Preliminary Investigation on Micro-Needling with Low-Level LED Therapy and Growth Factors in Hair Loss Related to COVID-19
Abstract
1. Introduction
2. Methods
2.1. Inclusion and Exclusion Criteria
2.2. Study Overview
2.3. Endpoint Definition
2.4. Micro-Needling (MND) Combined with Low-Level Light Therapy (LLLT) and Growth Factors (GFs): Protocol and Device
2.5. Phototrichograms Collection
2.6. Statistical Analysis
3. Results
3.1. Patient Assessment
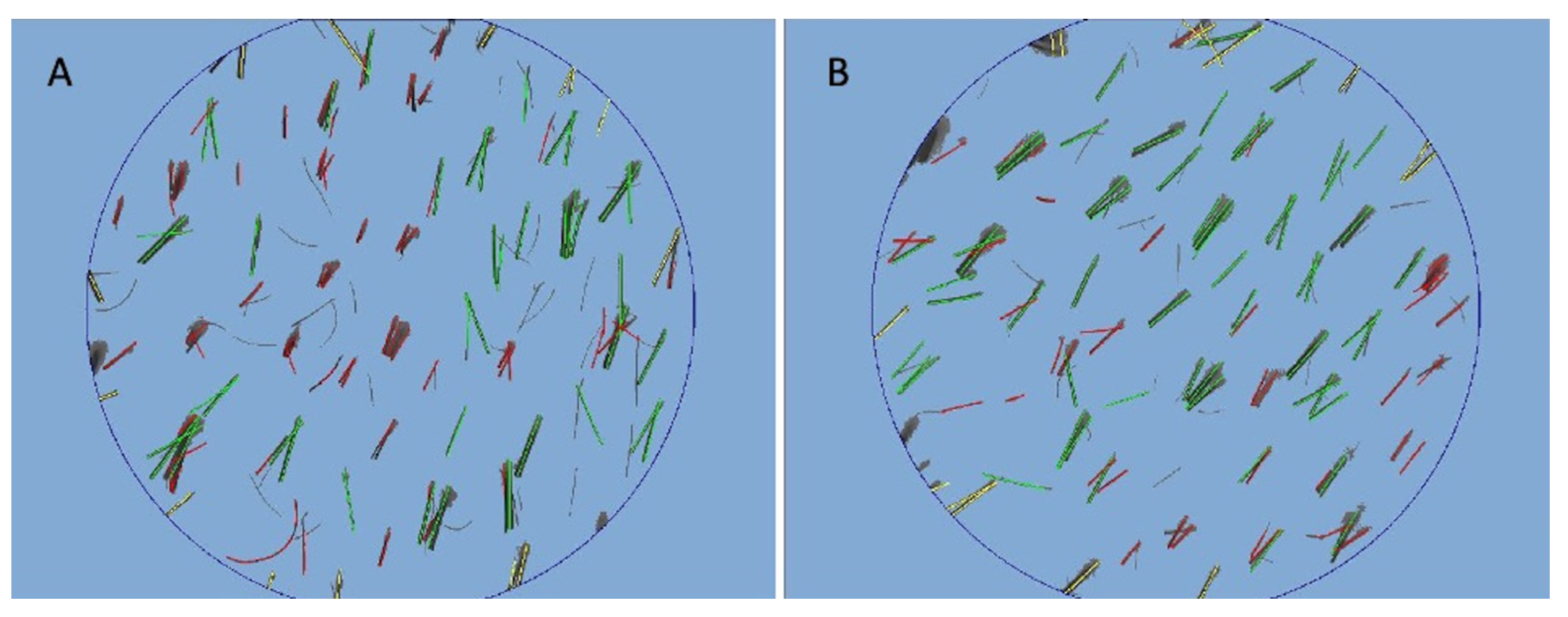
3.2. Trichoscopy Analysis
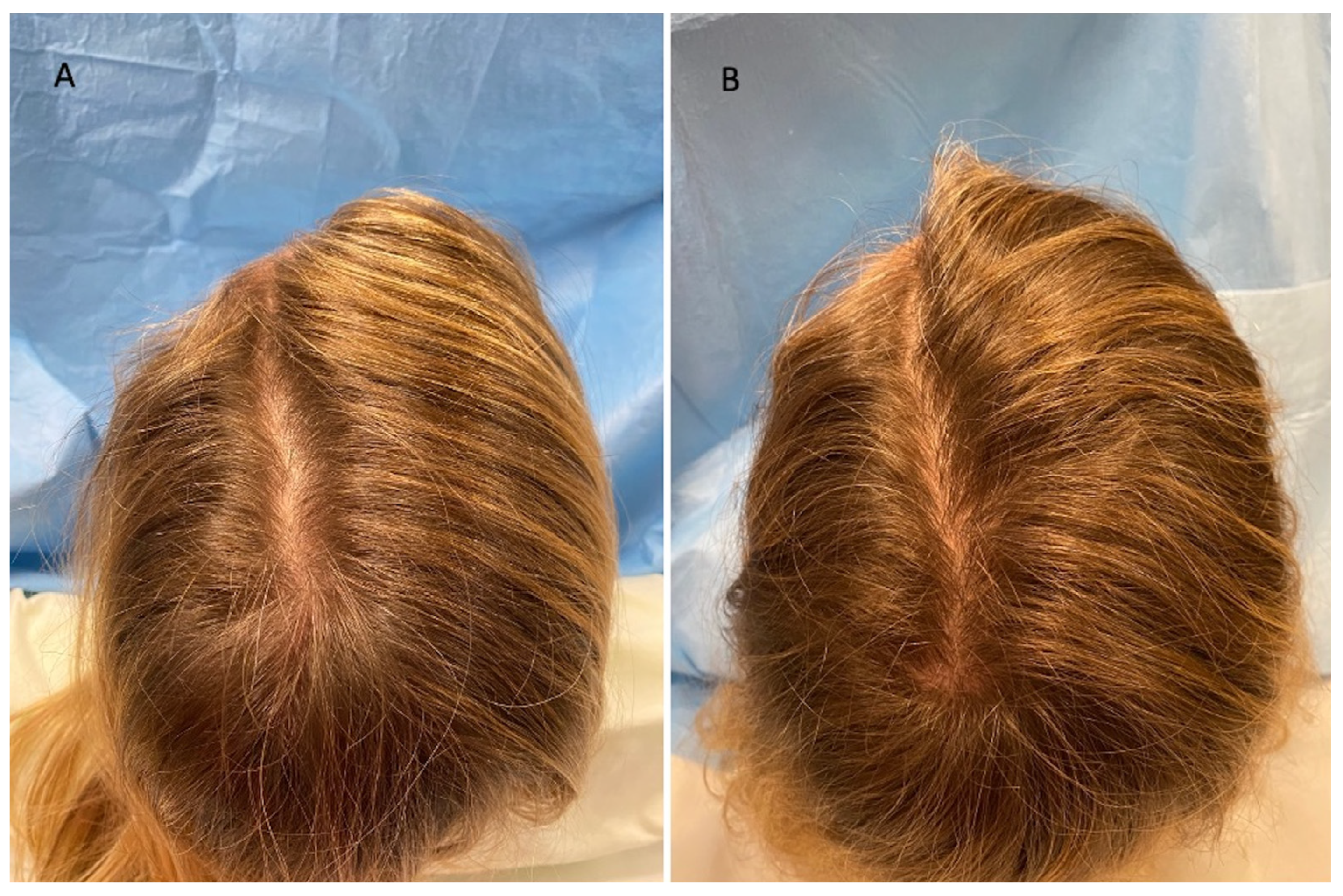
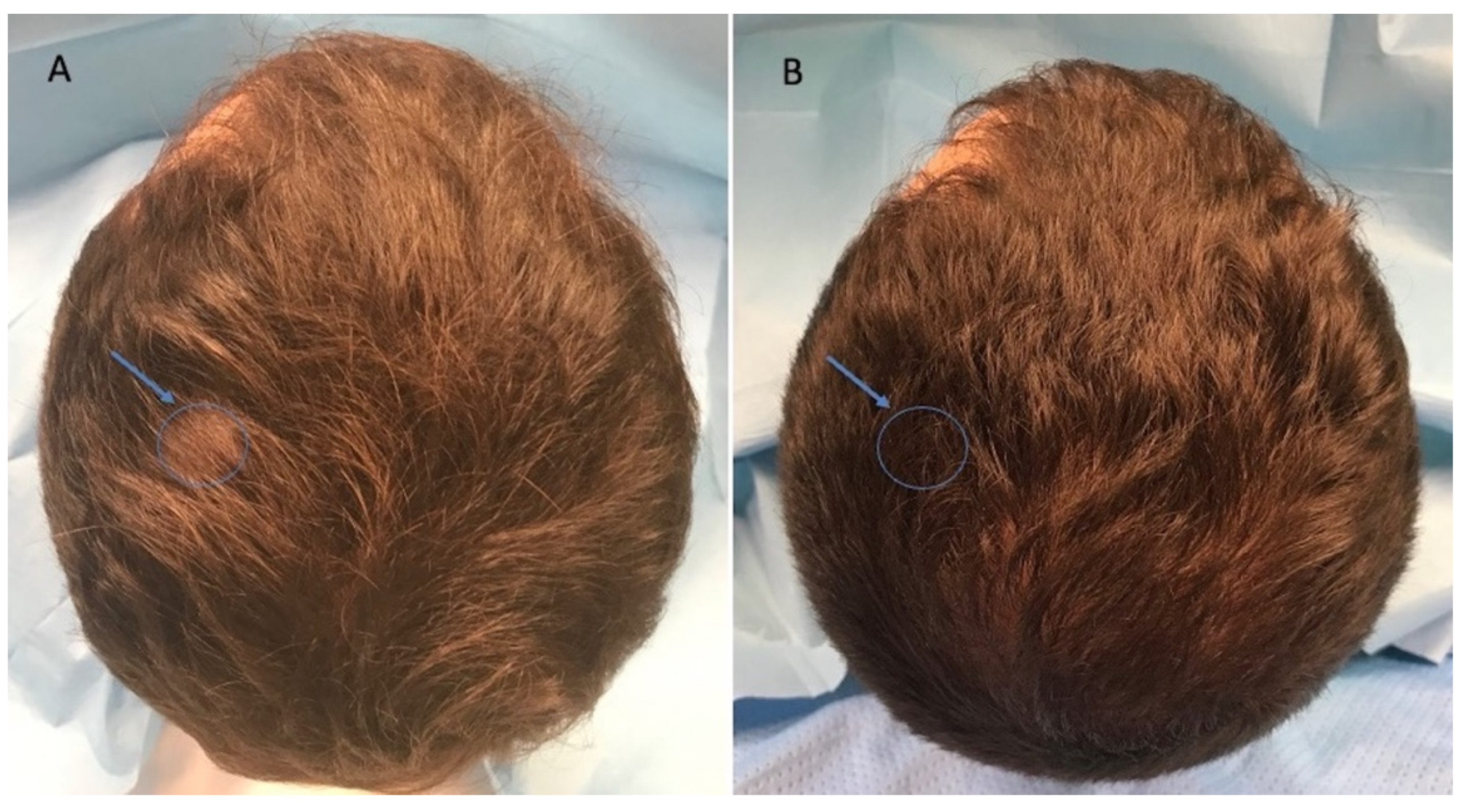
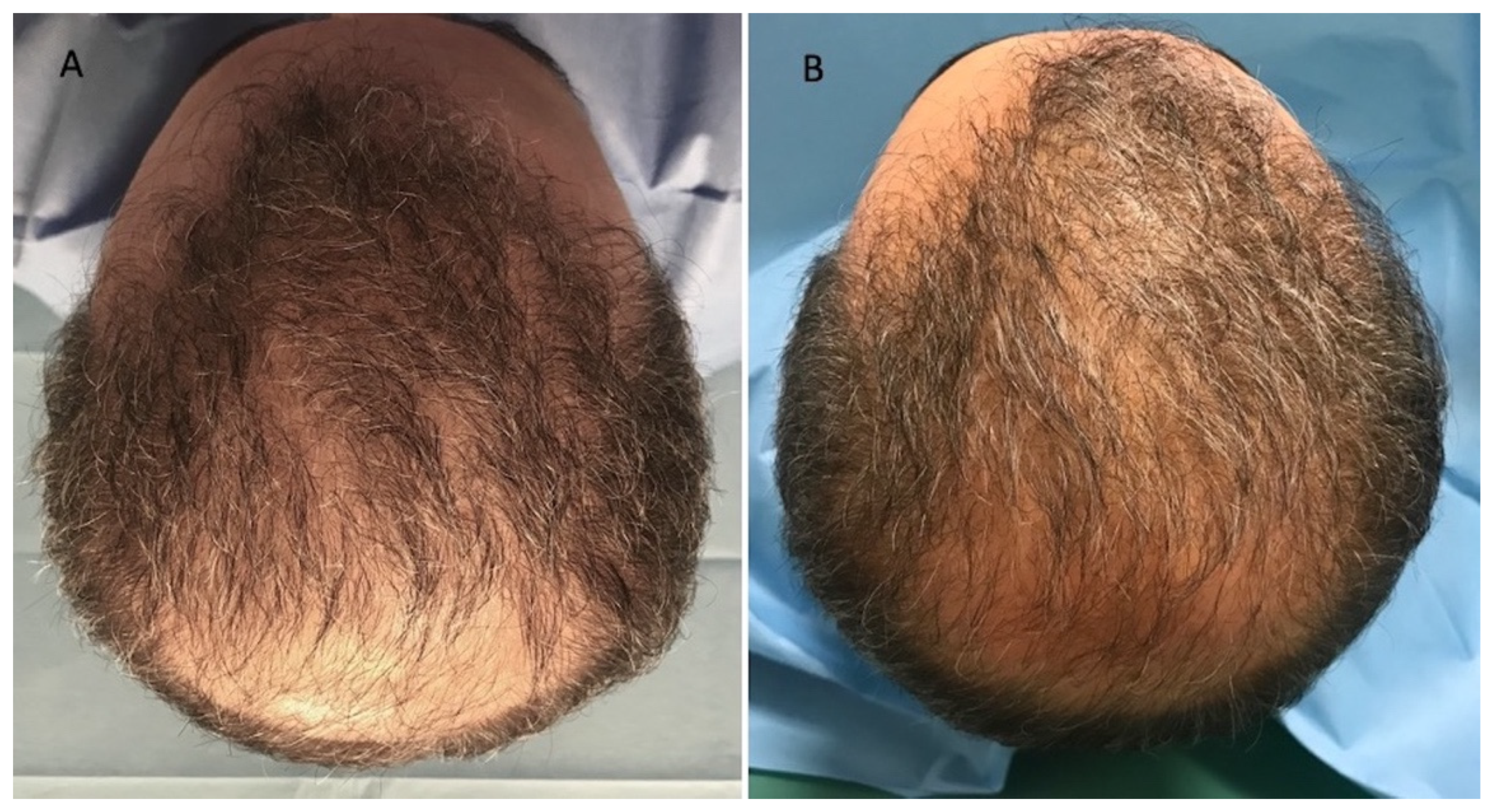
3.3. Clinical Evaluation
3.4. Limitations
4. Discussion
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Gentile, P.; Garcovich, S.; Scioli, M.G.; Bielli, A.; Orlandi, A.; Cervelli, V. Mechanical and Controlled PRP Injections in Patients Affected by Androgenetic Alopecia. J. Vis. Exp. 2018, 131, 56406. [Google Scholar] [CrossRef]
- Gentile, P.; Calabrese, C.; De Angelis, B.; Dionisi, L.; Pizzicannella, J.; Kothari, A.; De Fazio, D.; Garcovich, S. Impact of the Different Preparation Methods to Obtain Autologous Non-Activated Platelet-Rich Plasma (A-PRP) and Activated Platelet-Rich Plasma (AA-PRP) in Plastic Surgery: Wound Healing and Hair Regrowth Evaluation. Int. J. Mol. Sci. 2020, 21, 431. [Google Scholar] [CrossRef]
- Gentile, P.; Garcovich, S.; Bielli, A.; Scioli, M.G.; Orlandi, A.; Cervelli, V. The effect of platelet-rich plasma in hair regrowth: A randomized placebo-controlled trial. Stem. Cells Transl. Med. 2015, 4, 1317–1323. [Google Scholar] [CrossRef]
- Gentile, P.; Cole, J.P.; Cole, M.A.; Garcovich, S.; Bielli, A.; Scioli, M.G.; Orlandi, A.; Insalaco, C.; Cervelli, V. Evaluation of Not-Activated and Activated PRP in Hair Loss Treatment: Role of Growth Factor and Cytokine Concentrations Obtained by Different Collection Systems. Int. J. Mol. Sci. 2017, 18, 408. [Google Scholar] [CrossRef]
- Gentile, P.; Garcovich, S. Autologous activated Platelet-rich plasma (AA-PRP) and non-activated (A-PRP) in hair growth: A retrospective, blinded, randomized evaluation in androgenetic alopecia. Expert Opin. Biol. Ther. 2020, 20, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Gentile, P.; Scioli, M.G.; Bielli, A.; De Angelis, B.; De Sio, C.; De Fazio, D.; Ceccarelli, G.; Trivisonno, A.; Orlandi, A.; Cervelli, V.; et al. Platelet-Rich Plasma and Micrografts Enriched with Autologous Human Follicle Mesenchymal Stem Cells Improve Hair Re-Growth in Androgenetic Alopecia. Biomolecular Pathway Analysis and Clinical Evaluation. Biomedicines 2019, 8, 27. [Google Scholar] [CrossRef]
- Gentile, P. Autologous Cellular Method Using Micrografts of Human Adipose Tissue Derived Follicle Stem Cells in Androgenic Alopecia. Int. J. Mol. Sci. 2019, 13, 20. [Google Scholar] [CrossRef] [PubMed]
- Gentile, P.; Dionisi, L.; Pizzicannella, J.; de Angelis, B.; de Fazio, D.; Garcovich, S. A randomized blinded retrospective study: The combined use of micro-needling technique, low-level laser therapy, and autologous non-activated platelet-rich plasma improves hair re-growth in patients with androgenic alopecia. Expert Opin. Biol. Ther. 2020, 20, 1099–1109. [Google Scholar] [CrossRef] [PubMed]
- Gentile, P.; Garcovich, S.; Lee, S.-I.; Han, S. Regenerative biotechnologies in plastic surgery: A multicentric, retrospective, case-series study on the use of micro-needling with low-level light/laser therapy as a hair growth boost in patients affected by androgenetic alopecia. Appl. Sci. 2022, 12, 217. [Google Scholar] [CrossRef]
- Gentile, P.; Garcovich, S. The Effectiveness of Low-Level Light/Laser Therapy on Hair Loss. Facial Plast. Surg. Aesthet. Med. 2021. [Google Scholar] [CrossRef]
- Hoffmann, M.; Kleine-Weber, H.; Schroeder, S.; Krüger, N.; Herrler, T.; Erichsen, S.; Schiergens, T.S.; Herrler, G.; Wu, N.H.; Nitsche, A.; et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell 2020, 181, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Wiersinga, W.J.; Rhodes, A.; Cheng, A.C.; Peacock, S.J.; Prescott, H.C. Pathophysiology, Transmission, Diagnosis, and Treatment of Coronavirus Disease 2019 (COVID-19): A Review. JAMA 2020, 324, 782–793. [Google Scholar] [CrossRef] [PubMed]
- Goren, A.V.; Vano-Galvan, S.; Wambier, C.G.; McCoy, J.; Gomez-Zubiaur, A.; Moreno-Arrones, O.M.; Shapiro, J.; Sinclair, R.D.; Gold, M.H.; Kovacevic, M.; et al. A preliminary observation: Male pattern hair loss among hospitalized COVID-19 patients in Spain—A potential clue to the role of androgens in COVID-19 severity. J. Cosmet. Dermatol. 2020, 19, 1545–1547. [Google Scholar] [CrossRef]
- Wambier, C.G.; Vaño-Galván, S.; McCoy, J.; Gomez-Zubiaur, A.; Herrera, S.; Hermosa-Gelbard, Á.; Moreno-Arrones, O.M.; Jiménez-Gómez, N.; González-Cantero, A.; Fonda-Pascual, P.; et al. Androgenetic alopecia present in most patients hospitalized with COVID-19: The “Gabrin sign”. J. Am. Acad. Dermatol. 2020, 83, 680–682. [Google Scholar] [CrossRef] [PubMed]
- Wambier, C.G. Reply to “Comment on androgenetic alopecia present in the majority of patients hospitalized with COVID-19”. J. Am. Acad. Dermatol. 2020, 84, e53–e54. [Google Scholar] [CrossRef] [PubMed]
- Wambier, C.G.; Vaño-Galván, S.; McCoy, J.; Pai, S.; Dhurat, R.; Goren, A. Androgenetic alopecia in COVID-19: Compared to age-matched epidemiologic studies and hospital outcomes with or without the Gabrin sign. J. Am. Acad. Dermatol. 2020, 83, e453–e454. [Google Scholar] [CrossRef]
- Schuklenk, U.; Ashcroft, R. International research ethics. Bioethics 2000, 14, 158–172. [Google Scholar] [CrossRef]
- Von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P.; Strobe Initiative. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. J. Clin. Epidemiol. 2008, 61, 344–349. [Google Scholar] [CrossRef]
- Xiong, Q.; Xu, M.; Li, J.; Liu, Y.; Zhang, J.; Xu, Y.; Dong, W. Clinical sequelae of COVID-19 survivors in Wuhan, China: A single-center longitudinal study. Clin. Microbiol. Infect. 2020, 27, 89–95. [Google Scholar] [CrossRef]
- Kato, H.; Kinoshita, K.; Saito, N.; Kanayama, K.; Mori, M.; Asahi, N.; Sunaga, A.; Yoshizato, K.; Itami, S.; Yoshimura, K. The Effects of Ischemia and Hyperoxygenation on Hair Growth and Cycle. Organogenesis 2020, 16, 83–94. [Google Scholar] [CrossRef]
- Suchonwanit, P.; Chalermroj, N.; Khunkhet, S. Low-level laser therapy for the treatment of androgenetic alopecia in Thai men and women: A 24-week, randomized, double-blind, sham device-controlled trial. Lasers Med. Sci. 2018, 34, 1107–1114. [Google Scholar] [CrossRef] [PubMed]

| Patients | Gender | Hamilton–Norwood Degree | Ludwig Degree | Targeted Area | Age | Race | Doses | Vaccine |
|---|---|---|---|---|---|---|---|---|
| 1 | Male | III-vertex | - | Frontal, temporal, parietal, vertex | 23 | Caucasian | 1 dose | Pfizer |
| 2 | Male | III-vertex | - | Frontal, temporal, parietal, vertex | 43 | Caucasian | 2 doses | Pfizer |
| 3 | Male | III-vertex | - | Frontal, temporal, parietal, vertex | 68 | Caucasian | 3 doses | Pfizer |
| 4 | Male | IIa | - | Frontal, temporal, parietal, | 45 | Caucasian | 3 doses | Pfizer |
| 5 | Male | II | - | Frontal, temporal, parietal | 32 | Caucasian | 2 doses | Pfizer |
| 6 | Male | I | - | Frontal, parietal | 38 | Caucasian | 3 doses | Pfizer |
| 7 | Female | - | II | Frontal, temporal, parietal, vertex | 41 | Caucasian | 2 doses | Pfizer |
| 8 | Female | - | II | Frontal, temporal, parietal, vertex | 46 | Caucasian | 2 doses | Pfizer |
| 9 | Female | - | II | Frontal, temporal, parietal, vertex | 54 | Caucasian | 3 doses | Pfizer |
| 10 | Female | - | I | Frontal, parietal | 31 | Caucasian | 1 dose | Pfizer |
| Patients | Procedure | Hair Density (T0) | Hair Density (T1—16 wks) |
|---|---|---|---|
| 1 | DTSMG MTS stamp + HR3 matrix | 34 ± 2 hairs/cm2 | 45 ± 2 hairs/cm2 |
| 2 | DTSMG MTS stamp + HR3 matrix | 45 ± 2 hairs/cm2 | 56 ± 2 hairs/cm2 |
| 3 | DTSMG MTS stamp + HR3 matrix | 61 ± 2 hairs/cm2 | 72 ± 2 hairs/cm2 |
| 4 | DTSMG MTS stamp + HR3 matrix | 30 ± 2 hairs/cm2 | 41 ± 2 hairs/cm2 |
| 5 | DTSMG MTS stamp + HR3 matrix | 65 ± 2 hairs/cm2 | 76 ± 2 hairs/cm2 |
| 6 | DTSMG MTS stamp + HR3 matrix | 33 ± 2 hairs/cm2 | 44 ± 2 hairs/cm2 |
| 7 | DTSMG MTS stamp + HR3 matrix | 63 ± 2 hairs/cm2 | 74 ± 2 hairs/cm2 |
| 8 | DTSMG MTS stamp + HR3 matrix | 48 ± 2 hairs/cm2 | 59 ± 2 hairs/cm2 |
| 9 | DTSMG MTS stamp + HR3 matrix | 45 ± 2 hairs/cm2 | 56 ± 2 hairs/cm2 |
| 10 | DTSMG MTS stamp + HR3 matrix | 39 ± 2 hairs/cm2 | 50 ± 2 hairs/cm2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gentile, P. Preliminary Investigation on Micro-Needling with Low-Level LED Therapy and Growth Factors in Hair Loss Related to COVID-19. J. Clin. Med. 2022, 11, 5760. https://doi.org/10.3390/jcm11195760
Gentile P. Preliminary Investigation on Micro-Needling with Low-Level LED Therapy and Growth Factors in Hair Loss Related to COVID-19. Journal of Clinical Medicine. 2022; 11(19):5760. https://doi.org/10.3390/jcm11195760
Chicago/Turabian StyleGentile, Pietro. 2022. "Preliminary Investigation on Micro-Needling with Low-Level LED Therapy and Growth Factors in Hair Loss Related to COVID-19" Journal of Clinical Medicine 11, no. 19: 5760. https://doi.org/10.3390/jcm11195760
APA StyleGentile, P. (2022). Preliminary Investigation on Micro-Needling with Low-Level LED Therapy and Growth Factors in Hair Loss Related to COVID-19. Journal of Clinical Medicine, 11(19), 5760. https://doi.org/10.3390/jcm11195760







