Risk Factors for Adhesion-Related Readmission and Abdominal Reoperation after Gynecological Surgery: A Nationwide Cohort Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Research Design and Data-Retrieval
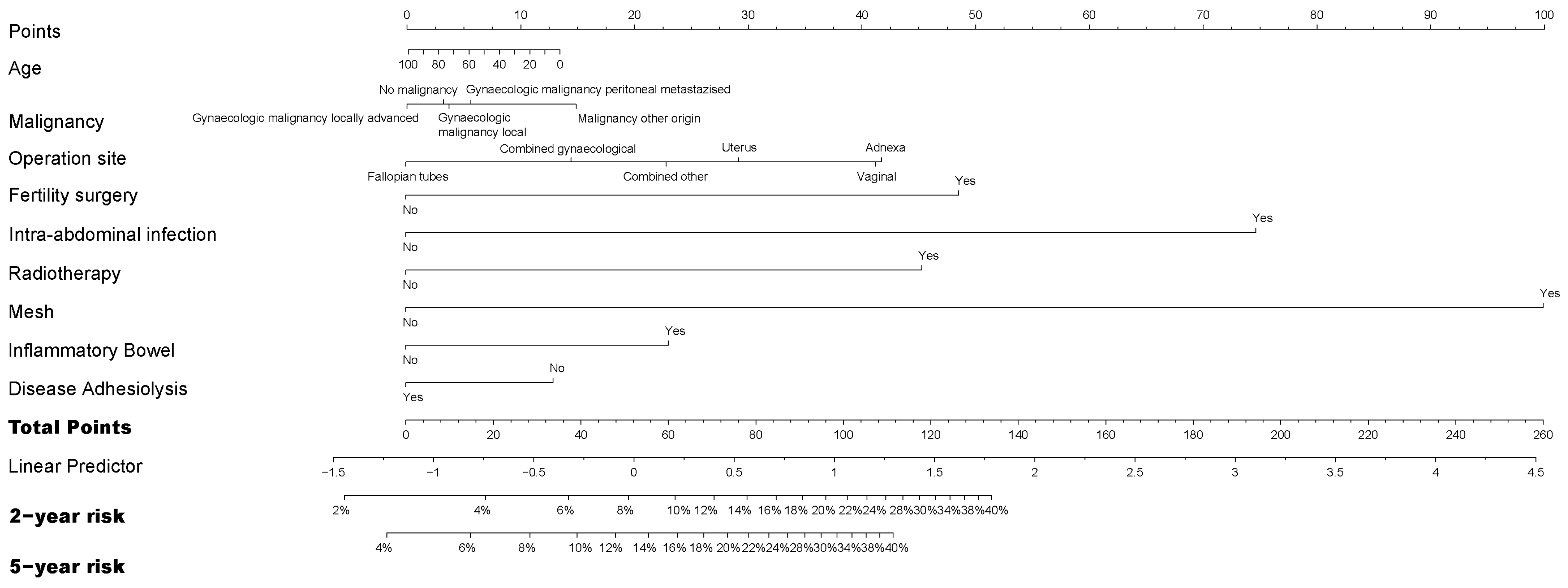
2.2. Statistical Analysis
3. Results
3.1. Risk Factors for Readmission Directly Related to Adhesions
3.2. Risk Factors for Possibly Adhesion-Related Readmissions
3.3. Risk Factors for Reoperation after Gynecological Surgery
4. Discussion
4.1. Main Findings
4.2. Strengths and Limitations
4.3. Interpretation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Krielen, P.; Stommel, M.W.J.; Pargmae, P.; Bouvy, N.D.; Bakkum, E.A.; Ellis, H.; Parker, M.C.; Griffiths, E.A.; van Goor, H.; Ten Broek, R.P.G. Adhesion-related readmissions after open and laparoscopic surgery: A retrospective cohort study (SCAR update). Lancet 2020, 395, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Nunoo-Mensah, J.W.; Rosen, M.; Chan, L.S.; Wasserberg, N.; Beart, R.W. Prevalence of intra-abdominal surgery: What is an individual’s lifetime risk? South Med. J. 2009, 102, 25–29. [Google Scholar] [CrossRef] [PubMed]
- Ten Broek, R.P.; Issa, Y.; van Santbrink, E.J.; Bouvy, N.D.; Kruitwagen, R.F.; Jeekel, J.; Bakkum, E.A.; Rovers, M.M.; van Goor, H. Burden of adhesions in abdominal and pelvic surgery: Systematic review and met-analysis. BMJ 2013, 347, f5588. [Google Scholar] [CrossRef] [PubMed]
- Scotland, N. Life Expectancy in Scotland, 2017–2019; NRS Scotland: Edinburgh, Scotland, 2020. Available online: https://www.nrscotland.gov.uk/ (accessed on 18 October 2021).
- Strik, C.; Stommel, M.W.; Ten Broek, R.P.; van Goor, H. Adhesiolysis in Patients Undergoing a Repeat Median Laparotomy. Dis. Colon. Rectum. 2015, 58, 792–798. [Google Scholar] [CrossRef]
- Okabayashi, K.; Ashrafian, H.; Zacharakis, E.; Hasegawa, H.; Kitagawa, Y.; Athanasiou, T.; Darzi, A. Adhesions after abdominal surgery: A systematic review of the incidence, distribution and severity. Surg. Today 2014, 44, 405–420. [Google Scholar] [CrossRef]
- Lundorff, P.; Brölmann, H.; Koninckx, P.R.; Mara, M.; Wattiez, A.; Wallwiener, M.; Trew, G.; Crowe, A.M.; De Wilde, R.L. Predicting formation of adhesions after gynaecological surgery: Development of a risk score. Arch. Gynecol. Obstet. 2015, 292, 931–938. [Google Scholar] [CrossRef]
- Lower, A.M.; Hawthorn, R.J.; Clark, D.; Boyd, J.H.; Finlayson, A.R.; Knight, A.D.; Crowe, A.M. Adhesion-related readmissions following gynaecological laparoscopy or laparotomy in Scotland: An epidemiological study of 24,046 patients. Hum. Reprod. 2004, 19, 1877–1885. [Google Scholar] [CrossRef]
- Ellis, H.; Moran, B.J.; Thompson, J.N.; Parker, M.C.; Wilson, M.S.; Menzies, D.; McGuire, A.; Lower, A.M.; Hawthorn, R.J.; O’Brien, F.; et al. Adhesion-related hospital readmissions after abdominal and pelvic surgery: A retrospective cohort study. Lancet 1999, 353, 1476–1480. [Google Scholar] [CrossRef]
- In, J.; Lee, D.K. Survival analysis: Part II-applied clinical data analysis. Korean J. Anesth. 2019, 72, 441–457. [Google Scholar] [CrossRef]
- Harrell, F.E., Jr. rms: Regression modeling strategies. R Package Version 2016, 5, 103–126. [Google Scholar]
- Steyerberg, E.W.; Vickers, A.J.; Cook, N.R.; Gerds, T.; Gonen, M.; Obuchowski, N.; Pencina, M.J.; Kattan, M.W. Assessing the Performance of Prediction Models: A Framework for Traditional and Novel Measures. Epidemiology 2010, 21, 128–138. [Google Scholar] [CrossRef]
- Tanvetyanon, T.; Finley, D.J.; Fabian, T.; Riquet, M.; Voltolini, L.; Kocaturk, C.; Bryant, A.; Robinson, L. Prognostic nomogram to predict survival after surgery for synchronous multiple lung cancers in multiple lobes. J. Thorac. Oncol. 2015, 10, 338–345. [Google Scholar] [CrossRef]
- Iba, K.; Shinozaki, T.; Maruo, K.; Noma, H. Re-evaluation of the comparative effectiveness of bootstrap-based optimism correction methods in the development of multivariable clinical prediction models. BMC Med. Res. Methodol. 2021, 21, 9. [Google Scholar] [CrossRef]
- Ten Broek, R.P.G.; Krielen, P.; Di Saverio, S.; Coccolini, F.; Biffl, W.L.; Ansaloni, L.; Velmahos, G.C.; Sartelli, M.; Fraga, G.P.; Kelly, M.D.; et al. Bologna guidelines for diagnosis and management of adhesive small bowel obstruction (ASBO): 2017 update of the evidence-based guidelines from the world society of emergency surgery ASBO working group. World J. Emerg. Surg. 2018, 13, 24. [Google Scholar] [CrossRef]
- Iglesia, C.B.; Fenner, D.E.; Brubaker, L. The use of mesh in gynecologic surgery. Int. Urogynecol. J. Pelvic. Floor Dysfunct. 1997, 8, 105–115. [Google Scholar] [CrossRef]
- Hegde, C.V. Use of mesh in vaginal pelvic organ prolapse surgery: Prolapse surgery-an overview. J. Obstet. Gynaecol. India 2011, 61, 381–383. [Google Scholar] [CrossRef][Green Version]
- Arabkhazaeli, M.; Keltz, J.; Eisenberg, R.; Levie, M.; Yettaw Luts, H. A Retrospective Study of Risk Factors for Small Bowel Obstruction After Hysterectomy. JSLS 2020, 24, e2020.00070. [Google Scholar] [CrossRef]
- Antosh, D.D.; Grimes, C.L.; Smith, A.L.; Friedman, S.; McFadden, B.L.; Crisp, C.C.; Allen, A.M.; Gutman, R.E.; Rogers, R.G. A case-control study of risk factors for ileus and bowel obstruction following benign gynecologic surgery. Int. J. Gynaecol. Obstet. 2013, 122, 108–111. [Google Scholar] [CrossRef]
- Salvador, C.; Sikirica, M.; Evans, A.; Pizzi, L.; Goldfarb, N. Clinical and Economic Outcomes of Prolonged Postoperative Ileus in Patients Undergoing Hysterectomy and Hemicolectomy; P&T: Hong Kong, China, 2004; Volume 30, pp. 590–595. [Google Scholar]
- Meuleman, T.; Schreinemacher, M.H.; van Goor, H.; Bakkum, E.A.; Dorr, P.J. Adhesion awareness: A nationwide survey of gynaecologists. Eur. J. Obstet. Gynecol. Reprod. Biol. 2013, 169, 353–359. [Google Scholar] [CrossRef]
- Trew, G.; Cooke, I.; Lower, A.; McVeigh, E. Post-operative abdominal adhesions—Awareness of UK gynaecologists—A survey of members of the Royal College of Obstetricians and Gynaecologists. Gynecol. Surg. 2009, 6, 25–37. [Google Scholar] [CrossRef]
- Mäkinen, J.; Johansson, J.; Tomás, C.; Tomás, E.; Heinonen, P.K.; Laatikainen, T.; Kauko, M.; Heikkinen, A.M.; Sjöberg, J. Morbidity of 10 110 hysterectomies by type of approach. Hum. Reprod. 2001, 16, 1473–1478. [Google Scholar] [CrossRef] [PubMed]
- Moss, E.L.; Morgan, G.; Martin, A.P.; Sarhanis, P.; Ind, T. Surgical trends, outcomes and disparities in minimal invasive surgery for patients with endometrial cancer in England: A retrospective cohort study. BMJ Open 2020, 10, e036222. [Google Scholar] [CrossRef] [PubMed]
- Zwanenburg, E.S.; Wisselink, D.D.; Klaver, C.E.L.; Brandt, A.R.M.; Bremers, A.J.A.; Burger, J.W.A.; van Grevenstein, W.M.U.; Hemmer, P.H.J.; de Hingh, I.; Kok, N.F.M.; et al. Adhesion formation after surgery for locally advanced colonic cancer in the COLOPEC trial. Br. J. Surg. 2022, 109, 315–318. [Google Scholar] [CrossRef]
- Fortin, C.N.; Saed, G.M.; Diamond, M.P. Predisposing factors to post-operative adhesion development. Hum. Reprod. Update 2015, 21, 536–551. [Google Scholar] [CrossRef]
- Reis, F.M.; Petraglia, F.; Taylor, R.N. Endometriosis: Hormone regulation and clinical consequences of chemotaxis and apoptosis. Hum. Reprod. Update 2013, 19, 406–418. [Google Scholar] [CrossRef] [PubMed]
- Sheyn, D.; Bretschneider, C.E.; Mahajan, S.T.; Ridgeway, B.; Davenport, A.; Pollard, R. Incidence and risk factors of early postoperative small bowel obstruction in patients undergoing hysterectomy for benign indications. Am. J. Obstet. Gynecol. 2019, 220, 251.e251–251.e259. [Google Scholar] [CrossRef]
- Montz, F.J. Small bowel obstruction following radical hysterectomy: Risk factors, incidence, and operative findings. Gynecol. Oncol. 1994, 53, 114–120. [Google Scholar] [CrossRef]
- Ten Broek, R.P.; Stommel, M.W.; Strik, C.; van Laarhoven, C.J.; Keus, F.; van Goor, H. Benefits and harms of adhesion barriers for abdominal surgery: A systematic review and meta-analysis. Lancet 2014, 383, 48–59. [Google Scholar] [CrossRef]
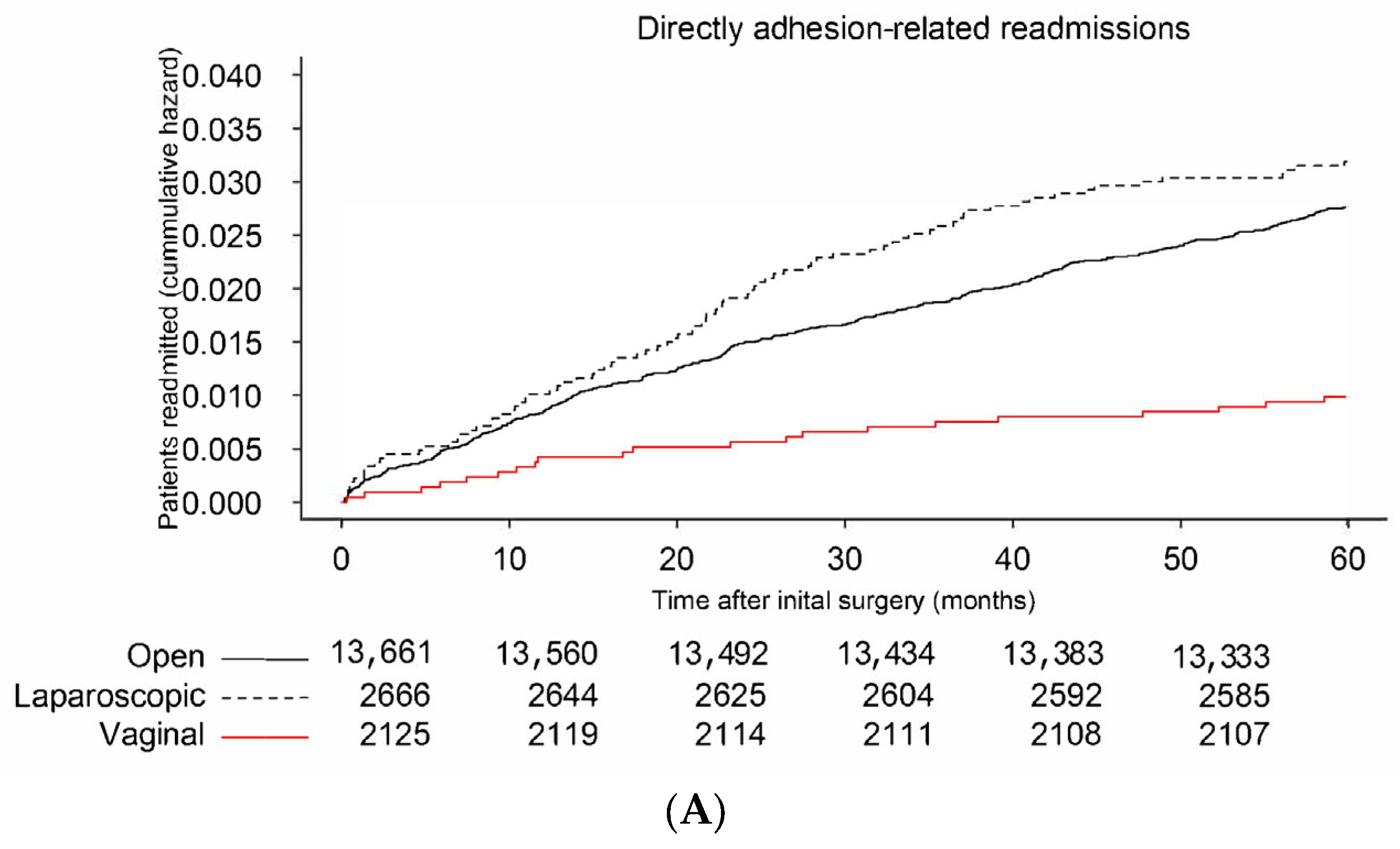
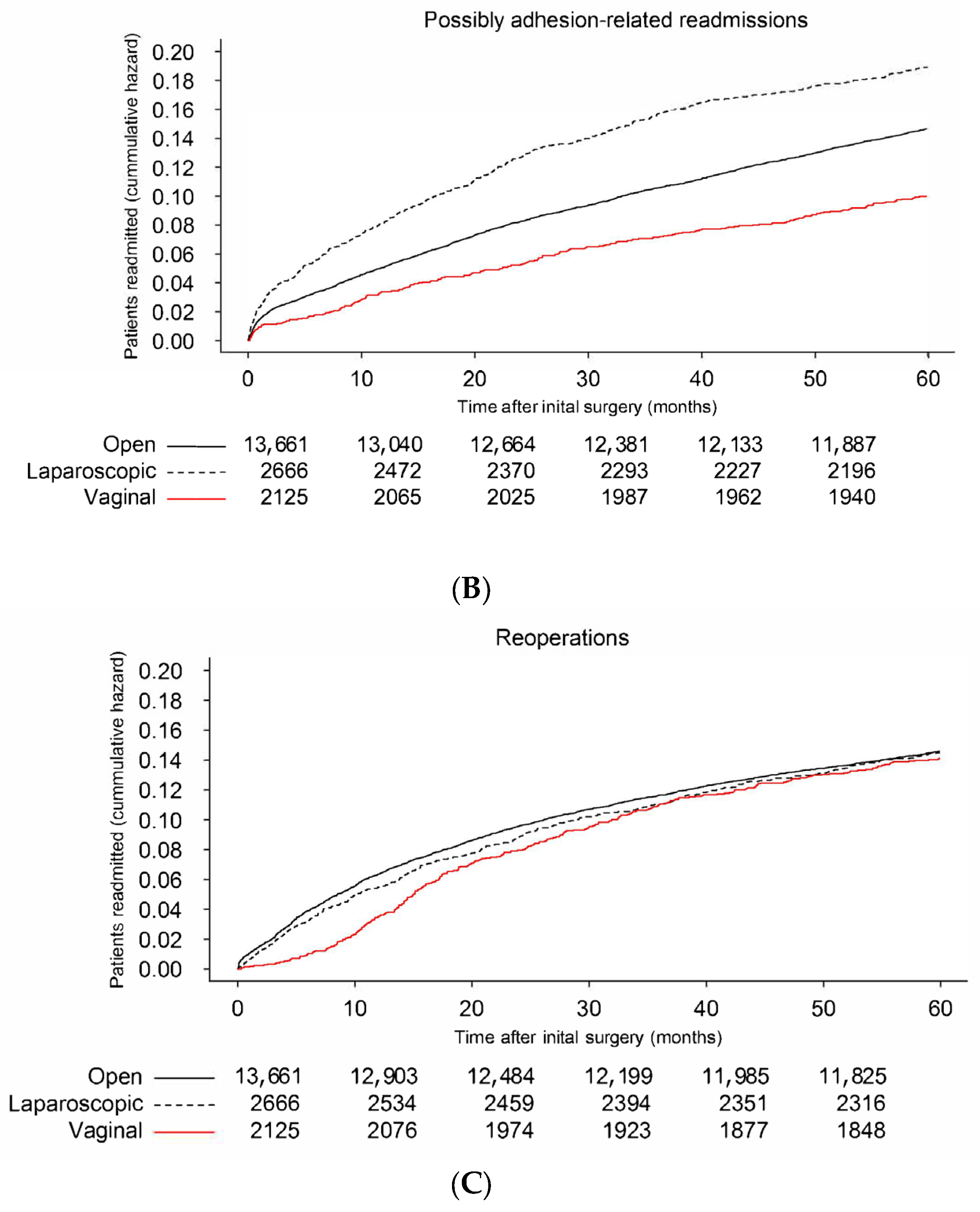
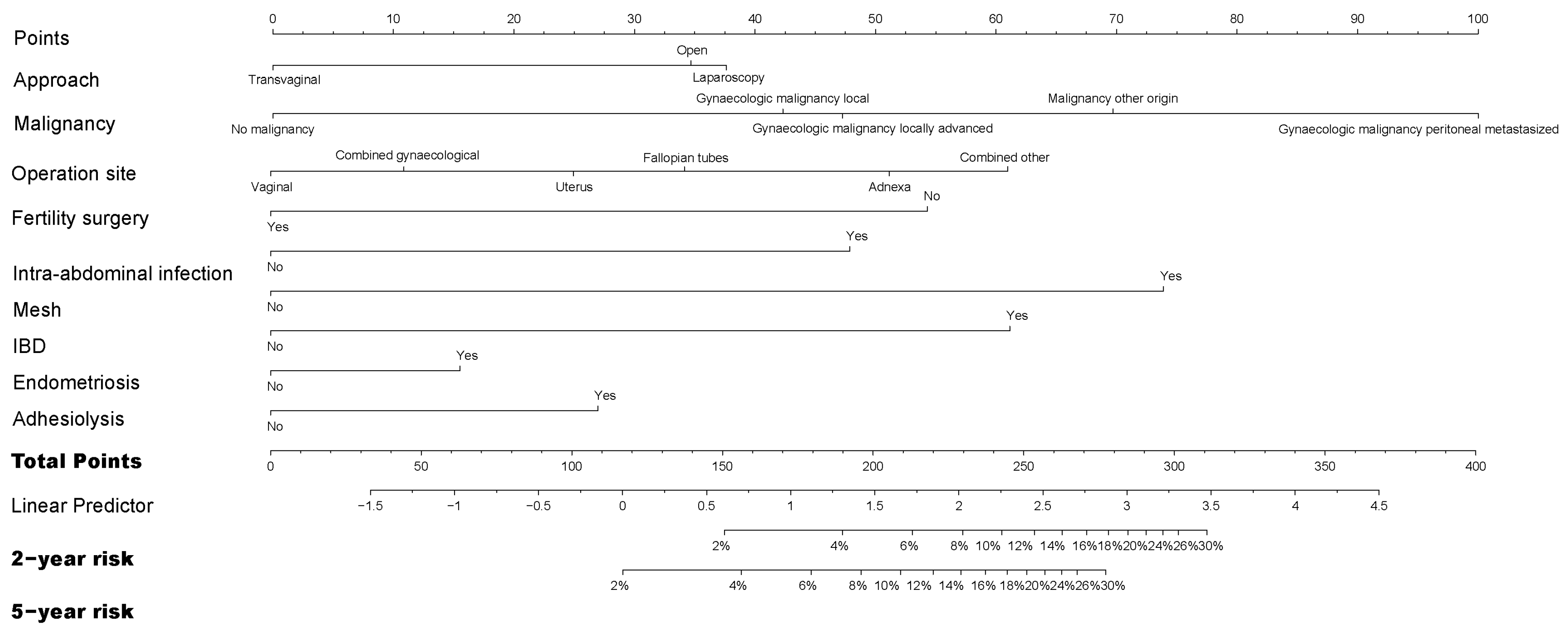
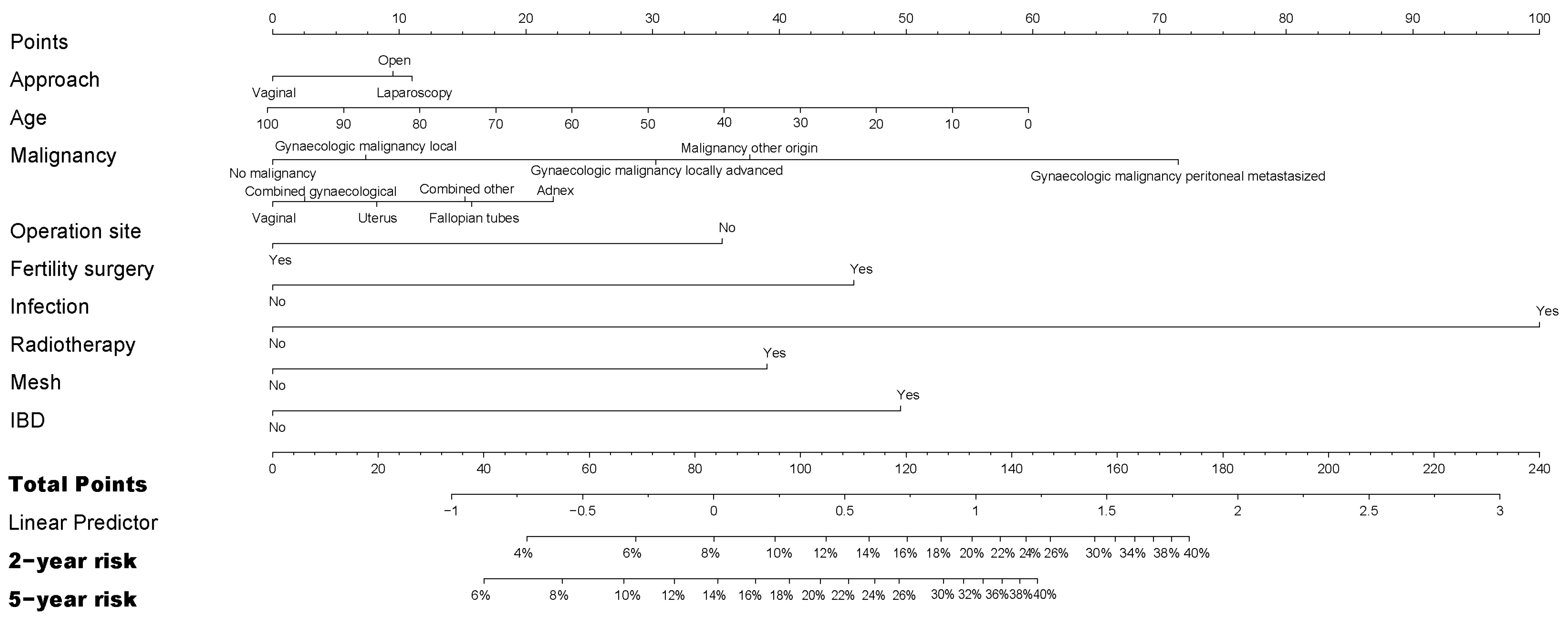
| Laparotomy N = 13,661 | Laparoscopy N = 2666 | Transvaginal N = 2125 | Total N = 18,452 | |
|---|---|---|---|---|
| Number women of readmitted | 2003 (14.6%) | 2003 (14.6%) | 212 (10.0%) | 2719 (14.7%) |
| Ovary | 170 (14.3%) | 170 (14.3%) | - | |
| Fallopian tubes | 128 (20.9%) | 128 (20.9%) | - | |
| Vagina | 388 (11.0%) | 388 (11.0%) | - | |
| Uterus | 318 (14.2%) | 318 (14.2%) | 72 (17.8%) | |
| Combined gynecological | 518 (14.2%) | 518 (14.2%) | 133 (8.4%) | |
| Other combined | 482 (19.6%) | 482 (19.6%) | 7 (5.4%) | |
| Number of total readmissions | 3033 | 896 | 274 | 4203 |
| Ovary | 258 | 504 | - | |
| Fallopian tubes | 204 | 121 | - | |
| Vagina | 498 | - | - | |
| Uterus | 510 | 37 | 102 | |
| Combined gynecological | 797 | 111 | 163 | |
| Other combined | 766 | 123 | 9 | |
| Number of women undergoing reoperation | 1993 (14.6%) | 386 (11.3%) | 300 (14.1%) | 2679 (14.5%) |
| Ovary | 377 (31.8%) | 205 (18.8%) | - | |
| Fallopian tubes | 55 (9.0%) | 27 (6.6%) | - | |
| Vagina | 611 (17.3%) | - | - | |
| Uterus | 316 (14.1%) | 19 (11.1%) | 59 (14.6%) | |
| Combined gynecological | 317 (8.7%) | 81 (11.6%) | 221 (13.9%) | |
| Other combined | 317 (12.9%) | 54 (17.8%) | 20 (15.4%) |
| Laparotomy | Laparoscopy | Transvaginal | Total | Sig. | |
|---|---|---|---|---|---|
| Age (years) | p < 0.001 | ||||
| Min | 0 | 0 | 25 | 0 | |
| Max | 94 | 99 | 89 | 99 | |
| Mean (SD) | 51.27 (14.69) | 39.17 (14.44) | 59.38 (12.10) | 50.46 (15.31) | |
| Operation site | p < 0.001 | ||||
| Ovary | 1186 (8.7%) | 1088 (40.8%) | 0 | 2274 (12.3%) | |
| Fallopian tubes | 611 (4.5%) | 407 (15.3%) | 0 | 1018 (5.5%) | |
| Vagina | 3523 (25.8%) | 0 | 0 | 3523 (19.1%) | |
| Uterus | 316 (14.1%) | 171 (6.4%) | 404 (19.0%) | 2815 (15.3%) | |
| Combined gynecological | 317 (8.7%) | 696 (26.1%) | 1591 (74.9%) | 5928 (32.1%) | |
| Combined other | 317 (12.9%) | 304 (11.4%) | 130 (6.1%) | 2894 (15.7%) | |
| Malignancy | p < 0.001 | ||||
| No malignancy | 11,923 (87.3%) | 2453 (92.0%) | 2079 (97.8%) | 16,455 (89.2%) | |
| Gynecological malignancy local | 853 (6.2%) | 115 (4.3%) | 32 (1.5%) | 1000 (5.4%) | |
| Gyn malignancy locally advanced | 547 (4.0%) | 32 (1.2%) | 6 (0.3%) | 585 (3.2%) | |
| Gyn malignancy peritoneal metastasized | 71 (0.5%) | 10 (0.4%) | 0 | 81 (0.4%) | |
| Malignancy other origin | 267 (2.0%) | 56 (2.1%) | 8 (0.4%) | 331 (1.8%) | |
| Fertility enhancing surgery | p < 0.001 | ||||
| No | 12,899 (94.4%) | 2583 (96.9%) | 2125 (100%) | 17,607 (95.4%) | |
| Yes | 762 (5.6%) | 83 (3.1%) | 0 | 845 (4.6%) | |
| Intra-abdominal infection | p = 0.214 | ||||
| No | 13,603 (99.6%) | 2653 (99.5%) | 2121 (99.8%) | 18,377 (99.6%) | |
| Yes | 58 (0.4%) | 13 (0.5%) | 4 (0.2%) | 75 (0.4%) | |
| History of radiotherapy | p = 0.309 | ||||
| No | 13,645 (99.9%) | 2664 (99.9%) | 2125 (100%) | 18,434 (99.9%) | |
| Yes | 16 (0.1%) | 2 (0.1%) | 0 | 18 (0.1%) | |
| Mesh placement | p < 0.001 | ||||
| No | 13,487 (98.7%) | 2641 (99.1%) | 2119 (99.7%) | 18,247 (98.9%) | |
| Yes | 174 (1.3%) | 25 (0.9%) | 6 (0.3%) | 205 (1.1%) | |
| IBD | p = 0.285 | ||||
| No | 13,603 (99.6%) | 2657 (99.7%) | 2121 (99.8%) | 18,381 (99.6%) | |
| Yes | 58 (0.4%) | 9 (0.3%) | 4 (0.2%) | 71 (0.4%) | |
| Endometriosis | p = 0.004 | ||||
| No | 12,747 (93.3%) | 2302 (86.3%) | 1999 (94.1%) | 17,048 (92.4%) | |
| Yes | 914 (6.7%) | 364 (13.7%) | 126 (5.9%) | 1404 (7.6%) | |
| Adhesiolysis | p < 0.001 | ||||
| No | 13,276 (97.%) | 2649 (99.4%) | 2122 (99.9%) | 18,047 (97.8%) | |
| Yes | 385 (2.8%) | 17 (0.6%) | 3 (0.1%) | 204 (2.2%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Toneman, M.; Groenveld, T.; Krielen, P.; Hooker, A.; de Wilde, R.; Torres-de la Roche, L.A.; Di Spiezio Sardo, A.; Koninckx, P.; Cheong, Y.; Nap, A.; et al. Risk Factors for Adhesion-Related Readmission and Abdominal Reoperation after Gynecological Surgery: A Nationwide Cohort Study. J. Clin. Med. 2023, 12, 1351. https://doi.org/10.3390/jcm12041351
Toneman M, Groenveld T, Krielen P, Hooker A, de Wilde R, Torres-de la Roche LA, Di Spiezio Sardo A, Koninckx P, Cheong Y, Nap A, et al. Risk Factors for Adhesion-Related Readmission and Abdominal Reoperation after Gynecological Surgery: A Nationwide Cohort Study. Journal of Clinical Medicine. 2023; 12(4):1351. https://doi.org/10.3390/jcm12041351
Chicago/Turabian StyleToneman, Masja, Tjitske Groenveld, Pepijn Krielen, Angelo Hooker, Rudy de Wilde, Luz Angela Torres-de la Roche, Atillio Di Spiezio Sardo, Philippe Koninckx, Ying Cheong, Annemiek Nap, and et al. 2023. "Risk Factors for Adhesion-Related Readmission and Abdominal Reoperation after Gynecological Surgery: A Nationwide Cohort Study" Journal of Clinical Medicine 12, no. 4: 1351. https://doi.org/10.3390/jcm12041351
APA StyleToneman, M., Groenveld, T., Krielen, P., Hooker, A., de Wilde, R., Torres-de la Roche, L. A., Di Spiezio Sardo, A., Koninckx, P., Cheong, Y., Nap, A., van Goor, H., Pargmae, P., & ten Broek, R. (2023). Risk Factors for Adhesion-Related Readmission and Abdominal Reoperation after Gynecological Surgery: A Nationwide Cohort Study. Journal of Clinical Medicine, 12(4), 1351. https://doi.org/10.3390/jcm12041351








