Bioinformatic Interrogation of 5p-arm and 3p-arm Specific miRNA Expression Using TCGA Datasets
Abstract
:1. Introduction
2. Experimental Section
2.1. Arm Feature Assignment of Human Mature miRNAs
2.2. miRNA-Seq Datasets from TCGA
2.3. Bioinformatic Analysis with Comprehensive Arm-Feature-Annotated miRNA
3. Results and Discussion
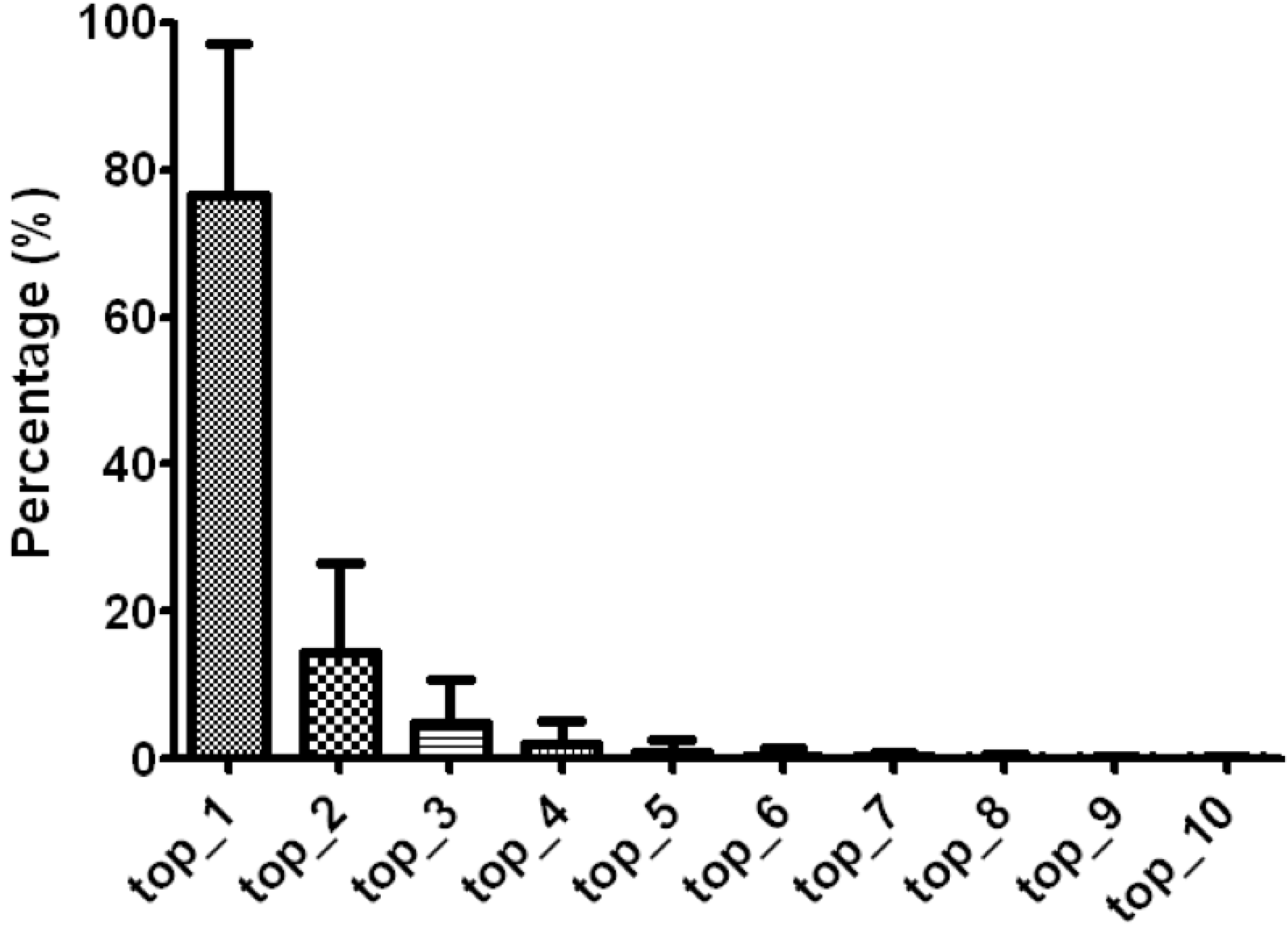
3.1. Arm Features and isomiR Quantifications

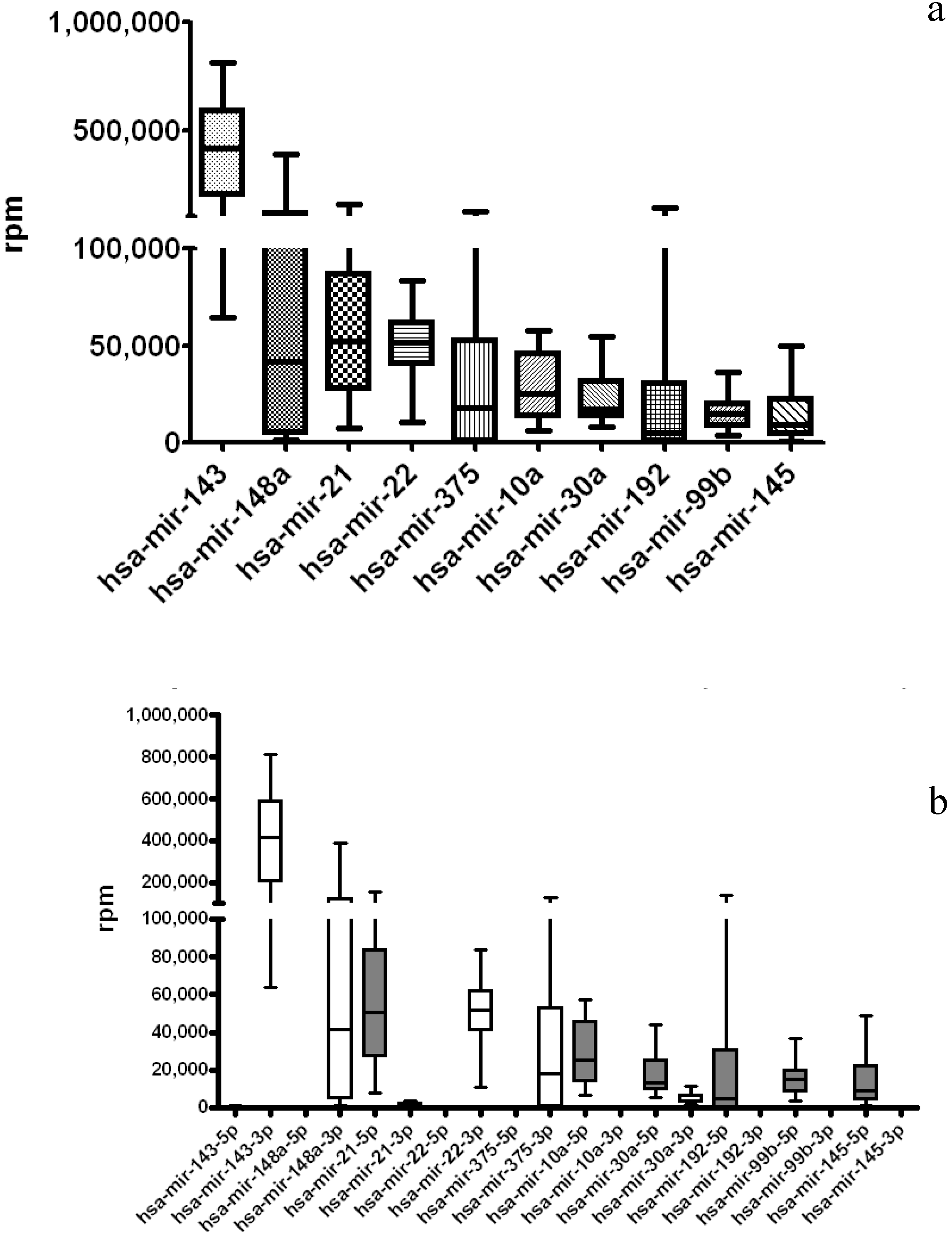
3.2. TCGA miRNA-Seq Analysis: STAD Gastric Cancer Dataset
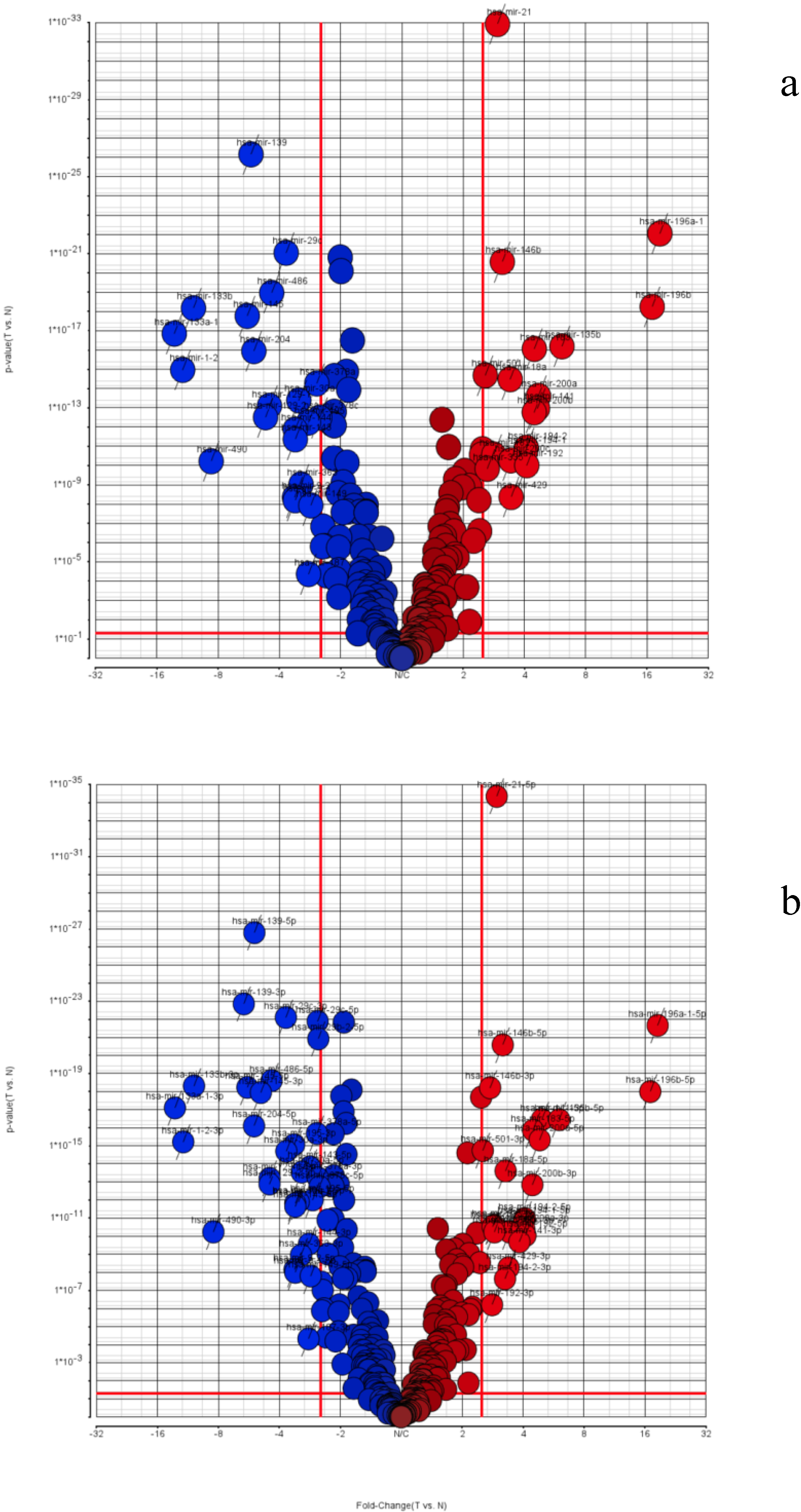
| TCGA STAD (Stomach Adenocarcinoma) | |||
|---|---|---|---|
| Overall miRNA Precursors | Separate 5p-arm and 3p-arm miRNAs | ||
| Down regulated | Up regulated | Down regulated | Up regulated |
| hsa-miR-139 | hsa-miR-21 | hsa-miR-139-5p | hsa-miR-21-5p |
| hsa-miR-29c | hsa-miR-196a-1 | hsa-miR-139-3p | hsa-miR-196a-1-5p |
| hsa-miR-486 | hsa-miR-146b | hsa-miR-29c-3p | hsa-miR-146b-5p |
| hsa-miR-133b | hsa-miR-196b | hsa-miR-29c-5p | hsa-miR-146b-3p |
| hsa-miR-145 | hsa-miR-135b | hsa-miR-29b-2-5p | hsa-miR-196b-5p |
| hsa-miR-133a-1 | hsa-miR-183 | hsa-miR-486-5p | hsa-miR-141-5p |
| hsa-miR-204 | hsa-miR-501 | hsa-miR-133b-3p | hsa-miR-135b-5p |
| hsa-miR-1-2 | hsa-miR-18a | hsa-miR-145-5p | hsa-miR-183-5p |
| hsa-miR-378a | hsa-miR-200a | hsa-miR-145-3p | hsa-miR-200a-5p |
| hsa-miR-30a | hsa-miR-141 | hsa-miR-133a-1-3p | hsa-miR-501-3p |
| hsa-miR-129-1 | hsa-miR-200b | hsa-miR-204-5p | hsa-miR-18a-5p |
| hsa-miR-129-2 | hsa-miR-194-2 | hsa-miR-378a-5p | hsa-miR-200b-3p |
| hsa-miR-378c | hsa-miR-194-1 | hsa-miR-1-2-3p | hsa-miR-194-2-5p |
| hsa-miR-195 | hsa-miR-182 | hsa-miR-195-3p | hsa-miR-194-1-5p |
| hsa-miR-144 | hsa-miR-200c | hsa-miR-30a-3p | hsa-miR-335-3p |
| hsa-miR-143 | hsa-miR-192 | hsa-miR-143-5p | hsa-miR-182-5p |
| hsa-miR-490 | hsa-miR-335 | hsa-miR-30a-5p | hsa-miR-200a-3p |
| hsa-miR-363 | hsa-miR-429 | hsa-miR-378a-3p | hsa-miR-200c-3p |
| hsa-miR-9-2 | hsa-miR-129-1-5p | hsa-miR-708-5p | |
| hsa-miR-9-1 | hsa-miR-129-2-5p | hsa-miR-192-5p | |
| hsa-miR-149 | hsa-miR-378c-5p | hsa-miR-141-3p | |
| hsa-miR-187 | hsa-miR-195-5p | hsa-miR-429-3p | |
| hsa-miR-30c-2-3p | hsa-miR-194-2-3p | ||
| hsa-miR-144-5p | hsa-miR-192-3p | ||
| hsa-miR-143-3p | |||
| hsa-miR-490-3p | |||
| hsa-miR-144-3p | |||
| hsa-miR-363-3p | |||
| hsa-miR-9-2-5p | |||
| hsa-miR-9-1-5p | |||
| hsa-miR-149-5p | |||
| hsa-miR-187-3p | |||
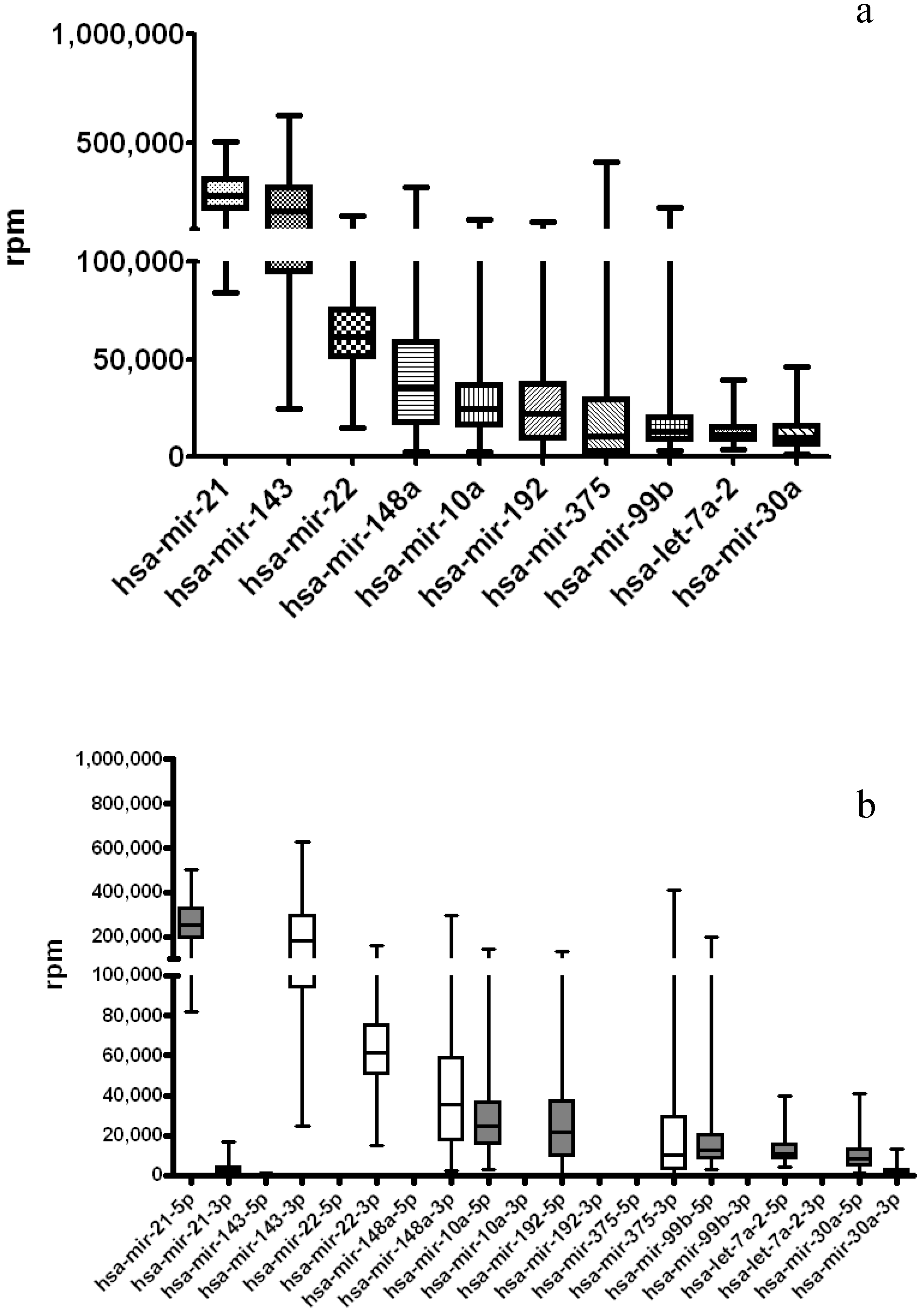
3.3. miRNA-Seq Analysis on Additional 12 TCGA Cancer Types
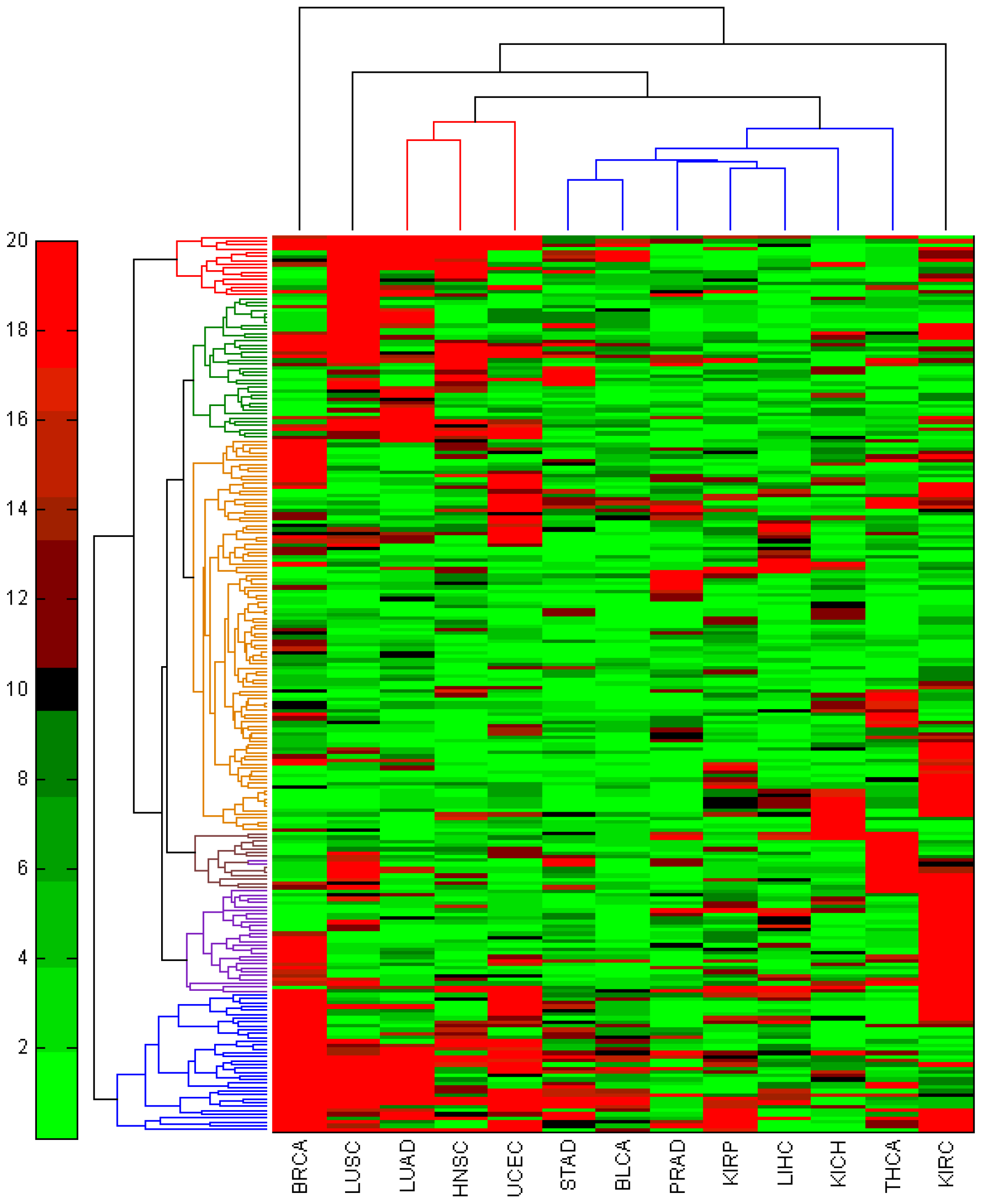
4. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- He, C.Z.; Zhang, K.H.; Li, Q.; Liu, X.H.; Hong, Y.; Lv, N.H. Combined use of AFP, CEA, CA125 and CAL9-9 improves the sensitivity for the diagnosis of gastric cancer. BMC Gastroenterol. 2013, 13. [Google Scholar] [CrossRef] [PubMed]
- Liao, Y.L.; Tsai, K.W.; Lin, W.C. miRNAs in gastric cancer. In Gastric Carcinoma—Molecular Aspects and Current Advances; Lotfy, D.M., Ed.; InTech Open: Rijeka, Croatia, 2011; pp. 87–104. [Google Scholar]
- Perez-Rivas, L.G.; Jerez, J.M.; Carmona, R.; de Luque, V.; Vicioso, L.; Claros, M.G.; Viguera, E.; Pajares, B.; Sanchez, A.; Ribelles, N.; et al. A microRNA signature associated with early recurrence in breast cancer. PLoS ONE 2014, 9, e91884. [Google Scholar] [CrossRef] [PubMed]
- Tsai, K.W.; Liao, Y.L.; Wu, C.W.; Hu, L.Y.; Li, S.C.; Chan, W.C.; Ho, M.R.; Lai, C.H.; Kao, H.W.; Fang, W.L.; et al. Aberrant expression of miR-196a in gastric cancers and correlation with recurrence. Genes Chromosomes Cancer 2012, 51, 394–401. [Google Scholar] [CrossRef] [PubMed]
- Kosaka, N.; Iguchi, H.; Ochiya, T. Circulating microRNA in body fluid: A new potential biomarker for cancer diagnosis and prognosis. Cancer Sci. 2010, 101, 2087–2092. [Google Scholar] [CrossRef] [PubMed]
- Bartel, D.P. MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell 2004, 116, 281–297. [Google Scholar] [CrossRef]
- Bartel, D.P. MicroRNAs: Target recognition and regulatory functions. Cell 2009, 136, 215–233. [Google Scholar] [CrossRef] [PubMed]
- Lujambio, A.; Lowe, S.W. The microcosmos of cancer. Nature 2012, 482, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Rosenfeld, N.; Aharonov, R.; Meiri, E.; Rosenwald, S.; Spector, Y.; Zepeniuk, M.; Benjamin, H.; Shabes, N.; Tabak, S.; Levy, A.; et al. MicroRNAs accurately identify cancer tissue origin. Nat. Biotechnol. 2008, 26, 462–469. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, R.U.; Miyazaki, H.; Ochiya, T. The role of microRNAs in the regulation of cancer stem cells. Front. Genet. 2014, 4. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, R.U.; Miyazaki, H.; Ochiya, T. The roles of microRNAs in breast cancer. Cancers (Basel) 2015, 7, 598–616. [Google Scholar] [CrossRef] [PubMed]
- Kosaka, N.; Iguchi, H.; Yoshioka, Y.; Takeshita, F.; Matsuki, Y.; Ochiya, T. Secretory mechanisms and intercellular transfer of microRNAs in living cells. J. Biol. Chem. 2010, 285, 17442–17452. [Google Scholar] [CrossRef] [PubMed]
- Schwarzenbach, H.; Nishida, N.; Calin, G.A.; Pantel, K. Clinical relevance of circulating cell-free microRNAs in cancer. Nat. Rev. Clin. Oncol. 2014, 11, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Tominaga, N.; Kosaka, N.; Ono, M.; Katsuda, T.; Yoshioka, Y.; Tamura, K.; Lotvall, J.; Nakagama, H.; Ochiya, T. Brain metastatic cancer cells release microRNA-181c-containing extracellular vesicles capable of destructing blood-brain barrier. Nat. Commun. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Sugimachi, K.; Matsumura, T.; Hirata, H.; Uchi, R.; Ueda, M.; Ueo, H.; Shinden, Y.; Iguchi, T.; Eguchi, H.; Shirabe, K.; et al. Identification of a bona fide microRNA biomarker in serum exosomes that predicts hepatocellular carcinoma recurrence after liver transplantation. Br. J. Cancer 2015, 112, 532–538. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, T.; Sugimachi, K.; Iinuma, H.; Takahashi, Y.; Kurashige, J.; Sawada, G.; Ueda, M.; Uchi, R.; Ueo, H.; Takano, Y.; et al. Exosomal microRNA in serum is a novel biomarker of recurrence in human colorectal cancer. Br. J. Cancer 2015, 113, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Kuo, W.T.; Lai, C.H.; Wu, C.W.; Fang, W.L.; Huang, K.H.; Lin, W.C. Urine miR-21-5p as a potential non-invasive biomarker for gastric cancer. Oncol. Lett. 2015, in press. [Google Scholar]
- Osaki, M.; Kosaka, N.; Okada, F.; Ochiya, T. Circulating microRNAs in drug safety assessment for hepatic and cardiovascular toxicity: The latest biomarker frontier? Mol. Diagn. Ther. 2014, 18, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Haldrup, C.; Kosaka, N.; Ochiya, T.; Borre, M.; Hoyer, S.; Orntoft, T.F.; Sorensen, K.D. Profiling of circulating microRNAs for prostate cancer biomarker discovery. Drug Deliv. Transl. Res. 2014, 4, 19–30. [Google Scholar] [CrossRef] [PubMed]
- Gregory, R.I.; Chendrimada, T.P.; Cooch, N.; Shiekhattar, R. Human RISC couples microRNA biogenesis and posttranscriptional gene silencing. Cell 2005, 123, 631–640. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Lee, Y.; Yeom, K.H.; Nam, J.W.; Heo, I.; Rhee, J.K.; Sohn, S.Y.; Cho, Y.; Zhang, B.T.; Kim, V.N. Molecular basis for the recognition of primary microRNAs by the Drosha-DGCR8 complex. Cell 2006, 125, 887–901. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Ahn, C.; Han, J.; Choi, H.; Kim, J.; Yim, J.; Lee, J.; Provost, P.; Radmark, O.; Kim, S.; et al. The nuclear RNAse III Drosha initiates microRNA processing. Nature 2003, 425, 415–419. [Google Scholar] [CrossRef] [PubMed]
- Khvorova, A.; Reynolds, A.; Jayasena, S.D. Functional siRNAs and miRNAs exhibit strand bias. Cell 2003, 115, 209–216. [Google Scholar] [CrossRef]
- Li, S.C.; Liao, Y.L.; Ho, M.R.; Tsai, K.W.; Lai, C.H.; Lin, W.C. miRNA arm selection and isomiR distribution in gastric cancer. BMC Genomics 2012, 13 (Suppl. 1). [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Arcila, M.L.; Li, Z.; Lee, E.J.; Henzler, C.; Liu, J.; Rana, T.M.; Kosik, K.S. Deep annotation of mouse iso-miR and iso-moR variation. Nucleic Acids Res. 2012, 40, 5864–5875. [Google Scholar] [CrossRef] [PubMed]
- Kuo, W.T.; Ho, M.R.; Wu, C.W.; Fang, W.L.; Huang, K.H.; Lin, W.C. Interrogation of microRNAs involved in gastric cancer using 5p-arm and 3p-arm annotated microRNAs. Anticancer Res. 2015, 35, 1345–1352. [Google Scholar] [PubMed]
- The Cancer Genome Atlas Data Portal. Available online: https://tcga-data.nci.nih.gov/tcga/tcgaHome2.jsp (accessed on 13 September 2015).
- Griffiths-Jones, S.; Grocock, R.J.; van Dongen, S.; Bateman, A.; Enright, A.J. miRBase: MicroRNA sequences, targets and gene nomenclature. Nucleic Acids Res. 2006, 34, D140–D144. [Google Scholar] [CrossRef] [PubMed]
- Griffiths-Jones, S.; Saini, H.K.; van Dongen, S.; Enright, A.J. miRBase: Tools for microRNA genomics. Nucleic Acids Res. 2008, 36, D154–D158. [Google Scholar] [CrossRef] [PubMed]
- Gailhouste, L.; Ochiya, T. Cancer-related microRNAs and their role as tumor suppressors and oncogenes in hepatocellular carcinoma. Histol. Histopathol. 2013, 28, 437–451. [Google Scholar] [PubMed]
- Neilsen, C.T.; Goodall, G.J.; Bracken, C.P. IsomiRs—The overlooked repertoire in the dynamic microRNAome. Trends Genet. 2012, 28, 544–549. [Google Scholar] [CrossRef] [PubMed]
- Chan, S.H.; Wu, C.W.; Li, A.F.; Chi, C.W.; Lin, W.C. miR-21 microRNA expression in human gastric carcinomas and its clinical association. Anticancer Res. 2008, 28, 907–911. [Google Scholar] [PubMed]
- Cancer Genome Atlas Research Network. Comprehensive molecular characterization of gastric adenocarcinoma. Nature 2014, 513, 202–209. [Google Scholar]
- Kang, S.M.; Choi, J.W.; Hong, S.H.; Lee, H.J. Up-regulation of microRNA* strands by their target transcripts. Int. J. Mol. Sci. 2013, 14, 13231–13240. [Google Scholar] [CrossRef] [PubMed]
- Ro, S.; Park, C.; Young, D.; Sanders, K.M.; Yan, W. Tissue-dependent paired expression of miRNAs. Nucleic Acids Res. 2007, 35, 5944–5953. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, D.S.; Hutvagner, G.; Du, T.; Xu, Z.; Aronin, N.; Zamore, P.D. Asymmetry in the assembly of the RNAi enzyme complex. Cell 2003, 115, 199–208. [Google Scholar] [CrossRef]
- Guo, L.; Zhang, H.; Zhao, Y.; Yang, S.; Chen, F. Selected isomiR expression profiles via arm switching? Gene 2014, 533, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Griffiths-Jones, S.; Hui, J.H.; Marco, A.; Ronshaugen, M. MicroRNA evolution by arm switching. EMBO Rep. 2011, 12, 172–177. [Google Scholar] [CrossRef] [PubMed]
- Auyeung, V.C.; Ulitsky, I.; McGeary, S.E.; Bartel, D.P. Beyond secondary structure: Primary-sequence determinants license pri-miRNA hairpins for processing. Cell 2013, 152, 844–858. [Google Scholar] [CrossRef] [PubMed]
- Larne, O.; Ostling, P.; Haflidadottir, B.S.; Hagman, Z.; Aakula, A.; Kohonen, P.; Kallioniemi, O.; Edsjo, A.; Bjartell, A.; Lilja, H.; et al. miR-183 in prostate cancer cells positively regulates synthesis and serum levels of prostate-specific antigen. Eur. Urol. 2015, 68, 581–588. [Google Scholar] [CrossRef] [PubMed]
- Noh, J.H.; Chang, Y.G.; Kim, M.G.; Jung, K.H.; Kim, J.K.; Bae, H.J.; Eun, J.W.; Shen, Q.; Kim, S.J.; Kwon, S.H.; et al. miR-145 functions as a tumor suppressor by directly targeting histone deacetylase 2 in liver cancer. Cancer Lett. 2013, 335, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Tsai, K.W.; Hu, L.Y.; Wu, C.W.; Li, S.C.; Lai, C.H.; Kao, H.W.; Fang, W.L.; Lin, W.C. Epigenetic regulation of miR-196b expression in gastric cancer. Genes Chromosomes Cancer 2010, 49, 969–980. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.H.; Yeh, C.T.; Ho, J.Y.; Ng, K.F.; Chen, T.C. OncomiR miR-96 and miR-182 promote cell proliferation and invasion through targeting ephrinA5 in hepatocellular carcinoma. Mol. Carcinog. 2015. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.D.; Jiang, L.H.; Sun, D.W.; Li, J.; Tang, J.H. miR-139-5p: Promising biomarker for cancer. Tumour. Biol. 2015, 36, 1355–1365. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, K.; Steptoe, A.L.; Martin, H.C.; Pattabiraman, D.R.; Nones, K.; Waddell, N.; Mariasegaram, M.; Simpson, P.T.; Lakhani, S.R.; Vlassov, A.; et al. miR-139-5p is a regulator of metastatic pathways in breast cancer. RNA 2013, 19, 1767–1780. [Google Scholar] [CrossRef] [PubMed]
- Uchino, K.; Takeshita, F.; Takahashi, R.U.; Kosaka, N.; Fujiwara, K.; Naruoka, H.; Sonoke, S.; Yano, J.; Sasaki, H.; Nozawa, S.; et al. Therapeutic effects of microRNA-582-5p and -3p on the inhibition of bladder cancer progression. Mol. Ther. 2013, 21, 610–619. [Google Scholar] [CrossRef] [PubMed]
- Baraniskin, A.; Birkenkamp-Demtroder, K.; Maghnouj, A.; Zollner, H.; Munding, J.; Klein-Scory, S.; Reinacher-Schick, A.; Schwarte-Waldhoff, I.; Schmiegel, W.; Hahn, S.A. miR-30a-5p suppresses tumor growth in colon carcinoma by targeting DTL. Carcinogenesis 2012, 33, 732–739. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuo, W.-T.; Su, M.-W.; Lee, Y.L.; Chen, C.-H.; Wu, C.-W.; Fang, W.-L.; Huang, K.-H.; Lin, W.-c. Bioinformatic Interrogation of 5p-arm and 3p-arm Specific miRNA Expression Using TCGA Datasets. J. Clin. Med. 2015, 4, 1798-1814. https://doi.org/10.3390/jcm4091798
Kuo W-T, Su M-W, Lee YL, Chen C-H, Wu C-W, Fang W-L, Huang K-H, Lin W-c. Bioinformatic Interrogation of 5p-arm and 3p-arm Specific miRNA Expression Using TCGA Datasets. Journal of Clinical Medicine. 2015; 4(9):1798-1814. https://doi.org/10.3390/jcm4091798
Chicago/Turabian StyleKuo, Wei-Ting, Ming-Wei Su, Yungling Leo Lee, Chien-Hsiun Chen, Chew-Wun Wu, Wen-Liang Fang, Kuo-Hung Huang, and Wen-chang Lin. 2015. "Bioinformatic Interrogation of 5p-arm and 3p-arm Specific miRNA Expression Using TCGA Datasets" Journal of Clinical Medicine 4, no. 9: 1798-1814. https://doi.org/10.3390/jcm4091798
APA StyleKuo, W.-T., Su, M.-W., Lee, Y. L., Chen, C.-H., Wu, C.-W., Fang, W.-L., Huang, K.-H., & Lin, W.-c. (2015). Bioinformatic Interrogation of 5p-arm and 3p-arm Specific miRNA Expression Using TCGA Datasets. Journal of Clinical Medicine, 4(9), 1798-1814. https://doi.org/10.3390/jcm4091798





