Bacterial Osteomyelitis in the Lower Extremities: Analysis of Histology and MRI Findings in a Case-Control Pilot Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Patient Population and Data Collection
2.3. Diagnostic Procedures
2.4. Statistic Analysis
3. Results
3.1. Microbiology
3.2. MRI
3.3. Histology
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CM | Contrast medium |
| COPD | Chronic obstructive pulmonary disease |
| Fx | Fracture |
| MARS | Metal artifact reduction sequence |
| mDIXON | Modified DIXON |
| MSSA | Methicillin-susceptible Staphylococcus aureus |
| NSA | Number of signal averages |
| OM | Osteomyelitis |
| PAD | Peripheral artery disease |
| PI | Parallel imaging |
| PDw | Proton density-weighted |
| SPIR | Spectral presaturation with inversion recovery |
| STIR | Short Tau Inversion Recovery |
| TSE | Turbo spin echo |
| T1w | T1-weighted |
| T2w | T2-weighted |
References
- Waldvogel, F.A.; Medoff, G.; Swartz, M.N. Osteomyelitis: A review of clinical features, therapeutic considerations and unusual aspects. N. Engl. J. Med. 1970, 282, 198–206. [Google Scholar] [CrossRef] [PubMed]
- Lew, D.P.; Waldvogel, F.A. Osteomyelitis. Lancet 2004, 364, 369–379. [Google Scholar] [CrossRef] [PubMed]
- Hofstee, M.I.; Muthukrishnan, G.; Atkins, G.J.; Riool, M.; Thompson, K.; Morgenstern, M.; Stoddart, M.J.; Richards, R.G.; Zaat, S.A.J.; Moriarty, T.F. Current concepts of osteomyelitis: From pathologic mechanisms to advanced research methods. Am. J. Pathol. 2020, 190, 1151–1163. [Google Scholar] [CrossRef] [PubMed]
- Moldovan, F. Role of serum biomarkers in differentiating periprosthetic joint infections from aseptic failures after total hip arthroplasties. J. Clin. Med. 2024, 13, 5716. [Google Scholar] [CrossRef]
- Li, M.; Hou, Q.; Zhong, L.; Zhao, Y.; Fu, X. Macrophage related chronic inflammation in non-healing wounds. Front. Immunol. 2021, 12, 681710. [Google Scholar] [CrossRef]
- McNally, M.; Nagarajah, K. (iv) Osteomyelitis. Orthop. Trauma 2010, 24, 416–429. [Google Scholar]
- Walter, N.; Baertl, S.; Alt, V.; Rupp, M. What is the burden of osteomyelitis in Germany? An analysis of inpatient data from 2008 through 2018. BMC Infect. Dis. 2021, 21, 550. [Google Scholar] [CrossRef]
- Kremers, H.M.; Nwojo, M.E.; Ransom, J.E.; Wood-Wentz, C.M.; Melton, L.J., 3rd; Huddleston, P.M., 3rd. Trends in the epidemiology of osteomyelitis: A population-based study, 1969 to 2009. J. Bone Jt. Surg. Am. 2015, 97, 837–845. [Google Scholar] [CrossRef]
- Laurent, E.; Gras, G.; Druon, J.; Rosset, P.; Baron, S.; Le-Louarn, A.; Rusch, E.; Bernard, L.; Grammatico-Guillon, L. Key features of bone and joint infections following the implementation of reference centers in France. Med. Mal. Infect. 2018, 48, 256–262. [Google Scholar] [CrossRef]
- Ferguson, J.; Alexander, M.; Bruce, S.; O’Connell, M.; Beecroft, S.; McNally, M. A retrospective cohort study comparing clinical outcomes and healthcare resource utilisation in patients undergoing surgery for osteomyelitis in England: A case for reorganising orthopaedic infection services. J. Bone Jt. Infect. 2021, 6, 151–163. [Google Scholar] [CrossRef]
- Rao, N.; Ziran, B.H.; Lipsky, B.A. Treating osteomyelitis: Antibiotics and surgery. Plast. Reconstr. Surg. 2011, 127, 177S–187S. [Google Scholar] [CrossRef] [PubMed]
- Hackenberg, R.K.; Schmitt-Sanchez, F.; Endler, C.; Tischler, V.; Surendar, J.; Welle, K.; Kabir, K.; Schildberg, F.A. Value of diagnostic tools in the diagnosis of osteomyelitis: Pilot study to establish an osteomyelitis score. J. Clin. Med. 2023, 12, 3057. [Google Scholar] [CrossRef]
- Buckwalter, K.A.; Lin, C.; Ford, J.M. Managing postoperative artifacts on computed tomography and magnetic resonance imaging. Semin. Musculoskelet. Radiol. 2011, 15, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Massel, D.H.; Jenkins, N.W.; Rush, A.J., 3rd; Trapana, J.E.; Foremny, G.B.; Donnally, C.J., 3rd; Subhawong, T.; Aiyer, A. MRI and clinical risk indicators for osteomyelitis. Foot Ankle Spec. 2021, 14, 415–426. [Google Scholar] [CrossRef]
- Park, B.N.; Hong, S.J.; Yoon, M.A.; Oh, J.K. MRI diagnosis for post-traumatic osteomyelitis of extremities using conventional metal-artifact reducing protocols: Revisited. Acad. Radiol. 2019, 26, e317–e323. [Google Scholar] [CrossRef]
- Govaert, G.A.; IJpma, F.F.; McNally, M.; McNally, E.; Reininga, I.H.; Glaudemans, A.W. Accuracy of diagnostic imaging modalities for peripheral post-traumatic osteomyelitis—A systematic review of the recent literature. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 1393–1407. [Google Scholar] [CrossRef]
- Tiemann, A.; Hofmann, G.O.; Krukemeyer, M.G.; Krenn, V.; Langwald, S. Histopathological Osteomyelitis Evaluation Score (HOES)—An innovative approach to histopathological diagnostics and scoring of osteomyelitis. GMS Interdiscip. Plast. Reconstr. Surg. DGPW 2014, 3, Doc08. [Google Scholar] [CrossRef]
- Tardaguila-Garcia, A.; Sanz-Corbalan, I.; Garcia-Morales, E.; Garcia-Alvarez, Y.; Molines-Barroso, R.J.; Lazaro-Martinez, J.L. Diagnostic accuracy of bone culture versus biopsy in diabetic foot osteomyelitis. Adv. Skin Wound Care 2021, 34, 204–208. [Google Scholar] [CrossRef]
- Ledermann, H.P.; Morrison, W.B.; Schweitzer, M.E. MR image analysis of pedal osteomyelitis: Distribution, patterns of spread, and frequency of associated ulceration and septic arthritis. Radiology 2002, 223, 747–755. [Google Scholar] [CrossRef]
- Lee, Y.J.; Sadigh, S.; Mankad, K.; Kapse, N.; Rajeswaran, G. The imaging of osteomyelitis. Quant. Imaging Med. Surg. 2016, 6, 184–198. [Google Scholar] [CrossRef]
- Germann, C.; Filli, L.; Jungmann, P.M.; Graf, D.N.; Fritz, J.; Pfirrmann, C.W.A.; Sutter, R. Prospective and longitudinal evolution of postoperative periprosthetic findings on metal artifact-reduced MR imaging in asymptomatic patients after uncemented total hip arthroplasty. Skelet. Radiol. 2021, 50, 1177–1188. [Google Scholar] [CrossRef]


| Parameter | Pre-/Post-Contrast T1w TSE Axial | T2x mDIXON TSE Axial | STIR TSE Coronal | Post-Contrast T1w SPIR TSE Sagittal |
|---|---|---|---|---|
| TR/TE (ms) | 687/8 | 4120/80 | 4596/65 | 615/10 |
| Flip angle (degrees) | 90 | 90 | 90 | 90 |
| Field of view (mm) | 380 × 380 | 380 × 380 | 480 × 480 | 460 × 460 |
| Matrix | 377 × 508 | 431 × 544 | 248 × 368 | 563 × 768 |
| Reconstructed Voxel size (mm) | 0.59 × 0.59 × 6.00 | 0.44 × 0.44 × 6.00 | 0.71 × 0.71 × 4.00 | 0.40 × 0.40 × 3.0 |
| Slices | 74 | 74 | 36 | 30 |
| TSE factor | 6 | 18 | 24 | 5 |
| Acquisition time (min:s) | 3:39.9 | 5:13.1 | 2:08.7 | 3:46.3 |
| PI reduction factor (SENSE) | 1.7 | 2.0 | 1.0 | 1.0 |
| NSA | 1 | 1 | 1 | 1 |
| Comorbidities | OM Group | Control Group | |
|---|---|---|---|
| Cardiovascular disorders | Arterial hypertension | n = 2 | n = 1 |
| Cardiac arrhythmia | n = 1 | n = 1 | |
| Coronary heart disease | n = 0 | n = 1 | |
| Vascular disorders | PAD 1 | n = 1 | n = 1 |
| Chronic venous insufficiency | n = 1 | n = 0 | |
| Pulmonary disorders | COPD 2 | n = 1 | n = 0 |
| Metabolic/endocrinological disorders | Type 2 diabetes mellitus | n = 1 | n = 0 |
| Type 1 diabetes mellitus | n = 0 | n = 1 | |
| Autoimmune disorders | Psoriasis | n = 0 | n = 1 |
| Malignant tumors | Pancreatic/ovarian cancer | n = 0 | n = 2 |
| Lymphadenopathy | n = 1 | n = 0 | |
| Others | Chronic renal insufficiency | n = 1 | n = 0 |
| Hypercholesterolemia | n = 0 | n = 1 | |
| Neurological/psychiatric illness | n = 1 | n = 1 | |
| Alcohol abuse | n = 0 | n = 1 | |
| Nicotine abuse | n = 0 | n = 1 | |
| Drug addiction | n = 1 | n = 1 | |
| OM Group | Pathogen in Bone | Other Pathogens |
|---|---|---|
| 1 | S. haemolyticus | Escherichia coli |
| S. aureus (MSSA *) | ||
| 2 | S. epidermidis | - |
| Pseudomonas aeruginosa | ||
| 3 | S. aureus (MSSA *) | S. epidermidis |
| Bacillus cereus | ||
| 4 | S. epidermidis | - |
| 5 | S. aureus (MSSA *) | S. epidermidis |
| S. saccharolyticus | S. pettenkoferi |
| Pat., Fig. No. | Affected Localization (OM, Fx) | MRI Findings of the Bone | Interpretation | Histopathologic Findings | Interpretation |
|---|---|---|---|---|---|
| Osteomyelitis group | |||||
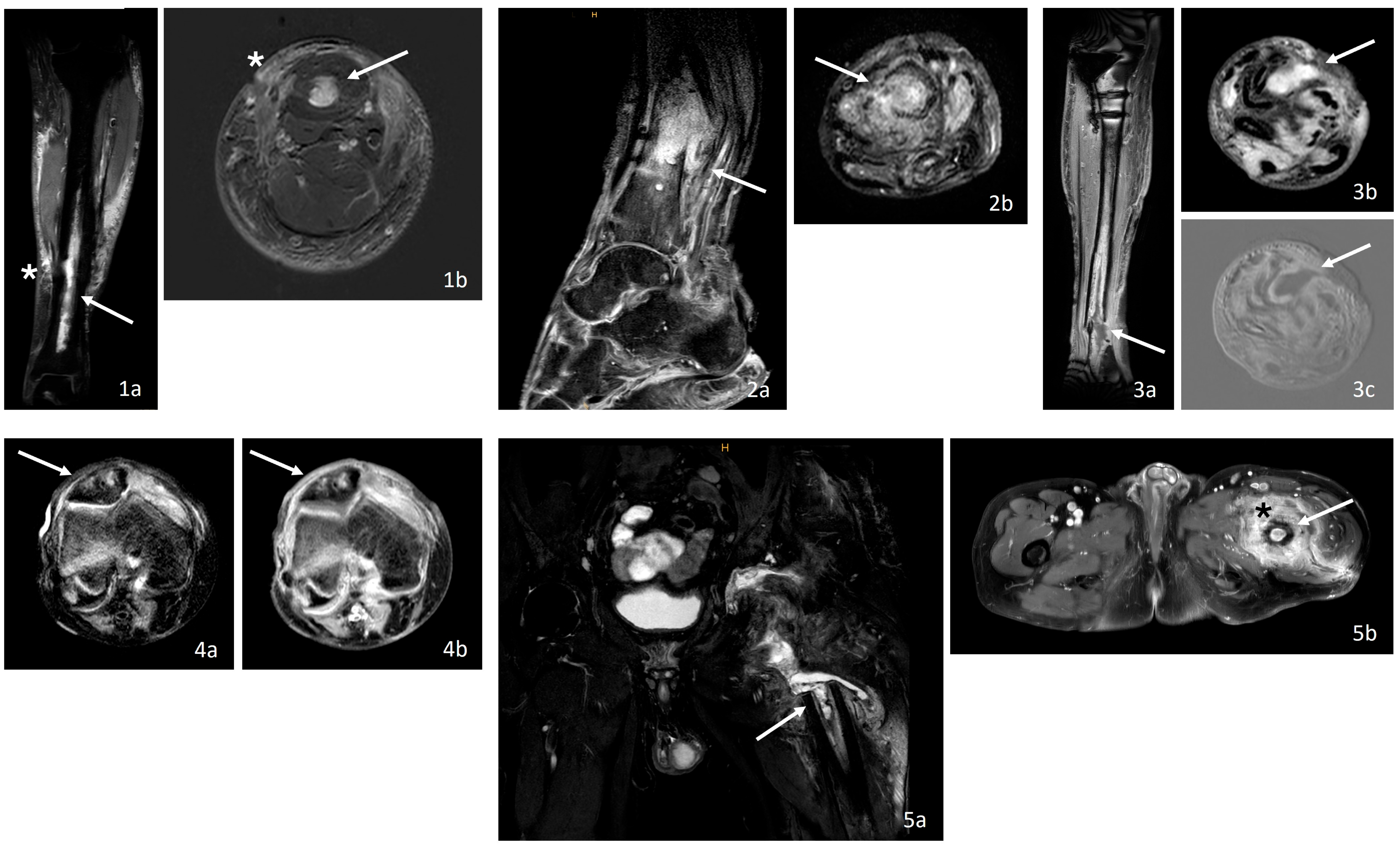
| 1, Figure 1.1 | OM: Left lower leg, Fx: Lower leg shaft fracture | Long-distance circumscribed CM enhancement of the bone marrow in the tibial shaft (↑) and the periosteal soft tissue with edema (*) | Characteristic of OM | Spongy bone components with fragmented trabeculae and mildly increased bone remodeling, edema in the marrow spaces, as well as fibrosis and lymphoid cell accumulations | Chronic granulating OM |
| 2, Figure 1.2 | OM: Left ankle, Fx: Lower leg shaft fracture | CM enhancement of the bone marrow adjacent to the fracture zone (↑) in the sense of bone edema and intraosseous abscess with marked soft tissue reaction | Characteristic of OM | Vital, focally avital corticospongious bone with signs of remodeling, reactive trabecular bone formation, and marrow fibrosis with acute and chronically granulating inflammation in the soft tissue and marrow | Active and chronic granulating OM |
| 3, Figure 1.3 | OM: Left lower leg, Fx: Proximal tibia and lower leg shaft fracture | Intramedullary fluid accumulation with CM enhancement as a sign of an intramedullary abscess of the distal tibia (↑) | Characteristic of OM | Acute and chronically granulating inflammation in the periosteal soft tissue and marrow, partially vital, partially avital bone with remodeling processes, marrow fibrosis, and dense infiltrates of neutrophilic granulocytes, lymphocytes, and plasma cells, along with areas of tissue necrosis and fibrin deposits | Active and chronic granulating OM |
| 4, Figure 1.4 | OM: Right knee and patella, Fx: Patella fracture | Intrapatellar inhomogeneous CM enhancement as a sign of bone edema with peripheral enhancement (↑) in the context of an intraosseous abscess | Characteristic of OM | Acute purulent inflammation of the soft tissue and bone with loose medullary fibrosis, necrobiotic spongy lamellar bone with signs of remodeling, and infiltration of neutrophilic granulocytes | Chronic granulating OM |
| 5, Figure 1.5 | OM: Left pelvis and hip, Fx: Proximal femur fracture | Pronounced bone marrow edema with intramedullary abscess (↑) and soft tissue reaction coronary as well as long-distance CM enhancement intramedullary (↑) and in the periosteal tissue (*) axial | Characteristic of OM | Chronic granulomatous inflammation in bone and soft tissue with fragmented bone tissue undergoing remodeling processes | Chronic granulating OM |
| Control group | |||||
| 6, Figure 2.1 | Right lower leg, Fx: Proximal tibia fracture | Low-grade intramedullary edema without BM accumulation in the context of postoperative changes in periosteal soft tissue reaction with edema and BM accumulation (↑) as a sign of soft tissue infection, but no signs of intramedullary sequestration or abscesses | Unremarkable | Periprosthetic wear-type membrane in the soft tissue, with no evidence of inflammatory cells in the bone | Soft tissue infection |
| 7, Figure 2.2 | Left lower leg, Fx: Ankle and proximal fibula fracture | Pronounced edematous distension of the lower leg muscles (*) with partial muscle necrosis (↑) (axial differences in sT1w and T2w), slight bone marrow edema of the tibial shaft, but no relevant CM enhancement, no bone necrosis or inflammatory changes | Unremarkable | Chronically granulating soft tissue inflammation with partially non-vital, atrophic skeletal muscle and focal acute granulocytic inflammation, without evidence of inflammatory cells in the bone | Soft tissue infection |
| 8, Figure 2.3 | Right ankle, Fx: Ankle fracture | Minimal CM enhancement of the superficial cortex and discrete bone marrow edema with CM enhancement of the distal fibula (↑) and adjacent soft tissues, primarily reactive postoperatively | Non-specific | Partially vital and non-vital, fragmented, inflammation-free bone tissue with remodeling processes, at most focal cell residues within otherwise empty osteocytic lacunae, interspersed with fibrin strands, gangrene of skin/subcutaneous tissue, and chronic granulomatous and fibrosing soft tissue inflammation | Soft tissue infection, osteonecrosis |
| 9, Figure 2.4 | Right ankle, Fx: Talus and distal fibula fracture | Soft tissue edema (↑) and focal bone edema with diffuse contrast enhancement, but no signs of cortical destruction, abscesses, or sequestra in the context of a subluxated ankle joint, most likely indicative of a stress reaction | Non-specific | Bone tissue with medullary fibrosis but without inflammatory infiltration | Osteonecrosis |
| 10, Figure 2.5 | Right lower leg, Fx: Tibia shaft fracture | Soft tissue reaction without osseous involvement in terms of bone edema, no contrast enhancement, no sequestra, abscesses, or cortical reactions | Unremarkable | Focal purulent inflammatory reaction in the soft tissue without a corresponding inflammatory response in the bone | Soft tissue infection |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hackenberg, R.K.; Schmitt-Sánchez, F.; Endler, C.; Tischler, V.; Surendar, J.; Kabir, K.; Welle, K.; Burger, C.; Wirtz, D.C.; Schildberg, F.A. Bacterial Osteomyelitis in the Lower Extremities: Analysis of Histology and MRI Findings in a Case-Control Pilot Study. J. Clin. Med. 2025, 14, 4877. https://doi.org/10.3390/jcm14144877
Hackenberg RK, Schmitt-Sánchez F, Endler C, Tischler V, Surendar J, Kabir K, Welle K, Burger C, Wirtz DC, Schildberg FA. Bacterial Osteomyelitis in the Lower Extremities: Analysis of Histology and MRI Findings in a Case-Control Pilot Study. Journal of Clinical Medicine. 2025; 14(14):4877. https://doi.org/10.3390/jcm14144877
Chicago/Turabian StyleHackenberg, Roslind K., Fabio Schmitt-Sánchez, Christoph Endler, Verena Tischler, Jayagopi Surendar, Koroush Kabir, Kristian Welle, Christof Burger, Dieter C. Wirtz, and Frank A. Schildberg. 2025. "Bacterial Osteomyelitis in the Lower Extremities: Analysis of Histology and MRI Findings in a Case-Control Pilot Study" Journal of Clinical Medicine 14, no. 14: 4877. https://doi.org/10.3390/jcm14144877
APA StyleHackenberg, R. K., Schmitt-Sánchez, F., Endler, C., Tischler, V., Surendar, J., Kabir, K., Welle, K., Burger, C., Wirtz, D. C., & Schildberg, F. A. (2025). Bacterial Osteomyelitis in the Lower Extremities: Analysis of Histology and MRI Findings in a Case-Control Pilot Study. Journal of Clinical Medicine, 14(14), 4877. https://doi.org/10.3390/jcm14144877








