Serum Autoantibodies in Patients with Dry and Wet Age-Related Macular Degeneration
Abstract
1. Introduction
2. Materials and Methods
2.1. Seldi-TOF Analysis
2.2. Protagen Arrays
2.3. Analysis
2.4. Antigen Microarrays
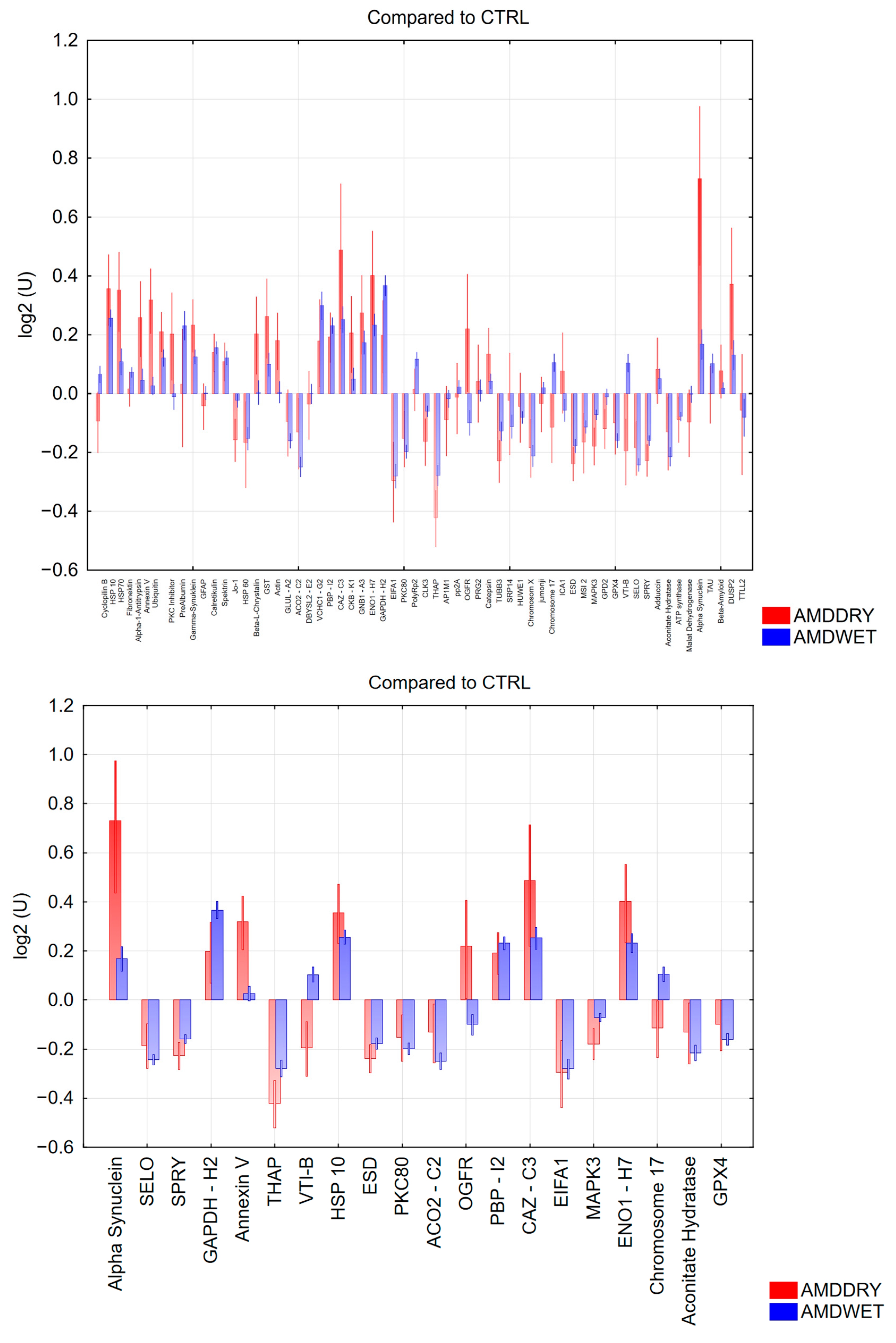
3. Results
Comparison of Immunoreactivities in Dry and Wet AMD
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mitchell, P.; Liew, G.; Gopinath, B.; Wong, T.Y. Age-related macular degeneration. Lancet 2018, 392, 1147–1159. [Google Scholar] [CrossRef] [PubMed]
- Ferris, F.L., 3rd; Wilkinson, C.P.; Bird, A.; Chakravarthy, U.; Chew, E.; Csaky, K.; Sadda, S.R. Clinical classification of age-related macular degeneration. Ophthalmology 2013, 120, 844–851. [Google Scholar] [CrossRef] [PubMed]
- Wong, W.L.; Su, X.; Li, X.; Cheung, C.M.; Klein, R.; Cheng, C.Y.; Wong, T.Y. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: A systematic review and meta-analysis. Lancet Glob. Health 2014, 2, e106–e116. [Google Scholar] [CrossRef] [PubMed]
- Ambati, J.; Ambati, B.K.; Yoo, S.H.; Ianchulev, S.; Adamis, A.P. Age-Related Macular Degeneration: Etiology, Pathogenesis, and Therapeutic Strategies. Surv. Ophthalmol. 2003, 48, 257–293. [Google Scholar] [CrossRef] [PubMed]
- Lim, L.S.; Mitchell, P.; Seddon, J.M.; Holz, F.G.; Wong, T.Y. Age-related macular degeneration. Lancet 2012, 379, 1728–1738. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, E.; Matsubara, J.; Cao, S.; Cui, J.; Forooghian, F. Antigen-specificity of antiretinal antibodies in patients with noninfectious uveitis. Can. J. Ophthalmol. 2017, 52, 463–467. [Google Scholar] [CrossRef] [PubMed]
- Heckenlively, J.R.; Fawzi, A.A.; Oversier, J.; Jordan, B.L.; Aptsiauri, N. Autoimmune retinopathy: Patients with antirecoverin immunoreactivity and panretinal degeneration. Arch. Ophthalmol. 2000, 118, 1525–1533. [Google Scholar] [CrossRef]
- Savchenko, M.S.; Bazhin, A.V.; Shifrina, O.N.; Demoura, S.A.; Kogan, E.A.; Chuchalin, A.G.; Philippov, P.P. Antirecoverin autoantibodies in the patient with non-small cell lung cancer but without cancer-associated retinopathy. Lung Cancer 2003, 41, 363–367. [Google Scholar] [CrossRef]
- Adamus, G.; Ren, G.; Weleber, R.G. Autoantibodies against retinal proteins in paraneoplastic and autoimmune retinopathy. BMC Ophthalmol. 2004, 4, 5. [Google Scholar] [CrossRef]
- Galbraith, G.M.; Emerson, D.; Fudenberg, H.H.; Gibbs, C.J.; Gajdusek, D.C. Antibodies to neurofilament protein in retinitis pigmentosa. J. Clin. Investig. 1986, 78, 865–869. [Google Scholar] [CrossRef]
- Galbraith, G.M.; Fudenberg, H.H. One subset of patients with retinitis pigmentosa has immunologic defects. Clin Immunol. Immunopathol. 1984, 31, 254–260. [Google Scholar] [CrossRef] [PubMed]
- Sim, S.S.; Wong, C.W.; Hoang, Q.V.; Lee, S.Y.; Wong, T.Y.; Cheung, C.M.G. Anti-retinal autoantibodies in myopic macular degeneration: A pilot study. Eye 2020, 35, 2254–2259. [Google Scholar] [CrossRef] [PubMed]
- Penfold, P.L.; Provis, J.M.; Furby, J.H.; Gatenby, P.A.; Billson, F.A. Autoantibodies to retinal astrocytes associated with age-related macular degeneration. Graefes. Arch. Clin. Exp. Ophthalmol. 1990, 228, 270–274. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Wu, L.; Pan, S.; Wu, D.Z. An immunologic study on age-related macular degeneration. Yan Ke Xue Bao 1993, 9, 113–120. [Google Scholar] [PubMed]
- Gurne, D.H.; Tso, M.O.; Edward, D.P.; Ripps, H. Antiretinal antibodies in serum of patients with age-related macular degeneration. Ophthalmology 1991, 98, 602–607. [Google Scholar] [CrossRef]
- Cherepanoff, S.; Mitchell, P.; Wang, J.J.; Gillies, M.C. Retinal autoantibody profile in early age-related macular degeneration: Preliminary findings from the Blue Mountains Eye Study. Clin. Exp. Ophthalmol. 2006, 34, 590–595. [Google Scholar] [CrossRef]
- Adamus, G.; Chew, E.Y.; Ferris, F.L.; Klein, M.L. Prevalence of anti-retinal autoantibodies in different stages of Age-related macular degeneration. BMC Ophthalmol. 2014, 14, 154. [Google Scholar] [CrossRef]
- Iannaccone, A.; Giorgianni, F.; New, D.D.; Hollingsworth, T.J.; Umfress, A.; Alhatem, A.; Neeli, I.; Lenchik, N.I.; Jennings, B.J.; Calzada, J.I.; et al. Circulating Autoantibodies in Age-Related Macular Degeneration Recognize Human Macular Tissue Antigens Implicated in Autophagy, Immunomodulation, and Protection from Oxidative Stress and Apoptosis. PLoS ONE 2015, 10, e0145323. [Google Scholar] [CrossRef]
- Patel, N.; Ohbayashi, M.; Nugent, A.K.; Ramchand, K.; Toda, M.; Chau, K.-Y.; Bunce, C.; Webster, A.; Bird, A.C.; Ono, S.J.; et al. Circulating anti-retinal antibodies as immune markers in age-related macular degeneration. Immunology 2005, 115, 422–430. [Google Scholar] [CrossRef]
- Adamus, G. Can innate and autoimmune reactivity forecast early and advance stages of age-related macular degeneration? Autoimmune Rev. 2017, 16, 231–236. [Google Scholar] [CrossRef]
- Camelo, S. Potential Sources and Roles of Adaptive Immunity in Age-Related Macular Degeneration: Shall We Rename AMD into Autoimmune Macular Disease? Autoimmune Dis. 2014, 2014, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.; Meer, S.G.; Miyagi, M.; Rayborn, M.E.; Hollyfield, J.G.; Crabb, J.W.; Salomon, R.G. Carboxyethylpyrrole protein adducts and autoantibodies, biomarkers for age-related macular degeneration. J. Biol. Chem. 2003, 278, 42027–42035. [Google Scholar] [CrossRef] [PubMed]
- Joachim, S.C.; Bruns, K.; Lackner, K.J.; Pfeiffer, N.; Grus, F.H. Analysis of IgG antibody patterns against retinal antigens and antibodies to α-crystallin, GFAP, and α-enolase in sera of patients with “wet” age-related macular degeneration. Graefe′s Arch. Clin. Exp. Ophthalmol. 2006, 245, 619–626. [Google Scholar] [CrossRef] [PubMed]
- Grus, F.H.; Joachim, S.C.; Pfeiffer, N. Analysis of complex autoantibody repertoires by surface-enhanced laser desorption/ionization-time of flight mass spectrometry. Proteomics 2003, 3, 957–961. [Google Scholar] [CrossRef] [PubMed]
- Salomon, O.; Moisseiev, J.; Rosenberg, N.; Vidne, O.; Yassur, I.; Zivelin, A.; Treister, G.; Steinberg, D.M.; Seligsohn, U. Analysis of genetic polymorphisms related to thrombosis and other risk factors in patients with retinal vein occlusion. Blood Coagul. Fibrinolysis 1998, 9, 617–622. [Google Scholar] [CrossRef]
- Quintana, F.J.; Farez, M.F.; Viglietta, V.; Iglesias, A.H.; Merbl, Y.; Izquierdo, G.; Lucas, M.; Basso, A.S.; Khoury, S.J.; Lucchinetti, C.F.; et al. Antigen microarrays identify unique serum autoantibody signatures in clinical and pathologic subtypes of multiple sclerosis. Proc. Natl. Acad. Sci. USA 2008, 105, 18889–18894. [Google Scholar] [CrossRef]
- Song, Q.; Liu, G.; Hu, S.; Zhang, Y.; Tao, Y.; Han, Y.; Zeng, H.; Huang, W.; Li, F.; Chen, P.; et al. Novel Autoimmune Hepatitis-Specific Autoantigens Identified Using Protein Microarray Technology. J. Proteome Res. 2009, 9, 30–39. [Google Scholar] [CrossRef]
- Grus, F.H.; Kramann, C.; Bozkurt, N.; Wiegel, N.; Bruns, K.; Lackner, N.; Pfeiffer, N. Effects of multipurpose contact lens solutions on the protein composition of the tear film. Contact Lens Anterior Eye 2005, 28, 103–112. [Google Scholar] [CrossRef]
- Saeed, A.I.; Sharov, V.; White, J.; Li, J.; Liang, W.; Bhagabati, N.; Braisted, J.; Klapa, M.; Currier, T.; Thiagarajan, M.; et al. TM4: A Free, Open-Source System for Microarray Data Management and Analysis. Biotechniques 2003, 34, 374–378. [Google Scholar] [CrossRef]
- Saeed, A.I.; Bhagabati, N.K.; Braisted, J.C.; Liang, W.; Sharov, V.; Howe, E.A.; Li, J.; Thiagarajan, M.; White, J.A.; Quackenbush, J. TM4 Microarray Software Suite. Methods Enzymol. 2006, 411, 134–193. [Google Scholar] [CrossRef]
- Hamelinck, D.; Zhou, H.; Li, L.; Verweij, C.; Dillon, D.; Feng, Z.; Costa, J.; Haab, B.B. Optimized Normalization for Antibody Microarrays and Application to Serum-Protein Profiling. Mol. Cell Proteom. 2005, 4, 773–784. [Google Scholar] [CrossRef] [PubMed]
- Lambert, N.G.; ElShelmani, H.; Singh, M.K.; Mansergh, F.C.; Wride, M.A.; Padilla, M.; Keegan, D.; Hogg, R.E.; Ambati, B.K. Risk factors and biomarkers of age-related macular degeneration. Prog. Retin. Eye Res. 2016, 54, 64–102. [Google Scholar] [CrossRef] [PubMed]
- Li, W.-H.; Zhao, J.; Li, H.-Y.; Liu, H.; Li, A.-L.; Wang, H.-X.; Wang, J.; He, K.; Liang, B.; Yu, M.; et al. Proteomics-based identification of autoantibodies in the sera of healthy Chinese individuals from Beijing. Proteomics 2006, 6, 4781–4789. [Google Scholar] [CrossRef] [PubMed]
- Avrameas, S. Natural autoantibodies: From ‘horror autotoxicus’ to ‘gnothi seauton’. Immunol. Today 1991, 12, 154–159. [Google Scholar]
- Nóbrega, A.; Haury, M.; Grandien, A.; Malanchère, E.; Sundblad, A.; Coutinho, A. Global analysis of antibody repertoires. II. Evidence for specificity, self-selection and the immunological “homunculus” of antibodies in normal serum. Eur. J. Immunol. 1993, 23, 2851–2859. [Google Scholar] [CrossRef]
- Joachim, S.C.; Grus, F.H.; Pfeiffer, N. Analysis of autoantibody repertoires in sera of patients with glaucoma. Eur. J. Ophthalmol. 2003, 13, 752–758. [Google Scholar] [CrossRef]
- Grus, F.H.; Joachim, S.C.; Bruns, K.; Lackner, K.J.; Pfeiffer, N.; Wax, M.B. Serum Autoantibodies to α-Fodrin Are Present in Glaucoma Patients from Germany and the United States. Investig. Opthalmol. Vis. Sci. 2006, 47, 968–976. [Google Scholar] [CrossRef]
- Grus, F.H.; Joachim, S.C.; Hoffmann, E.M.; Pfeiffer, N. Diagnosis of Glaucoma by Analysis of Autoantibody Repertoires. Investig. Opthalmol. Vis. Sci. 2003, 44, 2100. [Google Scholar]
- Grus, F.H.; Joachim, S.C.; Hoffmann, E.M.; Pfeiffer, N. Complex autoantibody repertoires in patients with glaucoma. Mol. Vis. 2004, 10, 132–137. [Google Scholar]
- Grus, F.H.; Augustin, A.J.; Pfeiffer, N. Analysis of Autoantibodies in Tears of Dry-Eye Patients. Investig. Opthalmol. Vis. Sci. 2002, 43, 64. [Google Scholar]
- Deane, K.D. Preclinical Rheumatoid Arthritis (Autoantibodies): An Updated Review. Curr. Rheumatol. Rep. 2014, 16, 419. [Google Scholar] [CrossRef]
- Romero-Vazquez, S.; Llorens, V.; Soler-Boronat, A.; Figueras-Roca, M.; Adan, A.; Molins, B. Interlink between Inflammation and Oxidative Stress in Age-Related Macular Degeneration: Role of Complement Factor H. Biomedicines 2021, 9, 763. [Google Scholar] [CrossRef] [PubMed]
- Baksi, S.; Tripathi, A.K.; Singh, N. Alpha-synuclein modulates retinal iron homeostasis by facilitating the uptake of transferrin-bound iron: Implications for visual manifestations of Parkinson′s disease. Free. Radic. Biol. Med. 2016, 97, 292–306. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Navarrete, G.C.; Martin-Nieto, J.; Esteve-Rudd, J.; Angulo, A.; Cuenca, N. Alpha synuclein gene expression profile in the retina of vertebrates. Mol. Vis. 2007, 13, 949–961. [Google Scholar] [PubMed]
- Pfeiffer, R.L.; Marc, R.E.; Jones, B.W. Persistent remodeling and neurodegeneration in late-stage retinal degeneration. Prog. Retin. Eye Res. 2020, 74, 100771. [Google Scholar] [CrossRef] [PubMed]
- Zhao, T.; Guo, X.; Sun, Y. Iron Accumulation and Lipid Peroxidation in the Aging Retina: Implication of Ferroptosis in Age-Related Macular Degeneration. Aging Dis. 2021, 12, 529–551. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Zheng, Y.; Wang, C.; Liu, Y. Glutathione depletion induces ferroptosis, autophagy, and premature cell senescence in retinal pigment epithelial cells. Cell Death Dis. 2018, 9, 1–15. [Google Scholar] [CrossRef]
- Iaccarino, L.; Ghirardello, A.; Canova, M.; Zen, M.; Bettio, S.; Nalotto, L.; Punzi, L.; Doria, A. Anti-annexins autoantibodies: Their role as biomarkers of autoimmune diseases. Autoimmune Rev. 2011, 10, 553–558. [Google Scholar] [CrossRef]
- Schloer, S.; Pajonczyk, D.; Rescher, U. Annexins in Translational Research: Hidden Treasures to Be Found. Int. J. Mol. Sci. 2018, 19, 1781. [Google Scholar] [CrossRef]
- Yu, C.; Muñoz, L.E.; Mallavarapu, M.; Herrmann, M.; Finnemann, S.C. Annexin A5 regulates surface αvβ5 integrin for retinal clearance phagocytosis. J. Cell Sci. 2019, 132, jcs232439. [Google Scholar] [CrossRef]
- Lederman, M.; Weiss, A.; Chowers, I. Association of Neovascular Age-Related Macular Degeneration with Specific Gene Expression Patterns in Peripheral White Blood Cells. Investig. Opthalmol. Vis. Sci. 2010, 51, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Winiarczyk, M.; Winiarczyk, D.; Michalak, K.; Kaarniranta, K.; Adaszek, Ł.; Winiarczyk, S.; Mackiewicz, J. Dysregulated Tear Film Proteins in Macular Edema Due to the Neovascular Age-Related Macular Degeneration Are Involved in the Regulation of Protein Clearance, Inflammation, and Neovascularization. J. Clin. Med. 2021, 10, 3060. [Google Scholar] [CrossRef] [PubMed]
- Cai, R.; Xue, W.; Liu, S.; Petersen, R.B.; Huang, K.; Zheng, L. Overexpression of glyceraldehyde 3-phosphate dehydrogenase prevents neurovascular degeneration after retinal injury. FASEB J. 2015, 29, 2749–2758. [Google Scholar] [CrossRef] [PubMed]
- Kanwar, M.; Kowluru, R.A. Role of Glyceraldehyde 3-Phosphate Dehydrogenase in the Development and Progression of Diabetic Retinopathy. Diabetes 2009, 58, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.-L.; Hung, C.-T.; Keller, J.J.; Lin, H.-C.; Wu, Y.-J. Proteomic analysis of retinal pigment epithelium cells after exposure to UVA radiation. BMC Ophthalmol. 2019, 19, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Pawestri, A.R.; Arjkongharn, N.; Suvannaboon, R.; Tuekprakhon, A.; Srimuninnimit, V.; Udompunthurak, S.; Atchaneeyasakul, L.-O.; Koolvisoot, A.; Trinavarat, A. Autoantibody profiles and clinical association in Thai patients with autoimmune retinopathy. Sci. Rep. 2021, 11, 15047. [Google Scholar] [CrossRef]
- McLaughlin, P.J.; Sassani, J.W.; Zagon, I.S. Dysregulation of the OGF-OGFr Pathway and Associated Diabetic Complications. J. Diabetes Clin. Res. 2021, 3, 64–67. [Google Scholar] [CrossRef]
- Alvarez-Rivera, F.; Serro, A.P.; Silva, D.; Concheiro, A.; Alvarez-Lorenzo, C. Hydrogels for diabetic eyes: Naltrexone loading, release profiles and cornea penetration. Mater. Sci. Eng. C 2019, 105, 110092. [Google Scholar] [CrossRef]
- Zagon, I.S.; Sassani, J.W.; McLaughlin, P.J. Adaptation of Homeostatic Ocular Surface Epithelium to Chronic Treatment with the Opioid Antagonist Naltrexone. Cornea 2006, 25, 821–829. [Google Scholar] [CrossRef]
- McLaughlin, P.J.; Zagon, I.S. Duration of opioid receptor blockade determines biotherapeutic response. Biochem. Pharmacol. 2015, 97, 236–246. [Google Scholar] [CrossRef]
- Shen, D.; Cao, X.; Zhao, L.; Tuo, J.; Wong, W.T.; Chan, C.-C. Naloxone Ameliorates Retinal Lesions inCcl2/Cx3cr1Double-Deficient Mice via Modulation of Microglia. Investig. Opthalmol. Vis. Sci. 2011, 52, 2897–2904. [Google Scholar] [CrossRef] [PubMed]
| Group | N (Sample Size) | Gender (m/f) | Age—Means | Age—Std.Dev. | Age—Minimum | Age—Maximum |
|---|---|---|---|---|---|---|
| CTRL | 10 | f | 71.8 | 9.36453 | 54 | 82 |
| CTRL | 11 | m | 74.5 | 10.98511 | 55 | 90 |
| 17 | 14 | f | 80.9 | 8.54722 | 64 | 97 |
| 17 | 6 | m | 73.2 | 7.19491 | 63 | 82 |
| 18 | 20 | f | 81.0 | 7.93339 | 59 | 92 |
| 18 | 9 | m | 81.4 | 5.11503 | 72 | 88 |
| ID | MW [kDa] | Protein Name UniProt | Protein Name | Abbreviation in Study |
|---|---|---|---|---|
| P62937 | 18.0 | Peptidyl-prolyl cis-trans isomerase A (Cyclophilin A) | Cyclophilin A human | Cyclophilin B |
| P61604 | 10.9 | 10 kDa heat shock protein, mitochondrial (Hsp10) | Chaperonin 10, Recombinant, Human | HSP 10 |
| P00441 | 15.9 | Superoxide dismutase [Cu-Zn] | Superoxide Dismutase from bovine erythrocytes | SOD |
| P02686 | 33.1 | Myelin basic protein (MBP); Isoform 1 | Myelin Basic Protein from bovine brain | MBP |
| P04792 | 22.8 | Heat shock protein beta-1 (Heat shock 27 kDa protein; Hsp27) | Hsp27 Protein—Low Endotoxin | HSP 27 |
| P08107 | 70.1 | Heat shock 70 kDa protein 1A/1B (Hsp70.1/Hsp70.2) | Heat Shock Protein 70 from bovine brain | HSP 70 |
| P02751 | 262.6 | Fibronectin; Isoform 1 | Fibronectin from human plasma | Fibronektin |
| P01009 | 46.7 | Alpha-1-antitrypsin | α1-Antitrypsin from human plasma | Alpha-1-Antitrypsin |
| P08758 | 35.9 | Annexin A5 | Annexin V from human placenta | Annexin V |
| Q14694 | 87.1 | Ubiquitin carboxyl-terminal hydrolase 10 (USP10) | Ubiquitin human | Ubiquitin |
| P49773 | 13.8 | Histidine triad nucleotide-binding protein 1 (Protein kinase C inhibitor 1) | Protein Kinase C Inhibitor, Myristoylated | PKC Inhibitor |
| P02766 | 15.9 | Transthyretin (Prealbumin) | Prealbumin from human plasma | PreAlbumin |
| O76070 | 13.3 | Gamma-synuclein | γ-Synuclein human | Gamma-Synuklein |
| P14136 | 49.9 | Glial fibrillary acidic protein (GFAP) | Anti-Glial Fibrillary Acidic Protein | GFAP |
| P27797 | 48.1 | Calreticulin | Calreticulin from bovine liver | Calretikulin |
| P02549 | 280.0 | Spectrin alpha chain, erythrocyte | Spectrin from human erythrocytes | Spektrin |
| P12081 | 57.4 | Histidine-tRNA ligase, cytoplasmic (JO-1) | JO-1 human | Jo-1 |
| P10809 | 61.1 | 60 kDa heat shock protein, mitochondrial (Hsp60) | HSP60 (human), (recombinant) | HSP 60 |
| P53674 | 28.0 | Beta-crystallin B1 | βL-Crystallin from bovine eye lens | Beta-L-Chrystalin |
| P09211 | 23.4 | Glutathione S-transferase P | Glutathione S-Transferase from bovine liver | GST |
| P68133 | 42.1 | Actin, alpha skeletal muscle | Actin from bovine muscle | Actin |
| P15104 | 42.1 | Glutamine synthetase | Glutamine synthetase | GLUL |
| Q99798 | 83.4 | Aconitase 2, mitochondrial | aconitase 2, mitochondrial | ACO2 |
| E5RFU4 | 18.3 | Dihydropyrimidinase-like 2 | Dihydropyrimidinase-like 2 | DBYSL2 |
| P09936 | 24.8 | Ubiquitin carboxyl-terminal hydrolase isozyme L1 (UCHL1) | Ubiquitin carboxyl-terminal hydrolase isozyme L1 | VCHC1 |
| P30086 | 21.1 | Phosphatidylethanolamine-binding protein 1 | Phosphatidylethanolamine-binding protein 1 | PBP |
| P00918 | 29.2 | Carbonic anhydrase 2 | Carbonic Anhydrase II | CAZ |
| P12277 | 42.6 | Creatine kinase B-type | Creatine kinase B | CKB |
| P62873 | 37.4 | Guanine nucleotide-binding protein G(I)/G(S)/G(T) subunit beta-1 (GNB1) | Guanine nucleotide-binding protein G(1)/G(S)/G(T) subunit beta 1 | GNB1 |
| P06733 | 47.2 | Alpha-enolase | Alpha-Enolase | ENO1 |
| P04406 | 36.1 | Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) | Glyceraldeyde (3-)phosphate dehydrogenase | GAPDH |
| P60842 | 46.2 | Eukaryotic initiation factor 4A-I | Homo sapiens eukaryotic translation initiation factor 4A isoform 1 (EIF4A1) mRNA | EIFA1 |
| A8K318 | 59.2 | Protein kinase C substrate 80K-H | protein kinase C substrate 80K-H isoform 2 [Homo sapiens] | PKC80 |
| Q68Y55 | 34.9 | Poly(RC) binding protein 2 | poly(rC) binding protein 2 isoform g [Homo sapiens] | PolyRp2 |
| P49761 | 58.6 | CDC-like kinase 3 (CLK3), transcript variant phclk3, mRNA | Homo sapiens CDC-like kinase 3 (CLK3); transcript variant phclk3; mRNA | CLK3 |
| Q9P2Z0 | 28.4 | THAP domain-containing protein 10 | THAP domain containing 10 [Homo sapiens] | THAP |
| Q9BXS5 | 48.6 | AP-1 complex subunit mu-1 (AP1M1) | Homo sapiens adaptor-related protein complex 1; mu 1 subunit (AP1M1); mRNA | AP1M1 |
| P63330 | 35.6 | Serine/threonine-protein phosphatase 2A catalytic subunit alpha isoform | protein phosphatase type 2A catalytic subunit alpha isoform [Mus musculus] | pp2A |
| Q9NZT2 | 73.3 | Opioid growth factor receptor | Homo sapiens opioid growth factor receptor (OGFR). mRNA | OGFR |
| Homo sapiens plasticity-related gene 2 (PRG2) mRNA | PRG2 | |||
| P43235 | 37.0 | Cathepsin K | cathepsin K preproprotein [Homo sapiens] | Catepsin |
| Q53G92 | 50.4 | Tubulin beta-3 chain | Homo sapiens tubulin beta 3 (TUBB3) mRNA | TUBB3 |
| P37108 | 14.6 | Signal recognition particle 14 kDa protein | Homo sapiens signal recognition particle 14 kDa (homologous Alu RNA binding protein) (SRP14) mRNA | SRP14 |
| Q7Z6Z7 | 481.9 | E3 ubiquitin-protein ligase HUWE1; Isoform 1 | HUWE1 protein [Homo sapiens] | HUWE1 |
| Homo sapiens chromosome X genomic contig. reference assembly | ChromosomX | |||
| Q96S16 | 36.9 | JmjC domain-containing protein 8 (Jumonji domain-containing protein 8) | jumonji domain containing 8 [Homo sapiens] | jumonji |
| Q96N21 | 55.1 | Uncharacterized protein C17orf56 | Homo sapiens chromosome 17 open reading frame 56 (C17orf56). mRNA | Chromosome 17 |
| Q96HG3 | 54.6 | Islet cell autoantigen 1, 69 kDa | Homo sapiens islet cell autoantigen 1. 69 kDa (ICA1). transcript variant 2. mRNA | ICA1 |
| P10768 | 31.5 | S-formylglutathione hydrolase (Esterase D) | Homo sapiens esterase D/formylglutathione hydrolase (ESD) | ESD |
| P25325 | 33.2 | 3-mercaptopyruvate sulfurtransferase | mercaptopyruvate sulfurtransferase isoform 2 [Homo sapiens] | MSI 2 |
| P27361 | 43.1 | Mitogen-activated protein kinase 3; Isoform 1 | Homo sapiens mitogen-activated protein kinase 3 (MAPK3); transcript variant 1; mRNA | MAPK3 |
| P43304 | 80.9 | Glycerol-3-phosphate dehydrogenase, mitochondrial (GPD2); Isoform 1 | Homo sapiens glycerol-3-phosphate dehydrogenase 2 (mitochondrial) (GPD2); mRNA | GPD2 |
| P36969 | 22.2 | Phospholipid hydroperoxide glutathione peroxidase, mitochondrial (Glutathione peroxidase 4 | Homo sapiens glutathione peroxidase 4 (phospholipid hydroperoxidase) (GPX4). transcript variant 1. mRNA | GPX4 |
| B7Z4U7 | 65.1 | Sec1 family domain containing 1, isoform CRA_b | vesicle transport-related protein isoform b [Homo sapiens] | VTI-B |
| Q9BVL4 | 73.5 | Selenoprotein O | Homo sapiens selenoprotein O (SELO) mRNA | SELO |
| Q6PJ21 | 39.4 | SPRY domain-containing SOCS box protein 3 | SPRY domain-containing SOCS box protein SSB-3 | SPRX |
| P35611 | 81.0 | Alpha-adducin | adducin 1 (alpha) isoform c [Homo sapiens] | Adduccin |
| Q99798 | 85.4 | Aconitate hydratase, mitochondrial | Aconitate Hydratase 2 (mitochondrial) | Aconitate Hydratase |
| P06576 | 56.6 | ATP synthase subunit beta, mitochondrial | ATP synthase | ATP Synthase |
| P40926 | 35.5 | Malate dehydrogenase, mitochondrial | Malat dehydrogenase | Malat Dehydrogenase |
| P37840 | 14.5 | Alpha-synuclein | alpha-synuclein | Alpha Synuclein |
| P10636 | 78.9 | Microtubule-associated protein tau | tau | TAU |
| P05067 | 86.9 | Amyloid beta A4 protein (Alzheimer disease amyloid protein) | beta-amyloid | Beta-Amyloid |
| Q05923 | 34.4 | Dual specificity protein phosphatase 2 | DUSP2 dual specificity phosphatase 2 [Homo sapiens] | DUSP2 |
| Q14166 | 74.4 | Tubulin-tyrosine ligase-like protein 12 | Homo sapiens tubulin tyrosine ligase-like family member 12 (TTLL12) mRNA | TTLL2 |
| ANOVA | |||||||
|---|---|---|---|---|---|---|---|
| CTRL-AV | CTRL-SE | AMDDRY-AV | AMDDRY-SE | AMDWET-AV | AMDWET-SE | ANOVA-p | |
| Alpha Synuclein | 6428 | 487 | 10.667 | 1974 | 7221 | 251 | 0.003 |
| SELO | 31.155 | 1755 | 27.412 | 1733 | 26.330 | 397 | 0.01 |
| SPRY | 24.905 | 1445 | 21.287 | 808 | 22.305 | 265 | 0.028 |
| GAPDH—H2 | 11.571 | 821 | 13.277 | 1137 | 14.926 | 360 | 0.031 |
| Annexin V | 14.977 | 1821 | 18.675 | 1422 | 15.258 | 321 | 0.034 |
| THAP | 14.422 | 1575 | 10.767 | 715 | 11.889 | 284 | 0.044 |
| VTI-B | 22.121 | 1816 | 19.333 | 1493 | 23.773 | 512 | 0.065 |
| HSP 10 | 16.099 | 1313 | 20.613 | 1716 | 19.235 | 385 | 0.071 |
| ESD | 29.416 | 1393 | 24.942 | 995 | 26.008 | 424 | 0.082 |
| PKC80 | 23.592 | 1798 | 21.236 | 1396 | 20.575 | 335 | 0.082 |
| ACO2—C2 | 19.238 | 1478 | 17.569 | 1462 | 16.185 | 376 | 0.089 |
| OGFR | 18.774 | 2383 | 21.865 | 3017 | 17.521 | 516 | 0.115 |
| PBP—I2 | 21.243 | 1455 | 24.279 | 1422 | 24.944 | 467 | 0.119 |
| CAZ—C3 | 5373 | 595 | 7534 | 1274 | 6402 | 196 | 0.148 |
| EIFA1 | 26.612 | 2773 | 21.695 | 2045 | 21.915 | 612 | 0.15 |
| MAPK3 | 28.505 | 1571 | 25.186 | 1110 | 27.123 | 315 | 0.15 |
| ENO1—H7 | 19.297 | 1419 | 25.493 | 2798 | 22.685 | 594 | 0.15 |
| Chromosome X reading frame 56 | 20.439 | 1587 | 18.881 | 1513 | 21.993 | 455 | 0.16 |
| Aconitate Hydratase | 20.584 | 1720 | 18.805 | 1622 | 17.738 | 391 | 0.163 |
| GPX4 | 19.207 | 1159 | 17.929 | 1277 | 17.190 | 283 | 0.177 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Korb, C.A.; Beck, S.; Wolters, D.; Lorenz, K.; Pfeiffer, N.; Grus, F.H. Serum Autoantibodies in Patients with Dry and Wet Age-Related Macular Degeneration. J. Clin. Med. 2023, 12, 1590. https://doi.org/10.3390/jcm12041590
Korb CA, Beck S, Wolters D, Lorenz K, Pfeiffer N, Grus FH. Serum Autoantibodies in Patients with Dry and Wet Age-Related Macular Degeneration. Journal of Clinical Medicine. 2023; 12(4):1590. https://doi.org/10.3390/jcm12041590
Chicago/Turabian StyleKorb, Christina A., Sabine Beck, Dominik Wolters, Katrin Lorenz, Norbert Pfeiffer, and Franz H. Grus. 2023. "Serum Autoantibodies in Patients with Dry and Wet Age-Related Macular Degeneration" Journal of Clinical Medicine 12, no. 4: 1590. https://doi.org/10.3390/jcm12041590
APA StyleKorb, C. A., Beck, S., Wolters, D., Lorenz, K., Pfeiffer, N., & Grus, F. H. (2023). Serum Autoantibodies in Patients with Dry and Wet Age-Related Macular Degeneration. Journal of Clinical Medicine, 12(4), 1590. https://doi.org/10.3390/jcm12041590






