Combined Trans-Arterial Embolization and Ablation for the Treatment of Large (>3 cm) Liver Metastases: Review of the Literature
Abstract
:1. Introduction
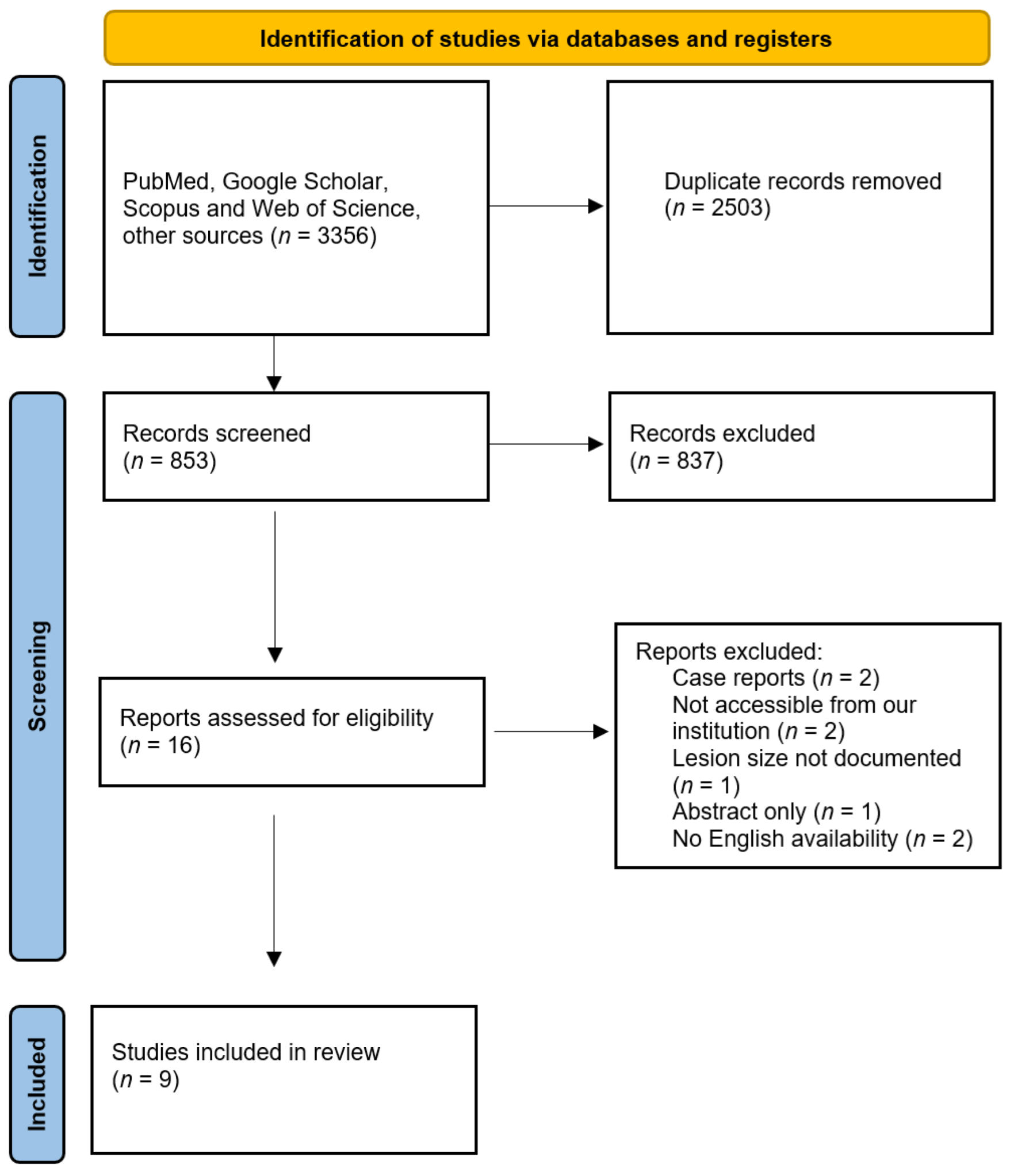
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tsilimigras, D.I.; Brodt, P.; Clavien, P.A.; Muschel, R.J.; D’Angelica, M.I.; Endo, I.; Parks, R.W.; Doyle, M.; de Santibañes, E.; Pawlik, T.M. Liver metastases. Nat. Rev. Dis. Primers 2021, 7, 27. [Google Scholar] [CrossRef]
- Horn, S.R.; Stoltzfus, K.C.; Lehrer, E.J.; Dawson, L.A.; Tchelebi, L.; Gusani, N.J.; Sharma, N.K.; Chen, H.; Trifiletti, D.M.; Zaorsky, N.G. Epidemiology of liver metastases. Cancer Epidemiol. 2020, 67, 101760. [Google Scholar] [CrossRef] [PubMed]
- Garden, O.J.; Rees, M.; Poston, G.J.; Mirza, D.; Saunders, M.; Ledermann, J.; Primrose, J.N.; Parks, R.W. Guidelines for resection of colorectal cancer liver metastases. Gut 2006, 55 (Suppl. 3), iii1–iii8. [Google Scholar] [CrossRef] [PubMed]
- Vera, R.; González-Flores, E.; Rubio, C.; Urbano, J.; Camps, M.V.; Ciampi-Dopazo, J.J.; Rincón, J.O.; Macías, V.M.; Braco, M.A.G.; Suarez-Artacho, G. Multidisciplinary management of liver metastases in patients with colorectal cancer: A consensus of SEOM, AEC, SEOR, SERVEI, and SEMNIM. Clin. Transl. Oncol. 2020, 22, 647–662. [Google Scholar] [CrossRef] [PubMed]
- Kagawa, T.; Koizumi, J.; Kojima, S.I.; Nagata, N.; Numata, M.; Watanabe, N.; Watanabe, T.; Mine, T. Transcatheter arterial chemoembolization plus radiofrequency ablation therapy for early stage hepatocellular carcinoma comparison with surgical resection. Cancer 2010, 116, 3638–3644. [Google Scholar] [CrossRef]
- Wáng, Y.X.; De Baere, T.; Idée, J.M.; Ballet, S. Transcatheter embolization therapy in liver cancer: An update of clinical evidences. Chin. J. Cancer Res. 2015, 27, 96–121. [Google Scholar] [PubMed]
- Izzo, F.; Granata, V.; Grassi, R.; Fusco, R.; Palaia, R.; Delrio, P.; Carrafiello, G.; Azoulay, D.; Petrillo, A.; Curley, S.A. Radiofrequency Ablation and Microwave Ablation in Liver Tumors: An Update. Oncologist 2019, 24, e990–e1005. [Google Scholar] [CrossRef]
- Vogl, T.J.; Gruber, T.; Balzer, J.O.; Eichler, K.; Hammerstingl, R.; Zangos, S. Repeated Transarterial Chemoembolization in the Treatment of Liver Metastases of Colorectal Cancer: Prospective Study. Radiology 2009, 250, 281–289. [Google Scholar] [CrossRef]
- Vogl, T.J.; Gruber, T.; Naguib, N.N.N.; Hammerstingl, R.; Nour-Eldin, N.-E.A. Liver Metastases of Neuroendocrine Tumors: Treatment With Hepatic Transarterial Chemotherapy Using Two Therapeutic Protocols. AJR Am. J. Roentgenol. 2009, 193, 941–947. [Google Scholar] [CrossRef]
- Giroux, M.-F.; Baum, R.A.; Soulen, M.C. Chemoembolization of Liver Metastasis from Breast Carcinoma. J. Vasc. Interv. Radiol. 2004, 15, 289–291. [Google Scholar] [CrossRef]
- Liu, C.; Li, T.; He, J.-T.; Shao, H. TACE combined with microwave ablation therapy vs. TACE alone for treatment of early- and intermediate-stage hepatocellular carcinomas larger than 5 cm: A meta-analysis. Diagn. Interv. Radiol. 2020, 26, 575–583. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Gadaleta, C.D.; Catino, A.; Ranieri, G.; Fazio, V.; Gadaleta-Caldarola, G.; Cramarossa, A.; Armenise, F.; Canniello, E.; Vinciarelli, G.; Laricchia, G.; et al. Single-step therapy—Feasibility and safety of simultaneous transarterial chemoembolization and radiofrequency ablation for hepatic malignancies. In Vivo 2009, 23, 813–820. [Google Scholar]
- Fong, Z.V.; Palazzo, F.; Needleman, L.; Brown, D.B.; Eschelman, D.J.; Chojnacki, K.A.; Yeo, C.J.; Rosato, E.L. Combined Hepatic Arterial Embolization and Hepatic Ablation for Unresectable Colorectal Metastases to the Liver. Am. Surg. 2012, 78, 1243–1248. [Google Scholar] [CrossRef] [PubMed]
- Kan, X.F.; Wang, Y.; Lin, G.C.; Xia, X.W.; Xiong, B.; Zhou, G.F.; Liang, H.M.; Feng, G.S.; Zheng, C.S. Radiofrequency ablation combined with transarterial chemoembolization for liver metastases from gastrointestinal cancers. J. Huazhong Univ. Sci. Technol. Med. Sci. 2016, 36, 200–204. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.-B.; Si, Z.-M.; Qian, S.; Liu, L.-X.; Qu, X.-D.; Zhou, B.; Zhang, W.; Wang, G.-Z.; Liu, R.; Wang, J.-H. Percutaneous microwave ablation combined with synchronous transcatheter arterial chemoembolization for the treatment of colorectal liver metastases: Results from a follow-up cohort. OncoTargets Ther. 2016, 9, 3783–3789. [Google Scholar] [CrossRef] [PubMed]
- Yamakado, K.; Inaba, Y.; Sato, Y.; Yasumoto, T.; Hayashi, S.; Yamanaka, T.; Nobata, K.; Takaki, H.; Nakatsuka, A. Radiofrequency Ablation Combined with Hepatic Arterial Chemoembolization Using Degradable Starch Microsphere Mixed with Mitomycin C for the Treatment of Liver Metastasis from Colorectal Cancer: A Prospective Multicenter Study. Cardiovasc. Interv. Radiol. 2017, 40, 560–567. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Liu, B.; Long, H.; Zhang, F.; Wang, S.; Li, F. Clinical study of radiofrequency ablation combined with TACE in the treatment of breast cancer with liver metastasis. Oncol. Lett. 2017, 14, 2699–2702. [Google Scholar] [CrossRef]
- Alexander, E.S.; Mick, R.; Nadolski, G.J.; Mondschein, J.I.; Stavropoulos, S.W.; Soulen, M.C. Combined chemoembolization and thermal ablation for the treatment of metastases to the liver. Abdom. Radiol. 2018, 43, 2859–2867. [Google Scholar] [CrossRef]
- Faiella, E.; Santucci, D.; Bernetti, C.; Schena, E.; Pacella, G.; Zobel, B.B.; Grasso, R.F. Combined trans-arterial embolisation and microwave ablation for the treatment of large unresectable hepatic metastases (>3 cm in maximal diameter). Int. J. Hyperth. 2020, 37, 1395–1403. [Google Scholar] [CrossRef]
- Kobe, A.; Tselikas, L.; Deschamps, F.; Roux, C.; Delpla, A.; Varin, E.; Hakime, A.; De Baère, T. Single-session transarterial chemoembolization combined with percutaneous thermal ablation in liver metastases 3 cm or larger. Diagn. Interv. Imaging 2022. [Google Scholar] [CrossRef] [PubMed]
- Acciuffi, S.; Meyer, F.; Bauschke, A.; Croner, R.; Settmacher, U.; Altendorf-Hofmann, A. Solitary colorectal liver metastasis: Overview of treatment strategies and role of prognostic factors. J. Cancer Res. Clin. Oncol. 2022, 148, 657–665. [Google Scholar] [CrossRef] [PubMed]
- Crocetti, L.; De Baére, T.; Pereira, P.L.; Tarantino, F.P. CIRSE Standards of Practice on Thermal Ablation of Liver Tumours. Cardiovasc. Interv. Radiol. 2020, 43, 951–962. [Google Scholar] [CrossRef] [PubMed]
- Lucatelli, P.; Burrel, M.; Guiu, B.; de Rubeis, G.; van Delden, O.; Helmberger, T. CIRSE Standards of Practice on Hepatic Transarterial Chemoembolisation. Cardiovasc. Interv. Radiol. 2021, 44, 1851–1867. [Google Scholar] [CrossRef]
- Kim, Y.-S.; Rhim, H.; Lim, H.K.; Choi, D.; Lee, W.J.; Kim, S.H. Hepatic Infarction after Radiofrequency Ablation of Hepatocellular Carcinoma with an Internally Cooled Electrode. J. Vasc. Interv. Radiol. 2007, 18, 1126–1133. [Google Scholar] [CrossRef]
| Authors | Publication Year | Journal | Country of Study | Study Design | Number of Patients | Age (Years) |
|---|---|---|---|---|---|---|
| Gadaleta et al. [13] | 2009 | In Vivo | Italy | Retrospective | 34 (21 with HCC, 13 with liver metastases) | 70 (range: 47–83) |
| Fong, Z.V. et al. [14] | 2012 | The American Surgeon | USA | Retrospective | 32 | 74.1 (range: 50–96) |
| Kan et al. [15] | 2016 | Journal of Huazhong University of Science and Technology | China | Retrospective | 19 | 61.8 (range: 34–82) |
| Wu et al. [16] | 2016 | OncoTargets and Therapy | China | Retrospective | 30 | 61.6 (range: 44–78) |
| Yamakado et al. [17] | 2017 | CardioVascular and Interventional Radiology | Japan | Prospective | 25 | 70.2 (range: 55–82) |
| Wang et al. [18] | 2017 | Oncology Letters | China | Prospective | 88 (control group = 50; observational group = 38) | 56.7 |
| Alexander, E.S. et al. [19] | 2018 | Abdominal Radiology | USA | Retrospective | 42 | 62 (range: 38–83) |
| Faiella et al. [20] | 2020 | International Journal of Hyperthermia | Italy | Retrospective | 22 | 58.5 (range: 43–81) |
| Kobe et al. [21] | 2022 | Diagnostic and Interventional Imaging | France | Retrospective | 39 | 55 (range: 28–77) |
| Authors | Average Maximum Lesion Diameter | Primary Tumor | Therapy | Median Survival | Major Complications |
|---|---|---|---|---|---|
| Gadaleta et al. [13] | 2.5 cm (1–6 cm) | Colorectal 9/13 (69%) Breast 3/13 (23%) Ovarian 1/13 (8%) | TACE + RFA | N/A | 1 died after acute liver failure |
| Fong, Z.V. et al. [14] | 4.4 cm (1.7–7.9 cm) | Colorectal 32/32 (100%) | TACE and 90Y + MWA | 36 months | 1 liver abscess, 2 postoperative ileus, 1 cholecystitis, 1 apical pneumothorax, and 1 portal vein thrombosis |
| Kan et al. [15] | 4.2 cm (1.5–7.8 cm) | Colorectal 12/19 (63%) Gastric 5/19 (26%) Esophagus 2/19 (11%) | TACE + RFA | 35.2 months | 1 died after bile duct injury, 1 segmental hepatic infarction |
| Wu et al. [16] | 4.4 ± 2.6 cm (1.4–10.0 cm) | Colorectal 30/30 (100%) | TACE + MWA | 11.0 months | None |
| Yamakado et al. [17] | 2.2 ± 0.9 cm (1.0–4.2 cm) | Colorectal 25/25 (100%) | TACE + RFA | 48.4 months | None |
| Wang et al. [18] | 3.5 ± 1.3 cm | Breast 88/88 (100%) | TACE + RFA | 15.6 months | 1 bone marrow suppression, 1 infection (non-specified), 1 severe digestive tract symptoms, and 1 liver and kidney damage |
| Alexander, E.S. et al. [19] | 4.7 cm (1.5–8.0 cm) | Colorectal 18/42 (43%) Sarcoma 5/42 (12%) Cholangiocarcinoma 5/42 (12%) Pancreatic neuroendocrine 4/42 (10%) Pancreatic adenocarcinoma 4/42 (10%) Breast 3/42 (7%) Appendiceal 1/42 (2%) Esophagus b1/42 (2%) Pyriform squamous 1/42 (2%) | TACE + RFA (32) or MWA (8) or CA (2) | 55% (95% CI 40–71%) at 1 year and 30% (95% CI 16–45%) at 2 years | 3 liver abscesses, 2 groin bleeds, 1 pseudoaneurysm, 1 portal vein thrombus with lobar infarct, 1 retroperitoneal hematoma, and 1 biliary fistula |
| Faiella et al. [20] | 3.7 cm (3.2–7.3 cm) | Breast 10/22 (45%) Colorectal 10/22 (45%) Neuroendocrine tumors 2/22 (5%) Leiomyosarcoma 2/22 (5%) | TAE + MWA | No residue/recurrence detected at the CT scan follow-up (2 years) | 1 bleeding |
| Kobe et al. [21] | 3.6 ± 0.6 cm (range: 3–5 cm) | Colorectal 8/46 (17%) Sarcoma 10/46 (22%) Adrenal carcinoma 10/46 (22%) Thyroid carcinoma 3/46 (7%) Neuroendocrine tumor 4/46 (9%) Breast 3/46 (7%) Other 8/46 (17%) | TACE + RFA (34) or MWA (11) or CA (1) | 95% at 1 year and 77% at 2 years | 1 pleural effusion, 1 segmental, 1 portal vein thrombosis, and 1 subcapsular hematoma |
| Authors | TAE/TACE Methods |
|---|---|
| Gadaleta et al. [13] | 23 patients: 35 mg epirubicin plus 15 mg mitomycin C 9 patients: 100 mg doxorubicin loaded on Dc-Beads (Biocompatibles, Farnham, UK) 1 patient: 100 mg doxorubicin loaded on Hepasphere (BioSphere Medical, Roissy, Cedex, France) 4 patients: 100 mg irinotecan loaded on Dc-Beads |
| Fong, Z.V. et al. [14] | 50 mg cisplatin, 50 mg adriamycin, and 10 mg mitomycin emulsified with Lipiodol (Guerbet, Bloomington, IN, USA) and Gelfoam slurry |
| Kan et al. [15] | 20–50 mg adriamycin or 4–10 mg mitomycin C emulsified with 2 to 12.5 mL Lipiodol UltraFluid, followed by embolization with gelatin sponge particles |
| Wu et al. [16] | 50–150 mg oxaliplatin, 10–50 mg epirubicin, and 1.5–10 mL Lipiodol, followed by embolization with gelatin sponge particles for insufficient embolization cases |
| Yamakado et al. [17] | A total of 2–6 mg mitomycin C was dissolved in 5 mL of distilled water and mixed with 300 mg degradable starch microspheres (Spherex; Yakult Co., Ltd., Tokyo, Japan). |
| Wang et al. [18] | Vinorelbine + capecitabine with or without trastuzumab, docetaxel + capecitabine with or without trastuzumab |
| Alexander, E.S. et al. [19] | Prior to 2010: 100 mg cisplatin, 50 mg doxorubicin, and 10 mg mitomycin C, followed by 150–250 µm Contour PVA Embolization Particle (Boston Scientific, Marlborough, MA, USA) From 2010 on: 50 mg doxorubicin, and 10 mg mitomycin C, followed by 100–300 µm Embospheres (Merit Medical, Salt Lake City, UT, USA) |
| Faiella et al. [20] | Embozene Microspheres 75 µm and 100 µm spheres (Embozene, Color-Advanced Microspheres; Celonova BioSciences, Peachtree City, GA, USA) |
| Kobe et al. [21] | Doxorubicin or irinotecan mixed with Lipiodol (32 lesions) (followed by embolization with gelatin sponge) or drug-eluting beads (Boston Scientific) (14 lesions) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Faiella, E.; Calabrese, A.; Santucci, D.; de Felice, C.; Pusceddu, C.; Fior, D.; Fontana, F.; Piacentino, F.; Moramarco, L.P.; Muraca, R.M.; et al. Combined Trans-Arterial Embolization and Ablation for the Treatment of Large (>3 cm) Liver Metastases: Review of the Literature. J. Clin. Med. 2022, 11, 5576. https://doi.org/10.3390/jcm11195576
Faiella E, Calabrese A, Santucci D, de Felice C, Pusceddu C, Fior D, Fontana F, Piacentino F, Moramarco LP, Muraca RM, et al. Combined Trans-Arterial Embolization and Ablation for the Treatment of Large (>3 cm) Liver Metastases: Review of the Literature. Journal of Clinical Medicine. 2022; 11(19):5576. https://doi.org/10.3390/jcm11195576
Chicago/Turabian StyleFaiella, Eliodoro, Alessandro Calabrese, Domiziana Santucci, Carlo de Felice, Claudio Pusceddu, Davide Fior, Federico Fontana, Filippo Piacentino, Lorenzo Paolo Moramarco, Rosa Maria Muraca, and et al. 2022. "Combined Trans-Arterial Embolization and Ablation for the Treatment of Large (>3 cm) Liver Metastases: Review of the Literature" Journal of Clinical Medicine 11, no. 19: 5576. https://doi.org/10.3390/jcm11195576
APA StyleFaiella, E., Calabrese, A., Santucci, D., de Felice, C., Pusceddu, C., Fior, D., Fontana, F., Piacentino, F., Moramarco, L. P., Muraca, R. M., & Venturini, M. (2022). Combined Trans-Arterial Embolization and Ablation for the Treatment of Large (>3 cm) Liver Metastases: Review of the Literature. Journal of Clinical Medicine, 11(19), 5576. https://doi.org/10.3390/jcm11195576






