Optimal Pharmacologic Treatment Strategies in Obesity and Type 2 Diabetes
Abstract
:1. Introduction
2. Development of Type 2 Diabetes
3. Treating Type 2 Diabetes
| Class & Medication | Trade Name | Dosage Availability | Weight Effect | A1c Reduction | Side Effects | Cost * | Notes & Considerations |
|---|---|---|---|---|---|---|---|
| Biguanide Metformin | Glucophage™ Glucophage ER™ | Oral tablet 500/850/1000 mg 500/750/1000 mg | Weight loss [24,25] 2–3 kg | 1.5%–2% [24,25] | GI 1, lactic acidosis | $0.70–1.44 $0.75–1.03 per tablet | First Line agent; Avoid in renal dysfunction |
| GLP 1 analogs Exenatide Liraglutide | Byetta™/Bydureon™ Victoza™ | Subcutaneous 5–10 mcg 0.6/1.2/1.8 mg | Weight loss [26] 2.87–3.84 kg | 0.9%–1% [27,28,29,30,31,32,33] | Class Effects: GI, acute pancreatitis, medullary thyroid cancer | Byetta™/Bydureon™—$197/$122 Victoza™—$71.42 (unit) $428 (package) | Injectable; Avoid in renal dysfunction & history of pancreatitis |
| DPP 4 inhibitors Sitagliptin Saxagliptin Linagliptin Alogliptin | Januvia™ Onglyza™ Tradjenta™ Nesina™ | Oral tablet 25/50/100 mg 2.5/5 mg 5 mg 6.25/12.5/25 mg | Weight neutral [34] | 0.7%–1% [34] | Class Effects: URI 2, anaphylaxis, acute pancreatitis | $11.35 $11.16 $11.35 $11.35 per tablet | Oral formulation; Renal dose adjustments needed; Avoid when history of pancreatitis |
| Amylin analog Pramlintide | Symlin™ | Subcutaneous 30/60/120 mcg | Weight loss [35] 2.57 kg | 0.3%–0.4% [35] | GI | $357.35 (120 mcg) $292.56 (60 mcg) (package) | Indicated for insulin dependent patients; Only used in the setting of insulin co-administration |
| SGLT2 inhibitors Canagliflozin Dapagliflozin | Invokana™ Farxiga™ | Oral tablet 100/300 mg 5/10 mg | Weight loss [36,37,38] 2–3 kg | 0.6%–0.9% [39] | Class Effects: Genital mycotic infection, UTI 3, increase in LDL-C 4, serum creatinine and K+ 5 | Invokana™ $11.57 per tablet Farxiga™—NA 6 | Extensive side effect profile |
| Miscellaneous Agents Acarbose/Miglitol Colesevelam Bromocriptine | Precose™/Glyset™ Welchol™ Cycloset™ | Oral tablet 25/50/100 mg 625 mg 1.6–4.8 mg | Weight loss—1.1 kg [40] Weight neutral [41,42,43,44] Weight neutral [45,46,47,48] | 1% [40] 0.5% [41,42,43,44] 0.6%–0.9% [45,46,47,48] | GI GI GI, asthenia, HA 7 | $0.89/0.97/1.16 $2.30 (625 mg) $2.75 per tablet | Colesevelam CI 8 when TG 9 > 500 mg/dL; Bromocriptine CI in uncontrolled HTN 10, syncopal migraine, breastfeeding women |
4. Medications for Type 2 Diabetes with Weight Neutral or Weight Loss Effects
4.1. Biguanide Medications
Metformin (Glucophage, Glucophage XR)
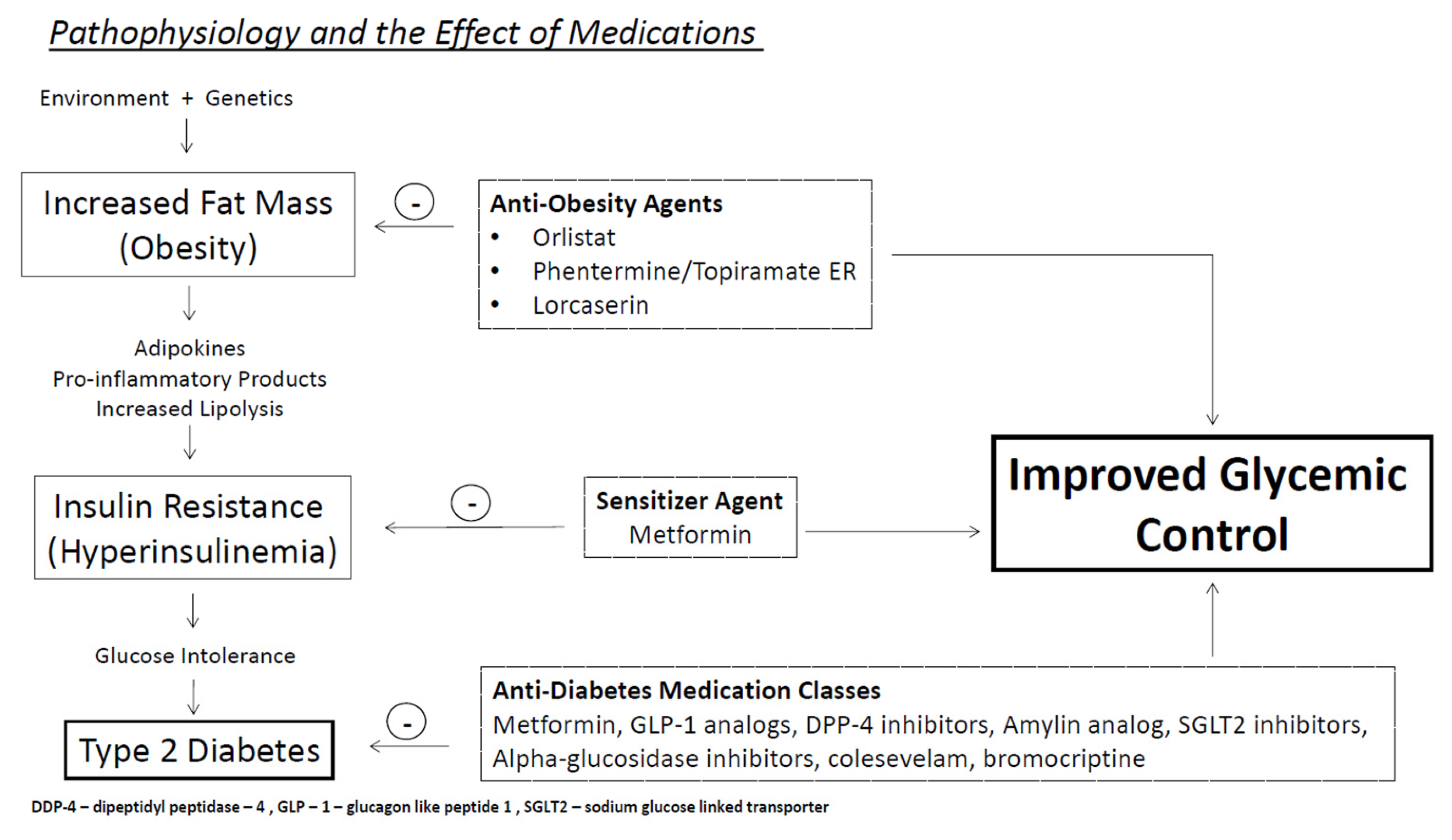
4.2. Role of Incretin Hormones
4.2.1. GLP-1 Analogs
Exenatide (Byetta, Bydureon), Liraglutide (Victoza)
4.2.2. Dipeptidyl Peptidase 4 (DPP-4) Inhibitors
Sitagliptin (Januvia), Saxagliptin (Onglyza), Linagliptin (Tradjenta), Alogliptin (Nesina)
4.3. Role of Amylin
Pramlintide (Symlin)
4.4. Role of Sodium-Glucose Linked Transporter 2 Inhibition
Canagliflozin (Invokana), Dapagliflozin (Farxiga)
4.5. Alpha-Glucosidase Inhibitors
Acarbose (Precose), Miglitol (Glyset)
4.6. Miscellaneous Agents
4.6.1. Colesevelam (Welchol)
4.6.2. Bromocriptine (Cycloset)
5. Anti-Obesity Agents
5.1. Orlistat (Alli, Xenical)
5.2. Phentermine/Topiramate Extended Release (Qsymia)
5.3. Lorcaserin (Belviq)
| Class & Medication | Trade Name | Dosage Availability | Weight Effect | A1c Reduction | Side Effects | Cost * | Notes & Considerations |
|---|---|---|---|---|---|---|---|
| Lipase Inhibitor Orlistat | Alli™ Xenical™ | Oral tablet 60 mg 120 mg | Weight loss [65,66,67] 2.9 kg | 0.29% [67] | GI 1 | $0.59 (Alli™) $6.26 (Xenical™) | Avoid in liver disease |
| Appetite Suppressant Phentermine/Topiramate ER | Qsymia™ | Oral tablet 3.75/23 mg 7.5/46 mg 11.25/69 mg 15/92 mg | Weight loss [68,69,70,71] 5.8kg | 0.2%–0.4% [70] | Metabolic acidosis tachycardia, congenital malformations | $5.44 $6.15 $7.38 $7.98 | Requires REMS 2 for congenital malformations; Sympathomimetic effects may exacerbate underlying cardiac disease |
| Serotonin-Receptor Agonist Lorcaserin | Belviq™ | Oral tablet 10 mg | Weight loss [73,74,75] 5 kg | 0.9%–1% [75] | GI, URI 3, HA 4, dizziness | $3.99 | Drug interactions with serotonergic agents |
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wing, R.R.; Lang, W.; Wadden, T.A.; Safford, M.; Knowler, W.C.; Bertoni, A.G.; Hill, J.O.; Brancati, F.L.; Peters, A.; Wagenknecht, L.; et al. Benefits of modest weight loss in improving cardiovascular risk factors in overweight and obese individuals with type 2 diabetes. Diabetes Care 2011, 34, 1481–1486. [Google Scholar] [CrossRef]
- Lim, S.S.; Vos, T.; Flaxman, A.D.; Danaei, G.; Shibuya, K.; Adair-Rohani, H.; Amann, M.; Anderson, H.R.; Andrews, K.G.; Aryee, M.; et al. A comparative risk assessment of burden of disease and injury attributable to 67 risk factors and risk clusters in 21 regions, 1990–2010: A systemic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2224–2260. [Google Scholar] [CrossRef]
- Kelly, T.; Yang, W.; Chen, C.S.; Reynolds, K.; He, J. Global burden of obesity in 2005 and projections to 2030. Int. J. Obes. 2008, 32, 1431–1437. [Google Scholar] [CrossRef]
- Chan, J.C.; Malik, V.; Jia, W.; Kadowaki, T.; Yajnik, C.S.; Yoon, K.H.; Hu, F.B. Diabetes in Asia: Epidemiology, risk factors, and pathophysiology. JAMA 2009, 301, 2129–2140. [Google Scholar] [CrossRef]
- Hu, F.B. Globalization of diabetes: The role of diet, lifestyle, and genes. Diabetes Care 2011, 34, 1249–1257. [Google Scholar] [CrossRef]
- Boden, G. Obesity, insulin resistance and free fatty acids. Curr. Opin. Endocrinol. Diabetes Obes. 2011, 18, 139–143. [Google Scholar] [CrossRef]
- Kershaw, E.E.; Flier, J.S. Adipose tissue as an endocrine organ. J. Clin. Endocrinol. Metab. 2004, 89, 2548–2556. [Google Scholar] [CrossRef]
- Hotamisligil, G.S.; Shargill, N.S.; Spiegelman, B.M. Adipose expression of tumor necrosis factor-alpha: Direct role in obesity-linked insulin resistance. Science 1993, 259, 87–91. [Google Scholar]
- Vgontzas, A.N.; Papanicolaou, D.A.; Bixler, E.O.; Kales, A.; Tyson, K.; Chrousos, G.P. Elevation of plasma cytokines in disorders of excessive daytime sleepiness: Role of sleep disturbance and obesity. J. Clin. Endocrinol. Metab. 1997, 82, 1313–1316. [Google Scholar] [CrossRef]
- Barzilai, N.; Wang, J.; Massilon, D.; Vuguin, P.; Hawkins, M.; Rosetti, L. Leptin selectively decreases visceral adiposity and enhances insulin action. J. Clin. Investig. 1997, 100, 3105–3110. [Google Scholar] [CrossRef]
- Reaven, G.M. Role of insulin resistance in human disease. Diabetes 1988, 37, 1595–1607. [Google Scholar] [CrossRef]
- Haffner, S.M.; Stern, M.P.; Dunn, J.; Mobley, M.; Blackwell, J.; Bergman, R.N. Diminished insulin sensitivity and increased insulin response in nonobese, nondiabetic Mexican Americans. Metabolism 1990, 39, 842–847. [Google Scholar] [CrossRef]
- Reaven, G.M.; Bernstein, R.; Davis, B.; Olefsky, J.M. Nonketotic diabetes mellitus: Insulin deficiency or insulin resistence? Am. J. Med. 1976, 60, 80–88. [Google Scholar] [CrossRef]
- Kahn, C.R. Insulin resistance, insulin sensitivity, and insulin unresponsiveness: A necessary distinction. Metabolism 1978, 27, 1893–1902. [Google Scholar] [CrossRef]
- DeFronzo, R.A.; Bonadonna, R.C.; Ferrannini, E. Pathogenesis of NIDDM, a balanced overview. Diabetes Care 1992, 15, 318–368. [Google Scholar]
- Warram, J.H.; Martin, B.C.; Krolewski, A.S.; Soeldner, J.S.; Kahn, C.R. Slow glucose removal rate and hyperinsulinemia precede the development of type II diabetes in offspring of diabetic patients. Ann. Intern. Med. 1990, 113, 909–915. [Google Scholar] [CrossRef]
- Garber, A.J.; Abrahamson, M.J.; Barzilary, J.I.; Blonde, L.; Bloomgarde, Z.T.; Bush, M.A.; Dagogo-Jack, S.; Davidson, M.B.; Einhorn, D.; Garvey, W.T.; et al. AACE comprehensive diabetes management algorithm 2013. Endocr. Pract. 2013, 19, 327–336. [Google Scholar]
- Inzucchi, S.E.; Bergenstal, R.M.; Buse, J.B.; Diamant, M.; Ferrannini, E.; Nauck, M.; Peters, A.L.; Tsapas, A.; Wender, R.; Matthews, D.R.; et al. Management of hyperglycemia in type 2 diabetes: A patient centered approach. Diabetes Care 2012, 35, 1364–1379. [Google Scholar] [CrossRef]
- Wolffenbuttel, B.H.R.; Landgraf, R. A 1-year multicenter randomized double-blind comparison of repaglinide and glyburide for the treatment of type 2 diabetes. Diabetes Care 1999, 22, 463–467. [Google Scholar] [CrossRef]
- Marbury, T.; Huang, W.; Strange, P.; Lebovitz, H. Repaglinide versus glyburide: A one year comparison trial. Diabetes Res. Clin. Parct. 1992, 15, 755–772. [Google Scholar]
- Aronoff, S.; Rosenblatt, S.; Egan, J.W.; Mathisen, A.L.; Schneider, R.L. Pioglitazone hydrochloride monotherapy improves glycemic control in the treatment of patients with type 2 diabetes: A 6-month randomized placebo-controlled dose-response study. Diabetes Care 2000, 23, 1605–1611. [Google Scholar] [CrossRef]
- Lebovitz, H.E.; Dole, J.F.; Patwardhan, R.; Rappaport, E.B.; Freed, M.I. Rosiglitazone monotherapy is effective in patients with type 2 diabetes. J. Clin. Endocrinol. Metab. 2001, 86, 280–288. [Google Scholar] [CrossRef]
- Dailey, G.; Admane, K.; Mercier, F.; Owens, D. Relationship of insulin dose, a1c lowering, and weight in type 2 diabetes: Comparing insulin Glargine and insulin detemir. Diabetes Technol. Ther. 2010, 12, 1019–1027. [Google Scholar]
- UK Prospective Diabetes Study (UKPDS) Group. Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). Lancet 1998, 352, 854–865. [Google Scholar] [CrossRef]
- Diabetes Prevention Program (DPP) Research Group. Long-term safety, tolerability, and weight loss associated with metformin in the diabetes prevention program outcome study. Diabetes Care 2012, 35, 731–737. [Google Scholar] [CrossRef]
- Buse, J.B.; Rosenstock, J.; Sesti, G.; Schmidt, W.E.; Montanya, E.; Brett, J.H.; Zychma, M.; Blonde, L. Liraglutide once a day versus exenatide twice a day for type 2 diabetes: A 26-week randomized, parallel-group, multinational, open-label trial (LEAD-6). Lancet 2009, 374, 39–47. [Google Scholar] [CrossRef]
- DeFronzo, R.A.; Ratner, R.E.; Han, J.; Kim, D.D.; Fineman, M.S.; Baron, A.D. Effects of exenatide (exendin-4) on glycemic control and weight over 30 weeks in metformin-treated patients with type 2 diabetes. Diabetes Care 2005, 28, 1092–1100. [Google Scholar] [CrossRef]
- Buse, J.B.; Henry, R.R.; Han, J.; Kim, D.D.; Fineman, M.S.; Baron, A.D. Effects of exenatide (exendin-4) on glycemic control over 30 weeks in sulfonylurea-treated patients with type 2 diabetes. Diabetes Care 2004, 27, 2628–2635. [Google Scholar] [CrossRef]
- Kendall, D.M.; Riddle, M.C.; Rosenstock, J.; Zhuang, D.; Kim, D.D.; Fineman, M.S.; Baron, A.D. Effects of exenatide (exendin-4) on glycemic control over 30 weeks in patients with type 2 diabetes treated with metformin and a sulfonylurea. Diabetes Care 2005, 28, 1083–1091. [Google Scholar] [CrossRef]
- Marre, M.; Shaw, J.; Brandle, M.; Bebakar, W.M.W.; Kamaruddin, N.A.; Strand, J.; Zdravkovic, M.; Le Thi, T.D.; Colagiuri, S.; LEAD-1 SU Study Group. Liraglutide, a once-daily human glp-1 analogue, added to a sulphonylurea over 26 weeks produces greater improvements in glycaemic and weight control compared with adding rosiglitazone or placebo in subjects with type 2 diabetes (LEAD-1 SU). Diabet. Med. 2009, 26, 268–278. [Google Scholar] [CrossRef]
- Nauck, M.; Frid, A.; Hermansen, K.; Shah, N.S.; Tankova, T.; Mitha, I.H.; Zdravkovic, M.; During, M.; Matthews, D.R.; LEAD-2 Study Group. Efficacy and safety comparison of liraglutide, glimepiride, and placebo, all in combination with metformin, in type 2 diabetes (LEAD-2 study). Diabetes Care 2009, 32, 84–90. [Google Scholar] [CrossRef]
- Garber, A.; Henry, R.; Ratner, R.; Garcia-Hernandez, P.A.; Rodriguez-Pattzi, H.; Olvera-Alvarez, I.; Hale, P.M.; Zdravkovic, M.; Bode, B.; LEAD-3 (Mono) Study Group. Liraglutide versus glimepiride monotherapy for type 2 diabetes (LEAD-3 mono): A randomized, 52-week, phase III, double-blind, parallel-treatment trial. Lancet 2009, 373, 473–481. [Google Scholar] [CrossRef]
- Zinman, B.; Gerich, J.; Buse, J.B.; Lewin, A.; Schwartz, S.; Raskin, P.; Hale, P.M.; Zdravkovic, M.; Blonde, L. Efficacy and safety of the human glucagon-like peptide-1 analog liraglutide in combination with metformin and thiazolidinedione in patients with type 2 diabetes (LEAD-4 MET+TZD). Diabetes Care 2009, 32, 1224–1230. [Google Scholar] [CrossRef]
- Nauck, M.A.; Meininger, G.; Sheng, D.; Terranella, L.; Stein, P.P. Efficacy and safety of the dipeptidyl peptidase-4 inhibitor, sitagliptin, compared with the sulfonylurea, glipizide, in patients with type 2 diabetes inadequately controlled on metformin alone: A randomized, double-blind, non-inferiority trial. Diabetes Obes. Metab. 2007, 9, 194–205. [Google Scholar] [CrossRef]
- Singh-Franco, D.; Perez, A.; Harrington, C. The effect of pramlintide acetate on glycemic control and weight in patients with type 2 diabetes mellitus and in obese patients without diabetes: A systematic review and meta-analysis. Diabetes Obes. Metab. 2011, 13, 169–180. [Google Scholar]
- List, J.F.; Woo, V.; Morales, E.; Tang, W.; Fiedorek, F.T. Sodium-glucose cotransport inhibition with dapagliflozin in type 2 diabetes. Diabetes Care 2009, 32, 650–657. [Google Scholar] [CrossRef]
- Ferrannini, E.; Seman, L.J.; Seewaldt-Becker, E.; Hantel, S.; Pinnetti, S.; Woerle, H.J. The potent and highly effective selective sodium-glucose co-transporter (sglt-2) inhibitor BI 10773 is safe and efficacious as monotherapy in patients with type 2 diabetes mellitus. Diabetologia 2010, 53, S351. [Google Scholar]
- Nauck, M.; del Prato, S.; Rohwedder, K.; Elze, M.; Parikh, S. Dapagliflozin vs. glipizide in patients with type 2 diabetes mellitus inadequately controlled on metformin: 52 week results of a double-blind, randomized controlled trial. Diabetologia 2010, 53, S107. [Google Scholar]
- Foote, C.; Perkovic, V.; Neal, B. Effects of sglt2 inhibitors on cardiovascular outcomes. Diabetes Vasc. Res. 2012, 9, 117–123. [Google Scholar] [CrossRef]
- Li, C.; Hung, Y.; Qamruddin, K.; Aziz, M.F.A.; Stein, H.; Schmidt, B. International noninterventional study of acarbose treatment in patients with type 2 diabetes mellitus. Diabetes Res. Clin. Pract. 2011, 92, 57–64. [Google Scholar] [CrossRef]
- Bays, H.E.; Goldberg, R.B.; Truitt, K.E.; Jones, M.R. Colesevelam hydrochloride therapy in patients with type 2 diabetes mellitus treated with metformin. Arch. Intern. Med. 2008, 168, 1975–1983. [Google Scholar] [CrossRef]
- Fonseca, V.A.; Rosenstock, J.; Wang, A.C.; Truitt, K.E.; Jones, M.R. Colesevelam hcl improves glycemic control and reduces ldl cholesterol in patients with inadequately controlled type 2 diabetes sulfonylurea-based therapy. Diabetes Care 2008, 31, 1479–1484. [Google Scholar] [CrossRef]
- Goldberg, R.B.; Fonseca, V.A.; Truitt, K.E.; Jones, M.R. Efficacy and safety of colesevelam in patients with type 2 diabetes mellitus and inadequate glycemic control receiving insulin-based therapy. Arch. Intern. Med. 2008, 168, 1531–1540. [Google Scholar] [CrossRef]
- Fonseca, V.A.; Handelsman, Y.; Staels, B. Colesevelam lowers glucose and lipid levels in type 2 diabetes: The clinical evidence. Diabetes Obes. Metab. 2010, 12, 384–392. [Google Scholar] [CrossRef]
- Cincotta, A.H.; Meier, A.H.; Cincotta, M., Jr. Bromocriptine improves glycaemic control and serum lipid profile in obese type 2 diabetic subjects: A new approach in the treatment of diabetes. Expert Opin. Investig. Drugs 1999, 8, 1683–1707. [Google Scholar] [CrossRef]
- Cincotta, A.H.; Gaziano, J.M.; Ezrokhi, M.; Scranton, R. Cycloset (quick-release bromobcriptine mesylate), a novel centrally acting treatment for type 2 diabetes. Diabetologia 2008, 51, S1–S588. [Google Scholar]
- Scranton, R.E.; Farwell, W.; Ezrokhi, M.; Gaziano, J.M.; Cincotta, A.H. Quick release bromocriptine (Cycloset) improves glycaemic control in patients with diabetes failing metformin/sulfonylurea combination therapy. Diabetologia 2008, 51, S1–S588. [Google Scholar] [CrossRef]
- DeFronzo, R.A. Bromocriptine: A sympatholytic, d2-dopamine agonist for the treatment of type 2 diabetes. Diabetes Care 2011, 34, 789–794. [Google Scholar] [CrossRef]
- American Diabetes Association. Standards of medical care in diabetes—2014 (Position Statement). Diabetes Care 2014, 37, s14–s80. [Google Scholar] [CrossRef]
- Nauck, M.; Stockmann, F.; Ebert, R.; Creutzfeldt, W. Reduced incretin effect in type 2 (non-insulin-dependant) diabetes. Diabetologia 1986, 29, 46–52. [Google Scholar] [CrossRef]
- Vilsboll, T.; Krarup, T.; Deacon, C.F.; Madsbad, S.; Holst, J.J. Reduced postprandial concentrations of intact biologically active glucagon-like peptide 1 in type 2 diabetic patients. Diabetes 2001, 50, 609–613. [Google Scholar] [CrossRef]
- Dungan, K.; Buse, J.B. Glucagon like peptide 1 based therapies for type 2 diabetes: A focus on exenatide. Clin. Diabetes 2005, 23, 56–62. [Google Scholar] [CrossRef]
- Demuth, H.U.; McIntosh, C.H.; Pederson, R.A. Type 2 diabetes—therapy with dipeptidyl peptide IV inhibitors. Biochim. Biophys. Acta 2005, 1751, 33–44. [Google Scholar] [CrossRef]
- Rosenstock, J.; Klaff, L.J.; Schwartz, S.; Northrup, J.; Holcombe, J.H.; Wilhelm, K.; Trautmann, M. Effects of exenatide and lifestyle modification on body weight and glucose tolerance in obese subjects with and without pre-diabetes. Diabetes Care 2010, 33, 1173–1175. [Google Scholar]
- Dushay, J.; Gao, C.; Gopalakrishnan, G.S.; Crawley, M.; Mitten, E.K.; Wilker, E.; Mullington, J.; Maratos-Flier, E. Short-term exenatide treatment leads to significant weight loss in a subset of obese women without diabetes. Diabetes Care 2012, 35, 4–11. [Google Scholar] [CrossRef]
- Astrup, A.; Rossner, S.; van Gaal, L.; Rissanen, A.; Niskanen, L.; Hakim, M.A.; Madsen, J.; Rasmussen, M.F.; Lean, M.E.J.; NN8022-1807 Study Group. Effects of liraglutide in the treatment of obesity: A randomized, double-blind, placebo-controlled study. Lancet 2009, 374, 1606–1616. [Google Scholar] [CrossRef]
- Singh, S.; Chang, H.; Richards, T.M.; Weiner, J.P.; Clark, J.M.; Segal, J.B. Glucagonlike peptide 1-based therapies and risk of hospitalization for acute pancreatitis in type 2 diabetes. JAMA Intern. Med. 2013, 173, 534–539. [Google Scholar] [CrossRef]
- Zuger, D.; Forster, K.; Lutz, T.A.; Riediger, T. Amylin and glp-1 target different populations of area postrema neurons that are both modulated by nutrient stimuli. Physiol. Behav. 2013, 61, 112–113. [Google Scholar]
- Asmar, M.; Bache, M.; Knop, F.K.; Madsbad, S.; Holst, J.J. Do the actions of glucagon like peptide 1 on gastric emptying, appetite and food intake involve release of amylin in humans? J. Clin. Endocrinol. Metab. 2010, 95, 2367–2375. [Google Scholar] [CrossRef]
- Wood, I.S.; Trayhurn, P. Glucose transporters (glut and sglt): Expanded families of sugar transport proteins. Br. J. Nutr. 2003, 89, 3–9. [Google Scholar] [CrossRef]
- Mather, A.; Pollock, C. Glucose handling by the kidney. Kideny Int. Suppl. 2011, 120, S1–S6. [Google Scholar] [CrossRef]
- Rahmoune, H.; Thompson, P.W.; Ward, J.M.; Smith, C.D.; Hong, G.; Brown, J. Glucose transporters in human renal proximal tubular cells isolated from the urine of patients with non-insulin-dependent diabetes. Diabetes 2005, 54, 3427–3434. [Google Scholar] [CrossRef]
- Bailey, C.J.; Gross, J.L.; Pieters, A.; Bastien, A.; List, J.F. Effect of dapagliflozin in patients with type 2 diabetes who have inadequate glycemic control with metformin: A randomized, double blind, placebo-controlled trial. Lancet 2010, 375, 2223–2233. [Google Scholar] [CrossRef]
- Gurabo, P.R. Invokana; Janssen Pharmaceuticals, Inc.: Titusville, NJ, USA, 2013. [Google Scholar]
- Davidson, M.H.; Hauptman, J.; DiGirolamo, M.; Foreyt, J.P.; Halsted, C.H.; Heber, D.; Heimburger, D.C.; Lucas, C.P.; Robbins, D.C.; Chung, J.; et al. Weight control and risk factor reduction in obsess subjects treated for 2 years with orlistat. JAMA 1999, 281, 235–242. [Google Scholar] [CrossRef]
- Torgerson, J.S.; Hauptman, J.; Boldrin, M.N.; Sjostrom, L. Xenical in the prevention of diabetes in obese subjects (xendos) study. Diabetes Care 2004, 27, 155–161. [Google Scholar] [CrossRef]
- Miles, J.M.; Leiter, L.; Hollander, P.; Wadden, T.; Anderson, J.W.; Doyle, M.; Foreyt, J.; Aronne, L.; Klein, S. Effect of orlistat in overweight and obese patients with type 2 diabetes treated with metformin. Diabetes Care 2002, 25, 1123–1128. [Google Scholar] [CrossRef]
- Allison, D.B.; Gadde, K.M.; Garvey, W.T.; Peterson, C.A.; Schwiers, M.L.; Najarian, T.; Tam, P.Y.; Troupin, B.; Day, W.W. Controlled-release phentermine/topiramate in severely obese adults: A randomized controlled trial (EQUIP). Obesity 2011, 20, 330–342. [Google Scholar]
- Gadde, K.M.; Allison, D.B.; Ryan, D.H.; Peterson, C.A.; Troupin, B.; Schwiers, M.L.; Day, W.W. Effects of low-dose, controlled-release, phentermine plus topiramate combination on weight and associated comorbidities in overweight and obese adults (CONQUER): A randomized, placebo-controlled, phase 3 trial. Lancet 2011, 377, 1341–1352. [Google Scholar] [CrossRef]
- Garvey, W.T.; Ryan, D.H.; Look, M.; Gadde, K.M.; Allison, D.B.; Peterson, C.A.; Schwiers, M.; Day, W.W.; Bowden, C.H. Two-year sustained weight loss and metabolic benefits with controlled-release phentermine/topiramate in obese and overweight adults (SEQUEL): A randomized, placebo-controlled, phase 3 extension study. Am. J. Clin. Nutr. 2012, 95, 297–308. [Google Scholar] [CrossRef]
- Garvey, W.T.; Ryan, D.H.; Henry, R.; Bohannon, N.J.; Toplak, H.; Schwiers, M.; Troupin, B.; Day, W.W. Prevention of type 2 diabetes in subjects with prediabetes and metabolic syndrome treated with phentermine and topiramate extended-release. Diabetes Care 2013, 37, 912–921. [Google Scholar]
- Leibowitz, S.F.; Alexander, J.T. Hypothalmic serotonin in control of eating behavior, meal size, and body weight. Biol. Psychiatry 1998, 44, 851–864. [Google Scholar] [CrossRef]
- Smith, S.R.; Weissman, N.J.; Anderson, C.M.; Sanchez, M.; Chuang, E.; Stubbe, S.; Bays, H.; Shanahan, W.R. Multicenter, placebo-controlled trial of lorcaserin for weight management. N. Engl. J. Med. 2010, 363, 245–256. [Google Scholar] [CrossRef]
- Fidler, M.C.; Sanchez, M.; Raether, B.; Weissman, N.J.; Smith, S.R.; Shanahan, W.R.; Anderson, C.M. A one-year randomized trial of lorcaserin for weight loss in obese and overweight adults: The BLOSSOM Trial. J. Clin. Endocrinol. Metab. 2011, 96, 3067–3077. [Google Scholar] [CrossRef]
- O’Neil, P.M.; Smith, S.R.; Weissman, N.J.; Fidler, M.C.; Sanchez, M.; Zhang, J.; Raether, B.; Anderson, C.M.; Shanahan, W.R. Randomized placebo-controlled clinical trial of lorcaserin for weight loss in type 2 diabetes mellitus: The BLOOM-DM Study. Obesity 2012, 20, 1426–1436. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Goswami, G.; Shinkazh, N.; Davis, N. Optimal Pharmacologic Treatment Strategies in Obesity and Type 2 Diabetes. J. Clin. Med. 2014, 3, 595-613. https://doi.org/10.3390/jcm3020595
Goswami G, Shinkazh N, Davis N. Optimal Pharmacologic Treatment Strategies in Obesity and Type 2 Diabetes. Journal of Clinical Medicine. 2014; 3(2):595-613. https://doi.org/10.3390/jcm3020595
Chicago/Turabian StyleGoswami, Gayotri, Nataliya Shinkazh, and Nichola Davis. 2014. "Optimal Pharmacologic Treatment Strategies in Obesity and Type 2 Diabetes" Journal of Clinical Medicine 3, no. 2: 595-613. https://doi.org/10.3390/jcm3020595
APA StyleGoswami, G., Shinkazh, N., & Davis, N. (2014). Optimal Pharmacologic Treatment Strategies in Obesity and Type 2 Diabetes. Journal of Clinical Medicine, 3(2), 595-613. https://doi.org/10.3390/jcm3020595




