Increased Natural Killer Cells Are Associated with Alcohol Liver Fibrosis and with T Cell and Cytotoxic Subpopulations Change
Abstract
1. Introduction
2. Materials and Methods
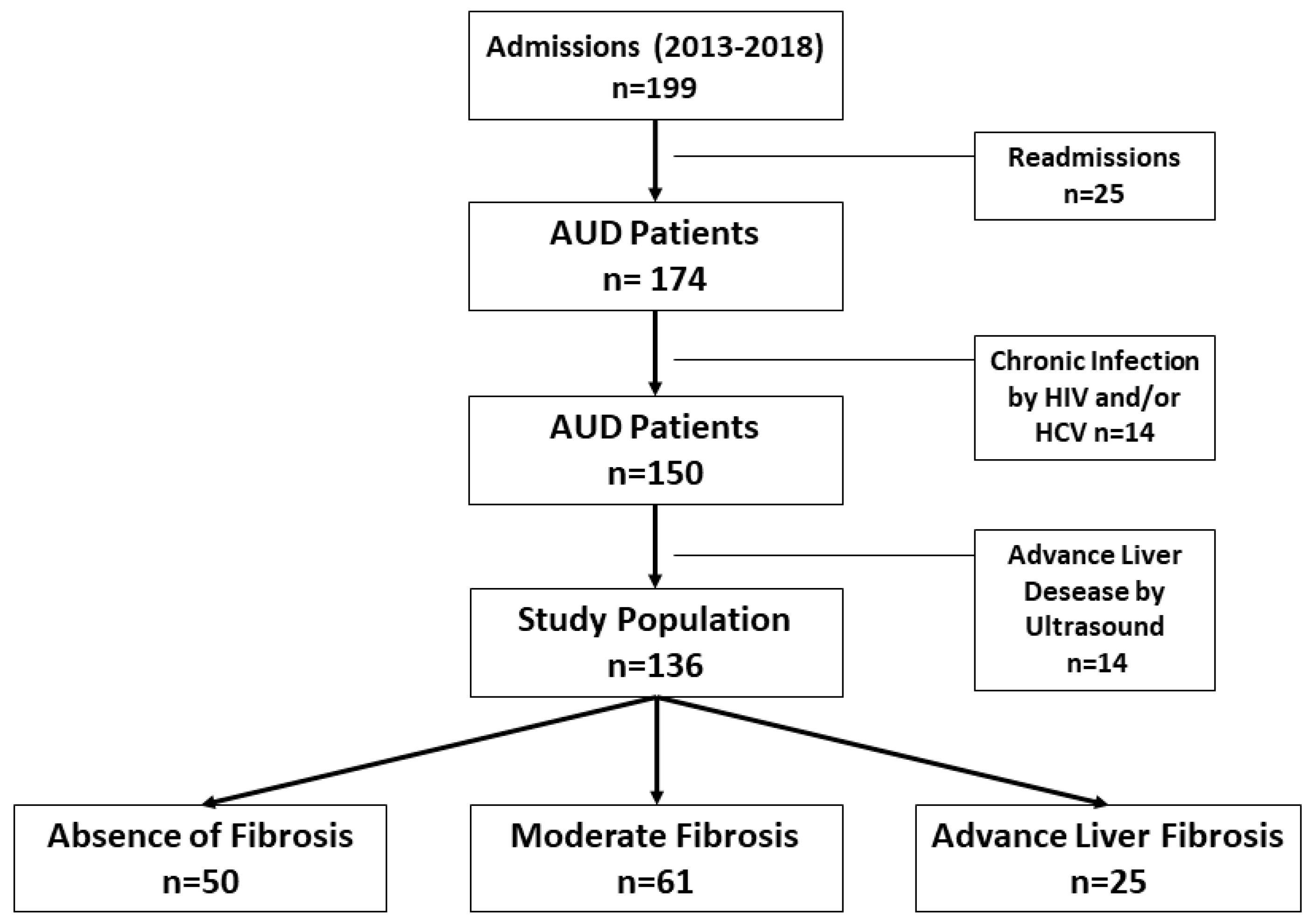
2.1. Patients
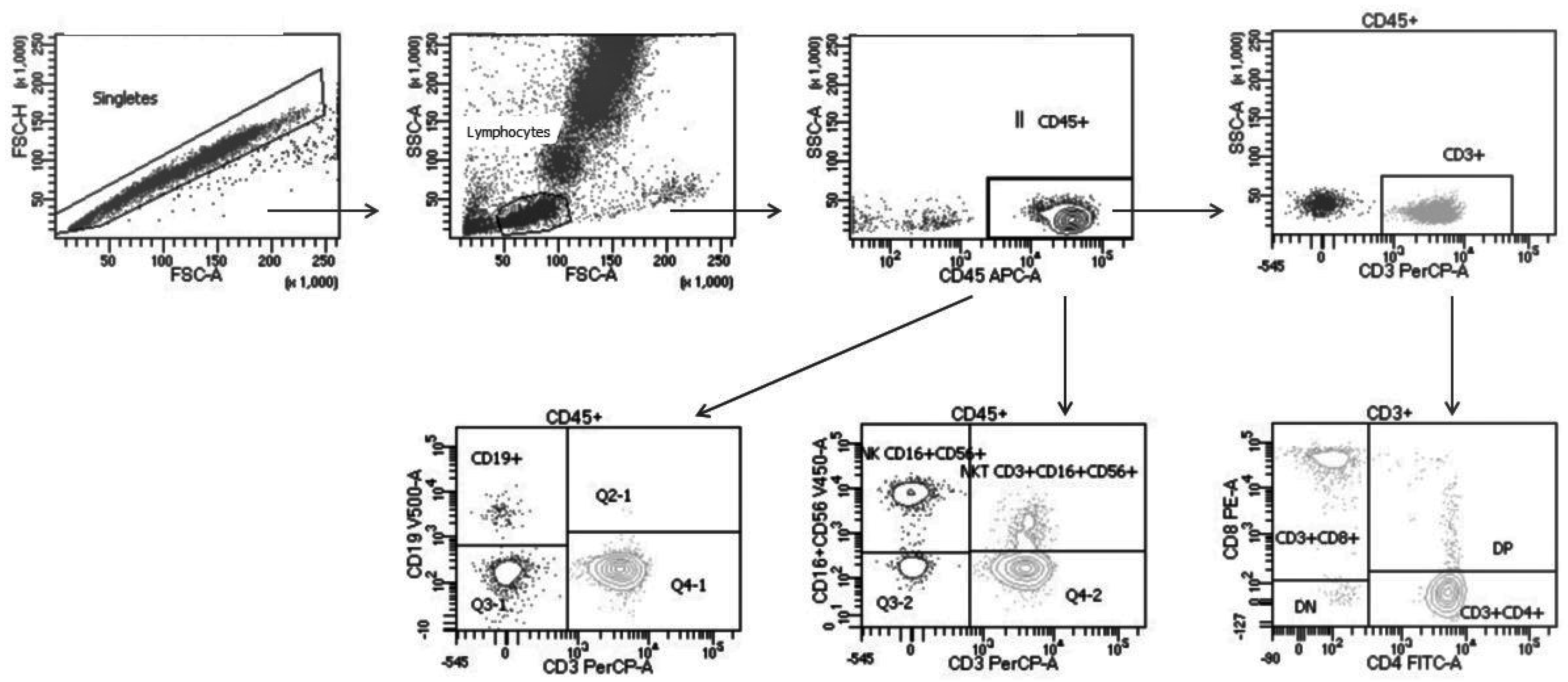
2.2. Flow Cytometry
2.3. Statistical Analysis
3. Results
3.1. Patients Details
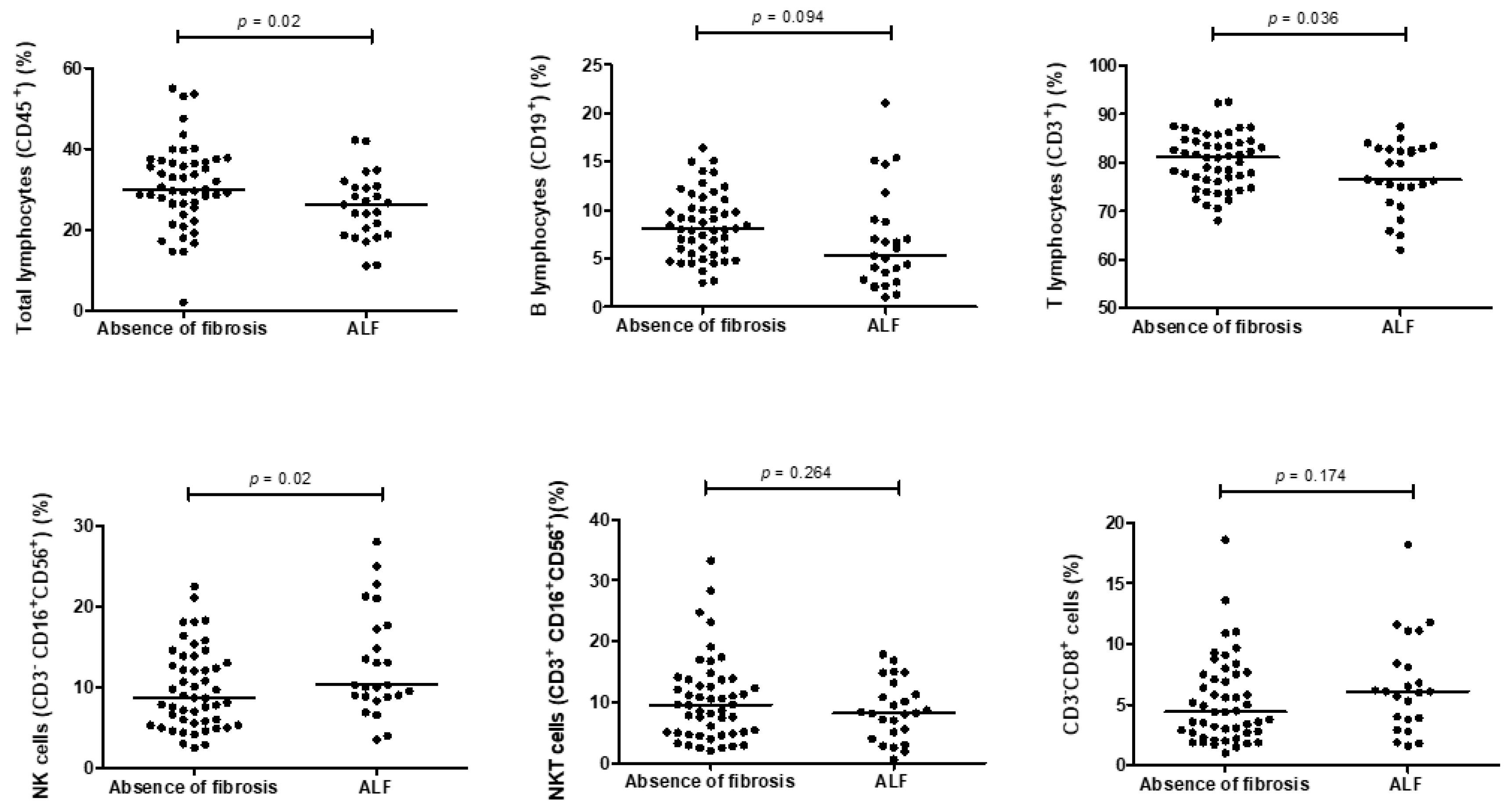
3.2. Count and Distribution of Lymphocyte Subpopulations
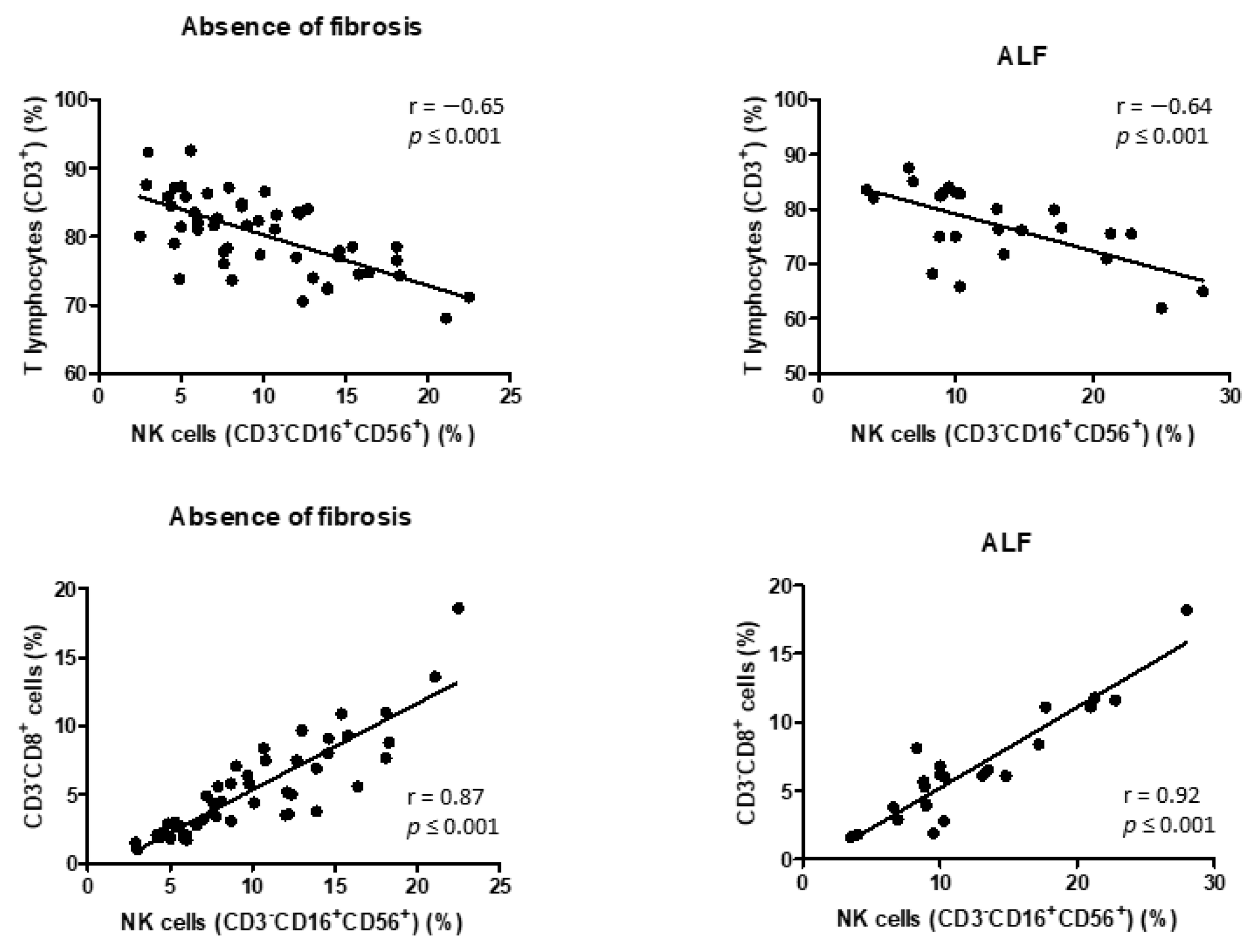
3.3. Correlation between NK Cells and Lymphocyte Subpopulations
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bataller, R.; Brenner, D. Liver fibrosis. J. Clin. Investig. 2005, 115, 209–218. [Google Scholar] [CrossRef]
- Higashi, T.; Friedman, S.L.; Hoshida, Y. Hepatic stellate cells as key target in liver fibrosis. Adv. Drug Deliv. Rev. 2017, 121, 27–42. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Tan, H.-Y.; Wang, N.; Feng, Y.; Wang, X.; Feng, Y. Recent Insights into the Role of Immune Cells in Alcoholic Liver Disease. Front. Immunol. 2019, 10, 1328. [Google Scholar] [CrossRef] [PubMed]
- Gao, B.; Radaeva, S.; Park, O. Liver natural killer and natural killer T cells: Immunobiology and emerging roles in liver diseases. J. Leukoc. Biol. 2009, 86, 513–528. [Google Scholar] [CrossRef]
- Glässner, A.; Eisenhardt, M.; Krämer, B.; Körner, C.; Coenen, M.; Sauerbruch, T.; Spengler, U.; Nattermann, J. NK cells from HCV-infected patients effectively induce apoptosis of activated primary human hepatic stellate cells in a TRAIL-, FasL- and NKG2D-dependent manner. Lab. Investig. 2012, 92, 967–977. [Google Scholar] [CrossRef] [PubMed]
- Abel, A.M.; Yang, C.; Thakar, M.S.; Malarkannan, S. Natural Killer Cells: Development, Maturation, and Clinical Utilization. Front. Immunol. 2018, 9, 1869. [Google Scholar] [CrossRef]
- Perney, P.; Portalès, P.; Corbeau, P.; Roques, V.; Blanc, F.; Clot, J. Specific Alteration of Peripheral Cytotoxic Cell Perforin Expression in Alcoholic Patients: A Possible Role in Alcohol-Related Diseases. Alcohol. Clin. Exp. Res. 2003, 27, 1825–1830. [Google Scholar] [CrossRef]
- Zhang, H.; Meadows, G.G. Chronic alcohol consumption perturbs the balance between thymus-derived and bone marrow-derived natural killer cells in the spleen. J. Leukoc. Biol. 2008, 83, 41–47. [Google Scholar] [CrossRef]
- Zhang, F.; Little, A.; Zhang, H. Chronic alcohol consumption inhibits peripheral NK cell development and maturation by decreasing the availability of IL-15. J. Leukoc. Biol. 2017, 101, 1015–1027. [Google Scholar] [CrossRef]
- Jeong, W.; Park, O.; Gao, B. Abrogation of the Antifibrotic Effects of Natural Killer Cells/Interferon-γ Contributes to Alcohol Acceleration of Liver Fibrosis. Gastroenterology 2008, 134, 248–258. [Google Scholar] [CrossRef]
- Jeong, W.-I.; Park, O.; Suh, Y.-G.; Byun, J.-S.; Park, S.-Y.; Choi, E.; Kim, J.-K.; Ko, H.; Wang, H.; Miller, A.M.; et al. Suppression of innate immunity (natural killer cell/interferon-γ) in the advanced stages of liver fibrosis in mice. Hepatology 2011, 53, 1342–1351. [Google Scholar] [CrossRef] [PubMed]
- Melsen, J.E.; Lugthart, G.; Lankester, A.C.; Schilham, M.W. Human Circulating and Tissue-Resident CD56bright Natural Killer Cell Populations. Front. Immunol. 2016, 7, 262. [Google Scholar] [CrossRef]
- Naude, C.E.; Bouic, P.; Senekal, M.; Kidd, M.; Ferrett, H.L.; Fein, G.; Carey, P.D. Lymphocyte measures in treatment-naïve 13–15-year old adolescents with alcohol use disorders. Alcohol 2011, 45, 507–514. [Google Scholar] [CrossRef][Green Version]
- Laso, F.J.; Almeida, J.; Torres, E.; Vaquero, J.M.; Marcos, M.; Orfao, A. Chronic Alcohol Consumption Is Associated with an Increased Cytotoxic Profile of Circulating Lymphocytes That May Be Related With the Development of Liver Injury. Alcohol. Clin. Exp. Res. 2010, 34, 876–885. [Google Scholar] [CrossRef]
- Støy, S.; Dige, A.; Sandahl, T.D.; Laursen, T.L.; Buus, C.; Hokland, M.; Vilstrup, H. Cytotoxic T lymphocytes and natural killer cells display impaired cytotoxic functions and reduced activation in patients with alcoholic hepatitis. Am. J. Physiol. Liver Physiol. 2015, 308, G269–G276. [Google Scholar] [CrossRef]
- Sehgal, R.; Kaur, S.; Shasthry, S.M.; Agrawal, T.; Dwivedi, V.; Seth, D.; Ramakrishna, G.; Sarin, S.K.; Trehanpati, N. Natural Killer Cells Contribute to Pathogenesis of Severe Alcoholic Hepatitis by Inducing Lysis of Endothelial Progenitor Cells. Alcohol. Clin. Exp. Res. 2019, 44, 78–86. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar]
- Sterling, R.K.; Lissen, E.; Clumeck, N.; Sola, R.; Correa, M.C.; Montaner, J.; Sulkowski, M.S.; Torriani, F.J.; Dieterich, D.T.; Thomas, D.L.; et al. Development of a simple noninvasive index to predict significant fibrosis in patients with HIV/HCV coinfection. Hepatology 2006, 43, 1317–1325. [Google Scholar] [CrossRef]
- Fuster, D.; Garcia-Calvo, X.; Zuluaga, P.; Rivas, I.; Sanvisens, A.; Tor, J.; Muga, R. Ultrasound findings of liver damage in a series of patients consecutively admitted for treatment of alcohol use disorder. Drug Alcohol Depend. 2018, 190, 195–199. [Google Scholar] [CrossRef] [PubMed]
- Singal, A.K.; Bataller, R.; Ahn, J.; Kamath, P.S.; Shah, V.H. ACG Clinical Guideline: Alcoholic Liver Disease. Am. J. Gastroenterol. 2018, 113, 175–194. [Google Scholar] [CrossRef] [PubMed]
- Apoil, P.A.; Puissant-Lubrano, B.; Congy, N.; Peres, M.; Tkaczuk, J.; Roubinet, F.; Blancher, A. Reference values for T, B and NK human lymphocyte subpopulations in adults. Data Brief 2017, 12, 400–404. [Google Scholar] [CrossRef]
- Melzer, S.; Zachariae, S.; Bocsi, J.; Engel, C.; Löffler, M.; Tárnok, A. Reference intervals for leukocyte subsets in adults: Results from a population-based study using 10-color flow cytometry. Cytom. Part B Clin. Cytom. 2015, 88, 270–281. [Google Scholar] [CrossRef] [PubMed]
- Apoil, P.; Puissant-Lubrano, B.; Congy, N.; Peres, M.; Tkaczuk, J.; Roubinet, F.; Blancher, A. Influence of age, sex and HCMV-serostatus on blood lymphocyte subpopulations in healthy adults. Cell. Immunol. 2017, 314, 42–53. [Google Scholar] [CrossRef]
- Langhans, B.; Alwan, A.W.; Krämer, B.; Glässner, A.; Lutz, P.; Strassburg, C.P.; Nattermann, J.; Spengler, U. Regulatory CD4+ T cells modulate the interaction between NK cells and hepatic stellate cells by acting on either cell type. J. Hepatol. 2015, 62, 398–404. [Google Scholar] [CrossRef]
- Glässner, A.; Eisenhardt, M.; Kokordelis, P.; Krämer, B.; Wolter, F.; Nischalke, H.D.; Boesecke, C.; Sauerbruch, T.; Rockstroh, J.K.; Spengler, U.; et al. Impaired CD4+ T cell stimulation of NK cell anti-fibrotic activity may contribute to accelerated liver fibrosis progression in HIV/HCV patients. J. Hepatol. 2013, 59, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Ma, P.-F.; Gao, C.-C.; Yi, J.; Zhao, J.-L.; Liang, S.-Q.; Zhao, Y.; Ye, Y.-C.; Bai, J.; Zheng, Q.-J.; Dou, K.-F.; et al. Cytotherapy with M1-polarized macrophages ameliorates liver fibrosis by modulating immune microenvironment in mice. J. Hepatol. 2017, 67, 770–779. [Google Scholar] [CrossRef]
- Shi, J.; Zhao, J.; Zhang, X.; Cheng, Y.; Hu, J.; Li, Y.; Zhao, X.; Shang, Q.; Sun, Y.; Tu, B.; et al. Activated hepatic stellate cells impair NK cell anti-fibrosis capacity through a TGF-β-dependent emperipolesis in HBV cirrhotic patients. Sci. Rep. 2017, 7, 44544. [Google Scholar] [CrossRef]
- Srour, E.F.; Leemhuis, T.; Jenski, L.; Redmond, R.; Jansen, J. Cytolytic activity of human natural killer cell subpopulations isolated by four-color immunofluorescence flow cytometric cell sorting. Cytometry 1990, 11, 442–446. [Google Scholar] [CrossRef]
- Addison, E.G.; North, J.; Bakhsh, I.; Marden, C.; Haq, S.; Al-Sarraj, S.; Malayeri, R.; Wickremasinghe, R.G.; Davies, J.K.; Lowdell, M.W. Ligation of CD8alpha on human natural killer cells prevents activation-induced apoptosis and enhances cytolytic activity. Immunology 2005, 116, 354–361. [Google Scholar] [CrossRef]
- Zuluaga, P.; Sanvisens, A.; Martinez-Cáceres, E.; Teniente, A.; Tor, J.; Muga, R. Over-expression of CD8 + T-cell activation is associated with decreased CD4 + cells in patients seeking treatment of Alcohol Use Disorder. Drug Alcohol Depend. 2017, 180, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Márquez, M.; Fernández-Gutiérrez, C.; Montes-De-Oca, M.; Blanco, M.J.; Brun, F.; Rodríguez-Ramos, C.; Girón-González, J.A. Chronic antigenic stimuli as a possible explanation for the immunodepression caused by liver cirrhosis. Clin. Exp. Immunol. 2009, 158, 219–229. [Google Scholar] [CrossRef]
- Zuluaga, P.; Sanvisens, A.; Serra, A.T.; el Ars, O.; Fuster, D.; Quirant-Sánchez, B.; Martínez-Cáceres, E.; Muga, R. Loss of naive T lymphocytes is associated with advanced liver fibrosis in alcohol use disorder. Drug Alcohol Depend. 2020, 213, 108046. [Google Scholar] [CrossRef]
- Pasala, S.; Barr, T.; Messaoudi, I. Impact of Alcohol Abuse on the Adaptive Immune System. Alcohol Res. Curr. Rev. 2015, 37, 185–197. [Google Scholar]
- Singh, V.; Keisham, A.; Bhalla, A.; Sharma, N.; Agarwal, R.; Sharma, R.; Singh, A. Efficacy of Granulocyte Colony-Stimulating Factor and N-Acetylcysteine Therapies in Patients with Severe Alcoholic Hepatitis. Clin. Gastroenterol. Hepatol. 2018, 16, 1650–1656.e2. [Google Scholar] [CrossRef]
- Garg, V.; Garg, H.; Khan, A.; Trehanpati, N.; Kumar, A.; Sharma, B.C.; Sakhuja, P.; Sarin, S.K. Granulocyte Colony–Stimulating Factor Mobilizes CD34+ Cells and Improves Survival of Patients With Acute-on-Chronic Liver Failure. Gastroenterology 2012, 142, 505–512. [Google Scholar] [CrossRef]
- Chrostek, L.; Przekop, D.; Gruszewska, E.; Gudowska-Sawczuk, M.; Cylwik, B. Noninvasive Indirect Markers of Liver Fibrosis in Alcoholics. BioMed Res. Int. 2019, 2019, 3646975. [Google Scholar] [CrossRef]
- Thiele, M.; Madsen, B.S.; Hansen, J.F.; Detlefsen, S.; Antonsen, S.; Krag, A. Accuracy of the Enhanced Liver Fibrosis Test vs FibroTest, Elastography, and Indirect Markers in Detection of Advanced Fibrosis in Patients with Alcoholic Liver Disease. Gastroenterology 2018, 154, 1369–1379. [Google Scholar] [CrossRef]
- Vallet-Pichard, A.; Mallet, V.; Nalpas, B.; Verkarre, V.; Nalpas, A.; Dhalluin-Venier, V.; Fontaine, H.; Pol, S. FIB-4: An inexpensive and accurate marker of fibrosis in HCV infection. comparison with liver biopsy and fibrotest. Hepatology 2007, 46, 32–36. [Google Scholar] [CrossRef]
- Nunes, D.; Fleming, C.; Offner, G.; Craven, D.; Fix, O.; Heeren, T.; Koziel, M.J.; Graham, C.; Tumilty, S.; Skolnik, P.; et al. Noninvasive Markers of Liver Fibrosis Are Highly Predictive of Liver-Related Death in a Cohort of HCV-Infected Individuals with and Without HIV Infection. Am. J. Gastroenterol. 2010, 105, 1346–1353. [Google Scholar] [CrossRef] [PubMed]
- Rhodes, F.A.; Trembling, P.; Panovska-Griffiths, J.; Tanwar, S.; Westbrook, R.H.; Rodger, A.; Rosenberg, W.M. Systematic review: Investigating the prognostic performance of four non-invasive tests in alcohol-related liver disease. J. Gastroenterol. Hepatol. 2021, 36, 1435–1449. [Google Scholar] [CrossRef] [PubMed]
- Sanvisens, A.; Muñoz, A.; Bolao, F.; Zuluaga, P.; Farré, M.; Jarrin, I.; Tor, J.; Muga, R. Do serum markers of liver fibrosis vary by HCV infection in patients with alcohol use disorder? Drug Alcohol Depend. 2018, 188, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, D.N.; Thiele, M.; Johansen, S.; Kjærgaard, M.; Lindvig, K.P.; Israelsen, M.; Antonsen, S.; Detlefsen, S.; Krag, A.; Anastasiadou, E.; et al. Prognostic performance of 7 biomarkers compared to liver biopsy in early alcohol-related liver disease. J. Hepatol. 2021, 75, 1017–1025. [Google Scholar] [CrossRef] [PubMed]
| Variables | Total n = 136 Median (RIQ) | Absence of Fibrosis n = 50 Median (RIQ) | Moderate Fibrosis n = 60 Median (RIQ) | ALF n = 25 Median (RIQ) | * p-Value |
|---|---|---|---|---|---|
| n (%) | 104 (76) | 32 (64) | 83.6 (81) | 21 (84) | 0.073 |
| Age (years) | 49 (44–56) | 47 (40–53) | 51 (46–57) | 52 (48–59) | <0.01 |
| Body Mass Index (kg/m2, n = 106) | 26 (23–29) | 26 (22–29) | 26 (23–28) | 25 (22–28) | 0.241 |
| Daily alcohol consumption (g/day, n = 132) | 140 (100–200) | 138 (100–200) | 160 (100–200) | 140 (100–200) | 0.975 |
| Age at starting alcohol consumption (years, n = 96) | 17 (15–20) | 17 (15–20) | 17 (16–20) | 16 (15–18) | 0.230 |
| Duration of alcohol use disorder (years, n = 104) | 15 (7–20) | 9 (5–17) | 17 (10–25) | 15 (8–20) | 0.03 |
| Tobacco (%) | 102 (79) | 38 (79) | 49 (84) | 15 (65) | 0.206 |
| Cocaine use in the last 30 days (%, n = 133) | 17 (13) | 9 (18) | 4 (7) | 4 (16) | 0.888 |
| Total n = 136 Median (RIQ) | Absence of Fibrosis n = 50 Median (RIQ) | Moderate Fibrosis n = 60 Median (RIQ) | ALF n = 25 Median (RIQ) | * p-Value | |
|---|---|---|---|---|---|
| Lymphocyte Populations | |||||
| Total Lymphocytes (cel/µL) | 1800 (1400–2300) | 2050 (1700–2700) | 1700 (1400–2300) | 1500 (1100–1600) | <0.01 |
| T Cells (cel/µL) | 1461 (1092–1917) | 1623 (1331–2252) | 1457 (1014–1835) | 1105 (729–1321) | <0.01 |
| CD4+ T Cells (cel/µL) | 925 (627–1154) | 1072 (818–1350) | 946 (610–1193) | 602 (448–837) | <0.01 |
| CD8+ T Cells (cel/µL) | 416 (287–592) | 448 (360–729) | 424 (290–595) | 329 (211–451) | <0.01 |
| B Cells (cel/µL) | 129 (84–223) | 177 (124–259) | 129 (88–190) | 77 (39–106) | <0.01 |
| NK Cells (cel/µL) | 173 (110–276) | 196 (124–264) | 172 (105–282) | 135 (94–196) | 0.154 |
| NKT Cells (cel/µL) | 116 (74–195) | 146 (76–226) | 107 (74–190) | 76 (37–137) | <0.01 |
| CD3-CD8+ Cells (cel/µL) | 66 (40–114) | 76 (48–129) | 72 (39–114) | 59 (32–94) | 0.161 |
| Laboratory Parameters | |||||
| Hemoglobin (mg/dL) | 14.3 (12.9–15.5) | 13.8 (12.4–15.2) | 14.8 (13.4–15.4) | 13.8 (13.1–15) | 0.779 |
| Platelets (×109/L) | 194 (153–240) | 241 (207–288) | 188 (157–222) | 128 (105–151) | <0.01 |
| AST (U/L) | 38 (23–64) | 23 (17–33) | 41 (27–64) | 90 (51–134) | <0.01 |
| ALT (U/L) | 28 (17–57) | 23 (16–33) | 32 (20–46) | 58 (27–86) | <0.01 |
| AST/ALT | 1.19 (0.80–1.72) | 0.88 (0.65–1.25) | 1.26 (0.93–1.73) | 1.9 (1.45–2.98) | <0.01 |
| AST/ALT > 2, n (%) | 21 (15) | 1 (2) | 10 (16) | 10 (40) | <0.01 |
| GGT ** (U/L, n = 134) | 133 (37–284) | 57 (31–132) | 133 (52–273) | 352 (77–728) | <0.01 |
| Bilirubin (mg/dL, n = 135) | 0.72 (0.47–1.08) | 0.6 (0.43–0.9) | 0.75 (0.47–1.1) | 0.99 (0.82–1.2) | <0.01 |
| Albumin (mg/dL) | 39.1 (36.3–41.8) | 39.1 (36–41.4) | 39.1 (36.2–42) | 39.4 (38.2–41.4) | 0.650 |
| Prothrombin Rate (%) (n = 133) | 100 (92–100) | 100 (95–100) | 100 (89–100) | 98 (87.5–100) | 0.172 |
| C-reactive Protein (mg/L) (n = 135) | 3.1 (1–7.5) | 2.6 (0.8–6.7) | 4.1 (1.8–8.4) | 1.7 (0.8–5.4) | 0.584 |
| Cholesterol total (mg/dL) (n = 135) | 198 (163–239) | 209 (168–247) | 190 (153–224) | 193 (179–239) | 0.456 |
| Triglycerides (mg/dL) (n = 135) | 121 (83–183) | 151 (99–206) | 112.5 (82–175) | 90 (61–121) | 0.567 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zuluaga, P.; Teniente-Serra, A.; Fuster, D.; Quirant-Sánchez, B.; Hernandez-Rubio, A.; Martínez-Cáceres, E.; Muga, R. Increased Natural Killer Cells Are Associated with Alcohol Liver Fibrosis and with T Cell and Cytotoxic Subpopulations Change. J. Clin. Med. 2022, 11, 305. https://doi.org/10.3390/jcm11020305
Zuluaga P, Teniente-Serra A, Fuster D, Quirant-Sánchez B, Hernandez-Rubio A, Martínez-Cáceres E, Muga R. Increased Natural Killer Cells Are Associated with Alcohol Liver Fibrosis and with T Cell and Cytotoxic Subpopulations Change. Journal of Clinical Medicine. 2022; 11(2):305. https://doi.org/10.3390/jcm11020305
Chicago/Turabian StyleZuluaga, Paola, Aina Teniente-Serra, Daniel Fuster, Bibiana Quirant-Sánchez, Anna Hernandez-Rubio, Eva Martínez-Cáceres, and Roberto Muga. 2022. "Increased Natural Killer Cells Are Associated with Alcohol Liver Fibrosis and with T Cell and Cytotoxic Subpopulations Change" Journal of Clinical Medicine 11, no. 2: 305. https://doi.org/10.3390/jcm11020305
APA StyleZuluaga, P., Teniente-Serra, A., Fuster, D., Quirant-Sánchez, B., Hernandez-Rubio, A., Martínez-Cáceres, E., & Muga, R. (2022). Increased Natural Killer Cells Are Associated with Alcohol Liver Fibrosis and with T Cell and Cytotoxic Subpopulations Change. Journal of Clinical Medicine, 11(2), 305. https://doi.org/10.3390/jcm11020305






