The Role of Malassezia spp. in Atopic Dermatitis
Abstract
:1. Introduction
2. Malassezia spp. As Part of the Normal and Atopic Skin Flora
3. Sensitization to Malassezia spp. May Correlate with the Severity of Atopic Dermatitis
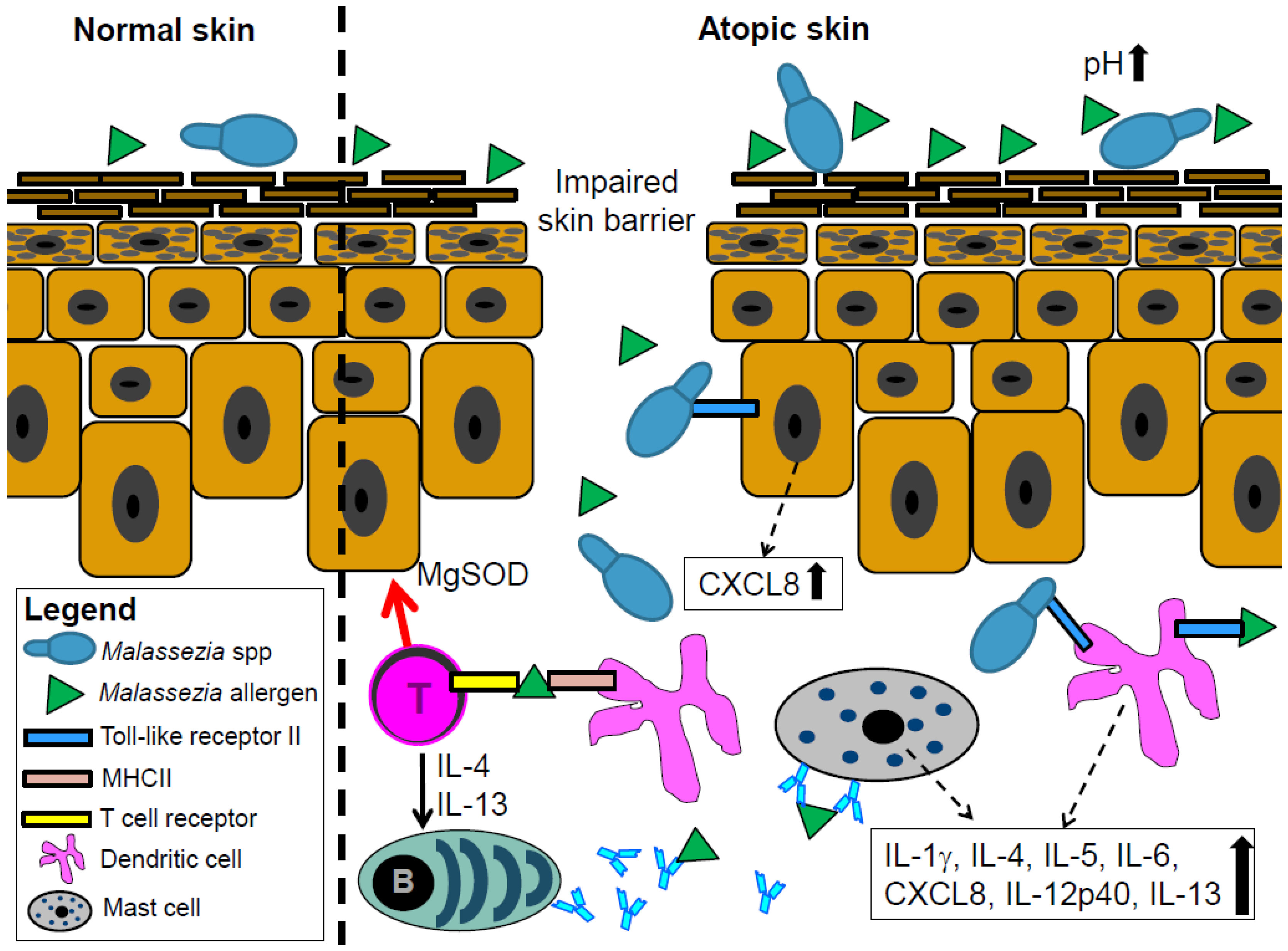
4. Malassezia spp. Interacts with the Skin Immune System
Therapeutic Approaches with Antifungals in AD
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bieber, T. Atopic dermatitis. N. Engl. J. Med. 2008, 358, 1483–1494. [Google Scholar] [CrossRef] [PubMed]
- Williams, H.; Flohr, C. How epidemiology has challenged 3 prevailing concepts about atopic dermatitis. J. Allergy Clin. Immunol. 2006, 118, 209–213. [Google Scholar] [CrossRef] [PubMed]
- De Benedetto, A.; Kubo, A.; Beck, L.A. Skin barrier disruption: A requirement for allergen sensitization? J. Investig. Dermatol. 2012, 132, 949–963. [Google Scholar] [CrossRef] [PubMed]
- Kuo, I.H.; Yoshida, T.; De Benedetto, A.; Beck, L.A. The cutaneous innate immune response in patients with atopic dermatitis. J. Allergy Clin. Immunol. 2013, 131, 266–278. [Google Scholar] [CrossRef] [PubMed]
- Howell, M.D.; Gallo, R.L.; Boguniewicz, M.; Jones, J.F.; Wong, C.; Streib, J.E.; Leung, D.Y. Cytokine milieu of atopic dermatitis skin subverts the innate immune response to vaccinia virus. Immunity 2006, 24, 341–348. [Google Scholar] [CrossRef] [PubMed]
- McGirt, L.Y.; Beck, L.A. Innate immune defects in atopic dermatitis. J. Allergy Clin. Immunol. 2006, 118, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Baker, B.S. The role of microorganisms in atopic dermatitis. Clin. Exp. Immunol. 2006, 144, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.; Byrd, A.L.; Deming, C.; Conlan, S.; Kong, H.H.; Segre, J.A. Biogeography and individuality shape function in the human skin metagenome. Nature 2014, 514, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Gaitanis, G.; Magiatis, P.; Hantschke, M.; Bassukas, I.D.; Velegraki, A. The Malassezia genus in skin and systemic diseases. Clin. Microbiol. Rev. 2012, 25, 106–141. [Google Scholar] [CrossRef] [PubMed]
- Saunders, C.W.; Scheynius, A.; Heitman, J. Malassezia fungi are specialized to live on skin and associated with dandruff, eczema, and other skin diseases. PLoS Pathog. 2012, 8, e1002701. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.A.; Hill, P.B. The biology of Malassezia organisms and their ability to induce immune responses and skin disease. Vet. Dermatol. 2005, 16, 4–26. [Google Scholar] [CrossRef] [PubMed]
- Gueho, E.; Midgley, G.; Guillot, J. The genus Malassezia with description of four new species. Antonie Van Leeuwenhoek 1996, 69, 337–355. [Google Scholar] [CrossRef] [PubMed]
- Findley, K.; Oh, J.; Yang, J.; Conlan, S.; Deming, C.; Meyer, J.A.; Schoenfeld, D.; Nomicos, E.; Park, M.; Kong, H.H.; et al. Topographic diversity of fungal and bacterial communities in human skin. Nature 2013, 498, 367–370. [Google Scholar] [CrossRef] [PubMed]
- Zomorodian, K.; Mirhendi, H.; Tarazooie, B.; Zeraati, H.; Hallaji, Z.; Balighi, K. Distribution of Malassezia species in patients with psoriasis and healthy individuals in Tehran, Iran. J. Cutan. Pathol. 2008, 35, 1027–1031. [Google Scholar] [CrossRef] [PubMed]
- Prohic, A. Distribution of Malassezia species in seborrhoeic dermatitis: Correlation with patients’ cellular immune status. Mycoses 2010, 53, 344–349. [Google Scholar] [PubMed]
- Gupta, A.K.; Kohli, Y. Prevalence of Malassezia species on various body sites in clinically healthy subjects representing different age groups. Med. Mycol. 2004, 42, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Ben Salah, S.; Makni, F.; Marrakchi, S.; Sellami, H.; Cheikhrouhou, F.; Bouassida, S.; Zahaf, A.; Ayadi, A. Identification of Malassezia species from Tunisian patients with pityriasis versicolor and normal subjects. Mycoses 2005, 48, 242–245. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.K.; Kohli, Y.; Summerbell, R.C.; Faergemann, J. Quantitative culture of Malassezia species from different body sites of individuals with or without dermatoses. Med. Mycol. 2001, 39, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Yim, S.M.; Kim, J.Y.; Ko, J.H.; Lee, Y.W.; Choe, Y.B.; Ahn, K.J. Molecular analysis of Malassezia microflora on the skin of the patients with atopic dermatitis. Ann. Dermatol. 2010, 22, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Sandstrom Falk, M.H.; Tengvall Linder, M.; Johansson, C.; Bartosik, J.; Back, O.; Sarnhult, T.; Wahlgren, C.F.; Scheynius, A.; Faergemann, J. The prevalence of Malassezia yeasts in patients with atopic dermatitis, seborrhoeic dermatitis and healthy controls. Acta Derm. Venereol. 2005, 85, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Tajima, M.; Sugita, T.; Nishikawa, A.; Tsuboi, R. Molecular analysis of Malassezia microflora in seborrheic dermatitis patients: Comparison with other diseases and healthy subjects. J. Investig. Dermatol. 2008, 128, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Kaga, M.; Sugita, T.; Nishikawa, A.; Wada, Y.; Hiruma, M.; Ikeda, S. Molecular analysis of the cutaneous Malassezia microbiota from the skin of patients with atopic dermatitis of different severities. Mycoses 2011, 54. [Google Scholar] [CrossRef] [PubMed]
- Oh, B.H.; Song, Y.C.; Lee, Y.W.; Choe, Y.B.; Ahn, K.J. Comparison of Nested PCR and RFLP for Identification and Classification of Malassezia Yeasts from Healthy Human Skin. Ann. Dermatol. 2009, 21, 352–357. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, T.; Makimura, K.; Abe, M.; Shiota, R.; Nakamura, Y.; Kano, R.; Hasegawa, A.; Sugita, T.; Shibuya, S.; Watanabe, S.; et al. Revised culture-based system for identification of Malassezia species. J. Clin. Microbiol. 2007, 45, 3737–3742. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, T.; Makimura, K.; Sugita, T.; Yamaguchi, H. Tween 40-based precipitate production observed on modified chromogenic agar and development of biological identification kit for Malassezia species. Med. Mycol. 2006, 44, 227–231. [Google Scholar] [CrossRef] [PubMed]
- Johansson, C.; Sandstrom, M.H.; Bartosik, J.; Sarnhult, T.; Christiansen, J.; Zargari, A.; Back, O.; Wahlgren, C.F.; Faergemann, J.; Scheynius, A.; et al. Atopy patch test reactions to Malassezia allergens differentiate subgroups of atopic dermatitis patients. Br. J. Dermatol. 2003, 148, 479–488. [Google Scholar] [CrossRef] [PubMed]
- Scalabrin, D.M.; Bavbek, S.; Perzanowski, M.S.; Wilson, B.B.; Platts-Mills, T.A.; Wheatley, L.M. Use of specific IgE in assessing the relevance of fungal and dust mite allergens to atopic dermatitis: A comparison with asthmatic and nonasthmatic control subjects. J. Allergy Clin. Immunol. 1999, 104, 1273–1279. [Google Scholar] [CrossRef]
- Scheynius, A.; Johansson, C.; Buentke, E.; Zargari, A.; Linder, M.T. Atopic eczema/dermatitis syndrome and Malassezia. Int. Arch. Allergy Immunol. 2002, 127, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Zargari, A.; Eshaghi, H.; Back, O.; Johansson, S.; Scheynius, A. Serum IgE reactivity to Malassezia furfur extract and recombinant M. furfur allergens in patients with atopic dermatitis. Acta Derm. Venereol. 2001, 81, 418–422. [Google Scholar] [CrossRef] [PubMed]
- Johansson, C.; Eshaghi, H.; Linder, M.T.; Jakobson, E.; Scheynius, A. Positive atopy patch test reaction to Malassezia furfur in atopic dermatitis correlates with a T helper 2-like peripheral blood mononuclear cells response. J. Investig. Dermatol. 2002, 118, 1044–1051. [Google Scholar] [CrossRef] [PubMed]
- Brodska, P.; Panzner, P.; Pizinger, K.; Schmid-Grendelmeier, P. IgE-mediated sensitization to Malassezia in atopic dermatitis: More common in male patients and in head and neck type. Dermatitis 2014, 25, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Lange, L.; Alter, N.; Keller, T.; Rietschel, E. Sensitization to Malassezia in infants and children with atopic dermatitis: Prevalence and clinical characteristics. Allergy 2008, 63, 486–487. [Google Scholar] [CrossRef] [PubMed]
- Kekki, O.M.; Scheynius, A.; Poikonen, S.; Koskinen, A.; Kautiainen, H.; Turjanmaa, K. Sensitization to Malassezia in children with atopic dermatitis combined with food allergy. Pediatr. Allergy Immunol. 2013, 24, 244–249. [Google Scholar] [CrossRef] [PubMed]
- Glatz, M.; Buchner, M.; von Bartenwerffer, W.; Schmid-Grendelmeier, P.; Worm, M.; Hedderich, J.; Folster-Holst, R. Malassezia spp.-specific Immunoglobulin E Level is a Marker for Severity of Atopic Dermatitis in Adults. Acta Derm. Venereol. 2015, 95, 191–196. [Google Scholar] [CrossRef] [PubMed]
- Sandstrom Falk, M.H.; Faergemann, J. Atopic dermatitis in adults: Does it disappear with age? Acta Derm. Venereol. 2006, 86, 135–139. [Google Scholar] [PubMed]
- Ramirez de Knott, H.M.; McCormick, T.S.; Kalka, K.; Skandamis, G.; Ghannoum, M.A.; Schluchter, M.; Cooper, K.D.; Nedorost, S.T. Cutaneous hypersensitivity to Malassezia sympodialis and dust mite in adult atopic dermatitis with a textile pattern. Contact Dermat. 2006, 54, 92–99. [Google Scholar]
- Faergemann, J. Atopic dermatitis and fungi. Clin. Microbiol. Rev. 2002, 15, 545–563. [Google Scholar] [CrossRef] [PubMed]
- Akdis, C.A.; Akdis, M.; Bieber, T.; Bindslev-Jensen, C.; Boguniewicz, M.; Eigenmann, P.; Hamid, Q.; Kapp, A.; Leung, D.Y.; Lipozencic, J.; et al. Diagnosis and treatment of atopic dermatitis in children and adults: European Academy of Allergology and Clinical Immunology/American Academy of Allergy, Asthma and Immunology/PRACTALL Consensus Report. Allergy 2006, 61, 969–987. [Google Scholar] [CrossRef] [PubMed]
- Zhang, E.; Tanaka, T.; Tajima, M.; Tsuboi, R.; Kato, H.; Nishikawa, A.; Sugita, T. Anti-Malassezia-Specific IgE Antibodies Production in Japanese Patients with Head and Neck Atopic Dermatitis: Relationship between the Level of Specific IgE Antibody and the Colonization Frequency of Cutaneous Malassezia Species and Clinical Severity. J. Allergy 2011, 2011. [Google Scholar] [CrossRef] [PubMed]
- Cotterill, J.A.; Cunliffe, W.J.; Williamson, B.; Bulusu, L. Age and sex variation in skin surface lipid composition and sebum excretion rate. Br. J. Dermatol. 1972, 87, 333–340. [Google Scholar] [CrossRef] [PubMed]
- Selander, C.; Zargari, A.; Mollby, R.; Rasool, O.; Scheynius, A. Higher pH level, corresponding to that on the skin of patients with atopic eczema, stimulates the release of Malassezia sympodialis allergens. Allergy 2006, 61, 1002–1008. [Google Scholar] [CrossRef] [PubMed]
- Balaji, H.; Heratizadeh, A.; Wichmann, K.; Niebuhr, M.; Crameri, R.; Scheynius, A.; Werfel, T. Malassezia sympodialis thioredoxin-specific T cells are highly cross-reactive to human thioredoxin in atopic dermatitis. J. Allergy Clin. Immunol. 2011, 128. [Google Scholar] [CrossRef] [PubMed]
- Schmid-Grendelmeier, P.; Fluckiger, S.; Disch, R.; Trautmann, A.; Wuthrich, B.; Blaser, K.; Scheynius, A.; Crameri, R. IgE-mediated and T cell-mediated autoimmunity against manganese superoxide dismutase in atopic dermatitis. J. Allergy Clin. Immunol. 2005, 115, 1068–1075. [Google Scholar] [CrossRef] [PubMed]
- Hiragun, T.; Ishii, K.; Hiragun, M.; Suzuki, H.; Kan, T.; Mihara, S.; Yanase, Y.; Bartels, J.; Schroder, J.M.; Hide, M.; et al. Fungal protein MGL_1304 in sweat is an allergen for atopic dermatitis patients. J. Allergy Clin. Immunol. 2013, 132, 608–615. [Google Scholar] [CrossRef] [PubMed]
- Hiragun, M.; Hiragun, T.; Ishii, K.; Suzuki, H.; Tanaka, A.; Yanase, Y.; Mihara, S.; Haruta, Y.; Kohno, N.; Hide, M.; et al. Elevated serum IgE against MGL_1304 in patients with atopic dermatitis and cholinergic urticaria. Allergol. Int. 2014, 63, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Buentke, E.; Scheynius, A. Dendritic cells and fungi. Apmis 2003, 111, 789–796. [Google Scholar] [CrossRef] [PubMed]
- Gehrmann, U.; Qazi, K.R.; Johansson, C.; Hultenby, K.; Karlsson, M.; Lundeberg, L.; Gabrielsson, S.; Scheynius, A. Nanovesicles from Malassezia sympodialis and host exosomes induce cytokine responses—Novel mechanisms for host-microbe interactions in atopic eczema. PLoS ONE 2011, 6, e21480. [Google Scholar] [CrossRef] [PubMed]
- Vilhelmsson, M.; Johansson, C.; Jacobsson-Ekman, G.; Crameri, R.; Zargari, A.; Scheynius, A. The Malassezia sympodialis allergen Mala s 11 induces human dendritic cell maturation, in contrast to its human homologue manganese superoxide dismutase. Int. Arch. Allergy Immunol. 2007, 143, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Selander, C.; Engblom, C.; Nilsson, G.; Scheynius, A.; Andersson, C.L. TLR2/MyD88-Dependent and -independent activation of mast cell IgE responses by the skin commensal yeast Malassezia sympodialis. J. Immunol. 2009, 182, 4208–4216. [Google Scholar] [CrossRef] [PubMed]
- Brasch, J.; Morig, A.; Neumann, B.; Proksch, E. Expression of antimicrobial peptides and toll-like receptors is increased in tinea and pityriasis versicolor. Mycoses 2014, 57, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Baroni, A.; Orlando, M.; Donnarumma, G.; Farro, P.; Iovene, M.R.; Tufano, M.A.; Buommino, E. Toll-like receptor 2 (TLR2) mediates intracellular signalling in human keratinocytes in response to Malassezia furfur. Arch. Dermatol. Res. 2006, 297, 280–288. [Google Scholar] [CrossRef] [PubMed]
- Novak, N.; Leung, D.Y. Advances in atopic dermatitis. Curr. Opin. Immunol. 2011, 23, 778–783. [Google Scholar] [CrossRef] [PubMed]
- Kistowska, M.; Fenini, G.; Jankovic, D.; Feldmeyer, L.; Kerl, K.; Bosshard, P.; Contassot, E.; French, L.E. Malassezia yeasts activate the NLRP3 inflammasome in antigen-presenting cells via Syk-kinase signaling. Exp. Dermatol. 2014, 23, 884–889. [Google Scholar] [CrossRef] [PubMed]
- Ribbing, C.; Engblom, C.; Lappalainen, J.; Lindstedt, K.; Kovanen, P.T.; Karlsson, M.A.; Lundeberg, L.; Johansson, C.; Nilsson, G.; Lunderius-Andersson, C.; et al. Mast cells generated from patients with atopic eczema have enhanced levels of granule mediators and an impaired Dectin-1 expression. Allergy 2011, 66, 110–119. [Google Scholar] [CrossRef] [PubMed]
- Darabi, K.; Hostetler, S.G.; Bechtel, M.A.; Zirwas, M. The role of Malassezia in atopic dermatitis affecting the head and neck of adults. J. Am. Acad. Dermatol. 2009, 60, 125–136. [Google Scholar] [CrossRef] [PubMed]
- Sugita, T.; Tajima, M.; Ito, T.; Saito, M.; Tsuboi, R.; Nishikawa, A. Antifungal activities of tacrolimus and azole agents against the eleven currently accepted Malassezia species. J. Clin. Microbiol. 2005, 43, 2824–2829. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, S.; Koike, A.; Kano, R.; Nagata, M.; Chen, C.; Hwang, C.Y.; Hasegawa, A.; Kamata, H. In vitro susceptibility of Malassezia pachydermatis isolates from canine skin with atopic dermatitis to ketoconazole and itraconazole in East Asia. J. Vet. Med. Sci. 2014, 76, 579–581. [Google Scholar] [CrossRef] [PubMed]
- Broberg, A.; Faergemann, J. Topical antimycotic treatment of atopic dermatitis in the head/neck area. A double-blind randomised study. Acta Derm. Venereol. 1995, 75, 46–49. [Google Scholar] [PubMed]
- Lintu, P.; Savolainen, J.; Kortekangas-Savolainen, O.; Kalimo, K. Systemic ketoconazole is an effective treatment of atopic dermatitis with IgE-mediated hypersensitivity to yeasts. Allergy 2001, 56, 512–517. [Google Scholar] [CrossRef] [PubMed]
- Svejgaard, E.; Larsen, P.O.; Deleuran, M.; Ternowitz, T.; Roed-Petersen, J.; Nilsson, J. Treatment of head and neck dermatitis comparing itraconazole 200 mg and 400 mg daily for 1 week with placebo. J. Eur. Acad. Dermatol. Venereol. 2004, 18, 445–449. [Google Scholar] [CrossRef] [PubMed]
- Back, O.; Bartosik, J. Systemic ketoconazole for yeast allergic patients with atopic dermatitis. J. Eur. Acad. Dermatol. Venereol. 2001, 15, 34–38. [Google Scholar] [CrossRef] [PubMed]
- Kaffenberger, B.H.; Mathis, J.; Zirwas, M.J. A retrospective descriptive study of oral azole antifungal agents in patients with patch test-negative head and neck predominant atopic dermatitis. J. Am. Acad. Dermatol. 2014, 71, 480–483. [Google Scholar] [CrossRef] [PubMed]
- Kanda, N.; Enomoto, U.; Watanabe, S. Anti-mycotics suppress interleukin-4 and interleukin-5 production in anti-CD3 plus anti-CD28-stimulated T cells from patients with atopic dermatitis. J. Investig. Dermatol. 2001, 117, 1635–1646. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, H.; Nakajima, S.; Sakata, I.; Iizuka, H. Antifungal effect of TONS504-photodynamic therapy on Malassezia furfur. J. Dermatol. 2014, 41, 895–897. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Glatz, M.; Bosshard, P.P.; Hoetzenecker, W.; Schmid-Grendelmeier, P. The Role of Malassezia spp. in Atopic Dermatitis. J. Clin. Med. 2015, 4, 1217-1228. https://doi.org/10.3390/jcm4061217
Glatz M, Bosshard PP, Hoetzenecker W, Schmid-Grendelmeier P. The Role of Malassezia spp. in Atopic Dermatitis. Journal of Clinical Medicine. 2015; 4(6):1217-1228. https://doi.org/10.3390/jcm4061217
Chicago/Turabian StyleGlatz, Martin, Philipp P. Bosshard, Wolfram Hoetzenecker, and Peter Schmid-Grendelmeier. 2015. "The Role of Malassezia spp. in Atopic Dermatitis" Journal of Clinical Medicine 4, no. 6: 1217-1228. https://doi.org/10.3390/jcm4061217
APA StyleGlatz, M., Bosshard, P. P., Hoetzenecker, W., & Schmid-Grendelmeier, P. (2015). The Role of Malassezia spp. in Atopic Dermatitis. Journal of Clinical Medicine, 4(6), 1217-1228. https://doi.org/10.3390/jcm4061217




