The Reflux and BariClip: Initial Results and Mechanism of Action
Abstract
:1. Introduction
2. Materials and Methods
Surgical Technique
3. Results
Post-Operative Complications
4. Discussion
- During the dissection, there is no anatomical damage of the ring and longitudinal fibers at the gastroesophageal junction. During the preclinical studies, a particular attention was paid to the closing pressure of the device. The BariClip is designed to minimize the closing force so that the limbs will simply oppose the anterior and posterior walls of the stomach to minimize the possibility of erosions and ischemia; the closing of the BariClip has been designed to be a low-pressure system.
- Minimal dissection of the His angle with no damage of the phreno-esophageal membrane. The posterior passage is realized with a minimal dissection in an avascular plane.
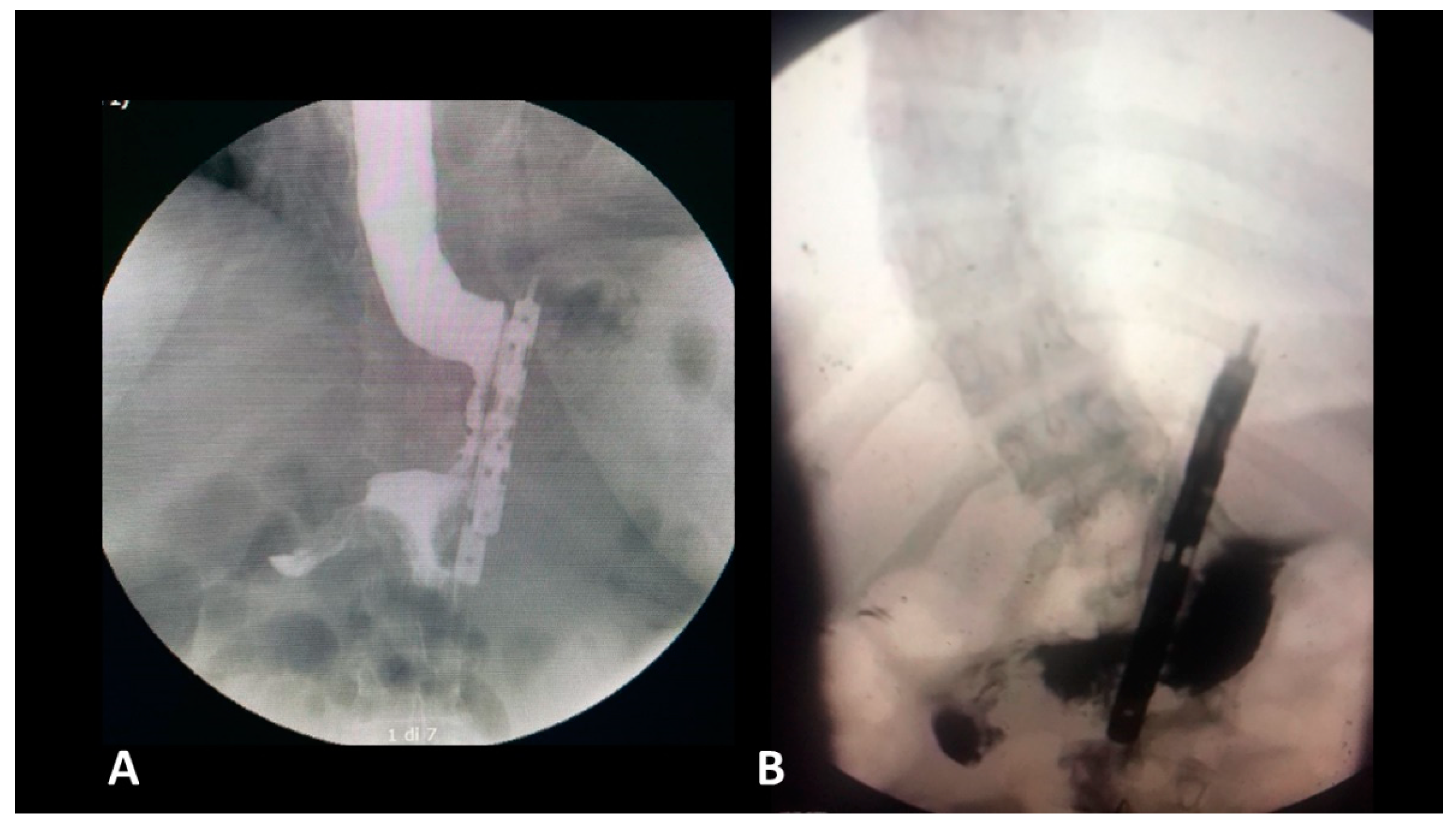
- The presence of a distal opening at the bottom of the BariClip makes the procedure a low-pressure system. It balances the pressures with the excluded part of the stomach, thus allowing for a possible acid or bile reflux to go towards the fundus, to the excluded portion of the stomach and not the esophagus, like in the illustrated upper GI swallow (Figure 2).
- A careful selection of patients is important. LBCG has not been performed in patients with large hiatal hernia.
- Compared with LSG, during LBCG, there is no section of the longitudinal fibers of the stomach. This type of gastric muscle fibers is involved in the gastric emptying. The shape of the gastric tube and the asymmetry between the anterior and the posterior parts could be an important factor for GERD. This risk factor is completely absent for LBCG.
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Angrisani, L.; Santonicola, A.; Iovino, P.; Ramos, A.; Shikora, S.; Kow, L. Bariatric Surgery Survey 2018: Similarities and Disparities Among the 5 IFSO Chapters. Obes. Surg. 2021, 31, 1937–1948. [Google Scholar] [CrossRef]
- Higa, K.; Ho, T.; Tercero, F.; Yunus, T.; Boone, K.B. Laparoscopic Roux-en-Y gastric bypass: 10-year follow-up. Surg. Obes. Relat. Dis. 2011, 7, 516–525. [Google Scholar] [CrossRef]
- Fezzi, M.; Kolotkin, R.L.; Nedelcu, M.; Jaussent, A.; Schaub, R.; Chauvet, M.A.; Cassafieres, C.; Lefebvre, P.; Renard, E.; Bringer, J.; et al. Improvement in quality of life after laparoscopic sleeve gastrectomy. Obes. Surg. 2011, 21, 1161–1167. [Google Scholar] [CrossRef] [PubMed]
- Arman, G.A.; Himpens, J.; Dhaenens, J.; Ballet, T.; Vilallonga, R.; Leman, G. Long-term (11+ years) outcomes in weight, patient satisfaction, comorbidities, and gastroesophageal reflux treatment after laparoscopic sleeve gastrectomy. Surg. Obes. Relat. Dis. 2016, 12, 1778–1786. [Google Scholar] [CrossRef] [PubMed]
- Genco, A.; Soricelli, E.; Casella, G.; Maselli, R.; Castagneto-Gissey, L.; Di Lorenzo, N.; Basso, N. Gastroesophageal reflux disease and Barrett’s esophagus after laparoscopic sleeve gastrectomy: A possible, underestimated long-term complication. Surg. Obes. Relat. Dis. 2017, 13, 568–574. [Google Scholar] [CrossRef]
- Assalia, A.; Gagner, M.; Nedelcu, M.; Ramos, A.C.; Nocca, D. Gastroesophageal Reflux and Laparoscopic Sleeve Gastrectomy: Results of the First International Consensus Conference. Obes. Surg. 2020, 30, 3695–3705. [Google Scholar] [CrossRef]
- Bou Daher, H.; Sharara, A.I. Gastroesophageal reflux disease, obesity and laparoscopic sleeve gastrectomy: The burning questions. World J. Gastroenterol. 2019, 25, 4805–4813. [Google Scholar] [CrossRef] [PubMed]
- Guzman-Pruneda, F.A.; Brethauer, S.A. Gastroesophageal Reflux After Sleeve Gastrectomy. J. Gastrointest. Surg. 2021, 25, 542–550. [Google Scholar] [CrossRef] [PubMed]
- Stenard, F.; Iannelli, A. Laparoscopic sleeve gastrectomy and gastroesophageal reflux. World J. Gastroenterol. 2015, 28, 10348–10357. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, M.; Zundel, N.; Plasencia, G.; Rodriguez-Pumarol, P.; Gomez, E.; Leithead, J., 3rd. A Vertically Placed Clip for Weight Loss: A 39-Month Pilot Study. Obes. Surg. 2017, 27, 1174–1181. [Google Scholar] [CrossRef] [PubMed]
- Manos, T.; Noel, P.; Bastid, C.; Vilallonga, R.; Nedelcu, M.; Nedelcu, A. Endoscopic Gastroplasty. Initial Experience. Chirurgia 2019, 114, 747–752. [Google Scholar] [CrossRef] [PubMed]
- Pizzicannella, M.; Fiorillo, C.; Barberio, M.; Rodriguez-Luna, M.R.; Vix, M.; Mutter, D.; Marescaux, J.; Costamagna, G.; Swantrom, L.; Perretta, S. Endoscopic assessment of morphological and histopathological upper gastrointestinal changes after endoscopic sleeve gastroplasty. Surg. Obes. Relat. Dis. 2021, 17, 1294–1301. [Google Scholar] [CrossRef] [PubMed]
- Noel, P.; Nedelcu, A.M.; Eddbali, I.; Zundel, N. Laparoscopic vertical clip gastroplasty-quality of life. Surg. Obes. Relat. Dis. 2018, 14, 1587–1593. [Google Scholar] [CrossRef] [PubMed]
- Jones, R.; Junghard, O.; Dent, J.; Vakil, N.; Halling, K.; Wernersson, B.; Lind, T. Development of the GerdQ, a tool for the diagnosis and management of gastro-oesophageal reflux disease in primary care. Aliment. Pharm. Ther. 2009, 30, 1030–1038. [Google Scholar] [CrossRef] [PubMed]
- Sebastianelli, L.; Benois, M.; Vanbiervliet, G.; Bailly, L.; Robert, M.; Turrin, N.; Gizard, E.; Foletto, M.; Bisello, M.; Albanese, A.; et al. Systematic Endoscopy 5 Years After Sleeve Gastrectomy Results in a High Rate of Barrett’s Esophagus: Results of a Multicenter Study. Obes Surg. 2019, 21, 1462–1469. [Google Scholar] [CrossRef] [PubMed]
- Nocca, D.; Skalli, E.M.; Boulay, E.; Nedelcu, M.; Michel Fabre, J.; Loureiro, M. Nissen Sleeve (N-Sleeve) operation: Preliminary results of a pilot study. Surg. Obes. Relat. Dis. 2016, 12, 1832–1837. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.; Liu, G.; Miller, L.; Ma, C.; Xu, W.; Schlachta, C.M.; Darling, G. Lack of correlation between a self-administered subjective GERD questionnaire and pathologic GERD diagnosed by 24-h esophageal pH monitoring. J. Gastrointest. Surg. J. Soc. Surg. Aliment. Tract. 2010, 14, 427–436. [Google Scholar] [CrossRef] [PubMed]
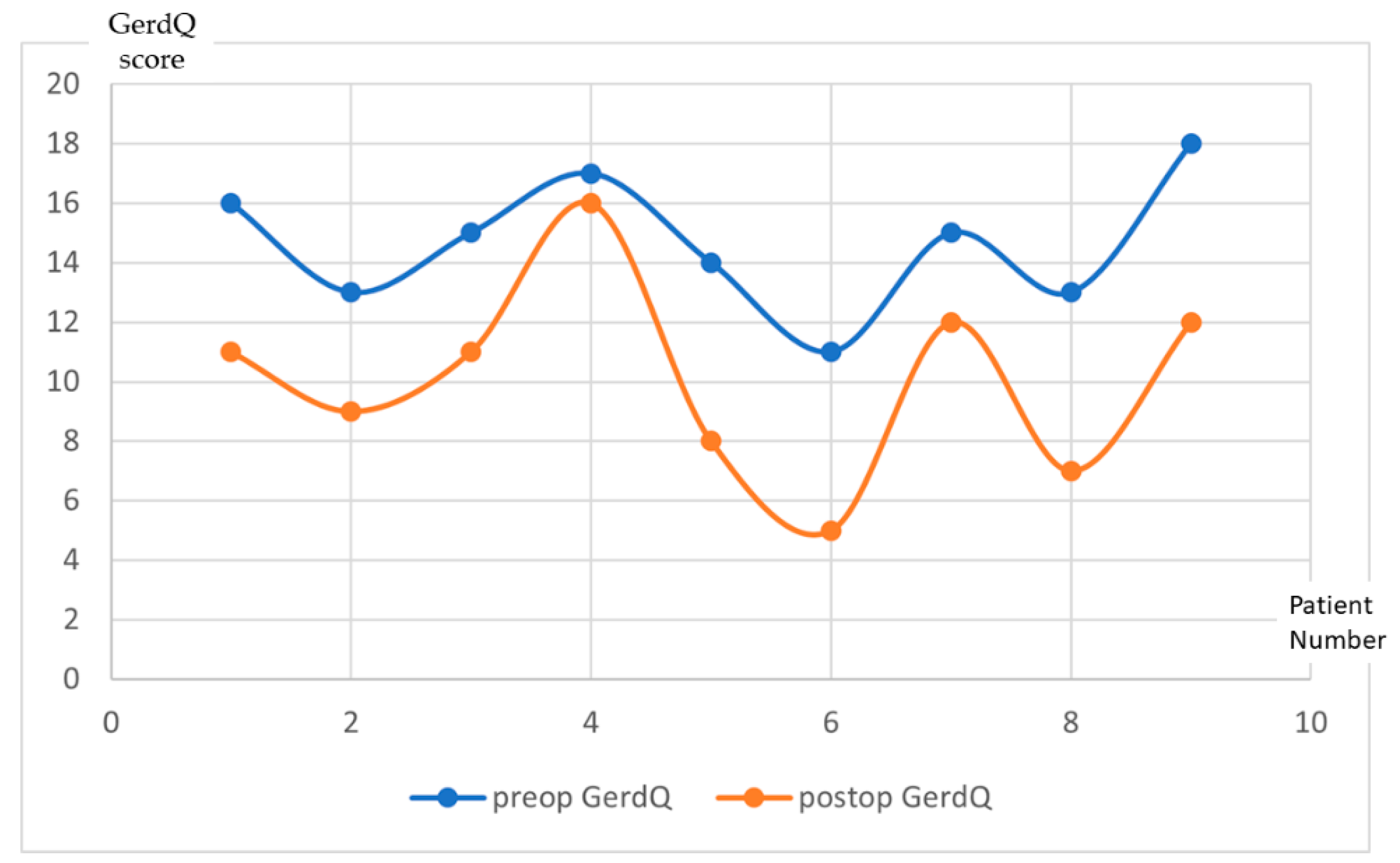
| Preoperatively | One Year Postoperatively | |
|---|---|---|
| GERD with systematic PPI use | 8 patients | 1 patient |
| GERD with occasional PPI use | 4 patients | 6 patients |
| De novo GERD | - | 1 patient |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Noel, P.; Layani, L.; Manos, T.; Adala, M.; Carandina, S.; Nedelcu, A.; Nedelcu, M. The Reflux and BariClip: Initial Results and Mechanism of Action. J. Clin. Med. 2022, 11, 6698. https://doi.org/10.3390/jcm11226698
Noel P, Layani L, Manos T, Adala M, Carandina S, Nedelcu A, Nedelcu M. The Reflux and BariClip: Initial Results and Mechanism of Action. Journal of Clinical Medicine. 2022; 11(22):6698. https://doi.org/10.3390/jcm11226698
Chicago/Turabian StyleNoel, Patrick, Laurent Layani, Thierry Manos, Mourad Adala, Sergio Carandina, Anamaria Nedelcu, and Marius Nedelcu. 2022. "The Reflux and BariClip: Initial Results and Mechanism of Action" Journal of Clinical Medicine 11, no. 22: 6698. https://doi.org/10.3390/jcm11226698
APA StyleNoel, P., Layani, L., Manos, T., Adala, M., Carandina, S., Nedelcu, A., & Nedelcu, M. (2022). The Reflux and BariClip: Initial Results and Mechanism of Action. Journal of Clinical Medicine, 11(22), 6698. https://doi.org/10.3390/jcm11226698






