Carotid–Femoral Pulse Wave Velocity Could Be a Marker to Predict Cardiovascular and All-Cause Mortality of Hemodialysis Patients
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Anthropometric Analysis
2.3. Biochemical Investigations
2.4. Carotid–Femoral Pulse Wave Velocity Measurements
2.5. Follow-Up and Endpoints
2.6. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bloembergen, W.E.; Port, F.K.; Mauger, E.A.; Wolfe, R.A. Causes of death in dialysis patients: Racial and gender differences. J. Am. Soc. Nephrol. 1994, 5, 1231–1242. [Google Scholar] [CrossRef]
- Lindner, A.; Charra, B.; Sherrard, D.J.; Scribner, B.H. Accelerated atherosclerosis in prolonged maintenance hemodialysis. N. Engl. J. Med. 1974, 290, 697–701. [Google Scholar] [CrossRef] [PubMed]
- Cheung, A.K.; Sarnak, M.J.; Yan, G.; Berkoben, M.; Heyka, R.; Kaufman, A.; Lewis, J.; Rocco, M.; Toto, R.; Windus, D.; et al. Cardiac diseases in maintenance hemodialysis patients: Results of the HEMO Study. Kidney Int. 2004, 65, 2380–2389. [Google Scholar] [CrossRef] [PubMed]
- Foley, R.N.; Parfrey, P.S.; Sarnak, M.J. Clinical epidemiology of cardiovascular disease in chronic renal disease. Am. J. Kidney Dis. 1998, 32 (Suppl. S3), S112–S119. [Google Scholar] [CrossRef]
- Foley, R.N.; Parfrey, P.S.; Sarnak, M.J. Epidemiology of cardiovascular disease in chronic renal disease. J. Am. Soc. Nephrol. 1998, 9 (Suppl. S12), S16–S23. [Google Scholar] [CrossRef]
- Jankowski, J.; Floege, J.; Fliser, D.; Böhm, M.; Marx, N. Cardiovascular disease in chronic kidney disease. Circulation 2021, 143, 1157–1172. [Google Scholar] [CrossRef]
- Collins, A.J.; Foley, R.N.; Gilbertson, D.T.; Chen, S.C. United States Renal Data System public health surveillance of chronic kidney disease and end-stage renal disease. Kidney Int. Suppl. 2015, 5, 2–7. [Google Scholar] [CrossRef] [PubMed]
- Carrero, J.J.; de Jager, D.J.; Verduijn, M.; Ravani, P.; De Meester, J.; Heaf, J.G.; Finne, P.; Hoitsma, A.J.; Pascual, J.; Jarraya, F.; et al. Cardiovascular and noncardiovascular mortality among patients starting dialysis. Clin. J. Am. Soc. Nephrol. 2011, 6, 1722–1730. [Google Scholar] [CrossRef]
- Guérin, A.P.; Pannier, B.; Métivier, F.; Marchais, S.J.; London, G.M. Assessment and significance of arterial stiffness in patients with chronic kidney disease. Curr. Opin. Nephrol. Hypertens. 2008, 17, 635–641. [Google Scholar] [CrossRef]
- Angoff, R.; Mosarla, R.C.; Tsao, C.W. Aortic stiffness: Epidemiology, risk factors, and relevant biomarkers. Front. Cardiovasc. Med. 2021, 8, 709396. [Google Scholar] [CrossRef]
- Cavalcante, J.L.; Lima, J.A.; Redheuil, A.; Al-Mallah, M.H. Aortic stiffness: Current understanding and future directions. J. Am. Coll. Cardiol. 2011, 57, 1511–1522. [Google Scholar] [CrossRef]
- Fortier, C.; Mac-Way, F.; Desmeules, S.; Marquis, K.; De Serres, S.A.; Lebel, M.; Boutouyrie, P.; Agharazii, M. Aortic-brachial stiffness mismatch and mortality in dialysis population. Hypertension 2015, 65, 378–384. [Google Scholar] [CrossRef] [PubMed]
- Karras, A.; Haymann, J.P.; Bozec, E.; Metzger, M.; Jacquot, C.; Maruani, G.; Houillier, P.; Froissart, M.; Stengel, B.; Guardiola, P.; et al. Large artery stiffening and remodeling are independently associated with all-cause mortality and cardiovascular events in chronic kidney disease. Hypertension 2012, 60, 1451–1457. [Google Scholar] [CrossRef]
- Pannier, B.; Guérin, A.P.; Marchais, S.J.; Safar, M.E.; London, G.M.J.H. Stiffness of capacitive and conduit arteries: Prognostic significance for end-stage renal disease patients. Hypertension 2005, 45, 592–596. [Google Scholar] [CrossRef] [PubMed]
- Sarafidis, P.A.; Loutradis, C.; Karpetas, A.; Tzanis, G.; Piperidou, A.; Koutroumpas, G.; Raptis, V.; Syrgkanis, C.; Liakopoulos, V.; Efstratiadis, G.; et al. Ambulatory pulse wave velocity is a stronger predictor of cardiovascular events and all-cause mortality than office and ambulatory blood pressure in hemodialysis patients. Hypertension 2017, 70, 148–157. [Google Scholar] [CrossRef] [PubMed]
- Reference Values for Arterial Stiffness ‘Collaboration. Determinants of pulse wave velocity in healthy people and in the presence of cardiovascular risk factors: ’establishing normal and reference values’. Eur. Heart J. 2010, 31, 2338–2350. [Google Scholar] [CrossRef]
- Laurent, S.; Boutouyrie, P.; Asmar, R.; Gautier, I.; Laloux, B.; Guize, L.; Ducimetiere, P.; Benetos, A. Aortic stiffness is an independent predictor of all-cause and cardiovascular mortality in hypertensive patients. Hypertension 2001, 37, 1236–1341. [Google Scholar] [CrossRef] [PubMed]
- Ben-Shlomo, Y.; Spears, M.; Boustred, C.; May, M.; Anderson, S.G.; Benjamin, E.J.; Boutouyrie, P.; Cameron, J.; Chen, C.H.; Cruickshank, J.K.; et al. Aortic pulse wave velocity improves cardiovascular event prediction: An individual participant meta-analysis of prospective observational data from 17,635 subjects. J. Am. Coll. Cardiol. 2014, 63, 636–646. [Google Scholar] [CrossRef]
- Unger, T.; Borghi, C.; Charchar, F.; Khan, N.A.; Poulter, N.R.; Prabhakaran, D.; Ramirez, A.; Schlaich, M.; Stergiou, G.S.; Tomaszewski, M.; et al. 2020 International society of hypertension global hypertension practice guidelines. Hypertension 2020, 75, 1334–1357. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.S.; Wang, C.H.; Lai, Y.H.; Kuo, C.H.; Lin, Y.L.; Hsu, B.G.; Tsai, J.P. Serum malondialdehyde-modified low-density lipoprotein is a risk factor for central arterial stiffness in maintenance hemodialysis patients. Nutrients 2020, 12, 2160. [Google Scholar] [CrossRef]
- Chiu, L.T.; Hung, C.D.; Lin, L.; Lin, Y.L.; Hsu, B.G. Serum fibroblast growth factor 21 level is associated with aortic stiffness in patients on maintenance hemodialysis. Int. J. Hypertens. 2022, 2022, 7098458. [Google Scholar] [CrossRef]
- Williams, B.; Mancia, G.; Spiering, W.; Agabiti Rosei, E.; Azizi, M.; Burnier, M.; Clement, D.L.; Coca, A.; de Simone, G.; Dominiczak, A.; et al. 2018 ESC/ESH guidelines for the management of arterial hypertension. Eur. Heart J. 2018, 39, 3021–3104. [Google Scholar] [CrossRef]
- Lioufas, N.; Hawley, C.M.; Cameron, J.D.; Toussaint, N.D. Chronic kidney disease and pulse wave velocity: A narrative review. Int. J. Hypertens. 2019, 2019, 9189362. [Google Scholar] [CrossRef]
- Tsai, J.P.; Hsu, B.G. Arterial stiffness: A brief review. Tzu Chi Med. J. 2020, 33, 115–121. [Google Scholar] [PubMed]
- Zhang, Y.; Lacolley, P.; Protogerou, A.D.; Safar, M.E. Arterial stiffness in hypertension and function of large arteries. Am. J. Hypertens. 2020, 33, 291–296. [Google Scholar] [CrossRef]
- Inserra, F.; Forcada, P.; Castellaro, A.; Castellaro, C. Chronic kidney disease and arterial stiffness: A two-way path. Front. Med. 2021, 8, 765924. [Google Scholar] [CrossRef]
- Blacher, J.; Safar, M.E.; Guerin, A.P.; Pannier, B.; Marchais, S.J.; London, G.M. Aortic pulse wave velocity index and mortality in end-stage renal disease. Kidney Int. 2003, 63, 1852–1860. [Google Scholar] [CrossRef] [PubMed]
- Sutton-Tyrrell, K.; Najjar, S.S.; Boudreau, R.M.; Venkitachalam, L.; Kupelian, V.; Simonsick, E.M.; Havlik, R.; Lakatta, E.G.; Spurgeon, H.; Kritchevsky, S.; et al. Elevated aortic pulse wave velocity, a marker of arterial stiffness, predicts cardiovascular events in well-functioning older adults. Circulation 2005, 111, 3384–3390. [Google Scholar] [CrossRef]
- Zhong, Q.; Hu, M.J.; Cui, Y.J.; Liang, L.; Zhou, M.M.; Yang, Y.W.; Huang, F. Carotid-femoral pulse wave velocity in the prediction of cardiovascular events and mortality: An updated systematic review and meta-analysis. Angiology 2018, 69, 617–629. [Google Scholar] [CrossRef]
- Vlachopoulos, C.; Aznaouridis, K.; Stefanadis, C. Prediction of cardiovascular events and all-cause mortality with arterial stiffness: A systematic review and meta-analysis. J. Am. Coll. Cardiol. 2010, 55, 1318–1327. [Google Scholar] [CrossRef] [PubMed]
- Sequí-Domínguez, I.; Cavero-Redondo, I.; Álvarez-Bueno, C.; Pozuelo-Carrascosa, D.P.; Nuñez de Arenas-Arroyo, S.; Martínez-Vizcaíno, V. Accuracy of pulse wave velocity predicting cardiovascular and all-cause mortality. A systematic review and meta-analysis. J. Clin. Med. 2020, 9, 2080. [Google Scholar] [CrossRef] [PubMed]
- Townsend, R.R.; Anderson, A.H.; Chirinos, J.A.; Feldman, H.I.; Grunwald, J.E.; Nessel, L.; Roy, J.; Weir, M.R.; Wright, J.T., Jr.; Bansal, N.; et al. Association of pulse wave velocity with chronic kidney disease progression and mortality: Findings from the CRIC study (Chronic Renal Insufficiency Cohort). Hypertension 2018, 71, 1101–1107. [Google Scholar] [CrossRef] [PubMed]
- Chirinos, J.A.; Khan, A.; Bansal, N.; Dries, D.L.; Feldman, H.I.; Ford, V.; Anderson, A.H.; Kallem, R.; Lash, J.P.; Ojo, A.; et al. Arterial stiffness, central pressures, and incident hospitalized heart failure in the chronic renal insufficiency cohort study. Circ. Heart Fail. 2014, 7, 709–716. [Google Scholar] [CrossRef]
- Verbeke, F.; Van Biesen, W.; Honkanen, E.; Wikström, B.; Jensen, P.B.; Krzesinski, J.M.; Rasmussen, M.; Vanholder, R.; Rensma, P.L.; CORD Study Investigators. Prognostic value of aortic stiffness and calcification for cardiovascular events and mortality in dialysis patients: Outcome of the calcification outcome in renal disease (CORD) study. Clin. J. Am. Soc. Nephrol. 2011, 6, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Blacher, J.; Guerin, A.P.; Pannier, B.; Marchais, S.J.; Safar, M.E.; London, G.M. Impact of aortic stiffness on survival in end-stage renal disease. Circulation 1999, 99, 2434–2439. [Google Scholar] [CrossRef]
- Tripepi, G.; Agharazii, M.; Pannier, B.; D’Arrigo, G.; Mallamaci, F.; Zoccali, C.; London, G. Pulse wave velocity and prognosis in end-stage kidney disease. Hypertension 2018, 71, 1126–1132. [Google Scholar] [CrossRef]
- Guerin, A.P.; Blacher, J.; Pannier, B.; Marchais, S.J.; Safar, M.E.; London, G.M. Impact of aortic stiffness attenuation on survival of patients in end-stage renal failure. Circulation 2001, 103, 987–992. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Zhao, S. Risk factors for mortality in patients undergoing hemodialysis: A systematic review and meta-analysis. Int. J. Cardiol. 2017, 238, 151–158. [Google Scholar] [CrossRef]
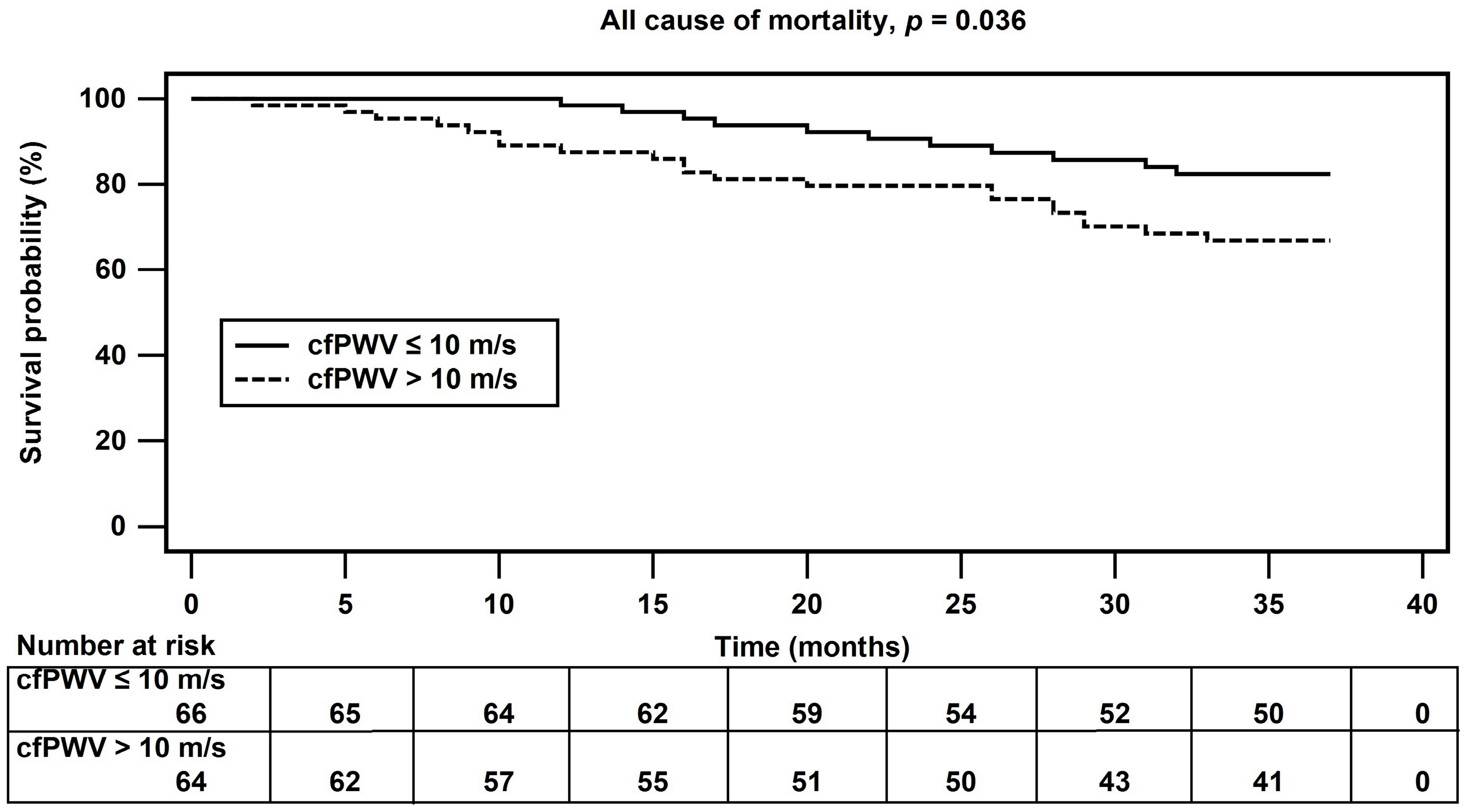
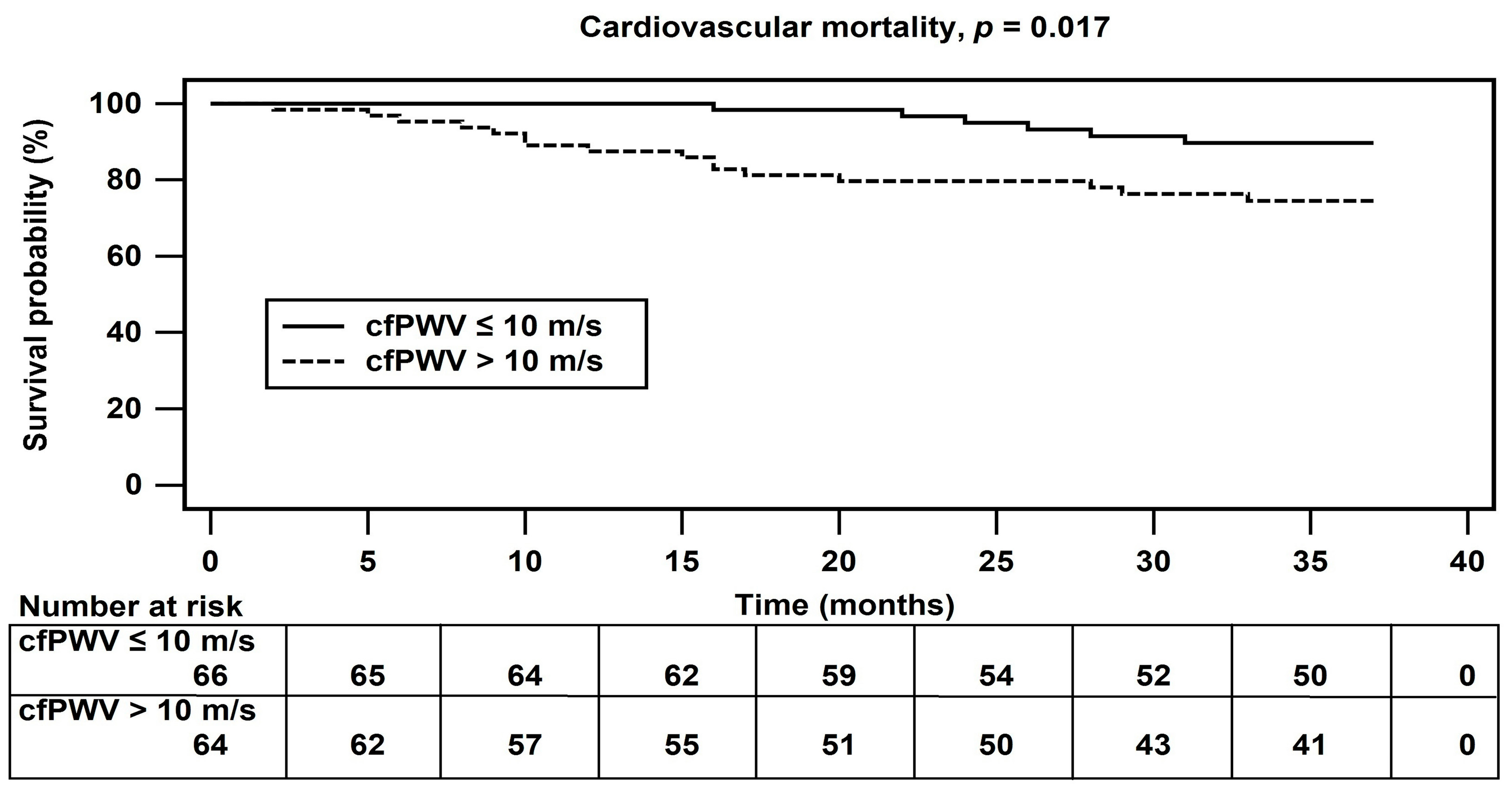
| Variables | All Participants (n = 130) | Control Group (n = 66) | Arterial Stiffness Group (n = 64) | p Value |
|---|---|---|---|---|
| Age (years) | 63.17 ± 12.94 | 60.95 ± 12.97 | 65.45 ± 12.59 | 0.047 * |
| HD vintage (months) | 55.92 (23.40–115.68) | 60.06 (21.99–136.80) | 54.54 (24.99–99.30) | 0.325 |
| Height (cm) | 160.47 ± 8.19 | 160.23 ± 8.46 | 160.72 ± 7.97 | 0.734 |
| Pre-HD body weight (kg) | 64.49 ± 14.63 | 63.76 ± 15.38 | 65.24 ± 13.90 | 0.566 |
| Post-HD body weight (kg) | 62.32 ± 14.12 | 61.68 ± 14.74 | 62.98 ± 13.54 | 0.601 |
| Body mass index (kg/m2) | 25.11 ± 4.81 | 24.88 ± 5.07 | 25.35 ± 4.56 | 0.573 |
| Carotid-femoral PWV (m/s) | 10.43 ± 3.28 | 7.82 ± 1.23 | 13.13 ± 2.44 | <0.001 * |
| Systolic blood pressure (mmHg) | 143.27 ± 26.58 | 136.58 ± 26.31 | 150.17 ± 25.24 | 0.003 * |
| Diastolic blood pressure (mmHg) | 77.47 ± 16.35 | 76.68 ± 15.48 | 78.28 ± 17.29 | 0.579 |
| Hemoglobin (g/dL) | 10.45 ± 1.12 | 10.27 ± 1.21 | 10.63 ± 1.00 | 0.067 |
| Total cholesterol (mg/dL) | 145.83 ± 34.61 | 149.61 ± 38.78 | 141.94 ± 29.51 | 0.208 |
| Triglyceride (mg/dL) | 118.00 (87.00–185.50) | 113.00 (84.00–205.50) | 124.50 (89.25–175.75) | 0.872 |
| Albumin (g/dL) | 4.10 (3.90–4.40) | 4.20 (3.98–4.40) | 4.10 (3.90–4.30) | 0.214 |
| Glucose (mg/dL) | 131.00 (108.50–169.00) | 126.50 (103.00–148.75) | 138.00 (114.25–185.00) | 0.020 * |
| Blood urea nitrogen (mg/dL) | 61.36 ± 13.80 | 61.27 ± 13.53 | 61.45 ± 14.17 | 0.941 |
| Creatinine (mg/dL) | 9.55 ± 2.04 | 9.76 ± 2.09 | 9.33 ± 1.99 | 0.239 |
| Total calcium (mg/dL) | 9.03 ± 0.79 | 8.94 ± 0.78 | 9.12 ± 0.80 | 0.209 |
| Phosphorus (mg/dL) | 4.72 ± 1.29 | 4.67 ± 1.33 | 4.77 ± 1.26 | 0.644 |
| iPTH (pg/mL) | 197.95 (69.38–440.68) | 245.95 (102.40–375.85) | 161.70 (57.28–463.85) | 0.299 |
| Urea reduction rate | 0.73 ± 0.04 | 0.73 ± 0.04 | 0.73 ± 0.04 | 0.987 |
| Kt/V (Gotch) | 1.34 ± 0.17 | 1.34 ± 0.17 | 1.34 ± 0.16 | 0.935 |
| Female, n (%) | 62 (47.7) | 33 (50.0) | 29 (45.3) | 0.593 |
| Diabetes mellitus, n (%) | 52 (40.0) | 13 (19.7) | 39 (60.9) | <0.001 * |
| Hypertension, n (%) | 69 (53.1) | 29 (43.9) | 40 (62.5) | 0.034 * |
| ARB, n (%) | 37 (28.5) | 18 (27.3) | 19 (29.7) | 0.760 |
| β-blocker, n (%) | 41 (31.5) | 20 (30.3) | 21 (32.8) | 0.758 |
| CCB, n (%) | 51 (39.2) | 29 (43.9) | 22 (34.4) | 0.264 |
| Statin, n (%) | 20 (15.4) | 6 (9.1) | 14 (21.9) | 0.043 * |
| Fibrate, n (%) | 16 (12.3) | 9 (13.6) | 7 (10.9) | 0.640 |
| Variables | Participants without Mortality (n = 98) | Participants with Mortality (n = 32) | p Value |
|---|---|---|---|
| Age (years) | 61.43 ± 12.95 | 68.50 ± 11.52 | 0.007 * |
| HD vintage (months) | 57.00 (21.69–125.10) | 51.48 (26.43–81.43) | 0.693 |
| Height (cm) | 161.08 ± 8.50 | 158.59 ± 6.96 | 0.136 |
| Pre-HD body weight (kg) | 65.59 ± 14.18 | 61.12 ± 15.69 | 0.134 |
| Post-HD body weight (kg) | 63.34 ± 13.71 | 59.18 ± 15.09 | 0.149 |
| Body mass index (kg/m2) | 25.30 ± 4.70 | 24.54 ± 5.18 | 0.440 |
| Carotid-femoral PWV (m/s) | 9.67 ± 2.69 | 12.75 ± 3.85 | <0.001 * |
| Systolic blood pressure (mmHg) | 142.64 ± 25.35 | 145.19 ± 30.39 | 0.640 |
| Diastolic blood pressure (mmHg) | 78.59 ± 16.51 | 74.03 ± 15.60 | 0.172 |
| Hemoglobin (g/dL) | 10.45 ± 1.15 | 10.45 ± 1.07 | 0.982 |
| Total cholesterol (mg/dL) | 149.03 ± 34.94 | 136.03 ± 32.13 | 0.065 |
| Triglyceride (mg/dL) | 119.50 (88.50–197.75) | 106.50 (87.00–140.00) | 0.218 |
| Albumin (g/dL) | 4.20 (4.00–4.50) | 4.10 (3.70–4.18) | 0.003 * |
| Glucose (mg/dL) | 128.50 (103.75–162.00) | 143.00 (120.75–184.00) | 0.060 |
| Blood urea nitrogen (mg/dL) | 62.12 ± 13.34 | 59.03 ± 15.08 | 0.273 |
| Creatinine (mg/dL) | 9.93 ± 1.94 | 8.38 ± 1.94 | <0.001 * |
| Total calcium (mg/dL) | 9.08 ± 0.79 | 8.89 ± 0.80 | 0.233 |
| Phosphorus (mg/dL) | 4.78 ± 1.29 | 4.53 ± 1.27 | 0.342 |
| iPTH (pg/mL) | 200.25 (73.05–416.65) | 189.40 (60.48–466.90) | 0.791 |
| Urea reduction rate | 0.73 ± 0.04 | 0.74 ± 0.05 | 0.380 |
| Kt/V (Gotch) | 1.33 ± 0.16 | 1.36 ± 0.19 | 0.346 |
| Female, n (%) | 48 (49.0) | 14 (43.8) | 0.607 |
| Diabetes mellitus, n (%) | 32 (32.7) | 20 (62.5) | 0.003 * |
| Hypertension, n (%) | 50 (51.0) | 19 (59.4) | 0.411 |
| ARB, n (%) | 28 (28.6) | 9 (28.1) | 0.961 |
| β-blocker, n (%) | 33 (33.7) | 8 (25.0) | 0.359 |
| CCB, n (%) | 42 (42.9) | 9 (28.1) | 0.138 |
| Statin, n (%) | 17 (17.3) | 3 (9.4) | 0.278 |
| Fibrate, n (%) | 12 (12.2) | 4 (12.5) | 0.970 |
| Carotid-Femoral PWV (m/s) | Unadjusted | Model 1 | Model 2 | Model 3 | ||||
|---|---|---|---|---|---|---|---|---|
| HR (95% CI) | p Value | HR (95% CI) | p Value | HR (95% CI) | p Value | HR (95% CI) | p Value | |
| Per 1 m/s carotid-femoral PWV increase | 1.267 (1.144–1.403) | <0.001 * | 1.241 (1.120–1.377) | <0.001 * | 1.179 (1.044–1.332) | 0.008 * | 1.170 (1.033–1.326) | 0.013 * |
| Variables | Participants without CV Mortality (n = 108) | Participants with CV Mortality (n = 22) | p Value |
|---|---|---|---|
| Age (years) | 62.39 ± 13.37 | 67.00 ± 9.95 | 0.128 |
| HD vintage (months) | 56.52 (22.23–123.60) | 51.48 (27.69–81.21) | 0.675 |
| Height (cm) | 160.42 ± 8.64 | 160.73 ± 5.62 | 0.872 |
| Pre-HD body weight (kg) | 64.45 ± 14.34 | 64.68 ± 16.33 | 0.946 |
| Post-HD body weight (kg) | 62.26 ± 13.84 | 62.58 ± 15.75 | 0.924 |
| Body mass index (kg/m2) | 25.06 ± 4.65 | 25.37 ± 5.64 | 0.787 |
| Carotid-femoral PWV (m/s) | 9.75 ± 2.78 | 13.78 ± 3.56 | <0.001 * |
| Systolic blood pressure (mmHg) | 142.87 ± 26.12 | 145.23 ± 29.30 | 0.706 |
| Diastolic blood pressure (mmHg) | 77.96 ± 16.47 | 75.04 ± 15.90 | 0.448 |
| Hemoglobin (g/dL) | 10.46 ± 1.12 | 10.41 ± 1.18 | 0.850 |
| Total cholesterol (mg/dL) | 147.36 ± 34.74 | 138.32 ± 33.75 | 0.266 |
| Triglyceride (mg/dL) | 119.50 (87.75–195.00) | 106.50 (71.75–160.75) | 0.290 |
| Albumin (g/dL) | 4.10 (3.93–4.40) | 4.10 (3.70–4.20) | 0.079 |
| Glucose (mg/dL) | 128.50 (104.25–162.00) | 148.50 (124.50–182.00) | 0.059 |
| Blood urea nitrogen (mg/dL) | 61.56 ± 13.68 | 60.41 ± 14.66 | 0.724 |
| Creatinine (mg/dL) | 9.74 ± 2.02 | 8.63 ± 1.96 | 0.020 * |
| Total calcium (mg/dL) | 9.06 ± 0.79 | 8.90 ± 0.80 | 0.406 |
| Phosphorus (mg/dL) | 4.71 ± 1.29 | 4.75 ± 1.33 | 0.896 |
| iPTH (pg/mL) | 195.20 (57.53–401.13) | 230.65 (88.10–577.33) | 0.203 |
| Urea reduction rate | 0.74 ± 0.04 | 0.73 ± 0.05 | 0.445 |
| Kt/V (Gotch) | 1.34 ± 0.16 | 1.31 ± 0.18 | 0.454 |
| Female, n (%) | 54 (50.0) | 8 (36.4) | 0.243 |
| Diabetes mellitus, n (%) | 36 (33.3) | 16 (72.7) | 0.001 * |
| Hypertension, n (%) | 54 (50.0) | 15 (68.2) | 0.119 |
| ARB, n (%) | 31 (28.7) | 6 (27.3) | 0.892 |
| β-blocker, n (%) | 33 (30.6) | 8 (36.4) | 0.593 |
| CCB, n (%) | 44 (40.7) | 7 (31.8) | 0.435 |
| Statin, n (%) | 17 (15.7) | 3 (13.6) | 0.803 |
| Fibrate, n (%) | 14 (13.0) | 2 (9.1) | 0.614 |
| Carotid-Femoral PWV (m/s) | Unadjusted | Model 1 | Model 2 | Model 3 | ||||
|---|---|---|---|---|---|---|---|---|
| HR (95% CI) | p Value | HR (95% CI) | p Value | HR (95% CI) | p Value | HR (95% CI) | p Value | |
| Per 1 m/s carotid-femoral PWV increase | 1.390 (1.234–1.567) | <0.001 * | 1.396 (1.227–1.588 | <0.001 * | 1.336 (1.149–1.552) | <0.001 * | 1.317 (1.130–1.534) | <0.001 * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ng, X.-N.; Tsai, J.-P.; Wang, C.-H.; Hsu, B.-G. Carotid–Femoral Pulse Wave Velocity Could Be a Marker to Predict Cardiovascular and All-Cause Mortality of Hemodialysis Patients. J. Clin. Med. 2023, 12, 2509. https://doi.org/10.3390/jcm12072509
Ng X-N, Tsai J-P, Wang C-H, Hsu B-G. Carotid–Femoral Pulse Wave Velocity Could Be a Marker to Predict Cardiovascular and All-Cause Mortality of Hemodialysis Patients. Journal of Clinical Medicine. 2023; 12(7):2509. https://doi.org/10.3390/jcm12072509
Chicago/Turabian StyleNg, Xin-Ning, Jen-Pi Tsai, Chih-Hsien Wang, and Bang-Gee Hsu. 2023. "Carotid–Femoral Pulse Wave Velocity Could Be a Marker to Predict Cardiovascular and All-Cause Mortality of Hemodialysis Patients" Journal of Clinical Medicine 12, no. 7: 2509. https://doi.org/10.3390/jcm12072509
APA StyleNg, X.-N., Tsai, J.-P., Wang, C.-H., & Hsu, B.-G. (2023). Carotid–Femoral Pulse Wave Velocity Could Be a Marker to Predict Cardiovascular and All-Cause Mortality of Hemodialysis Patients. Journal of Clinical Medicine, 12(7), 2509. https://doi.org/10.3390/jcm12072509









