Hyponatremia in Patients with Spontaneous Intracerebral Hemorrhage
Abstract
:1. Introduction
2. Experimental Section
2.1. Patients
2.2. Patient Demographics and Clinical Variables
2.3. Study Definitions
2.4. Statistical Analysis
3. Results and Discussion
3.1. Incidence
| Characteristic | No HN (n = 75) | HN (n = 24) | p-Value |
|---|---|---|---|
| Age, years, mean ± SD | 58.6 ± 10.4 | 59.4 ± 12.1 | 0.73 |
| Male gender, n (%) | 43 (57) | 18 (75) | 0.12 |
| African American, n (%) | 59 (77) | 17 (75) | 0.71 |
| Past Medical History, n (%) | |||
| Neurological Injury * | 14 (19) | 1 (4) | 0.11 |
| Seizure Disorder ** | 1 (1) | 1 (4) | 0.43 |
| Hypertension | 59 (79) | 20 (83) | 0.77 |
| Diabetes | 17 (23) | 4 (17) | 0.53 |
| Heart Failure | 8 (11) | 1 (4) | 0.45 |
| CKD or ESRD | 16 (21) | 2 (8) | 0.23 |
| Social History, n (%) ** | |||
| Tobacco | 24 (32) | 10 (42) | 0.39 |
| Alcohol | 19 (25) | 10 (42) | 0.13 |
| Illicit Drugs | 22 (29) | 5 (21) | 0.81 |
| Medications Prior to Admission | |||
| Diuretic | 13 (17) | 3 (13) | 1.0 |
| NSAIDs and COX-2 Inhibitors | 3 (4) | 1 (4) | 1.0 |
| SSRI | 1 (1) | 0 (0) | 1.0 |
| Laboratory Parameters on Admission | |||
| Sodium (mmol/L) | 143 ± 3 | 140 ± 3 | 0.002 |
| Potassium (mEq/L) | 4.1 ± 0.8 | 4.2 ± 0.7 | 0.63 |
| Serum Creatinine (mg/dL) | 1.7 ± 1.83 | 1.11 ± 0.52 | 0.13 |
| Glucose (mg/dL) | 145 ± 66 | 155 ± 70 | 0.51 |
| GCS, median (IQR) | 14 (10, 15) | 14 (7, 15) | 0.85 |
| Location of ICH, n (%) | |||
| Lobar | 36 (48) | 11 (46) | 0.85 |
| Deep | 28 (37) | 13 (54) | 0.15 |
| Brainstem/Cerebellar | 11 (15) | 0 (0) | 0.06 |
| Surgical Intervention, n (%) | |||
| Craniectomy | 1 (1) | 3 (13) | 0.01 |
| Craniotomy | 2 (3) | 1 (4) | 0.67 |
| Stereotaxy Aspiration | 1 (1) | 0 (0) | 0.58 |
| ICH Volume (mL), median (IQR) | 22 (8, 44) | 32 (9, 89) | 0.30 |
3.2. Description of Hyponatremia
| Variable | No HN (n = 75) | HN (n = 24) |
|---|---|---|
| Sodium Nadir (mmol/L), mean ± SD | 139 ± 3 | 130 ± 3 |
| Average Sodium (mmol/L), mean ± SD | 143 ± 4 | 139 ± 4 |
| Time from Admission to Sodium <135 mmol/L (days), mean ± SD | - | 3.9 ± 5.7 |
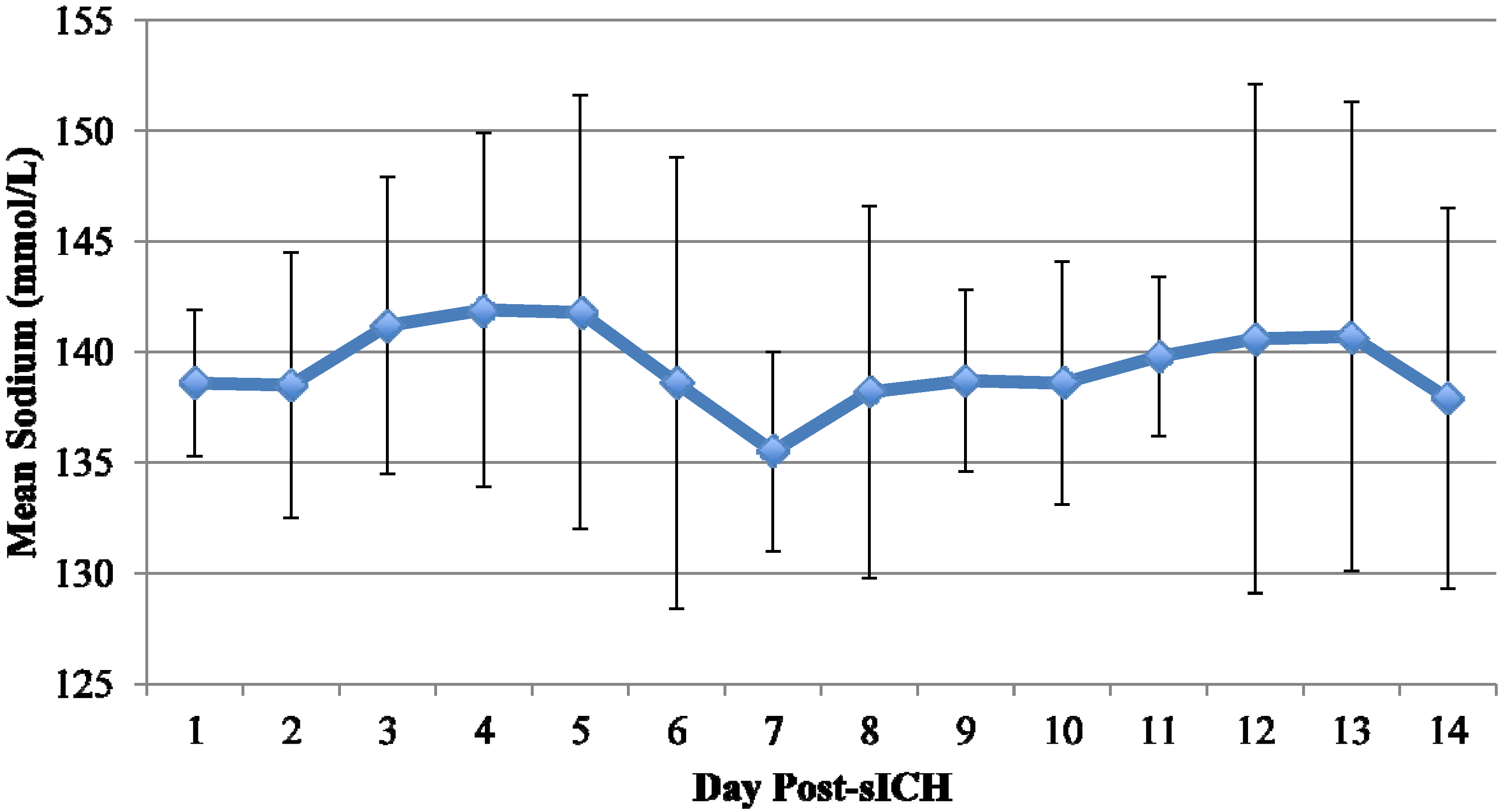
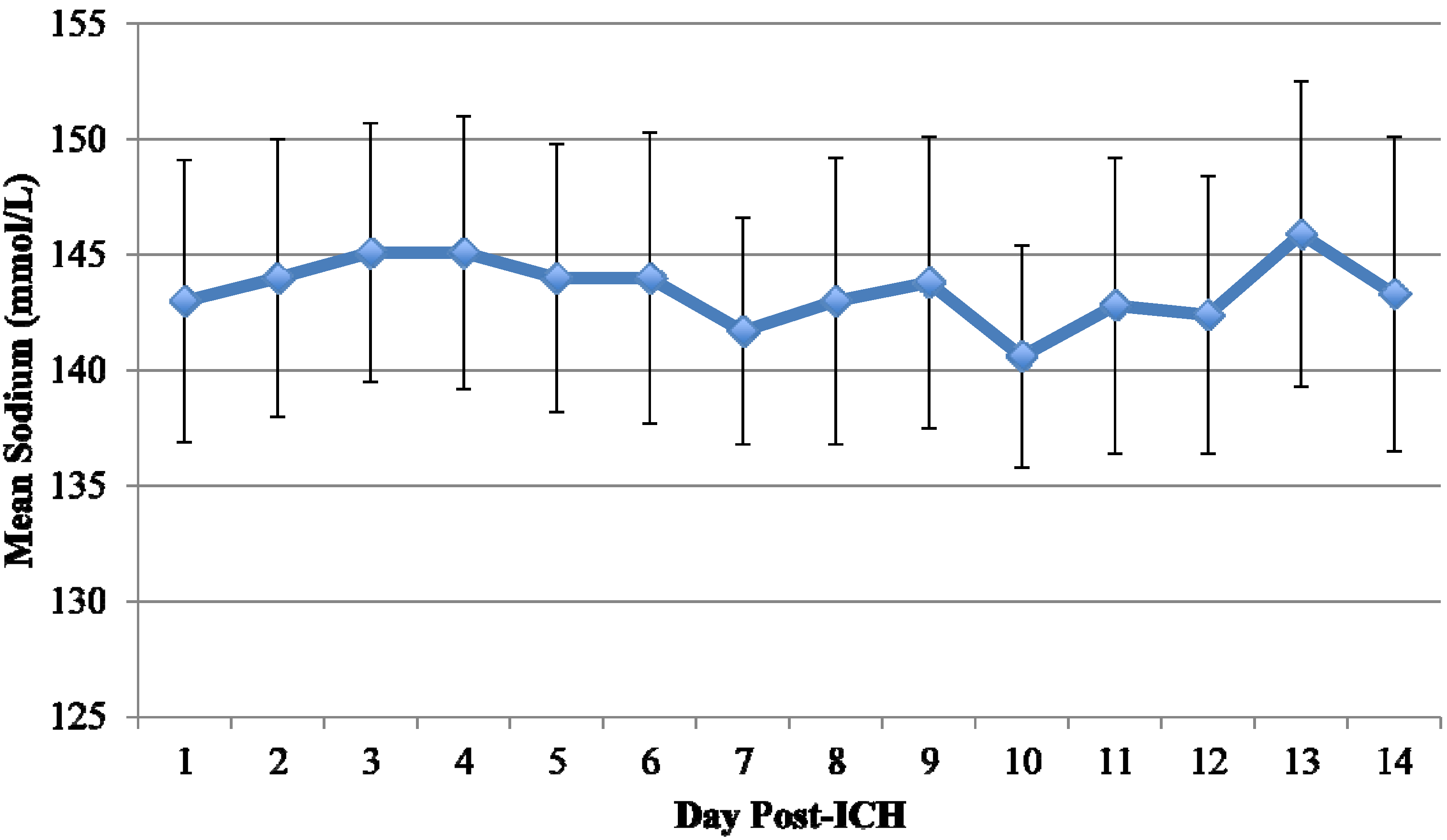
3.3. Etiology of Hyponatremia
3.4. Clinical Outcomes, Complications, and Mortality
| Variable | No HN (n = 75) | HN (n = 24) | p-Value |
|---|---|---|---|
| Clinical Outcomes | |||
| Hospital LOS (days), median (IQR) | 6 (3, 9) | 14 (8, 25) | <0.001 |
| ICU LOS (days), median (IQR) | 4 (2, 9) | 10 (7, 20) | 0.91 |
| In-Hospital Mortality, n (%) | 24 (32) | 6 (25) | 0.52 |
| Complications, n (%) | |||
| Seizures | 4 (5) | 3 (13) | 0.36 |
| Cerebral Edema | 43 (57) | 17 (71) | 0.24 |
| Fever | 17 (23) | 12 (50) | 0.01 |
| Infection | 21 (28) | 14 (58) | 0.007 |
| Thrombocytopenia | 1 (1) | 4 (17) | 0.01 |
| Inpatient Medications, n (%) | |||
| Furosemide | 19 (25) | 14 (58) | 0.01 |
| ACEi | 30 (40) | 15 (63) | 0.21 |
| Carbamazepine | 0 (0) | 2 (8) | 0.06 |
| Antidepressants | 2 (3) | 0 (0) | 1.0 |
| Opioids | 2 (3) | 1 (4) | 0.57 |
| Hypertonic Saline | 3 (4) | 2 (8) | 0.62 |
| Mannitol | 8 (11) | 2 (8) | 0.72 |
3.5. Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tierney, W.M.; Martin, D.K.; Greenlee, M.C.; Zerbe, R.L.; McDonald, C.J. The prognosis of hyponatremia at hospital admission. J. Gen. Intern. Med. 1986, 1, 380–385. [Google Scholar] [PubMed]
- Anderson, R.J.; Chung, H.M.; Kluge, R.; Schrier, R.W. Hyponatremia: A prospective analysis of its epidemiology and the pathogenetic role of vasopressin. Ann. Intern. Med. 1985, 102, 164–168. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.; Friedman, W.A. Hyponatremia in neurosurgical patients: Clinical guidelines development. Neurosurgery 2009, 65, 925–935. [Google Scholar] [CrossRef] [PubMed]
- Sherlock, M.; O’Sullivan, E.; Agha, A.; Behan, L.A.; Rawluk, D.; Brennan, P.; Tormey, W.; Thompson, C.J. The incidence and pathophysiology of hyponatraemia after subarachnoid haemorrhage. Clin. Endocrinol. (Oxf.) 2006, 64, 250–254. [Google Scholar] [CrossRef]
- Bhardwaj, A. Neurological impact of vasopressin dysregulation and hyponatremia. Ann. Neurol. 2006, 59, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Fraser, J.F.; Stieg, P.E. Hyponatremia in the neurosurgical patient: Epidemiology, pathophysiology, diagnosis, and management. Neurosurgery 2006, 59, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Kao, L.; Al-Lawati, Z.; Vavao, J.; Steinberg, G.K.; Katznelson, L. Prevalence and clinical demographics of cerebral salt wasting in patients with aneurysmal subarachnoid hemorrhage. Pituitary 2009, 12, 347–351. [Google Scholar] [CrossRef] [PubMed]
- Unterberg, A.; Kiening, K.; Schmiedek, P.; Lanksch, W. Long-term observations of intracranial pressure after severe head injury. The phenomenon of secondary rise of intracranial pressure. Neurosurgery 1993, 32, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Moro, N.; Katayama, Y.; Igarashi, T.; Mori, T.; Kawamata, T.; Kojima, J. Hyponatremia in patients with traumatic brain injury: Incidence, mechanism, and response to sodium supplementation or retention therapy with hydrocortisone. Surg. Neurol. 2007, 68, 387–393. [Google Scholar] [CrossRef] [PubMed]
- Wijdicks, E.F.; Vermeulen, M.; Hijdra, A.; van Gijn, J. Hyponatremia and cerebral infarction in patients with ruptured intracranial aneurysms: Is fluid restriction harmful? Ann. Neurol. 1985, 17, 137–140. [Google Scholar] [CrossRef] [PubMed]
- Waikar, S.S.; Mount, D.B.; Curhan, G.C. Mortality after hospitalization with mild, moderate, and severe hyponatremia. Am. J. Med. 2009, 122, 857–865. [Google Scholar] [CrossRef] [PubMed]
- Janicic, N.; Verbalis, J.G. Evaluation and management of hypo-osmolality in hospitalized patients. Endocrinol. Metab. Clin. North Am. 2003, 32, 459–481. [Google Scholar] [CrossRef] [PubMed]
- Rabinstein, A.A.; Wijdicks, E.F. Hyponatremia in critically ill neurological patients. Neurologist 2003, 9, 290–300. [Google Scholar] [CrossRef] [PubMed]
- Diringer, M.N.; Zazulia, A.R. Hyponatremia in neurologic patients: Consequences and approaches to treatment. Neurologist 2006, 12, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Kuramatsu, J.B.; Bobinger, T.; Volbers, B.; Staykov, D.; Lücking, H.; Kloska, S.P.; Köhrmann, M.; Huttner, H.B. Hyponatremia is an independent predictor of in-hospital mortality in spontaneous intracerebral hemorrhage. Stroke 2014, 45, 1285–1291. [Google Scholar] [CrossRef] [PubMed]
- Amin, A.; Deitelzweig, S.; Christian, R.; Friend, K.; Lin, J.; Belk, K.; Baumer, B.; Lowe, T.J. Evaluation of incremental healthcare resource burden and readmission rates associated with hospitalized hyponatremic patients in the US. J. Hosp. Med. 2012, 7, 634–639. [Google Scholar] [CrossRef] [PubMed]
- Wald, R.; Jaber, B.L.; Price, L.L.; Upadhyay, A.; Madias, N.E. Impact of hospital-associated hyponatremia on selected outcomes. Arch. Intern. Med. 2010, 170, 294–302. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gray, J.R.; Morbitzer, K.A.; Liu-DeRyke, X.; Parker, D., Jr.; Zimmerman, L.H.; Rhoney, D.H. Hyponatremia in Patients with Spontaneous Intracerebral Hemorrhage. J. Clin. Med. 2014, 3, 1322-1332. https://doi.org/10.3390/jcm3041322
Gray JR, Morbitzer KA, Liu-DeRyke X, Parker D Jr., Zimmerman LH, Rhoney DH. Hyponatremia in Patients with Spontaneous Intracerebral Hemorrhage. Journal of Clinical Medicine. 2014; 3(4):1322-1332. https://doi.org/10.3390/jcm3041322
Chicago/Turabian StyleGray, Jaime Robenolt, Kathryn A. Morbitzer, Xi Liu-DeRyke, Dennis Parker, Jr., Lisa Hall Zimmerman, and Denise H. Rhoney. 2014. "Hyponatremia in Patients with Spontaneous Intracerebral Hemorrhage" Journal of Clinical Medicine 3, no. 4: 1322-1332. https://doi.org/10.3390/jcm3041322
APA StyleGray, J. R., Morbitzer, K. A., Liu-DeRyke, X., Parker, D., Jr., Zimmerman, L. H., & Rhoney, D. H. (2014). Hyponatremia in Patients with Spontaneous Intracerebral Hemorrhage. Journal of Clinical Medicine, 3(4), 1322-1332. https://doi.org/10.3390/jcm3041322




