The Association of Myocardial Infarction History and Geriatric Syndromes in the Elderly: Data from the Cross-Sectional Study EVKALIPT
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Limitations
5. Discussion
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Guzman, J. Ageing in the Twenty-First Century: A Celebration and A Challenge. 2012. Available online: https://www.unfpa.org/sites/default/files/pub-pdf/UNFPA-Exec-Summary.pdf (accessed on 20 August 2024).
- Beard, J.R.; Officer, A.; de Carvalho, I.A.; Sadana, R.; Pot, A.M.; Michel, J.-P.; Lloyd-Sherlock, P.; Epping-Jordan, J.E.; Peeters, G.M.E.E.G.; Mahanani, W.R.; et al. The World report on ageing and health: A policy framework for healthy ageing. Lancet 2016, 387, 2145–2154. [Google Scholar] [CrossRef] [PubMed]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Frailty in older adults: Evidence for a phenotype. J. Gerontol. A Biol. Sci. Med. Sci. 2001, 56, M146–M156. [Google Scholar] [CrossRef] [PubMed]
- Clegg, A.; Young, J.; Iliffe, S.; Rikkert, M.O.; Rockwood, K. Frailty in elderly people. Lancet 2013, 381, 752–762, Erratum in Lancet 2013, 382, 1328. [Google Scholar] [CrossRef] [PubMed]
- Rasiah, J.; Cummings, G.G.; Gruneir, A.; Oelke, N.D.; Estabrooks, C.; Holroyd-Leduc, J. Prefrailty in older adults: A concept analysis. Int. J. Nurs. Stud. 2020, 108, 103618. [Google Scholar] [CrossRef] [PubMed]
- Santos-Eggimann, B.; Cuénoud, P.; Spagnoli, J.; Junod, J. Prevalence of frailty in middle-aged and older community-dwelling Europeans living in 10 countries. J. Gerontol. A Biol. Sci. Med. Sci. 2009, 64, 675–681. [Google Scholar] [CrossRef]
- O’caoimh, R.; Sezgin, D.; O’donovan, M.R.; Molloy, D.W.; Clegg, A.; Rockwood, K.; Liew, A. Prevalence of frailty in 62 countries across the world: A systematic review and meta-analysis of population-level studies. Age Ageing 2021, 50, 96–104. [Google Scholar] [CrossRef]
- Spoon, D.B.; Psaltis, P.J.; Singh, M.; Holmes, D.R.; Gersh, B.J.; Rihal, C.S.; Lennon, R.J.; Moussa, I.D.; Simari, R.D.; Gulati, R. Trends in cause of death after percutaneous coronary intervention. Circulation 2014, 129, 1286–1294. [Google Scholar] [CrossRef]
- Singh, M.; Stewart, R.; White, H. Importance of frailty in patients with cardiovascular disease. Eur. Heart J. 2014, 35, 1726–1731. [Google Scholar] [CrossRef]
- Aïdoud, A.; Gana, W.; Poitau, F.; Debacq, C.; Leroy, V.; Nkodo, J.; Poupin, P.; Angoulvant, D.; Fougère, B. High Prevalence of Geriatric Conditions Among Older Adults with Cardiovascular Disease. J. Am. Heart Assoc. 2023, 12, e026850. [Google Scholar] [CrossRef]
- Vorobyeva, N.M.; Tkacheva, O.N.; Kotovskaya, Y.V.; Ovcharova, L.N.; Selezneva, E.V. Russian epidemiological study EVKALIPT: Protocol and basic characteristics of participants. Russ. J. Geriatr. Med. 2021, 1, 35–43. (In Russian) [Google Scholar] [CrossRef]
- Dudinskaya, E.N.; Vorobyeva, N.M.; Onuchina, J.S.; Machekhina, L.V.; Selezneva, E.V.; Ovcharova, L.N.; Kotovskaya, Y.V.; Tkacheva, O.N. The association of osteoporosis and geriatric syndromes in the elderly: Data from the Russian epidemiological study EVKALIPT. Arch. Osteoporos. 2023, 18, 30, Erratum in Arch. Osteoporos. 2023, 18, 40. [Google Scholar] [CrossRef] [PubMed]
- Shalnova, S.A.; Drapkina, O.M.; Kutsenko, V.A.; Kapustina, A.V.; Muromtseva, G.A.; Yarovaya, E.B.; Balanova, Y.A.; Evstifeeva, S.E.; Imaeva, A.E.; Shlyakhto, E.V.; et al. Myocardial infarction in the population of some Russian regions and its prognostic value. Russ. J. Cardiol. 2022, 27, 4952. (In Russian) [Google Scholar] [CrossRef]
- Salari, N.; Morddarvanjoghi, F.; Abdolmaleki, A.; Rasoulpoor, S.; Khaleghi, A.A.; Hezarkhani, L.A.; Shohaimi, S.; Mohammadi, M. The global prevalence of myocardial infarction: A systematic review and meta-analysis. BMC Cardiovasc. Disord. 2023, 23, 206. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Cai, Y.; Liu, H.; Fan, L.; Wu, C. Frailty as a predictor of all-cause mortality and readmission in older patients with acute coronary syndrome: A systematic review and meta-analysis. Wien. Klin. Wochenschr. 2020, 132, 301–309. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, M.K.; Marques, A.; Lobo, D.M.L.; Umeda, I.I.K.; Oliveira, M.F. Pre-Frailty Increases the Risk of Adverse Events in Older Patients Undergoing Cardiovascular Surgery. Arq. Bras. Cardiol. 2017, 109, 299–306. [Google Scholar] [CrossRef]
- Veronese, N.; Cereda, E.; Stubbs, B.; Solmi, M.; Luchini, C.; Manzato, E.; Sergi, G.; Manu, P.; Harris, T.; Fontana, L.; et al. Risk of cardiovascular disease morbidity and mortality in frail and pre-frail older adults: Results from a meta-analysis and exploratory meta-regression analysis. Ageing Res. Rev. 2017, 35, 63–73. [Google Scholar] [CrossRef]
- Marinus, N.; Vigorito, C.; Giallauria, F.; Haenen, L.; Jansegers, T.; Dendale, P.; Feys, P.; Meesen, R.; Timmermans, A.; Spildooren, J.; et al. Frailty is highly prevalent in specific cardiovascular diseases and females, but significantly worsens prognosis in all affected patients: A systematic review. Ageing Res. Rev. 2021, 66, 101233. [Google Scholar] [CrossRef]
- Bai, W.; Hao, B.; Meng, W.; Qin, J.; Xu, W.; Qin, L. Association between frailty and short- and long-term mortality in patients with critical acute myocardial infarction: Results from MIMIC-IV. Front. Cardiovasc. Med. 2022, 9, 1056037. [Google Scholar] [CrossRef]
- Patel, A.; Goodman, S.G.; Yan, A.T.; Alexander, K.P.; Wong, C.L.; Cheema, A.N.; Udell, J.A.; Kaul, P.; D’Souza, M.; Hyun, K.; et al. Frailty and Outcomes after Myocardial Infarction: Insights from the Concordance Registry. J. Am. Heart Assoc. 2018, 7, e009859. [Google Scholar] [CrossRef]
- Ekerstad, N.; Javadzadeh, D.; Alexander, K.P.; Bergström, O.; Eurenius, L.; Fredrikson, M.; Gudnadottir, G.; Held, C.; Ängerud, K.H.; Jahjah, R.; et al. Clinical Frailty Scale classes are independently associated with 6-month mortality for patients after acute myocardial infarction. Eur. Heart J. Acute Cardiovasc. Care 2022, 11, 89–98. [Google Scholar] [CrossRef]
- Bowling, C.B.; Fonarow, G.C.; Patel, K.; Zhang, Y.; Feller, M.A.; Sui, X.; Blair, S.N.; Alagiakrishnan, K.; Aban, I.B.; Love, T.E.; et al. Impairment of activities of daily living and incident heart failure in community-dwelling older adults. Eur. J. Heart Fail. 2012, 14, 581–587. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.V.; Pham, T.T.X.; Nguyen, T.N. The Burden of Cardiovascular Disease and Geriatric Syndromes in Older Patients Undergoing Chronic Hemodialysis. Int. J. Environ. Res. Public Health 2024, 21, 812. [Google Scholar] [CrossRef] [PubMed]
- Conroy, S.P.; Bardsley, M.; Smith, P.; Neuburger, J.; Keeble, E.; Arora, S.; Kraindler, J.; Ariti, C.; Sherlaw-Johnson, C.; Street, A.; et al. Comprehensive Geriatric Assessment for Frail Older People in Acute Hospitals: The HoW-CGA Mixed-Methods Study; NIHR Journals Library: Southampton, UK, 2019; Health Services and Delivery Research, No. 7.15.; Appendix 7, List of the Variables Used to Construct the Rockwood Index. Available online: https://www.ncbi.nlm.nih.gov/books/NBK540053/ (accessed on 20 August 2024).
- Searle, S.D.; Mitnitski, A.; Gahbauer, E.A.; Gill, T.M.; Rockwood, K. A standard procedure for creating a frailty index. BMC Geriatr. 2008, 8, 24. [Google Scholar] [CrossRef] [PubMed]
- Ferrucci, L.; Fabbri, E. Inflammageing: Chronic inflammation in ageing, cardiovascular disease, and frailty. Nat. Rev. Cardiol. 2018, 15, 505–522. [Google Scholar] [CrossRef] [PubMed]
- Libby, P. Inflammation in Atherosclerosis-No Longer a Theory. Clin. Chem. 2021, 67, 131–142. [Google Scholar] [CrossRef]
- Ruparelia, N.; Chai, J.T.; Fisher, E.A.; Choudhury, R.P. Inflammatory processes in cardiovascular disease: A route to targeted therapies. Nat. Rev. Cardiol. 2017, 14, 133–144. [Google Scholar] [CrossRef]
- Wang, H.; Liu, Z.; Shao, J.; Lin, L.; Jiang, M.; Wang, L.; Lu, X.; Zhang, H.; Chen, Y.; Zhang, R. Immune and Inflammation in Acute Coronary Syndrome: Molecular Mechanisms and Therapeutic Implications. J. Immunol. Res. 2020, 2020, 4904217. [Google Scholar] [CrossRef]
- Ryabov, V.V.; Vorobeva, D.A.; Kologrivova, I.V.; Suslova, T.E. Pro-Inflammatory Biomarkers and Progression of Atherosclerosis in Patients with Myocardial Infarction with Non-Obstructive Coronary Artery Disease: 1-Year Follow-Up. J. Pers. Med. 2023, 13, 1669. [Google Scholar] [CrossRef]
- Pascual, M.J.E.; Pais, J.L.; Coronel, B.I.; Ibanez, D.N.; Gil, D.G.; Alvarez, C.P.; Rodriguez, R.O.; Romero, R.A.; Vinues, C.M.; Sanz, A.F.; et al. Role of inflammation in Myocardial Infarction with Non-Obstructive Coronary Arteries. Eur. Heart J. 2021, 42 (Suppl. S1), ehab724.1296. [Google Scholar] [CrossRef]
- Pizzi, C.; Costa, G.M.; Santarella, L.; Flacco, M.E.; Capasso, L.; Bert, F.; Manzoli, L. Depression symptoms and the progression of carotid intima-media thickness: A 5-year follow-up study. Atherosclerosis 2014, 233, 530–536. [Google Scholar] [CrossRef]
- Koganti, S.; Karanasos, A.; Regar, E.; Rakhit, R.D. Association of systemic inflammatory biomarkers with morphological characteristics of coronary atherosclerotic plaque by intravascular optical coherence tomography. Hellenic. J. Cardiol. 2021, 62, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Birnhuber, A.; Fliesser, E.; Gorkiewicz, G.; Zacharias, M.; Seeliger, B.; David, S.; Welte, T.; Schmidt, J.; Olschewski, H.; Wygrecka, M.; et al. Between inflammation and thrombosis: Endothelial cells in COVID-19. Eur. Respir. J. 2021, 58, 2100377. [Google Scholar] [CrossRef] [PubMed]
- Döring, Y.; Soehnlein, O.; Weber, C. Neutrophil Extracellular Traps in Atherosclerosis and Atherothrombosis. Circ. Res. 2017, 120, 736–743. [Google Scholar] [CrossRef] [PubMed]
- Walston, J.; McBurnie, M.A.; Newman, A.; Tracy, R.P.; Kop, W.J.; Hirsch, C.H.; Gottdiener, J.; Fried, L.P. Cardiovascular Health Study. Frailty and activation of the inflammation and coagulation systems with and without clinical comorbidities: Results from the Cardiovascular Health Study. Arch. Intern. Med. 2002, 162, 2333–2341. [Google Scholar] [CrossRef]
- Manten, A.; de Winter, R.J.; Minnema, M.C.; Cate, H.T.; Lijmer, J.G.; Adams, R.; Peters, R.J.; van Deventer, S.J. Procoagulant and proinflammatory activity in acute coronary syndromes. Cardiovasc. Res. 1998, 40, 389–395. [Google Scholar] [CrossRef]
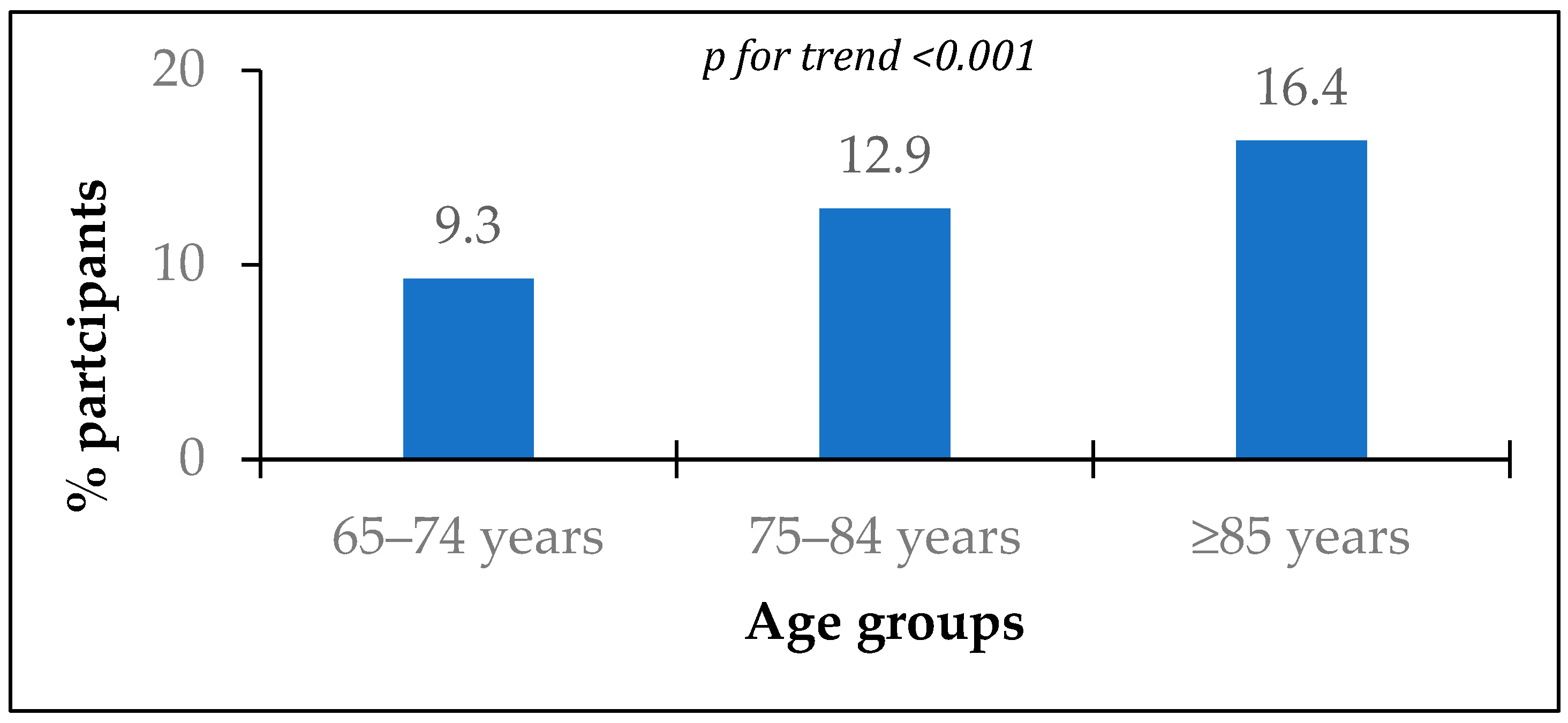
| Parameter | All Patients (n = 4295) | MI History | p | |
|---|---|---|---|---|
| Yes (n = 540) | No (n = 3755) | |||
| Age, years (M ± SD) | 78.3 ± 8.4 | 80.3 ± 8.3 | 78.0 ± 8.4 | <0.001 |
| Male sex, % | 29.7 | 59.8 | 28.2 | <0.001 |
| Height, m (M ± SD) | 1.63 ± 0.09 | 1.63 ± 0.09 | 1.63 ± 0.09 | 0.896 |
| Weight, kg (M ± SD) | 73.9 ± 14.3 | 74.5 ± 14.9 | 73.8 ± 14.1 | 0.178 |
| Body mass index, kg/m2 (M ± SD) | 27.9 ± 5.0 | 28.0 ± 5.2 | 27.9 ± 4.9 | 0.759 |
| Body mass, % | ||||
| Deficit | 1.3 | 2.6 | 1.1 | 0.004 |
| Normal | 27.6 | 26.9 | 27.7 | 0.688 |
| Overweight | 40.9 | 39.5 | 41.1 | 0.487 |
| Obesity | 30.2 | 31.0 | 30.1 | 0.668 |
| Degrees of obesity, % (n = 1264) | ||||
| I | 72.2 | 73.9 | 71.9 | 0.585 |
| II | 21.6 | 19.4 | 22.0 | 0.451 |
| III | 6.2 | 6.7 | 6.1 | 0.783 |
| Systolic BP, mm Hg (M ± SD) | 136.1 ± 16.5 | 136.7 ± 17.7 | 136.0 ± 16.3 | 0.219 |
| Systolic BP ≥ 140 mm Hg, % | 38.9 | 44.3 | 38.1 | 0.007 |
| Diastolic BP, mm Hg (M ± SD) | 80.2 ± 9.5 | 80.5 ± 10.1 | 80.2 ± 9.5 | 0.367 |
| Diastolic BP ≥ 90 mm Hg, % | 18.1 | 22.2 | 17.5 | 0.008 |
| Pulse BP, mm Hg (M ± SD) | 55.9 ± 13.0 | 56.3 ± 13.9 | 55.8 ± 12.8 | 0.891 |
| Heart rate, beats/min (M ± SD) | 72.7 ± 8.6 | 73.0 ± 8.9 | 72.6 ± 8.5 | 0.620 |
| Heart rate > 80 beats/min, % | 13.8 | 15.4 | 13.5 | 0.238 |
| Parameter | All Patients (n = 4295) | MI History | p | ||
|---|---|---|---|---|---|
| Yes (n = 540) | No (n = 3755) | ||||
| SPPB, points * | 6 (3; 9) | 5 (2; 8) | 6 (3; 9) | <0.001 | |
| Hand grip strength, kg * | Men | 22 (16; 30) | 20 (14; 28) | 22 (16; 30) | 0.004 |
| Women | 16 (11;21) | 12 (8;19) | 16 (11;21) | <0.001 | |
| Decrease in hand grip strength, % | 70.8 | 80.2 | 69.4 | <0.001 | |
| Gait velocity, m/s * | 0.60 (0.46;0.83) | 0.57 (0.44;0.80) | 0.63 (0.47;0.83) | 0.001 | |
| Decrease in gait velocity, % | 56.1 | 60.9 | 55.4 | 0.024 | |
| Mini-Cog test, points | <0.001 | ||||
| Me (25%; 75%) | 3 (2; 4) | 3 (1; 4) | 3 (2; 4) | ||
| M ± SD | 2.89 ± 1.51 | 2.61 ± 1.50 | 2.94 ± 1.50 | ||
| Geriatric Depression Scale, points * | 4 (2; 8) | 6 (3; 10) | 4 (2; 7) | <0.001 | |
| Basic Functional Activity Scale (Barthel Index), points | <0.001 | ||||
| Me (25%; 75%) | 95 (85; 100) | 95 (80; 100) | 95 (85; 100) | ||
| M ± SD | 88.5 ± 17.8 | 84.3 ± 20.6 | 89.1 ± 17.3 | ||
| Lawton Instrumental Activities of Daily Living Scale, points * | 7 (5; 8) | 6 (4; 8) | 7 (5; 8) | <0.001 | |
| MNA (Mini Nutritional Assessment) short-form, points * | 12 (10; 13) | 11.5 (10; 13) | 12 (10; 13) | <0.001 | |
| Quality of life self-assessment according to VAS, points * | 7 (5; 8) | 6 (5; 7) | 7 (5; 8) | <0.001 | |
| Health status self-assessment according to VAS, points | <0.001 | ||||
| Me (25%; 75%) | 5 (5; 7) | 5 (4; 6) | 5 (5; 7) | ||
| M ± SD | 5.6 ± 2.0 | 4.9 ± 2.0 | 5.7 ± 2.0 | ||
| Pain self-assessment according to VAS, points * | 3 (0; 5) | 4 (0; 5) | 3 (0; 5) | <0.001 | |
| Pain during the previous week self-assessment according to VAS, points * | 4 (2; 6) | 5 (3; 7) | 4 (2; 6) | <0.001 | |
| Parameter | MI History | p | |
|---|---|---|---|
| Yes (n = 540) | No (n = 3755) | ||
| Use of assistive devices, % | 95.9 | 92.1 | 0.001 |
| Number of assistive devices, Me (25%; 75%) | 3 (2; 3) | 2 (1; 3) | <0.001 |
| Glasses/lens, % | 82.6 | 78.8 | 0.045 |
| Hearing aid, % | 13.3 | 6.4 | <0.001 |
| Dentures, % | 66.9 | 58.7 | <0.001 |
| Walking stick, % | 45.6 | 30.8 | <0.001 |
| Crutches, % | 3.7 | 2.2 | 0.034 |
| Walkers, % | 6.9 | 3.6 | <0.001 |
| Wheelchair, % | 1.9 | 1.8 | 0.948 |
| Orthopedic shoes, % | 4.6 | 5.1 | 0.667 |
| Orthopedic insoles, % | 9.8 | 10.2 | 0.780 |
| Orthopedic corset, % | 5.9 | 4.5 | 0.152 |
| Urological pads, % | 16.9 | 13.3 | 0.027 |
| Diapers/underpads, % | 9.3 | 5.3 | <0.001 |
| Mobility aids (walking stick, crutches, walkers, wheelchair), % | 50.6 | 34.0 | <0.001 |
| Absorbent clothing for urinary/fecal incontinence (urological pads, diapers), % | 22.0 | 16.2 | 0.001 |
| Parameter | N | MI History | p | |
|---|---|---|---|---|
| Yes (n = 540) | No (n = 3755) | |||
| Chronic pain syndrome | 4295 | 90.6 | 86.7 | 0.013 |
| Impairment in basic activities of daily living | 4295 | 71.9 | 59.6 | <0.001 |
| Impairment in instrumental activities of daily living | 4295 | 71.9 | 51.7 | <0.001 |
| Frailty | 4295 | 70.2 | 61.6 | <0.001 |
| Cognitive impairment | 3537 | 69.1 | 59.5 | <0.001 |
| Depression | 4271 | 57.9 | 46.7 | <0.001 |
| Urinary incontinence | 4295 | 51.3 | 44.4 | 0.002 |
| Falls during the previous year | 4289 | 38.7 | 29.1 | <0.001 |
| Sensory deficit (any) | 4294 | 19.1 | 14.9 | 0.011 |
| Hearing loss | 4292 | 14.6 | 11.4 | 0.032 |
| Malnutrition | 4295 | 9.6 | 5.4 | <0.001 |
| Visual impairment | 4294 | 6.5 | 4.8 | 0.099 |
| Orthostatic hypotension | 3975 | 5.8 | 8.2 | 0.059 |
| Fecal incontinence | 4295 | 5.6 | 4.8 | 0.427 |
| Bedsores | 4293 | 3.9 | 2.0 | 0.006 |
| Geriatric Syndromes | N | OR | 95% CI | p |
|---|---|---|---|---|
| Urinary incontinence | 4295 | 1.32 | 1.10–1.58 | 0.002 |
| Sensory deficit | 4294 | 1.33 | 1.02–1.72 | 0.032 |
| Hearing loss | 4292 | 1.35 | 1.07–1.70 | 0.012 |
| Chronic pain syndrome | 4295 | 1.47 | 1.08–1.99 | 0.013 |
| Frailty | 4295 | 1.47 | 1.20–1.78 | <0.001 |
| Cognitive impairment | 3537 | 1.52 | 1.24–1.87 | <0.001 |
| Falls during the previous year | 4289 | 1.53 | 1.27–1.85 | <0.001 |
| Depression | 4271 | 1.57 | 1.31–1.89 | <0.001 |
| Impairment in basic activities of daily living | 4295 | 1.73 | 1.42–2.11 | <0.001 |
| Malnutrition | 4295 | 1.87 | 1.36–2.58 | <0.001 |
| Bedsores | 4293 | 1.96 | 1.20–3.20 | 0.007 |
| Impairment in instrumental activities of daily living | 4295 | 2.38 | 1.95–2.90 | <0.001 |
| Predictors | OR | 95% CI | p |
|---|---|---|---|
| Impairment in basic activities of daily living | 1.28 | 1.00–1.63 | 0.047 |
| Depression | 1.28 | 1.03–1.58 | 0.026 |
| Falls during the previous year | 1.45 | 1.18–1.79 | <0.001 |
| Impairment in instrumental activities of daily living | 1.86 | 1.47–2.36 | <0.001 |
| Male sex | 1.89 | 1.53–2.33 | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zakiev, V.; Vorobyeva, N.; Malaya, I.; Kotovskaya, Y.; Tkacheva, O. The Association of Myocardial Infarction History and Geriatric Syndromes in the Elderly: Data from the Cross-Sectional Study EVKALIPT. J. Clin. Med. 2024, 13, 6420. https://doi.org/10.3390/jcm13216420
Zakiev V, Vorobyeva N, Malaya I, Kotovskaya Y, Tkacheva O. The Association of Myocardial Infarction History and Geriatric Syndromes in the Elderly: Data from the Cross-Sectional Study EVKALIPT. Journal of Clinical Medicine. 2024; 13(21):6420. https://doi.org/10.3390/jcm13216420
Chicago/Turabian StyleZakiev, Vadim, Natalya Vorobyeva, Irina Malaya, Yulia Kotovskaya, and Olga Tkacheva. 2024. "The Association of Myocardial Infarction History and Geriatric Syndromes in the Elderly: Data from the Cross-Sectional Study EVKALIPT" Journal of Clinical Medicine 13, no. 21: 6420. https://doi.org/10.3390/jcm13216420
APA StyleZakiev, V., Vorobyeva, N., Malaya, I., Kotovskaya, Y., & Tkacheva, O. (2024). The Association of Myocardial Infarction History and Geriatric Syndromes in the Elderly: Data from the Cross-Sectional Study EVKALIPT. Journal of Clinical Medicine, 13(21), 6420. https://doi.org/10.3390/jcm13216420






