Oocyte Cryopreservation in Women with Ovarian Endometriosis
Abstract
:1. Introduction
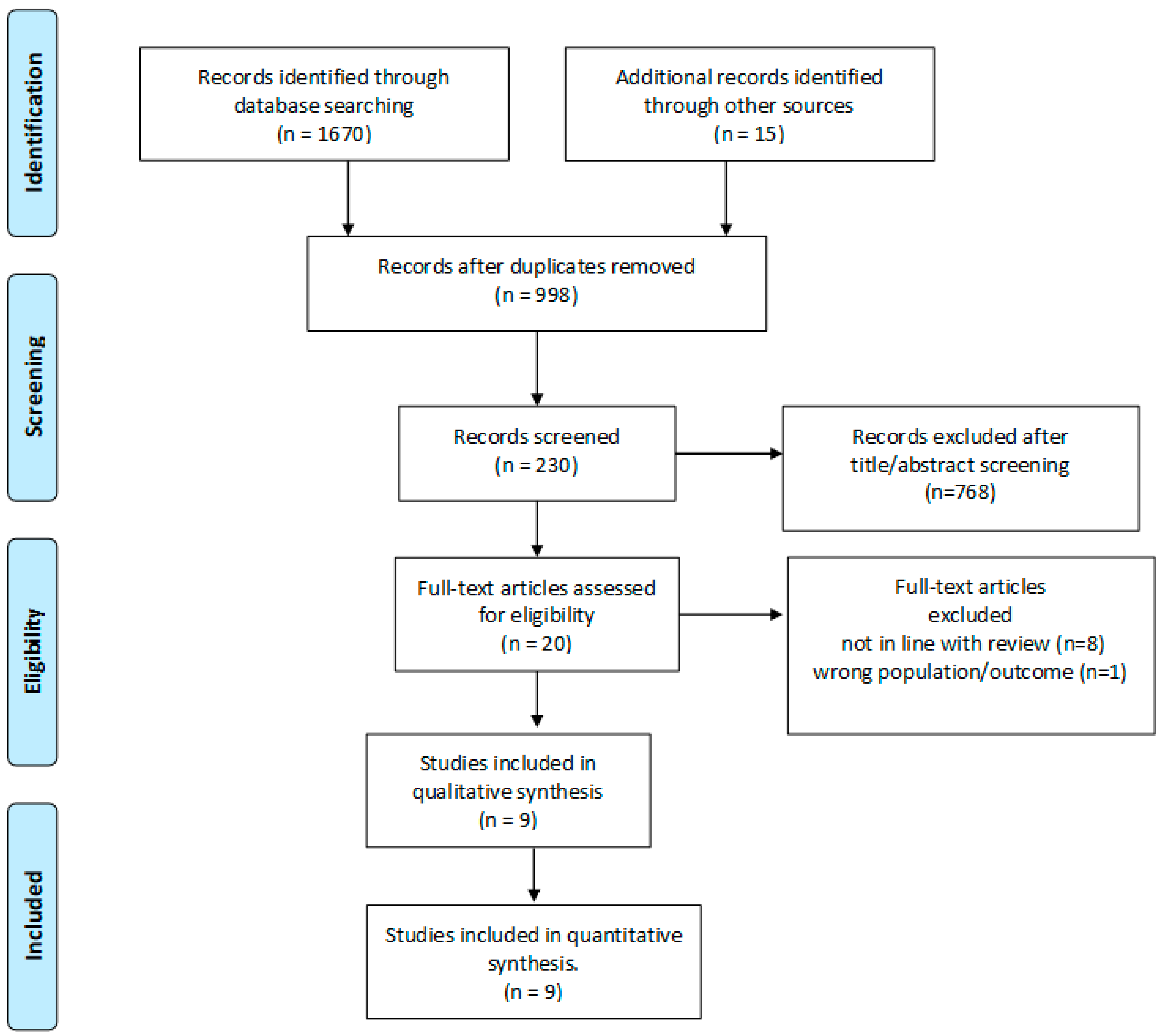
2. Materials and Methods
3. Ovarian Reserve in Endometriosis
4. Effects of Surgery for Ovarian Endometriosis on Ovarian Reserve
5. Surgical Techniques That May Help to Reduce Ovarian Damage in Endometrioma Surgery
6. Ovarian Stimulation in Women with Endometriosis
7. Outcomes of Fertility Preservation
8. Return Rate
9. Pregnancy Rates and Embryo Quality
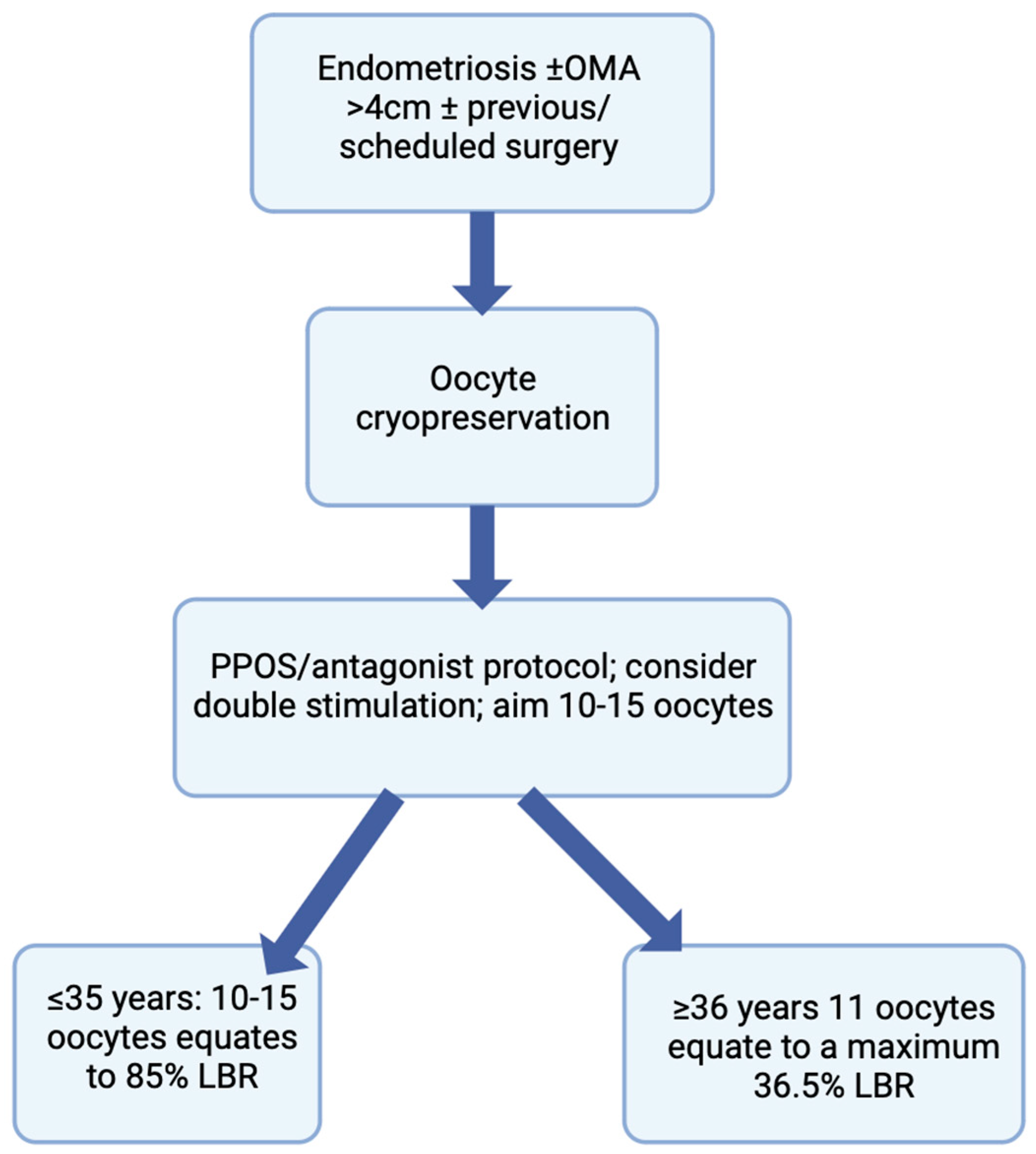
10. Number of Oocytes to Cryopreserve
11. Conclusions and Future Considerations
Author Contributions
Funding
Conflicts of Interest
References
- Meuleman, C.; Vandenabeele, B.; Fieuws, S.; Spiessens, C.; Timmerman, D.; D’Hooghe, T. High Prevalence of Endometriosis in Infertile Women with Normal Ovulation and Normospermic Partners. Fertil. Steril. 2009, 92, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Patel, B.G.; Lenk, E.E.; Lebovic, D.I.; Shu, Y.; Yu, J.; Taylor, R.N. Pathogenesis of Endometriosis: Interaction between Endocrine and Inflammatory Pathways. Best Pract. Res. Clin. Obstet. Gynaecol. 2018, 50, 50–60. [Google Scholar] [CrossRef]
- Gupta, S.; Goldberg, J.M.; Aziz, N.; Goldberg, E.; Krajcir, N.; Agarwal, A. Pathogenic Mechanisms in Endometriosis-Associated Infertility. Fertil. Steril. 2008, 90, 247–257. [Google Scholar] [CrossRef]
- Somigliana, E.; Vigano, P.; Filippi, F.; Papaleo, E.; Benaglia, L.; Candiani, M.; Vercellini, P. Fertility Preservation in Women with Endometriosis: For All, for Some, for None? Hum. Reprod. 2015, 30, 1280–1286. [Google Scholar] [CrossRef] [PubMed]
- Imperiale, L.; Nisolle, M.; Noël, J.C.; Fastrez, M. Three Types of Endometriosis: Pathogenesis, Diagnosis and Treatment. State of the Art. J. Clin. Med. 2023, 12, 994. [Google Scholar] [CrossRef] [PubMed]
- Nisolle, M.; Donnez, J. Peritoneal Endometriosis, Ovarian Endometriosis, and Adenomyotic Nodules of the Rectovaginal Septum Are Three Different Entities. Fertil. Steril. 1997, 68, 585–596. [Google Scholar] [CrossRef]
- Hudelist, G.; Ballard, K.; English, J.; Wright, J.; Banerjee, S.; Mastoroudes, H.; Thomas, A.; Singer, C.F.; Keckstein, J. Transvaginal Sonography vs. Clinical Examination in the Preoperative Diagnosis of Deep Infiltrating Endometriosis. Ultrasound Obstet. Gynecol. 2011, 37, 480–487. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, T.A.; Templeton, A. Prevalence and Genesis of Endometriosis. Hum. Reprod. 1991, 6, 544–549. [Google Scholar] [CrossRef] [PubMed]
- Santulli, P.; Lamau, M.C.; Marcellin, L.; Gayet, V.; Marzouk, P.; Borghese, B.; Lafay Pillet, M.-C.; Chapron, C. Endometriosis-Related Infertility: Ovarian Endometrioma per Se Is Not Associated with Presentation for Infertility. Hum. Reprod. 2016, 31, 1765–1775. [Google Scholar] [CrossRef]
- Vercellini, P.; Chapron, C.; De Giorgi, O.; Consonni, D.; Frontino, G.; Crosignani, P.G. Coagulation or Excision of Ovarian Endometriomas? Am. J. Obstet. Gynecol. 2003, 188, 606–610. [Google Scholar] [CrossRef] [PubMed]
- Vercellini, P.; Viganò, P.; Somigliana, E.; Fedele, L. Endometriosis: Pathogenesis and Treatment. Nat. Rev. Endocrinol. 2014, 10, 261–275. [Google Scholar] [CrossRef]
- Muzii, L.; Di Tucci, C.; Di Feliciantonio, M.; Galati, G.; Di Donato, V.; Musella, A.; Palaia, I.; Panici, P.B. Antimüllerian Hormone Is Reduced in the Presence of Ovarian Endometriomas: A Systematic Review and Meta-Analysis. Fertil. Steril. 2018, 110, 932–940.e1. [Google Scholar] [CrossRef] [PubMed]
- Younis, J.S.; Shapso, N.; Ben-Sira, Y.; Nelson, S.M.; Izhaki, I. Endometrioma Surgery—A Systematic Review and Meta-Analysis of the Effect on Antral Follicle Count and Anti-Müllerian Hormone. Am. J. Obstet. Gynecol. 2022, 226, 33–51.e7. [Google Scholar] [CrossRef] [PubMed]
- Karadağ, C.; Yoldemir, T.; Demircan Karadağ, S.; Turgut, A. The Effects of Endometrioma Size and Bilaterality on Ovarian Reserve. J. Obstet. Gynaecol. 2020, 40, 531–536. [Google Scholar] [CrossRef] [PubMed]
- Kasapoglu, I.; Ata, B.; Uyaniklar, O.; Seyhan, A.; Orhan, A.; Yildiz Oguz, S.; Uncu, G. Endometrioma-Related Reduction in Ovarian Reserve (ERROR): A Prospective Longitudinal Study. Fertil. Steril. 2018, 110, 122–127. [Google Scholar] [CrossRef]
- Yılmaz Hanege, B.; Güler Çekıç, S.; Ata, B. Endometrioma and Ovarian Reserve: Effects of Endometriomata per Se and Its Surgical Treatment on the Ovarian Reserve. Facts Views Vis. ObGyn 2019, 11, 151–157. [Google Scholar]
- Tan, Z.; Gong, X.; Li, Y.; Hung, S.W.; Huang, J.; Wang, C.C.; Chung, J.P.W. Impacts of Endometrioma on Ovarian Aging from Basic Science to Clinical Management. Front. Endocrinol. 2023, 13, 1073261. [Google Scholar] [CrossRef]
- Cornillie, F.J.; Oosterlynck, D.; Lauweryns, J.M.; Koninckx, P.R. Deeply Infiltrating Pelvic Endometriosis: Histology and Clinical Significance. Fertil. Steril. 1990, 53, 978–983. [Google Scholar] [CrossRef]
- Dunselman, G.A.; Vermeulen, N.; Becker, C.; Calhaz-Jorge, C.; D’Hooghe, T.; De Bie, B.; Heikinheimo, O.; Horne, A.W.; Kiesel, L.; Nap, A.; et al. ESHRE guideline: Management of women with endometriosis. Hum. Reprod. 2014, 29, 400–412. [Google Scholar] [CrossRef]
- Nadeem, A.; Habte, A.; Ahsan, A.; Tariq, R.; Basaria, A.A.A. Deep Infiltrating Endometriosis: A Pictorial Essay. J. Ultrasound Med. 2023. [Google Scholar] [CrossRef]
- Cozzolino, M.; Magro-Malosso, E.R.; Tofani, L.; Coccia, M.E. Evaluation of Sexual Function in Women with Deep Infiltrating Endometriosis. Sex. Reprod. Healthc. 2018, 16, 6–9. [Google Scholar] [CrossRef]
- Kvaskoff, M.; Mahamat-Saleh, Y.; Farland, L.V.; Shigesi, N.; Terry, K.L.; Harris, H.R.; Roman, H.; Becker, C.M.; As-Sanie, S.; Zondervan, K.T.; et al. Endometriosis and Cancer: A Systematic Review and Meta-Analysis. Hum. Reprod. Update 2021, 27, 393–420. [Google Scholar] [CrossRef] [PubMed]
- Cozzolino, M.; Nasioudis, D.; Sisti, G.; Coccia, M.E. Malignant Transformation of Vaginal Endometriosis—A Review of Literature. Gynecol. Obstet. Investig. 2017, 82, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Raffi, F.; Metwally, M.; Amer, S. The Impact of Excision of Ovarian Endometrioma on Ovarian Reserve: A Systematic Review and Meta-Analysis. J. Clin. Endocrinol. Metab. 2012, 97, 3146–3154. [Google Scholar] [CrossRef] [PubMed]
- Rasoul, N.S.; Al Allak, M.M. A Prospective Cohort Study on Laparoscopic Cystectomy of Endometrioma and Its Effects on Ovarian Reserve. J. Pak. Med. Assoc. 2021, 71 (Suppl. S9), S8–S11. [Google Scholar]
- Roman, H.; Tarta, O.; Pura, I.; Opris, I.; Bourdel, N.; Marpeau, L.; Sabourin, J.-C. Direct Proportional Relationship between Endometrioma Size and Ovarian Parenchyma Inadvertently Removed during Cystectomy, and Its Implication on the Management of Enlarged Endometriomas. Hum. Reprod. 2010, 25, 1428–1432. [Google Scholar] [CrossRef] [PubMed]
- Muzii, L.; Marana, R.; Angioli, R.; Bianchi, A.; Cucinella, G.; Vignali, M.; Benedetti Panici, P.; Busacca, M. Histologic Analysis of Specimens from Laparoscopic Endometrioma Excision Performed by Different Surgeons: Does the Surgeon Matter? Fertil. Steril. 2011, 95, 2116–2119. [Google Scholar] [CrossRef]
- Cobo, A.; García-Velasco, J.A.; Remohí, J.; Pellicer, A. Oocyte Vitrification for Fertility Preservation for Both Medical and Nonmedical Reasons. Fertil. Steril. 2021, 115, 1091–1101. [Google Scholar] [CrossRef]
- Chen, C. Pregnancy after Human Oocyte Cryopreservation. Lancet 1986, 1, 884–886. [Google Scholar] [CrossRef]
- Li, Z.; Wang, Y.A.; Ledger, W.; Edgar, D.H.; Sullivan, E.A. Clinical Outcomes Following Cryopreservation of Blastocysts by Vitrification or Slow Freezing: A Population-Based Cohort Study. Hum. Reprod. 2014, 29, 2794–2801. [Google Scholar] [CrossRef]
- Rienzi, L.; Gracia, C.; Maggiulli, R.; LaBarbera, A.R.; Kaser, D.J.; Ubaldi, F.M.; Vanderpoel, S.; Racowsky, C. Oocyte, Embryo and Blastocyst Cryopreservation in ART: Systematic Review and Meta-Analysis Comparing Slow-Freezing versus Vitrification to Produce Evidence for the Development of Global Guidance. Hum. Reprod. Update 2017, 23, 139–155. [Google Scholar] [CrossRef] [PubMed]
- Da Luz, C.M.; Caetano, M.A.; Berteli, T.S.; Vireque, A.A.; Navarro, P.A. The Impact of Oocyte Vitrification on Offspring: A Systematic Review. Reprod. Sci. 2022, 29, 3222–3234. [Google Scholar] [CrossRef] [PubMed]
- Walker, Z.; Lanes, A.; Ginsburg, E. Oocyte Cryopreservation Review: Outcomes of Medical Oocyte Cryopreservation and Planned Oocyte Cryopreservation. Reprod. Biol. Endocrinol. 2022, 20, 10. [Google Scholar] [CrossRef]
- Leung, A.Q.; Baker, K.; Vaughan, D.; Shah, J.S.; Korkidakis, A.; Ryley, D.A.; Sakkas, D.; Toth, T.L. Clinical Outcomes and Utilization from over a Decade of Planned Oocyte Cryopreservation. Reprod. Biomed. Online 2021, 43, 671–679. [Google Scholar] [CrossRef] [PubMed]
- Bourdon, M.; Maignien, C.; Pocate-Cheriet, K.; Plu Bureau, G.; Marcellin, L.; Patrat, C.; Chapron, C.; Santulli, P. The Freeze-All Strategy after IVF: Which Indications? Reprod. Biomed. Online 2021, 42, 529–545. [Google Scholar] [CrossRef] [PubMed]
- Cobo, A.; Giles, J.; Paolelli, S.; Pellicer, A.; Remohí, J.; García-Velasco, J.A. Oocyte Vitrification for Fertility Preservation in Women with Endometriosis: An Observational Study. Fertil. Steril. 2020, 113, 836–844. [Google Scholar] [CrossRef]
- Becker, C.M.; Bokor, A.; Heikinheimo, O.; Horne, A.; Jansen, F.; Kiesel, L.; King, K.; Kvaskoff, M.; Nap, A.; Petersen, K.; et al. ESHRE Guideline: Endometriosis. Hum. Reprod. Open. 2022, 2022, hoac009. [Google Scholar] [CrossRef]
- Fleming, R.; Seifer, D.B.; Frattarelli, J.L.; Ruman, J. Assessing Ovarian Response: Antral Follicle Count versus Anti-Müllerian Hormone. Reprod. Biomed. Online 2015, 31, 486–496. [Google Scholar] [CrossRef]
- Zhu, M.; Wang, S.; Yi, S.; Huang, X.; Meng, J.; Chen, L.; Sun, H.; Zhou, J. A Predictive Formula for Selecting Individual FSH Starting Dose Based on Ovarian Reserve Markers in IVF/ICSI Cycles. Arch. Gynecol. Obstet. 2019, 300, 441–446. [Google Scholar] [CrossRef]
- Romanski, P.A.; Brady, P.C.; Farland, L.V.; Thomas, A.M.; Hornstein, M.D. The Effect of Endometriosis on the Antimüllerian Hormone Level in the Infertile Population. J. Assist. Reprod. Genet. 2019, 36, 1179–1184. [Google Scholar] [CrossRef]
- Tian, Z.; Zhang, Y.; Zhang, C.; Wang, Y.; Zhu, H.-L. Antral Follicle Count Is Reduced in the Presence of Endometriosis: A Systematic Review and Meta-Analysis. Reprod. Biomed. Online 2021, 42, 237–247. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, A.M.; Viganò, P.; Somigliana, E.; Panina-Bordignon, P.; Vercellini, P.; Candiani, M. The Distinguishing Cellular and Molecular Features of the Endometriotic Ovarian Cyst: From Pathophysiology to the Potential Endometrioma-Mediated Damage to the Ovary. Hum. Reprod. Update 2014, 20, 217–230. [Google Scholar] [CrossRef] [PubMed]
- Kitajima, M.; Defrère, S.; Dolmans, M.-M.; Colette, S.; Squifflet, J.; Van Langendonckt, A.; Donnez, J. Endometriomas as a Possible Cause of Reduced Ovarian Reserve in Women with Endometriosis. Fertil. Steril. 2011, 96, 685–691. [Google Scholar] [CrossRef]
- Kitajima, M.; Dolmans, M.-M.; Donnez, O.; Masuzaki, H.; Soares, M.; Donnez, J. Enhanced Follicular Recruitment and Atresia in Cortex Derived from Ovaries with Endometriomas. Fertil. Steril. 2014, 101, 1031–1037. [Google Scholar] [CrossRef] [PubMed]
- Matsuzaki, S.; Pankhurst, M.W. Hyperactivation of Dormant Primordial Follicles in Ovarian Endometrioma Patients. Reproduction 2020, 160, R145–R153. [Google Scholar] [CrossRef] [PubMed]
- Dokras, A.; Habana, A.; Giraldo, J.; Jones, E. Secretion of Inhibin B during Ovarian Stimulation Is Decreased in Infertile Women with Endometriosis. Fertil. Steril. 2000, 74, 35–40. [Google Scholar] [CrossRef]
- Bianco, B.; Loureiro, F.A.; Trevisan, C.M.; Peluso, C.; Christofolini, D.M.; Montagna, E.; Laganà, A.S.; Barbosa, C.P. Effects of FSHR and FSHB Variants on Hormonal Profile and Reproductive Outcomes of Infertile Women With Endometriosis. Front. Endocrinol. 2021, 12, 760616. [Google Scholar] [CrossRef]
- Garcia-Velasco, J.A.; Domingo, J.; Cobo, A.; Martínez, M.; Carmona, L.; Pellicer, A. Five Years’ Experience Using Oocyte Vitrification to Preserve Fertility for Medical and Nonmedical Indications. Fertil. Steril. 2013, 99, 1994–1999. [Google Scholar] [CrossRef]
- Raad, J.; Sonigo, C.; Tran, C.; Sifer, C.; Durnerin, I.C.; Grynberg, M. Oocyte Vitrification for Preserving Fertility in Patients with Endometriosis: First Observational Cohort Study… and Many Unresolved Questions. Letter to the Editor. Eur. J. Obstet. Gynecol. Reprod. Biol. 2018, 220, 140–141. [Google Scholar] [CrossRef]
- Mathieu d’Argent, E.; Ferrier, C.; Zacharopoulou, C.; Ahdad-Yata, N.; Boudy, A.-S.; Cantalloube, A.; Levy, R.; Antoine, J.-M.; Daraï, E.; Bendifallah, S. Outcomes of Fertility Preservation in Women with Endometriosis: Comparison of Progestin-Primed Ovarian Stimulation versus Antagonist Protocols. J. Ovarian Res. 2020, 13, 18. [Google Scholar] [CrossRef]
- Kim, S.J.; Kim, S.K.; Lee, J.R.; Suh, C.S.; Kim, S.H. Oocyte Cryopreservation for Fertility Preservation in Women with Ovarian Endometriosis. Reprod. Biomed. Online 2020, 40, 827–834. [Google Scholar] [CrossRef]
- Santulli, P.; Bourdon, M.; Koutchinsky, S.; Maignien, C.; Marcellin, L.; Maitrot-Mantelet, L.; Pocate Cheriet, K.; Patrat, C.; Chapron, C. Fertility Preservation for Patients Affected by Endometriosis Should Ideally Be Carried out before Surgery. Reprod. Biomed. Online 2021, 43, 853–863. [Google Scholar] [CrossRef]
- Legrand, C.; Keller, L.; Collinet, P.; Barbotin, A.L.; Béhal, H.; Rubod, C.; Decanter, C. Oocyte Accumulation for Fertility Preservation in Women with Benign Ovarian Tumours with a History of Previous Surgery, Multiple or Large Cysts. Reprod. Biomed. Online 2021, 43, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.H.; Lee, H.K.; Kim, S.K.; Lee, J.R.; Suh, C.S. The Significance of Planned Fertility Preservation for Women With Endometrioma Before an Expected Ovarian Cystectomy. Front. Endocrinol. 2021, 12, 794117. [Google Scholar] [CrossRef] [PubMed]
- Elizur, S.E.; Aizer, A.; Yonish, M.; Shavit, T.; Orvieto, R.; Mashiach, R.; Cohen, S.B.; Berkowitz, E. Fertility Preservation for Women with Ovarian Endometriosis: Results from a Retrospective Cohort Study. Reprod. Biomed. Online 2023, 46, 332–337. [Google Scholar] [CrossRef] [PubMed]
- Nankali, A.; Kazeminia, M.; Jamshidi, P.K.; Shohaimi, S.; Salari, N.; Mohammadi, M.; Hosseinian-Far, A. The Effect of Unilateral and Bilateral Laparoscopic Surgery for Endometriosis on Anti-Mullerian Hormone (AMH) Level after 3 and 6 Months: A Systematic Review and Meta-Analysis. Health Qual. Life Outcomes 2020, 18, 314. [Google Scholar] [CrossRef]
- Moreno, J.A.; Romeral, C.; Niño, G.; Benavente, A.P. The Effect of Laparoscopic Endometrioma Surgery on Anti-Müllerian Hormone: A Systematic Review of the Literature and Meta-Analysis. JBRA Assist. Reprod. 2022, 26, 88. [Google Scholar] [CrossRef]
- Somigliana, E.; Berlanda, N.; Benaglia, L.; Viganò, P.; Vercellini, P.; Fedele, L. Surgical Excision of Endometriomas and Ovarian Reserve: A Systematic Review on Serum Antimüllerian Hormone Level Modifications. Fertil. Steril. 2012, 98, 1531–1538. [Google Scholar] [CrossRef]
- Guo, S.-W. Recurrence of Endometriosis and Its Control. Hum. Reprod. Update 2009, 15, 441–461. [Google Scholar] [CrossRef]
- Xu, X.; Chen, Y.; Yu, Q.; Wang, J.; Xu, P.; Zhu, L.; Xu, Q.; Zhang, J.; Cui, S.; Yu, K.; et al. The Presence of Living Endometrial Cells in Ovarian Endometriotic Cyst Fluid May Contribute to the Recurrence of Endometriosis after Surgical Excision of Endometriomas. J. Ovarian Res. 2022, 15, 89. [Google Scholar] [CrossRef]
- Ata, B.; Turkgeldi, E.; Seyhan, A.; Urman, B. Effect of Hemostatic Method on Ovarian Reserve Following Laparoscopic Endometrioma Excision; Comparison of Suture, Hemostatic Sealant, and Bipolar Dessication. A Systematic Review and Meta-Analysis. J. Minim. Invasive Gynecol. 2015, 22, 363–372. [Google Scholar] [CrossRef]
- Chung, J.P.W.; Law, T.S.M.; Mak, J.S.M.; Sahota, D.S.; Li, T.C. Ovarian Reserve and Recurrence 1 Year Post-Operatively after Using Haemostatic Sealant and Bipolar Diathermy for Haemostasis during Laparoscopic Ovarian Cystectomy. Reprod. Biomed. Online 2021, 43, 310–318. [Google Scholar] [CrossRef]
- Ferrero, S.; Scala, C.; Tafi, E.; Racca, A.; Venturini, P.L.; Leone Roberti Maggiore, U. Impact of Large Ovarian Endometriomas on the Response to Superovulation for in Vitro Fertilization: A Retrospective Study. Eur. J. Obstet. Gynecol. Reprod. Biol. 2017, 213, 17–21. [Google Scholar] [CrossRef] [PubMed]
- Muraoka, A.; Osuka, S.; Yabuki, A.; Bayasula; Yoshihara, M.; Tanaka, H.; Sonehara, R.; Miyake, N.; Murakami, M.; Yoshita, S.; et al. Impact of Perioperative Use of GnRH Agonist or Dienogest on Ovarian Reserve after Cystectomy for Endometriomas: A Randomized Controlled Trial. Reprod. Biol. Endocrinol. 2021, 19, 179. [Google Scholar] [CrossRef] [PubMed]
- Bourdon, M.; Santulli, P.; Bordonne, C.; Millisher, A.E.; Maitrot-Mantelet, L.; Maignien, C.; Marcellin, L.; Melka, L.; Chapron, C. Presence of Adenomyosis at MRI Reduces Live Birth Rates in ART Cycles for Endometriosis. Hum. Reprod. 2022, 37, 1470–1479. [Google Scholar] [CrossRef] [PubMed]
- Humaidan, P.; Garcia Velasco, J.A.; Cozzolino, M. Local Intraendometrial Estrogen Biosynthesis Leading to Progesterone Resistance Impacts Implantation in Adenomyosis and Endometriosis. Fertil. Steril. 2023, 120, 927. [Google Scholar] [CrossRef]
- Zimmermann, A.; Faust, C.; Miquel, L.; Berbis, J.; Perrin, J.; Courbiere, B. Impact of Moderate-to-Severe Endometriosis on IVF Cumulative Live Birth Rate: A Retrospective Matched Cohort Study. Reprod. Biomed. Online 2023, 47, 103186. [Google Scholar] [CrossRef]
- Dongye, H.; Tian, Y.; Qi, D.; Du, Y.; Yan, L. The Impact of Endometrioma on Embryo Quality in In Vitro Fertilization: A Retrospective Cohort Study. J. Clin. Med. 2023, 12, 2416. [Google Scholar] [CrossRef]
- Stoop, D.; Cobo, A.; Silber, S. Fertility Preservation for Age-Related Fertility Decline. Lancet 2014, 384, 1311–1319. [Google Scholar] [CrossRef]
- Cobo, A.; García-Velasco, J.A.; Coello, A.; Domingo, J.; Pellicer, A.; Remohí, J. Oocyte Vitrification as an Efficient Option for Elective Fertility Preservation. Fertil. Steril. 2016, 105, 755–764.e8. [Google Scholar] [CrossRef]
- Henry, L.; Vervier, J.; Boucher, A.; Brichant, G.; Gaspard, O.; Labied, S.; Munaut, C.; Ravet, S.; Nisolle, M. Oocyte Cryopreservation in Patients with Endometriosis: Current Knowledge and Number Needed to Treat. J. Clin. Med. 2022, 11, 4559. [Google Scholar] [CrossRef] [PubMed]
- Cecchino, G.N.; Cozzolino, M.; Roque, M.; García-Velasco, J.A. Endometrioma and Reproductive Issues: A Well-Informed Patient May Be the Driver for Change. Minerva Ginecol. 2020, 72, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Adamczyk, M.; Wender-Ozegowska, E.; Kedzia, M. Epigenetic Factors in Eutopic Endometrium in Women with Endometriosis and Infertility. Int. J. Mol. Sci. 2022, 23, 3804. [Google Scholar] [CrossRef] [PubMed]
| First Author, Year | Study Design | Number of Endometriosis Women; Number of Cycles | Stimulation Protocol | Number of MII Oocytes |
|---|---|---|---|---|
| Garcia-Velasco, 2013 [48] | Retrospective, multicenter, observational study | 38; N/A | Antagonist protocol | - |
| Raad, 2018 [49] | Retrospective Cohort | 49; 70 | Antagonist or long agonist protocols | 7.2 ± 4.9 |
| Cobo, 2020 [36] | Retrospective cohort study | 485; 840 | Antagonist or agonist protocols | 5.5 ± 5.2 |
| D’Argent, 2020 [50] | Prospective cohort study | 108; 108 | 54 women were stimulated with an antagonist protocol; 54 with a PPOS protocol | - |
| Kim, 2020 [51] | Retrospective Cohort | 34; 50 | Antagonist protocol | 4.1 ± 3.1 |
| Santulli, 2021 [52] | Retrospective Cohort | 146; 258 | Long agonist, short agonist or antagonist protocol | 6.0 ± 4.7 |
| Legrand, 2021 [53] | Retrospective cohort | 70; 113 | Agonist or antagonist protocol | - |
| Yeon Hee Hong, 2021 [54] | Retrospective Cohort | 62; 95 | Antagonist protocol | 3.0 (1.5–5.0) |
| Elizur, 2023 [55] | Retrospective Cohort | 71; 138 | Antagonist protocol | 6 (3–10) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mifsud, J.-M.; Pellegrini, L.; Cozzolino, M. Oocyte Cryopreservation in Women with Ovarian Endometriosis. J. Clin. Med. 2023, 12, 6767. https://doi.org/10.3390/jcm12216767
Mifsud J-M, Pellegrini L, Cozzolino M. Oocyte Cryopreservation in Women with Ovarian Endometriosis. Journal of Clinical Medicine. 2023; 12(21):6767. https://doi.org/10.3390/jcm12216767
Chicago/Turabian StyleMifsud, Judith-Marie, Livia Pellegrini, and Mauro Cozzolino. 2023. "Oocyte Cryopreservation in Women with Ovarian Endometriosis" Journal of Clinical Medicine 12, no. 21: 6767. https://doi.org/10.3390/jcm12216767
APA StyleMifsud, J.-M., Pellegrini, L., & Cozzolino, M. (2023). Oocyte Cryopreservation in Women with Ovarian Endometriosis. Journal of Clinical Medicine, 12(21), 6767. https://doi.org/10.3390/jcm12216767






