Involvement of Peripheral Monocytes with IL-1β in the Pathogenesis of West Syndrome
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.1.1. Inclusion Criteria
2.1.2. Exclusion Criteria
2.2. Blood Sampling, Isolation, and Activation of Lymphocytes
2.3. Flow Cytometry Analysis
2.4. Analysis of Cytokine Profiles in Plasma
2.5. Statistical Analysis
3. Results
3.1. Patient Demographics
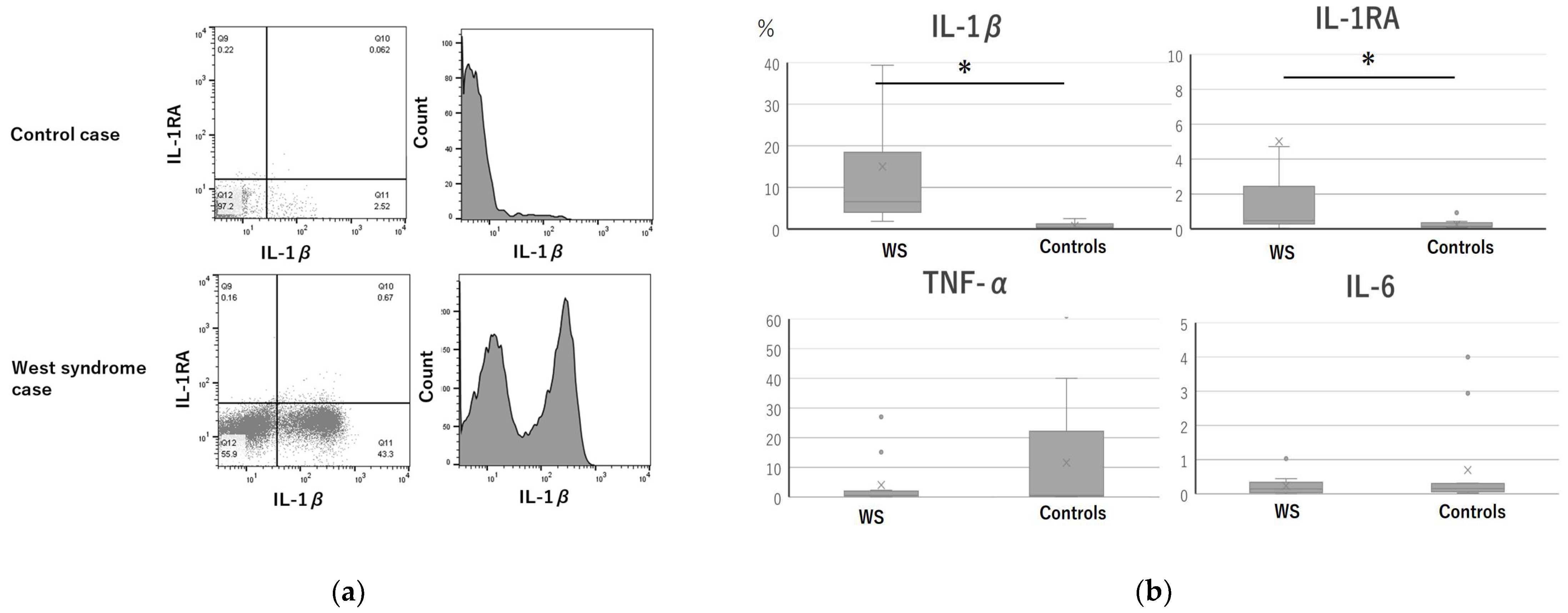
3.2. Intracellular Cytokine Expression
3.3. Plasma Cytokine Levels
4. Discussion
4.1. Pincipal Findings
4.2. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Riikonen, R. Long-Term Otucome of West Syndrome: A Study of Adults with a History of Infantile Spasms. Epilepsia 1996, 37, 367–372. [Google Scholar] [CrossRef]
- Shandra, O.; Moshé, S.L.; Galanopoulou, A.S. Inflammation in Epileptic Encephalopathies. Adv. Protein Chem. Struct. Biol. 2017, 108, 59–84. [Google Scholar] [CrossRef]
- Liu, Z.S.; Wang, Q.W.; Wang, F.L.; Yang, L.Z. Serum Cytokine Levels Are Altered in Patients with West Syndrome. Brain Dev. 2001, 23, 548–551. [Google Scholar] [CrossRef]
- Tekgul, H.; Polat, M.; Tosun, A.; Serdaroglu, G.; Kutukculer, N.; Gokben, S. Cerebrospinal Fluid Interleukin-6 Levels in Patients with West Syndrome. Brain Dev. 2006, 28, 19–23. [Google Scholar] [CrossRef]
- Haginoya, K.; Noguchi, R.; Zhao, Y.; Munakata, M.; Yokoyama, H.; Tanaka, S.; Hino-Fukuyo, N.; Uematsu, M.; Yamamoto, K.; Takayanagi, M.; et al. Reduced Levels of Interleukin-1 Receptor Antagonist in the Cerebrospinal Fluid in Patients with West Syndrome. Epilepsy Res. 2009, 85, 314–317. [Google Scholar] [CrossRef] [PubMed]
- Lombroso, C.T. A Prospective Study of Infantile Spasms: Clinical and Therapeutic Correlations. Epilepsia 1983, 24, 135–158. [Google Scholar] [CrossRef]
- Iwasaki, T.; Nonoda, Y.; Ishii, M. Remission Associated with Human Herpesvirus Infection in West Syndrome. J. Child Neurol. 2006, 21, 886–890. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, C.; Kidokoro, H.; Fukasawa, T.; Yamamoto, H.; Ishihara, N.; Ito, Y.; Sakaguchi, Y.; Okai, Y.; Ohno, A.; Nakata, T.; et al. Cytotoxic edema at onset in West syndrome of unknown etiology: A longitudinal diffusion tensor imaging study. Epilepsia 2018, 59, 440–448. [Google Scholar] [CrossRef] [PubMed]
- Vezzani, A.; Maroso, M.; Balosso, S.; Sanchez, M.A.; Bartfai, T. IL-1 Receptor/Toll-Like Receptor Signaling in Infection, Inflammation, Stress and Neurodegeneration Couples Hyperexcitability and Seizures. Brain Behav. Immun. 2011, 25, 1281–1289. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, G.; Takamatsu, T.; Morichi, S.; Yamazaki, T.; Mizoguchi, I.; Ohno, K.; Watanabe, Y.; Ishida, Y.; Oana, S.; Suzuki, S.; et al. Interleukin-1β in Peripheral Monocytes Is Associated with Seizure Frequency in Pediatric Drug-Resistant Epilepsy. J. Neuroimmunol. 2021, 352, 577475. [Google Scholar] [CrossRef]
- Vezzani, A.; Balosso, S.; Ravizza, T. The role of cytokines in the pathophysiology of epilepsy. Brain Behav. Immun. 2008, 22, 797–803. [Google Scholar] [CrossRef] [PubMed]
- Fumikazu, S.; Masao, A.; Schuichi, K. Reactive astrocyte-driven epileptogenesis is induced by microglia initially activated following status epilepticus. JCI Insight 2021, 6, e135391. [Google Scholar]
- Shiihara, T.; Miyashita, M.; Yoshizumi, M.; Watanabe, M.; Yamada, Y.; Kato, M. Peripheral Lymphocyte Subset and Serum Cytokine Profilesof Patients with West Syndrome. Brain Dev. 2010, 32, 695–702. [Google Scholar] [CrossRef]
- Yamanaka, G.; Kawashima, H.; Oana, S.; Ishida, Y.; Miyajima, T.; Kashiwagi, Y.; Hoshika, A. Increased Level of Serum Interleukin-1 Receptor Antagonist Subsequent to Resolution of Clinical Symptoms in Patients with West Syndrome. J. Neurol. Sci. 2010, 298, 106–109. [Google Scholar] [CrossRef] [PubMed]
- Ohya, T.; Nagai, T.; Araki, Y.; Yanagawa, T.; Tanabe, T.; Iyoda, K.; Kurihara, M.; Yamamoto, K.; Masunaga, K.; Iizuka, C.; et al. A pilot study on the changes in immunity after ACTH therapy in patients with West syndrome. Brain Dev. 2009, 31, 739–743. [Google Scholar] [CrossRef] [PubMed]
- Türe, E.; Kamaşak, T.; Cora, M.; Şahin, S.; Arslan, E.A.; Kaklıkaya, N.; Cansu, A. Comparison of the Serum Cytokine Levels Before and After Adrenocorticotropic Hormone (ACTH) Therapy in Patients with Infantile Spasm. Seizure 2016, 41, 112–115. [Google Scholar] [CrossRef][Green Version]
- de Vries, E.E.; van den Munckhof, B.; Braun, K.P.; van Royen-Kerkhof, A.; de Jager, W.; Jansen, F.E. Inflammatory Mediators in Human Epilepsy: A Systematic Review and Meta-Analysis. Neurosci. Biobehav. Rev. 2016, 63, 177–190. [Google Scholar] [CrossRef]
- Lehtimäki, K.A.; Keränen, T.; Palmio, J.; Mäkinen, R.; Hurme, M.; Honkaniemi, J.; Peltola, J. Increased Plasma Levels of Cytokines After Seizures in Localization-Related Epilepsy. Acta Neurol. Scand. 2007, 116, 226–230. [Google Scholar] [CrossRef]
- Alapirtti, T.; Rinta, S.; Hulkkonen, J.; Mäkinen, R.; Keränen, T.; Peltola, J. Interleukin-6, Interleukin-1 Receptor Antagonist and Interleukin-1Beta Production in Patients with Focal Epilepsy: A Video-EEG Study. J. Neurol. Sci. 2009, 280, 94–97. [Google Scholar] [CrossRef]
- Lehtimäki, K.A.; Keränen, T.; Palmio, J.; Peltola, J. Levels of IL-1Beta and IL-1RA in Cerebrospinal Fluid of Human Patients After Single and Prolonged Seizures. Neuroimmunomodulation 2010, 17, 19–22. [Google Scholar] [CrossRef]
- Jackman, K.; Kahles, T.; Lane, D.; Garcia-Bonilla, L.; Abe, T.; Capone, C.; Hochrainer, K.; Voss, H.; Zhou, P.; Ding, A.; et al. Progranulin Deficiency Promotes Post-Ischemic Blood–Brain Barrier Disruption. J. Neurosci. 2013, 33, 19579–19589. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, G.; Morishita, N.; Morichi, S.; Takeshita, M.; Tomomi, U.; Ishida, Y.; Tomoko, T.; Oana, S.; Watanabe, Y.; Go, S.; et al. Serial Analysis of Multiple Serum Cytokine Responses to Adrenocorticotropic Hormone Therapy in Patients with West Syndrome. J. Child Neurol. 2018, 33, 528–533. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Castejon, G.; Brough, D. Understanding the Mechanism of IL-1β Secretion. Cytokine Growth Factor Rev. 2011, 22, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Enjoji, M.; Yanai, N. Analytic Test for Development in Infancy and Childhood. Pediatr. Int. 1961, 4, 2–6. [Google Scholar] [CrossRef]
- Hulkkonen, J.; Koskikallio, E.; Rainesalo, S.; Keränen, T.; Hurme, M.; Peltola, J. The Balance of Inhibitory and Excitatory Cytokines Is Differently Regulated In Vivo and In Vitro Among Therapy Resistant Epilepsy Patients. Epilepsy Res. 2004, 59, 199–205. [Google Scholar] [CrossRef]
- Auvin, S.; Jeljeli, M.; Desnous, B.; Soussi-Yanicostas, N.; Dournaud, P.; Sterkers, G. Altered Vaccine-Induced Immunity in Children with Dravet Syndrome. Epilepsia 2018, 59, e45–e50. [Google Scholar] [CrossRef]
- Tian, D.S.; Peng, J.; Murugan, M.; Feng, L.J.; Liu, J.L.; Eyo, U.B.; Zhou, L.J.; Mogilevsky, R.; Wang, W.; Wu, L.-J. Chemokine CCL2-CCR2 Signaling Induces Neuronal Cell Death via STAT3 Activation and IL-1β Production After Status Epilepticus. J. Neurosci. 2017, 37, 7878–7892. [Google Scholar] [CrossRef]
- de Jager, W.; Hoppenreijs, E.P.; Wulffraat, N.M.; Wedderburn, L.R.; Kuis, W.; Prakken, B.J. Blood and Synovial Fluid Cytokine Signatures in Patients with Juvenile Idiopathic Arthritis: A Cross-Sectional Study. Ann. Rheum. Dis. 2007, 66, 589–598. [Google Scholar] [CrossRef]
- Kenney-Jung, D.L.; Vezzani, A.; Kahoud, R.J.; LaFrance-Corey, R.G.; Ho, M.L.; Muskardin, T.W.; Wirrell, E.C.; Howe, C.L.; Payne, E.T. Febrile Infection-Related Epilepsy Syndrome Treated with Anakinra. Ann. Neurol. 2016, 80, 939–945. [Google Scholar] [CrossRef]
- Park, Y.J.; Warnock, G.L.; Ao, Z.; Safikhan, N.; Meloche, M.; Asadi, A.; Kieffer, T.J.; Marzban, L. Dual Role of Interleukin-1Beta in Islet Amyloid Formation and Its Beta-Cell Toxicity: Implications for type 2 Diabetes and Islet Transplantation. Diabetes Obes. Metab. 2017, 19, 682–694. [Google Scholar] [CrossRef]
- Rosa, D.V.; Rezende, V.B.; Costa, B.S.; Mudado, F.; Schütze, M.; Torres, K.C.; Martins, L.C.; Moreira-Filho, C.A.; Miranda, D.M.; Romano-Silva, M.A. Epilepsy research 2016, Daniela Valadao Rosa. Circulating CD4 and CD8 T Cells Expressing Pro-Inflammatory Cytokines in a Cohort of Mesial Temporal Lobe Epilepsy Patients with Hippocampal Sclerosis. Epilepsy Res. 2016, 120, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Diez-Ruiz, A.; Tilz, G.P.; Zangerle, R.; Baier-Bitterlich, G.; Wachter, H.; Fuchs, D. Soluble receptors for tumour necrosis factor in clinical laboratory diagnosis. Eur. J. Haematol. 1995, 54, 1–8. [Google Scholar] [CrossRef]
- Daida, A.; Hamano, S.; Hayashi, K.; Nonoyama, H.; Ikemoto, S.; Hirata, Y.; Matsuura, R.; Koichihara, R.; Yamanaka, G.; Kikuchi, K. Comparison of adrenocorticotropic hormone efficacy between aetiologies of infantile spasms. Seizure Eur. J. Epilepsy 2020, 85, 6–11. [Google Scholar] [CrossRef]
- Xu, D.; Robinson, A.P.; Ishii, T.; Duncan, D.S.; Alden, T.D.; Goings, G.E.; Ifergan, I.; Podojil, J.R.; Penaloza-MacMaster, P.; Kearney, J.A.; et al. Peripherally Derived T Regulatory and γδ T Cells Have Opposing Roles in the Pathogenesis of Intractable Pediatric Epilepsy. J. Exp. Med. 2018, 215, 1169–1186. [Google Scholar] [CrossRef]
- Yamanaka, G.; Morichi, S.; Takamatsu, T.; Watanabe, Y.; Suzuki, S.; Ishida, Y.; Oana, S.; Yamazaki, T.; Takata, F.; Kawashima, H. Links Between Immune Cells from the Periphery and the Brain in the Pathogenesis of Epilepsy: A Narrative Review. Int. J. Mol. Sci. 2021, 22, 4395. [Google Scholar] [CrossRef]
- Aronica, E.; Bauer, S.; Bozzi, Y.; Caleo, M.; Dingledine, R.; Gorter, J.A.; Henshall, D.C.; Kaufer, D.; Koh, S.; Löscher, W.; et al. Neuroinflammatory Targets and Treatments for Epilepsy Validated in Experimental Models. Epilepsia 2017, 58, 27–38. [Google Scholar] [CrossRef]
- Himmerich, H.; Bartsch, S.; Hamer, H.; Mergl, R.; Schönherr, J.; Petersein, C.; Munzer, A.; Kirkby, K.C.; Bauer, K.; Sack, U. Impact of Mood Stabilizers and Antiepileptic Drugs on Cytokine Production In-Vitro. J. Psychiatr. Res. 2013, 47, 1751–1759. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Nowak, M.; Bauer, S.; Schlegel, K.; Stei, S.; Allenhöfer, L.; Waschbisch, A.; Tackenberg, B.; Höllerhage, M.; Höglinger, G.; et al. Levetiracetam but not valproate inhibits function of CD8+ T lymphocytes. Seizure 2013, 22, 462–466. [Google Scholar] [CrossRef] [PubMed]
| Case | Sex | Age (Month) | Cause of West Syndrome | Antiepileptic Drugs Taken During Blood Sampling | Antiepileptic Drugs Administered After Blood Sampling | Outcomes |
|---|---|---|---|---|---|---|
| 1 | M | 5 | None | Vit.B6 | ACTH, VGB | ND |
| 2 | F | 6 | None | Vit.B6 | ACTH, VGB | ND |
| 3 | M | 6 | Tuberous sclerosis | None | VGB | ND |
| 4 | F | 4 | Tuberous sclerosis | ZNS | VGB | MD |
| 5 | M | 5 | Tuberous sclerosis | ZNS | VGB, VPA | MD |
| 6 | M | 5 | Cerebral infarction | ZNS | ACTH | MD |
| 7 | M | 3 | Lissencephaly | VPA, ZNS | VGB, Keto milk | SD |
| 8 | M | 5 | Focal cortical dysplasia | Vit.B6, ZNS, LEV | ACTH | SD |
| 9 | M | 3 | Unexplained brain atrophy | Vit.B6 | ACTH, VGB, LTG | SD |
| 10 | M | 5 | Periventricular leukomalacia | VPA, ZNS | ACTH, Clonazepam | SD |
| 11 | M | 7 | Trisomy 21 | TPM, VPA | VGB | SD |
| 12 | M | 7 | Leigh syndrome | ZNS, LEV | None | SD |
| 13 | M | 5 | Cerebral infarction | Vit.B6 | ACTH | MD |
| WS Group | Control Group | p Value | |||
|---|---|---|---|---|---|
| Monocytes | |||||
| IL-1β | 6.5 | (4.0, 18.4) | 0.75 | (0.4, 1.2) | 0.000 * |
| IL-1RA | 0.47 | (0.28, 2.4) | 0.35 | (0.14, 0.43) | 0.039 * |
| IL-6 | 0.14 | (0.04, 0.34) | 0.15 | (0.02, 0.30) | 0.48 |
| TNF-α | 2.0 | (0.55, 2.3) | 0.55 | (0.35, 22.2) | 0.51 |
| CD4+ T cells | |||||
| IFN-γ | 0.01 | (0, 0.86) | 0.02 | (0.01, 0.64) | 0.62 |
| Granzyme A | 0.42 | (0.18, 0.73) | 0.58 | (0.33, 0.84) | 0.76 |
| IL-17 | 0.04 | (0.02, 0.5) | 0.09 | (0.12, 0.46) | 1.00 |
| IL-10 | 0.04 | (0.03, 1.29) | 0.19 | (0.11, 0.30) | 0.59 |
| IL-1β | 0.32 | (0.14, 0.84) | 0.51 | (0.19, 1.99) | 0.49 |
| IL-1RA | 2.4 | (0.80, 6.2) | 3.7 | (0.7, 8.4) | 0.29 |
| IL-6 | 0.05 | (0.01, 0.21) | 0.05 | (0.01, 0.07) | 0.38 |
| TNF-α | 0.3 | (0.1, 0.42) | 0.25 | (0.21, 0.48) | 0.96 |
| CD8+ T cells | |||||
| IFN-γ | 4.2 | (0.38, 6.2) | 1.9 | (0.12, 3.7) | 0.02 * |
| Granzyme A | 2.3 | (0.58, 9.5) | 1.9 | (0.60, 5.1) | 0.26 |
| IL-17 | 0.01 | (0, 0.08) | 0 | (0, 0.04) | 0.62 |
| IL-10 | 0.02 | (0.01, 0.26) | 0.1 | (0, 0.16) | 0.89 |
| IL-1β | 1.8 | (0.37, 1.9) | 0.26 | (0.09, 0.26) | 0.29 |
| IL-1RA | 1.5 | (1.1, 22.9) | 4.1 | (4.9, 0.3) | 0.68 |
| IL-6 | 0.24 | (0, 1.3) | 0.24 | (0, 1.3) | 0.34 |
| TNF-α | 0.3 | (0, 0.16) | 0.55 | (0.02, 0.23) | 0.71 |
| NKT-like cells | |||||
| IFN-γ | 0.04 | (0.02, 0.07) | 0.12 | (0, 0.13) | 0.690 |
| Granzyme A | 0.0 | (0, 0.06) | 0.01 | (0, 0.02) | 0.847 |
| NK cells | |||||
| IFN-γ | 0.69 | (0.58, 1,3) | 0.95 | (0.21, 4.2) | 0.405 |
| Granzyme A | 0.02 | (0.01, 0.09) | 0.28 | (0.05, 0.4) | 0.131 |
| B cells | |||||
| IFN-γ | 0.26 | (0.01, 0.76) | 0.25 | (0.02, 0.47) | 0.372 |
| Granzyme A | 0.02 | (0.01, 0.04) | 0.25 | (0.02, 0.47) | 0.846 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Takamatsu, T.; Yamanaka, G.; Ohno, K.; Hayashi, K.; Watanabe, Y.; Takeshita, M.; Suzuki, S.; Morichi, S.; Go, S.; Ishida, Y.; et al. Involvement of Peripheral Monocytes with IL-1β in the Pathogenesis of West Syndrome. J. Clin. Med. 2022, 11, 447. https://doi.org/10.3390/jcm11020447
Takamatsu T, Yamanaka G, Ohno K, Hayashi K, Watanabe Y, Takeshita M, Suzuki S, Morichi S, Go S, Ishida Y, et al. Involvement of Peripheral Monocytes with IL-1β in the Pathogenesis of West Syndrome. Journal of Clinical Medicine. 2022; 11(2):447. https://doi.org/10.3390/jcm11020447
Chicago/Turabian StyleTakamatsu, Tomoko, Gaku Yamanaka, Koko Ohno, Kanako Hayashi, Yusuke Watanabe, Mika Takeshita, Shinji Suzuki, Shinichiro Morichi, Soken Go, Yu Ishida, and et al. 2022. "Involvement of Peripheral Monocytes with IL-1β in the Pathogenesis of West Syndrome" Journal of Clinical Medicine 11, no. 2: 447. https://doi.org/10.3390/jcm11020447
APA StyleTakamatsu, T., Yamanaka, G., Ohno, K., Hayashi, K., Watanabe, Y., Takeshita, M., Suzuki, S., Morichi, S., Go, S., Ishida, Y., Oana, S., Kashiwagi, Y., & Kawashima, H. (2022). Involvement of Peripheral Monocytes with IL-1β in the Pathogenesis of West Syndrome. Journal of Clinical Medicine, 11(2), 447. https://doi.org/10.3390/jcm11020447






