Carcinogenesis: Biomarkers, Functions, and Therapy. Journal of Clinical Medicine, 7(5), 98. https://doi.org/10.3390/jcm7050098





