Vagus Nerve Stimulation for Drug Resistant Epilepsy: Clinical Outcome, Adverse Events, and Potential Prognostic Factors in a Single Center Experience
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Data Collection
2.2. Device Implantation and Settings
2.3. Statistical Analysis
- Age of onset of epilepsy: 1–51 years old;
- Age at implant: 5–54 years old;
- Duration of epilepsy prior to VNS: 0.25–39 years;
- Gender: female (n = 15), male (n = 30);
- Pre-implantation number of ASMs: ≥3 (n = 24), =2 (n = 21);
- Dominant seizure type: generalized (n = 28) and focal (n = 17).
3. Results
3.1. Demographic Data
3.2. Device Parameters
3.3. Clinical Outcomes
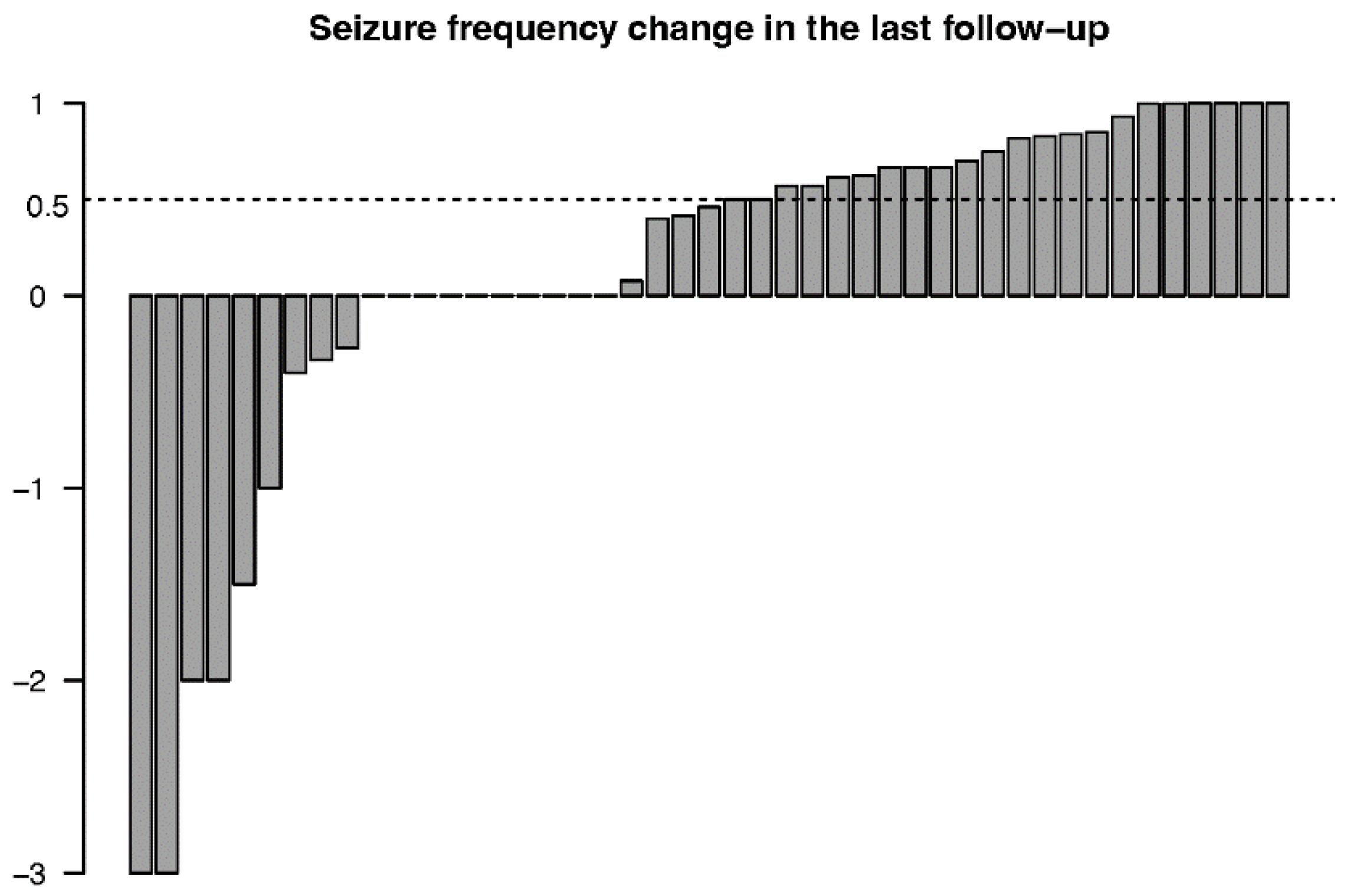
3.3.1. Seizure Outcomes
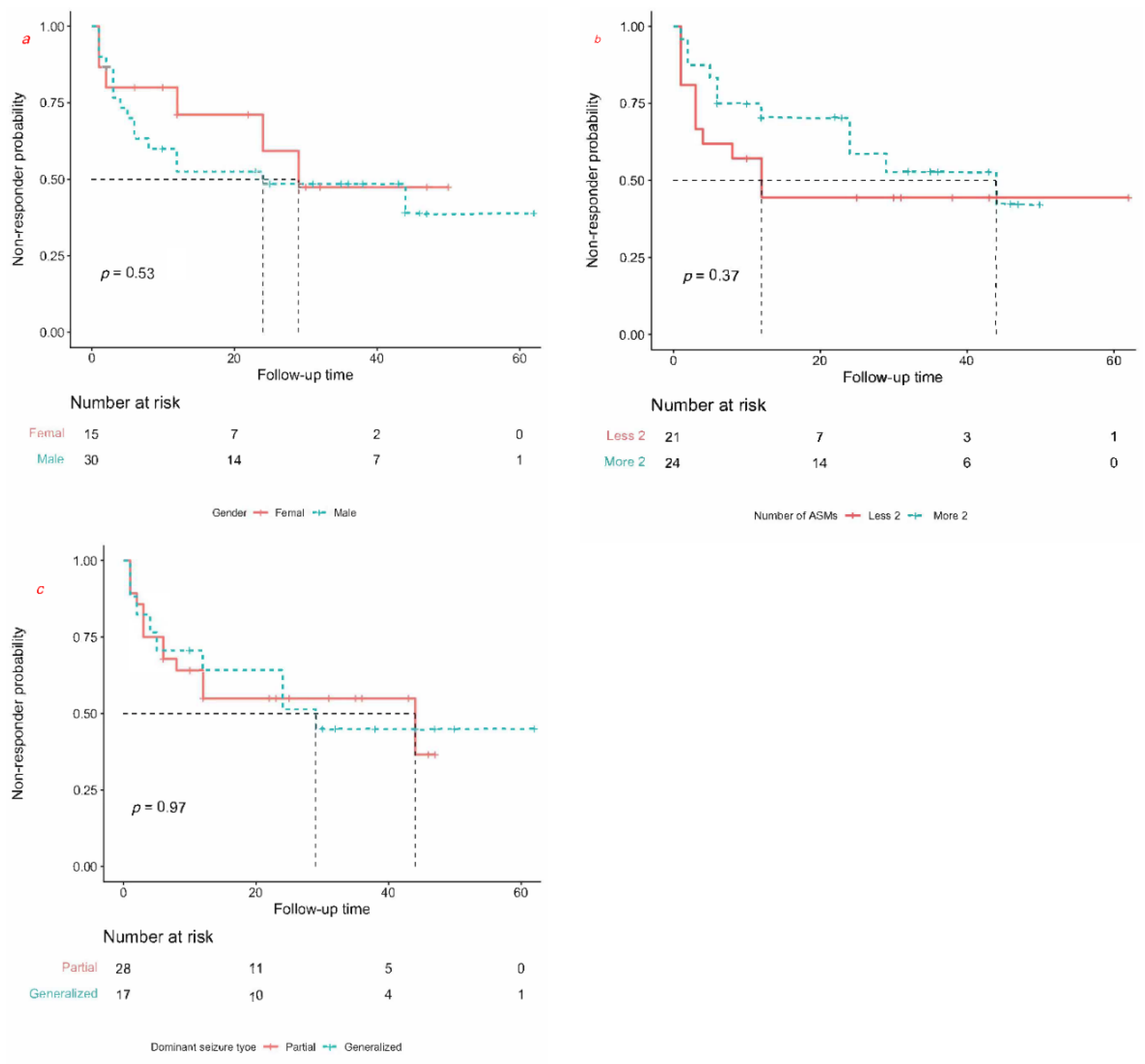
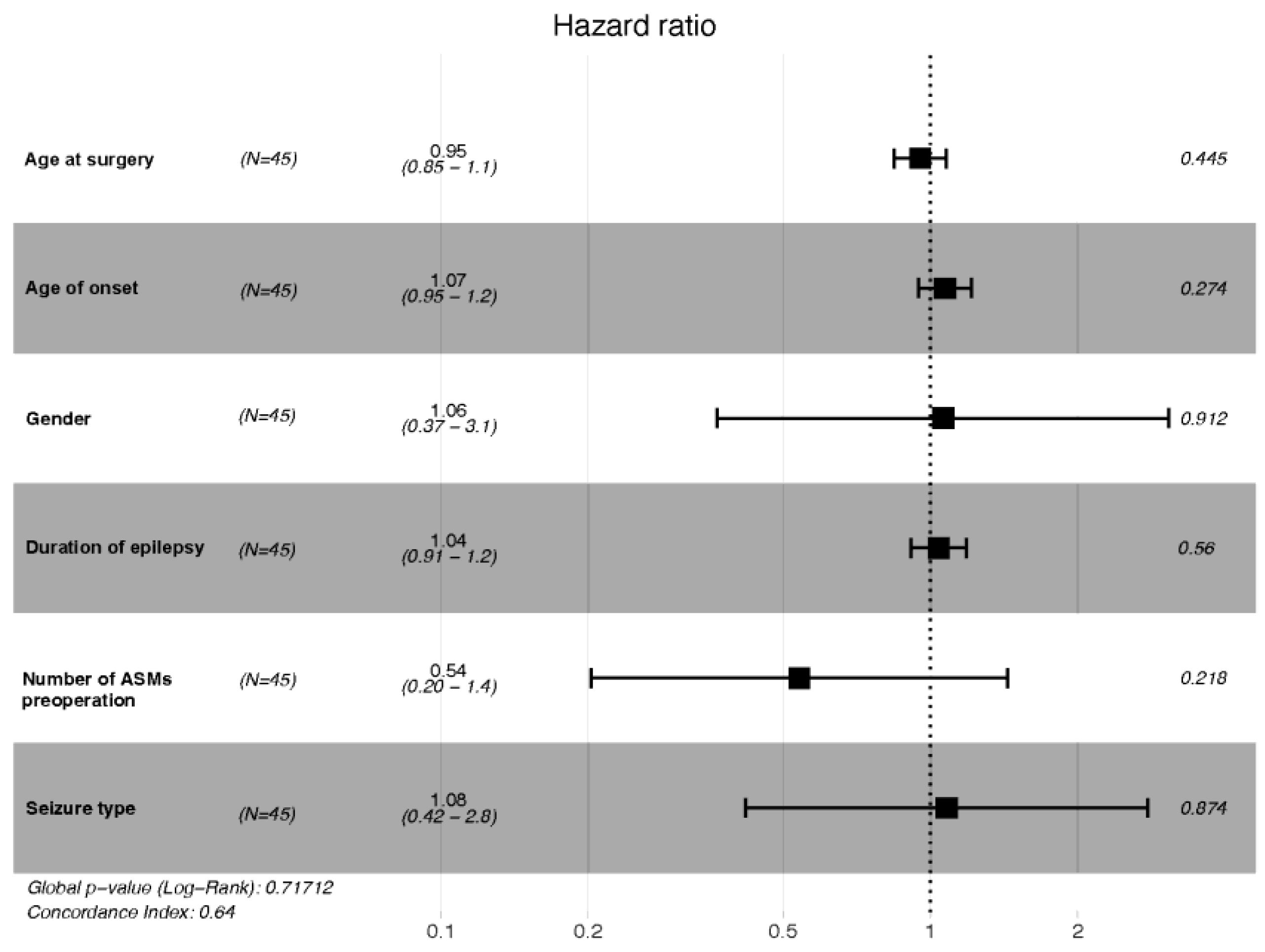
3.3.2. Prognostic Factors
3.3.3. Improvement in Quality of Life
3.3.4. Number of Anti-Seizure Medications
3.3.5. Adverse Events
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Beghi, E. The epidemiology of epilepsy. Neuroepidemiology 2020, 54, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Fisher, R.S.; van Emde Boas, W.; Blume, W.; Elger, C.; Genton, P.; Lee, P.; Engel, J., Jr. Epileptic seizures and epilepsy: Definitions proposed by the International League against Epilepsy (ILAE) and the International Bureau for Epilepsy (IBE). Epilepsia 2005, 46, 470–472. [Google Scholar] [CrossRef]
- Bastos, F.; Cross, J.H. Epilepsy. Handb. Clin. Neurol. 2020, 174, 137–158. [Google Scholar] [PubMed]
- Kwan, P.; Arzimanoglou, A.; Berg, A.T.; Brodie, M.J.; Allen Hauser, W.; Mathern, G.; Moshé, S.L.; Perucca, E.; Wiebe, S.; French, J. Definition of drug resistant epilepsy: Consensus proposal by the ad hoc Task Force of the ILAE Commission on Therapeutic Strategies. Epilepsia 2010, 51, 1069–1077. [Google Scholar] [CrossRef] [PubMed]
- Kawai, K.; Tanaka, T.; Baba, H.; Bunker, M.; Ikeda, A.; Inoue, Y.; Kameyama, S.; Kaneko, S.; Kato, A.; Nozawa, T.; et al. Outcome of vagus nerve stimulation for drug-resistant epilepsy: The first three years of a prospective Japanese registry. Epileptic Disord. 2017, 19, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Bodin, E.; Le Moing, A.G.; Bourel-Ponchel, E.; Querne, L.; Toussaint, P.; Berquin, P. Vagus nerve stimulation in the treatment of drug-resistant epilepsy in 29 children. Eur. J. Paediatr. Neurol. 2016, 20, 346–351. [Google Scholar] [CrossRef]
- Pakdaman, H.; Amini Harandi, A.; Abbasi, M.; Karimi, M.; Arami, M.; Mosavi, S.; Haddadian, K.; Rezaei, O.; Sadeghi, S.; Sharifi, G.; et al. Vagus nerve stimulation in drug-resistant epilepsy: The efficacy and adverse effects in a 5-year follow-up study in Iran. Neurol. Sci. 2016, 37, 1773–1778. [Google Scholar] [CrossRef]
- Kayyali, H.; Abdelmoity, S.; Bansal, L.; Kaufman, C.; Smith, K.; Fecske, E.; Pawar, K.; Hall, A.; Gustafson, M.; Abdelmoity, A.; et al. The efficacy and safety of rapid cycling vagus nerve stimulation in children with intractable epilepsy. Pediatr. Neurol. 2020, 109, 35–38. [Google Scholar] [CrossRef]
- Soleman, J.; Stein, M.; Knorr, C.; Datta, A.N.; Constantini, S.; Fried, I.; Guzman, R.; Kramer, U. Improved quality of life and cognition after early vagal nerve stimulator implantation in children. Epilepsy Behav. 2018, 88, 139–145. [Google Scholar]
- Englot, D.J.; Hassnain, K.H.; Rolston, J.D.; Harward, S.C.; Sinha, S.R.; Haglund, M.M. Quality-of-life metrics with vagus nerve stimulation for epilepsy from provider survey data. Epilepsy Behav. 2017, 66, 4–9. [Google Scholar] [CrossRef]
- Brodtkorb, E.; Samsonsen, C.; Jørgensen, J.V.; Helde, G. Epilepsy patients with and without perceived benefit from vagus nerve stimulation: A long-term observational single center study. Seizure 2019, 72, 28–32. [Google Scholar] [CrossRef] [PubMed]
- Arhan, E.; Serdaroglu, A.; Kurt, G.; Bilir, E.; Durdağ, E.; Erdem, A.; Aksakal, F.N.; Ozcelik, A.; Baykaner, K. The efficacy of vagal nerve stimulation in children with pharmacoresistant epilepsy: Practical experience at a Turkish tertiary referral center. Eur. J. Paediatr. Neurol. 2010, 14, 334–339. [Google Scholar] [CrossRef] [PubMed]
- Vale, F.L.; Ahmadian, A.; Youssef, A.S.; Tatum, W.O.; Benbadis, S.R. Long-term outcome of vagus nerve stimulation therapy after failed epilepsy surgery. Seizure 2011, 20, 244–248. [Google Scholar] [CrossRef] [PubMed]
- Arcos, A.; Romero, L.; Gelabert, M.; Prieto, A.; Pardo, J.; Osorio, X.R.; Arráez, M.A. Can we predict the response in the treatment of epilepsy with vagus nerve stimulation? Neurosurg. Rev. 2014, 37, 661–668. [Google Scholar] [CrossRef]
- Colicchio, G.; Montano, N.; Fuggetta, F.; Papacci, F.; Signorelli, F.; Meglio, M. Vagus nerve stimulation in drug-resistant epilepsies. Analysis of potential prognostic factors in a cohort of patients with long-term follow-up. Acta Neurochir. 2012, 154, 2237–2240. [Google Scholar] [CrossRef]
- Scheffer, I.E.; Berkovic, S.; Capovilla, G.; Connolly, M.B.; French, J.; Guilhoto, L.; Hirsch, E.; Jain, S.; Mathern, G.W.; Moshé, S.L.; et al. ILAE classification of the epilepsies: Position paper of the ILAE commission for classification and terminology. Epilepsia 2017, 58, 512–521. [Google Scholar] [CrossRef]
- Mao, H.; Chen, Y.; Ge, Q.; Ye, L.; Cheng, H. Short- and long-term response of vagus nerve stimulation therapy in drug-resistant epilepsy: A systematic review and meta-analysis. Neuromodulation 2022, 25, 327–342. [Google Scholar] [CrossRef]
- Lagae, L.; Verstrepen, A.; Nada, A.; Van Loon, J.; Theys, T.; Ceulemans, B.; Jansen, K. Vagus nerve stimulation in children with drug-resistant epilepsy: Age at implantation and shorter duration of epilepsy as predictors of better efficacy? Epileptic Disord. 2015, 17, 308–314. [Google Scholar] [CrossRef]
- Wang, H.-J.; Tan, G.; Zhu, L.-N.; Chen, D.; Xu, D.; Chu, S.-S.; Liu, L. Predictors of seizure reduction outcome after vagus nerve stimulation in T drug-resistant epilepsy. Seizure 2019, 66, 53–60. [Google Scholar] [CrossRef]
- Englot, D.J.; Rolston, J.D.; Wright, C.W.; Hassnain, K.H.; Chang, E.F. Rates and predictors of seizure freedom with vagus nerve stimulation for intractable epilepsy. Neurosurgery 2016, 79, 345–353. [Google Scholar] [CrossRef]
- Xu, C.; Lin, H.; Xu, J.; Zhang, X.; Hao, G.; Liu, Q.Q.; Ding, C.; Wang, S.; Zhao, Q.; Bai, X.; et al. Long-term outcomes and prognosis factors of vagus nerve stimulation in patients with refractory epilepsy. Acta Epileptol. 2022, 4, 38. [Google Scholar] [CrossRef]
- Englot, D.J.; Chang, E.F.; Auguste, K.I. Vagus nerve stimulation for epilepsy: A meta-analysis of efficacy and predictors of response. J. Neurosurg. 2011, 115, 1248–1255. [Google Scholar] [CrossRef] [PubMed]
- Ryvlin, P.; Gilliam, F.G.; Nguyen, D.K.; Colicchio, G.; Iudice, A.; Tinuper, P.; Zamponi, N.; Aguglia, U.; Wagner, L.; Minotti, L.; et al. The long-term effect of vagus nerve stimulation on quality of life in patients with pharmacoresistant focal epilepsy: The PuLsE (Open Prospective Randomized Long-term Effectiveness) trial. Epilepsia 2014, 55, 893–900. [Google Scholar] [CrossRef] [PubMed]
- Elliott, R.E.; Morsi, A.; Tanweer, O.; Grobelny, B.; Geller, E.; Carlson, C.; Devinsky, O.; Doyle, W.K. Efficacy of vagus nerve stimulation over time: Review of 65 consecutive patients with treatment-resistant epilepsy treated with VNS N10 years. Epilepsy Behav. 2011, 20, 478–483. [Google Scholar] [CrossRef]
- Orosz, I.; McCormick, D.; Zamponi, N.; Varadkar, S.; Feucht, M.; Parain, D.; Griens, R.; Vallée, L.; Boon, P.; Rittey, C.; et al. Vagus nerve stimulation for drug-resistant epilepsy: A European long-term study up to 24 months in 347 children. Epilepsia 2014, 55, 1576–1584. [Google Scholar] [CrossRef]
- Meng, F.G.; Jia, F.M.; Ren, X.H.; Ge, Y.; Wang, K.L.; Ma, Y.S.; Ge, M.; Zhang, K.; Hu, W.H.; Zhang, X.; et al. Vagus nerve stimulation for pediatric and adult patients with pharmaco-resistant epilepsy. Chin. Med. J. 2015, 128, 2599–2604. [Google Scholar] [CrossRef]

| Male/Female; n (%) | 30/15 (67.3%/32.7%) |
|---|---|
| Age at seizure onset (years); average ± SD | 15.5 ± 11.0 |
| Age at VNS implantation (years); average ± SD | 27.9 ± 11.9 |
| Duration of epilepsy (years); average ± SD | 11.8 ± 9.6 |
| Duration of follow-up period (months); range | 5.0–60.0 |
| Etiology; n (%) | |
| Genetic | 0 (0) |
| Structural | 20 (44.4%) |
| Infectious | 11 (24.4%) |
| Metabolic | 0(0) |
| Immune | 0 (0) |
| Unknown | 14(31.1%) |
| Dominant seizure type; n (%) | |
| Focal | 17 (37.8%) |
| Generalized | 28 (62.2%) |
| History of epilepsy-related surgery; n (%) | |
| Yes | 2 (4.4%) |
| No | 43 (95.6%) |
| Number of ASMs prior to VNS implantation; n (%) | |
| ≥3 | 24 (53.3%) |
| 2 | 21 (46.7%) |
| Etiology of Epilepsy | n |
|---|---|
| Post-traumatic | 6 |
| Meningoencephalitis | 11 |
| Brain tumor | 2 |
| Perinatal hypoxic ischemic encephalopathy | 1 |
| Hippocampal sclerosis | 3 |
| Cortical atrophy | 2 |
| Encephalomalacia foci | 6 |
| Unknown | 14 |
| McHugh Classification | Description | Number (%) |
|---|---|---|
| I | 80–100% reduction in seizure frequency | 11 (24.4%) |
| II | 50–79% reduction in seizure frequency | 11 (24.4%) |
| III | <50% reduction in seizure frequency | 4 (8.9%) |
| IV | Magnet benefit only | 10 (22.2%) |
| V | No benefit | 9 (20.0%) |
| Etiology | Responder (n = 22) | Total (n = 45) | Fisher’s Exact Test | |
|---|---|---|---|---|
| Yes | No | |||
| Post-traumatic | 5 | 1 | 6 | p = 0.096 |
| Meningoencephalitis | 6 | 5 | 11 | p = 0.738 |
| Brain tumor | 0 | 2 | 2 | p = 0.489 |
| Perinatal hypoxic ischemic encephalopathy | 0 | 1 | 1 | p > 0.999 |
| Hippocampal sclerosis | 2 | 1 | 3 | p = 0.608 |
| Cortical atrophy | 0 | 2 | 2 | p = 0.489 |
| Encephalomalacia | 1 | 5 | 6 | p = 0.187 |
| Unknown | 8 | 6 | 14 | p = 0.530 |
| Crude HR (95 CI%) | Adjust HR (95 CI%) | p (Wald’s Test) | p (LR-Test) | |
|---|---|---|---|---|
| Age at surgery | 1.01 (0.98, 1.04) | 0.95 (0.85, 1.08) | 0.445 | 0.380 |
| Age at onset | 1.02 (0.99, 1.05) | 1.07 (0.95, 1.22) | 0.274 | 0.180 |
| Gender | 0.74 (0.29, 1.9) | 1.06 (0.37, 3.08) | 0.912 | 0.912 |
| Duration of epilepsy | 0.99 (0.94, 1.04) | 1.04 (0.91, 1.19) | 0.560 | 0.519 |
| Number of ASMs | 0.68 (0.29, 1.56) | 0.54 (0.2, 1.44) | 0.218 | 0.216 |
| Seizure type | 1.02 (0.43, 2.41) | 1.08 (0.42, 2.78) | 0.874 | 0.874 |
| Adverse Event | Number (n = 26) | Incidence (%) |
|---|---|---|
| Hoarseness | 10 | 22.2% |
| Discomfort in surgical site | 5 | 11.1% |
| Coughing | 4 | 8.9% |
| Nausea | 2 | 4.4% |
| Paresthesia | 2 | 4.4% |
| Dyspnea | 2 | 4.4% |
| Cervical muscle spasm | 1 | 2.2% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shan, M.; Mao, H.; Xie, H.; Gan, Y.; Wu, D.; Song, J.; Bai, Y.; Zhang, J. Vagus Nerve Stimulation for Drug Resistant Epilepsy: Clinical Outcome, Adverse Events, and Potential Prognostic Factors in a Single Center Experience. J. Clin. Med. 2022, 11, 7536. https://doi.org/10.3390/jcm11247536
Shan M, Mao H, Xie H, Gan Y, Wu D, Song J, Bai Y, Zhang J. Vagus Nerve Stimulation for Drug Resistant Epilepsy: Clinical Outcome, Adverse Events, and Potential Prognostic Factors in a Single Center Experience. Journal of Clinical Medicine. 2022; 11(24):7536. https://doi.org/10.3390/jcm11247536
Chicago/Turabian StyleShan, Ming, Hongliang Mao, Hutao Xie, Yifei Gan, Delong Wu, Jian Song, Yutong Bai, and Jianguo Zhang. 2022. "Vagus Nerve Stimulation for Drug Resistant Epilepsy: Clinical Outcome, Adverse Events, and Potential Prognostic Factors in a Single Center Experience" Journal of Clinical Medicine 11, no. 24: 7536. https://doi.org/10.3390/jcm11247536
APA StyleShan, M., Mao, H., Xie, H., Gan, Y., Wu, D., Song, J., Bai, Y., & Zhang, J. (2022). Vagus Nerve Stimulation for Drug Resistant Epilepsy: Clinical Outcome, Adverse Events, and Potential Prognostic Factors in a Single Center Experience. Journal of Clinical Medicine, 11(24), 7536. https://doi.org/10.3390/jcm11247536





