How Often Do Comparative Randomised Controlled Trials in the Field of Eczema Fail to Directly Compare the Treatments Being Tested?
Abstract
:1. Introduction
2. Materials and Methods
2.1. Selection of Publications
2.2. Review of Papers
| Outcome | Data Distribution | No. Parallel Groups | Appropriate Analysis |
|---|---|---|---|
| Continuous | Normal | 2 | Student t-test or Multivariate linear regression |
| Normal | ≥2 | Analysis of variance or Analysis of Covariance | |
| Non-normal | 2 | Mann-Whitney U test | |
| Non-normal | >2 | Kruskal-Wallis test | |
| Categorical | N/A | ≥2 | Pearson Chi-squared test |
2.3. Citation per Article and Journal Impact Factor
2.4. Validation of the GREAT Database
2.5. Abstract Results
3. Results
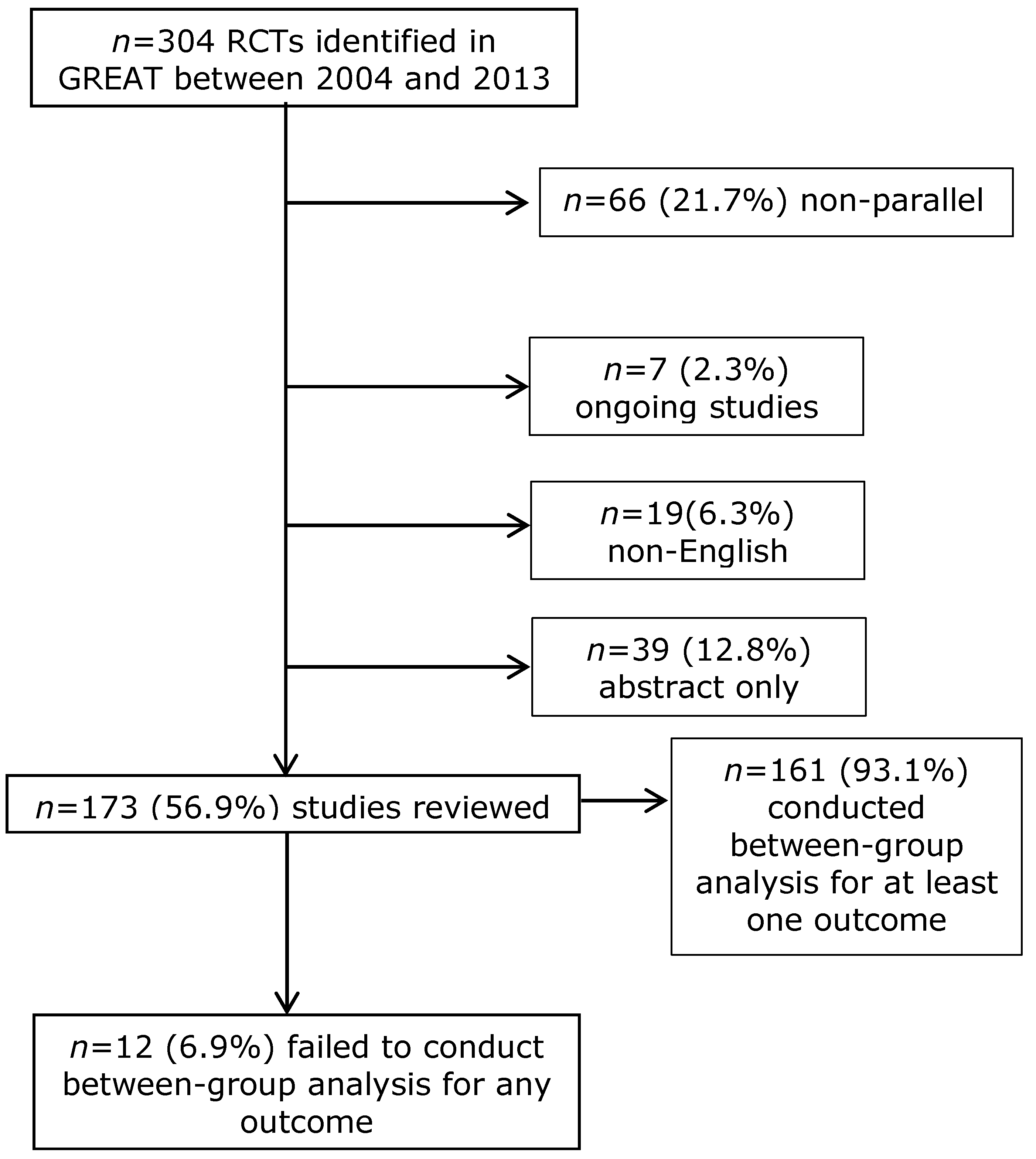
3.1. Selection of Studies
| Author | Title | Year | Size | Journal | Impact Factor | Citations | Funding | Conclusion |
|---|---|---|---|---|---|---|---|---|
| Draelos, Z [9] | Pharmacokinetics of topical calcineurin inhibitors in adult atopic dermatitis: A randomized, investigator-blind comparison | 2005 | 37 | Journal of the American Academy of Dermatology | 4.91 | 38 | Not reported | “Pimecrolimus appears to be associated with lower systemic drug exposure than tacrolimus.” |
| Taniuchi, S [10] | Administration of Bifidobacterium to infants with atopic dermatitis: Changes in fecal microflora and clinical symptoms | 2005 | 17 | Journal of Applied Research | 0 | 10 | Commercial | “Administration of bifidobacteria to infants with cow’s milk hypersensitivity with atopic dermatitis significantly increased the proportion of bifidobacteria in the fecal microflora and also might improve their allergic symptoms.” |
| Kimata, H [11] | Improvement of atopic dermatitis and reduction of skin allergic responses by oral intake of konjac ceramide | 2006 | 50 | Paediatric Dermatology | 1.52 | 23 | Government | “These results demonstrated that oral intake of konjac ceramide significantly improved skin symptoms in children with AD.” |
| Hennino, A [12] | Influence of measles vaccination on the progression of atopic dermatitis in infants | 2007 | 12 | Pediatric Allergy & Immunology | 3.86 | 2 | Commercial | “These data suggest that measles vaccination not only does not aggravate AD, but may also improve some of the immunological parameters of this allergic disease.” |
| Reitamo, S [13] | The pharmacokinetics of tacrolimus after first and repeated dosing with 0.03% ointment in infants with atopic dermatitis. | 2009 | 53 | International Journal of Dermatology | 1.23 | 12 | Not reported | “Treatment was well tolerated and led to considerable improvement.” |
| Yokoyama, Y [14] | Ethylene vinyl alcohol (EVOH) fiber compared to cotton underwear in the treatment of childhood atopic dermatitis: A Double-blind randomized study | 2009 | 21 | Indian Pediatrics | 1.04 | 2 | Not reported | “Ethylene vinyl alcohol fiber underwear might be useful for children with atopic dermatitis.” |
| Yoshida, Y [15] | Clinical effects of probiotic Bifidobacterium breve supplementation in adult patients with atopic dermatitis | 2010 | 24 | Yonago Acta medica | 0.27 | 3 | Commercial | “Our results suggest that B. breve may be beneficial for the treatment of atopic dermatitis.” |
| Byun, HJ [16] | Full-spectrum light phototherapy for atopic dermatitis | 2011 | 38 | International Journal of Dermatology | 1.23 | 11 | University | “We showed that FSL phototherapy can be an effective and safe treatment option in AD.” |
| Amestejani, MD [8] | Vitamin D supplementation in the treatment of atopic dermatitis: A clinical trial study | 2012 | 60 | Journal of Drugs in Dermatology | 1.95 | 41 | Not reported | “Supplementation with oral vitamin D dramatically improved disease severity in AD patients.” |
| Bae, B [17] | Progressive muscle relaxation therapy for atopic dermatitis: Objective assessment of efficacy | 2012 | 25 | Acta Dermato-Venereologica | 4.24 | 19 | Not reported | “Progressive muscle relaxation may be a useful adjunctive modality for the management of atopic dermatitis through the reduction of anxiety.” |
| Chung, BY [18] | Dose-dependent effects of evening primrose oil in children and adolescents with atopic dermatitis. | 2013 | 40 | Annals of dermatology | 0.95 | 1 | University | “The results of this study suggest that the 320 mg and 160 mg of primrose oil groups may be equally effective in treating AD patients.” |
| Iyengar, SR [19] | Immunologic effects of omalizumab in children with severe refractory atopic dermatitis: A randomized, placebo-controlled clinical trial | 2013 | 8 | International Archives of Allergy and Immunology | 2.43 | 21 | Non-profit organisation | “Patients on anti-IgE therapy had an improvement in clinical outcomes as measured by the SCORAD system; however, these effects were comparable to improvements in the control group.” |
3.2. Characteristics of Studies that Failed to Compare Their Hypothesis
3.3. Misleading Conclusions
3.4. Validation of the GREAT Database
3.5. Exploration of Abstracts for Studies that Had Conducted a Between-Group Analysis
4. Discussion
4.1. Main Findings
4.2. Strengths and Limitations
4.3. Comparison with Other Studies
4.4. Implications of Our Findings
Conclusions
Author Contributions
Conflicts of Interest
References
- Bigby, M.; Gadenne, A.-S. Understanding and evaluating clinical trials. J. Am. Acad. Dermatol. 1996, 34, 555–590. [Google Scholar] [CrossRef] [PubMed]
- Kunz, R.; Oxman, A.D. The unpredictability paradox: Review of empirical comparisons of randomised and non-randomised clinical trials. BMJ 1998, 317, 1185–1190. [Google Scholar] [CrossRef] [PubMed]
- Gray, J.A.M. Evidence-Based Healthcare; Churchill Livingstone Inc.: New York, NY, USA, 1997; pp. 78–86. [Google Scholar]
- Adetugbo, K.; Williams, H. How well are randomized controlled trials reported in the dermatology literature? Arch. Dermatol. 2000, 136, 381–385. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, F.; Meyer, N.; Gourraud, P.A.; Paul, C. CONSORT adoption and quality of reporting of randomized controlled trials: A systematic analysis in two dermatology journals. Br. J. Dermatol. 2009, 161, 1159–1165. [Google Scholar] [CrossRef] [PubMed]
- GREA. Available online: http://www.greatdatabase.org.uk (accessed on 24 February 2015).
- Drago, L.; Iemoli, E.; Rodighiero, V.; Nicola, L.; de Vecchi, E.; Piconi, S. Effects of Lactobacillus salivarius LS01 (DSM 22775) treatment on adult atopic dermatitis: A randomized placebo-controlled study. Int. J. Immunopathol. Pharmacol. 2011, 24, 1037–1048. [Google Scholar] [PubMed]
- Amestejani, M.; Salehi, B.S.; Vasigh, M.; Sobhkhiz, A.; Karami, M.; Alnia, H.; Kamrava, S.K.; Shamspour, N.; Ghalehbaghi, B.; Behzadi, A.H. Vitamin D supplementation in the treatment of atopic dermatitis: A clinical trial study. J. Drugs Dermatol. 2012, 11, 327–330. [Google Scholar] [PubMed]
- Draelos, Z.; Nayak, A.; Pariser, D.; Shupack, J.L.; Chon, K.; Abrams, B.; Paul, C.F. Pharmacokinetics of topical calcineurin inhibitors in adult atopic dermatitis: A randomized, investigator-blind comparison. J. Am. Acad. Dermatol. 2005, 53, 602–609. [Google Scholar] [CrossRef] [PubMed]
- Tanuichi, S.; Hattori, K.; Yamamoto, A.; Sasai, M.; Hatano, Y.; Takatsugu, K.; Kobayashi, Y.; Iwamoto, H.; Yaeshima, T. Administration of Bifidobacterium to infants with atopic dermatitis: Changes in fecal microflora and clinical symptoms. J. Appl. Res. 2005, 5, 387–396. [Google Scholar]
- Kimata, H. Improvement of atopic dermatitis and reduction of skin allergic responses by oral intake of konjac ceramide. Pediatr. Dermatol. 2006, 23, 386–389. [Google Scholar] [CrossRef] [PubMed]
- Hennino, A.; Cornu, C.; Rozieres, A.; Augey, F.; Villard-Truc, F.; Payot, F.; Lachaux, A.; Nicolas, J.F.; Horvat, B. Influence of measles vaccination on the progression of atopic dermatitis in infants. Pediatr. Allergy Immunol. 2007, 18, 385–390. [Google Scholar] [CrossRef] [PubMed]
- Reitamo, S.; Mandelin, J.; Rubins, A.; Remitz, A.; Mäkelä, M.; Cirule, K.; Rubins, S.; Zigure, S.; Ho, V.; Dickinson, J.; et al. The pharmacokinetics of tacrolimus after first and repeated dosing with 0.03% ointment in infants with atopic dermatitis. Int. J. Dermatol. 2009, 48, 348–355. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, Y.; Kimata, H.; Mitarai, S.; Hirano, S.; Shirakawa, T. Ethylene vinyl alcohol (EVOH) fiber comapred to cotton udewear in the treatment of childhood atopic dermatitis: A double-blind randomised study. Indian Pediatr. 2009, 46, 611–614. [Google Scholar] [PubMed]
- Yoshida, R.; Fukami, M.; Sasagawa, I.; Hasegawa, T.; Kamatani, N.; Ogata, T. Association of cryptorchidism with a specific haplotype of the estrogen receptor alpha gene: Implication for the susceptibility to estrogenic environmental endocrine disruptors. J. Clin. Endocrinol. Metab. 2005, 90, 4716–4721. [Google Scholar] [CrossRef] [PubMed]
- Byun, H.J.; Lee, H.I.; Kim, B.; Kim, M.N.; Hong, H.; Choi, Y.; Jo, Y.; Cho, K.H.; Mun, S.-K. Full-spectrum light phototherapy for atopic dermatitis. Int. J. Dermatol. 2011, 50, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Bae, B.G.; Oh, S.H.; Park, C.O.; Noh, S.; Noh, J.Y.; Kim, K.R.; Lee, K.H. Progressive muscle relaxation therapy for atopic dermatitis: Objective assessment of efficacy. Acta Derm. Venereol. 2012, 92, 57–61. [Google Scholar] [PubMed]
- Chung, B.Y.; Kim, J.H.; Cho, S.I.; Ahn, I.S.; Kim, H.O.; Park, C.W.; Lee, C.H. Dose-dependent effects of evening primrose oil in children and adolescents with atopic dermatitis. Ann. Dermatol. 2013, 25, 285–291. [Google Scholar] [CrossRef] [PubMed]
- Iyengar, S.R.; Hoyte, E.G.; Loza, A.; Bonaccorso, S.; Chiang, D.; Umetsu, D.T.; Nadeau, K.C. Immunologic effects of omalizumab in children with severe refractory atopic dermatitis: A randomized, placebo-controlled clinical trial. Int. Arch. Allergy Immunol. 2013, 162, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Yadav, M.; Mittal, K. Effect of Vitamin D Supplementation on moderate to severe bronchial asthma. Indian J. Pediatr. 2014, 81, 650–654. [Google Scholar] [CrossRef] [PubMed]
- Barman, M.; Jonsson, K.; Hesselmar, B.; Sandin, A.; Sandberg, A.-S.; Wold, A.E. No association between allergy and current 25-hydroxy vitamin D in serum or vitamin D intake. Acta Paediatrica 2015, 104, 405–413. [Google Scholar] [CrossRef] [PubMed]
- Camargo, C.A., Jr.; Ganmaa, D.; Sidbury, R.; Erdenedelger, K.; Radnaakhand, N.; Khandsuren, B. Randomized trial of vitamin D supplementation for winter-related atopic dermatitis in children. J. Allergy Clin. Immunol. 2014, 134, 831.e1–835.e1. [Google Scholar] [CrossRef] [PubMed]
- Boyle, R.J.; Bath-Hextall, F.J.; Leonardi-Bee, J.; Murrell, D.F.; Tang, M.L.K. Probiotics for treating eczema. Cochrane Database Syst. Rev. 2008. [Google Scholar] [CrossRef]
- Nankervis, H.; Baibergenova, A.; Williams, H.C.; Thomas, K.S. Prospective registration and outcome-reporting bias in randomized controlled trials of eczema treatments: A systematic review. J. Investig. Dermatol. 2012, 132, 2727–2734. [Google Scholar] [CrossRef] [PubMed]
- Dwan, K.; Altman, D.G.; Arnaiz, J.A.; Bloom, J.; Chan, A.-W.; Cronin, E.; Decullier, E.; Easterbrook, P.J.; Von Elm, E.; Gamble, C.; et al. Systematic review of the empirical evidence of study publication bias and outcome reporting bias. PLoS ONE 2008, 3, e3081. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Fortin, P.; Jadad, A.R.; Juni, P.; Klassen, T.; Le Lorier, J.; Liberati, A.; Linde, K.; Penne, A. Completeness of reporting trials published in langauges other than Englsih: Implications for conduct and reporting of systematic reviews. Lancet 1996, 10, 363–366. [Google Scholar] [CrossRef]
- Williams, H.C. Clinical Trials Submitted to the JID: Place your bet and show us your hand. Soc. Investig. Dermatol. 2015, 135, 325–327. [Google Scholar]
- Batchelor, J.; Spuls, P.I. Prospective registration of clinical trials published in the British Journal of Dermatology. Br. J. Dermatol. 2014, 171, 681–683. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ratib, S.; Wilkes, S.R.; Nankervis, H.; Thomas, K.S.; Williams, H.C. How Often Do Comparative Randomised Controlled Trials in the Field of Eczema Fail to Directly Compare the Treatments Being Tested? J. Clin. Med. 2015, 4, 1312-1324. https://doi.org/10.3390/jcm4061312
Ratib S, Wilkes SR, Nankervis H, Thomas KS, Williams HC. How Often Do Comparative Randomised Controlled Trials in the Field of Eczema Fail to Directly Compare the Treatments Being Tested? Journal of Clinical Medicine. 2015; 4(6):1312-1324. https://doi.org/10.3390/jcm4061312
Chicago/Turabian StyleRatib, Sonia, Sally R. Wilkes, Helen Nankervis, Kim S. Thomas, and Hywel C. Williams. 2015. "How Often Do Comparative Randomised Controlled Trials in the Field of Eczema Fail to Directly Compare the Treatments Being Tested?" Journal of Clinical Medicine 4, no. 6: 1312-1324. https://doi.org/10.3390/jcm4061312
APA StyleRatib, S., Wilkes, S. R., Nankervis, H., Thomas, K. S., & Williams, H. C. (2015). How Often Do Comparative Randomised Controlled Trials in the Field of Eczema Fail to Directly Compare the Treatments Being Tested? Journal of Clinical Medicine, 4(6), 1312-1324. https://doi.org/10.3390/jcm4061312





